chromatography
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What is chromatography used for? (1)
Chromatography is used to separate mixtures into their individual components
What are the two phases in chromatography? (2)
The mobile phase and the stationary phase.
What is the stationary phase in chromatography? (1)
A solid (or a liquid in gas-liquid chromatography) that the mixture travels across
How does the stationary phase affect the movement of the mixture? (1)
Components more attracted to the stationary phase move slower
What is the mobile phase in chromatography? (1)
A solvent that dissolves the mixture and carries its components across the stationary phase
How does solubility affect movement in the mobile phase? (1)
More soluble components move faster with the solvent.
How does polarity influence the interaction with the stationary phase and mobile phase? (4)
- A polar solvent dissolves polar molecules.
- A non-polar solvent dissolves non-polar molecules.
- A polar stationary phase attracts polar molecules.
- A non-polar stationary phase attracts non-polar molecules.
What is TLC? (1)
Thin layer chromatography
Draw and label the apparatus for TLC (5)
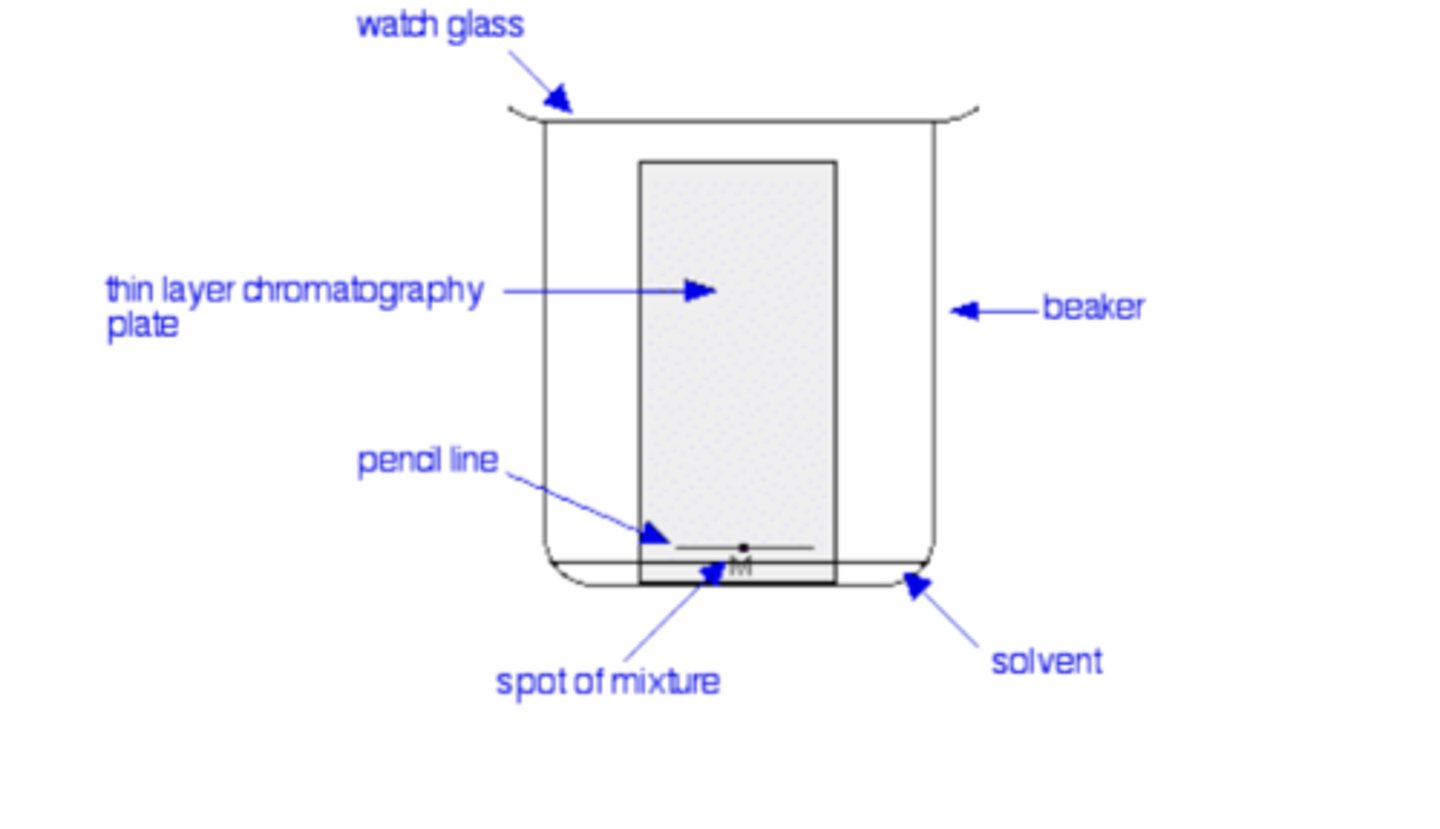
What is used as the stationary phase in thin layer chromatography? (1)
A thin, uniform layer of silica gel or alumina coated onto a rigid plastic piece
What is the mobile phase in thin layer chromatography? (1)
A suitable liquid solvent that contains the dissolved mixture
Why must a pencil line be used to mark the original position of the drop in thin layer chromatography? (1)
Pencil must be used because ink dyes could also move as the chromatogram develops
Why is it important for the solvent level to be below the line with the spot of mixture in thin layer chromatography? (1)
To prevent the mixture from dissolving off the TLC plate into the solvent mixture
How can separated spots be made visible after a TLC run? (1)
Use UV light or a developing agent such as ninhydrin
What is the equation to calculate Rf (retention factor) in chromatography? (2)

How are amino acids separated using TLC? (6)
1. The amino acid is 'spotted' on the start line.
2. The TLC plate is placed in a beaker coated with a polar solid.
3. A non-polar solvent is added to the beaker.
4. The solvent dissolves the amino acid and carries it up the plate.
5. The plate is removed before the solvent reaches the top.
6. The amino acid's position on the plate can be viewed using UV light or ninhydrin.
What determines the specific distance traveled by an amino acid on a TLC plate? (2)
- Adsorption on the plate (polar interactions)
- Solubility in the solvent (non-polar interactions)
How can the primary structure of a protein be determined using TLC? (5)
1. Hydrolyse the protein using concentrated acid (additional NH2 groups on amino acids make them more polar after hydrolysis)
2. Run a TLC plate
3. View the component spots (each amino acid or dipeptide travels a unique distance)
4. Calculate the Rf values
5. Compare the Rf values with known data
What is two-way paper chromatography used for? (1)
Two-way paper chromatography separates substances with very similar Rf values
How is a two-way paper chromatography experiment set up initially? (2)
1. A single spot of mixture is placed towards one end of the baseline.
2. The paper is stood in a solvent until the solvent front reaches close to the top of the paper.
What happens after the first solvent run in two-way paper chromatography? (2)
1. The position of the solvent front (SF1) is marked in pencil.
2. The paper is rotated 90° and developed again in a different solvent.
Why is it unlikely for two substances to have the same Rf value in both solvents during two-way paper chromatography? (1)
It is because the two substances will likely move by different amounts in the second solvent.
How are Rf values used in two-way paper chromatography? (1)
Rf values for each spot in both solvents are calculated and compared with known compounds under the same conditions
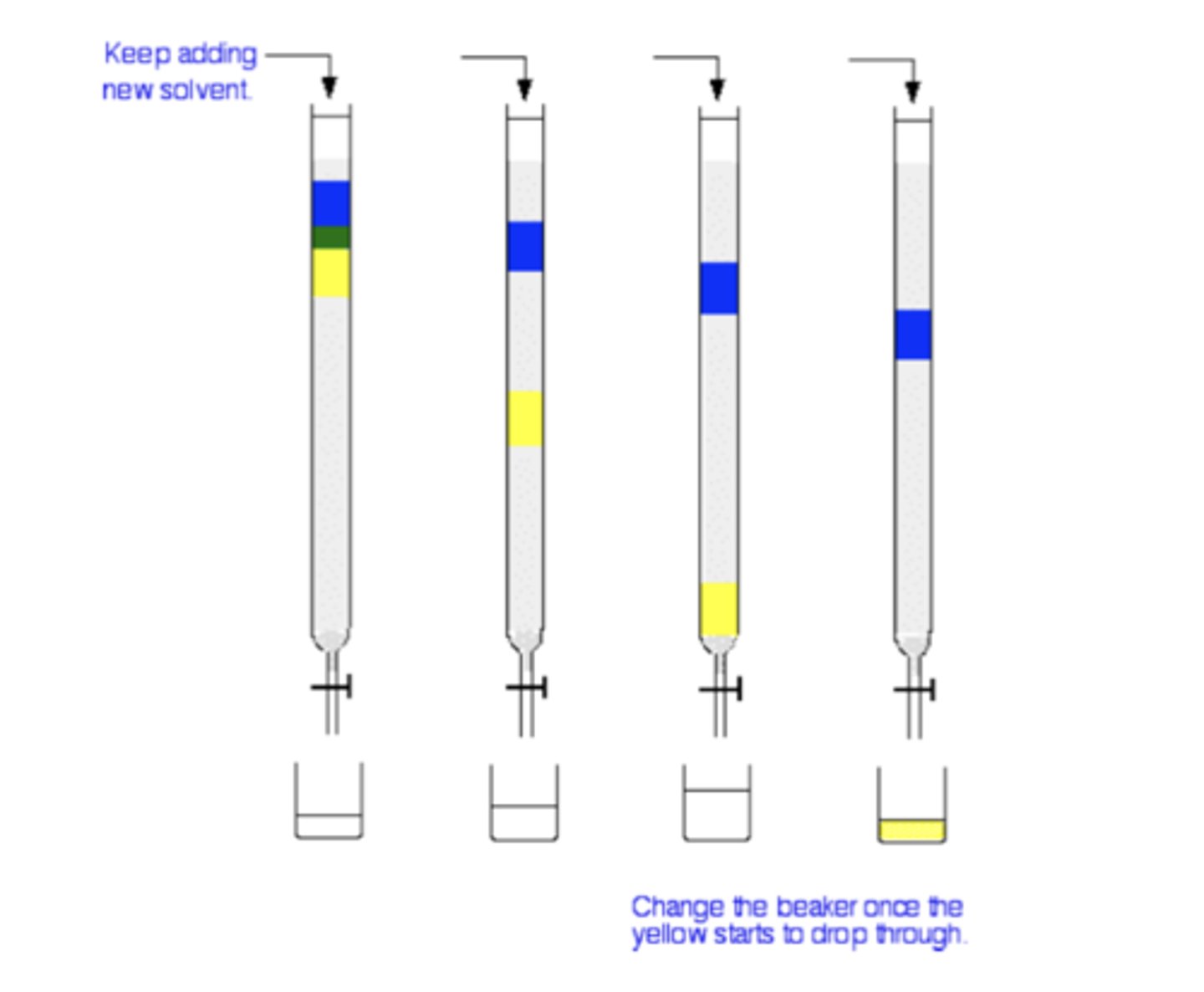
What is the stationary phase in column chromatography? (1)
A powder such as silica or aluminium oxide (alumina)
How is the stationary phase used in column chromatography? (1)
It is packed into a narrow column like a burette
How is the solvent (eluent) introduced in column chromatography? (1)
The solvent is added from the top of the column
What happens as the solvent moves down the column in column chromatography? (2)
1. The different components of the mixture separate as they move at different speeds.
2. The separated portions are collected in a beaker at the bottom
What can be done if a component of the mixture does not move down the column? (1)
A different solvent can be used to better match the polarity of the molecule
Draw the apparatus for column chromatography (3)
Where is the stationary phase located in gas chromatography? (1)
Inside a coiled column within the machine
What is the mobile phase in gas chromatography? (1)
An inert gas, such as nitrogen or helium
How is the sample carried in gas chromatography? (1)
The sample is carried with the gas as it is injected
Why do portions of the mixture have different retention times in gas chromatography? (2)
- Some portions of the sample are carried well and quickly by the gas, emerging first.
- Other portions interact differently and emerge later.
Draw an example graph produced from gas chromatography (3)
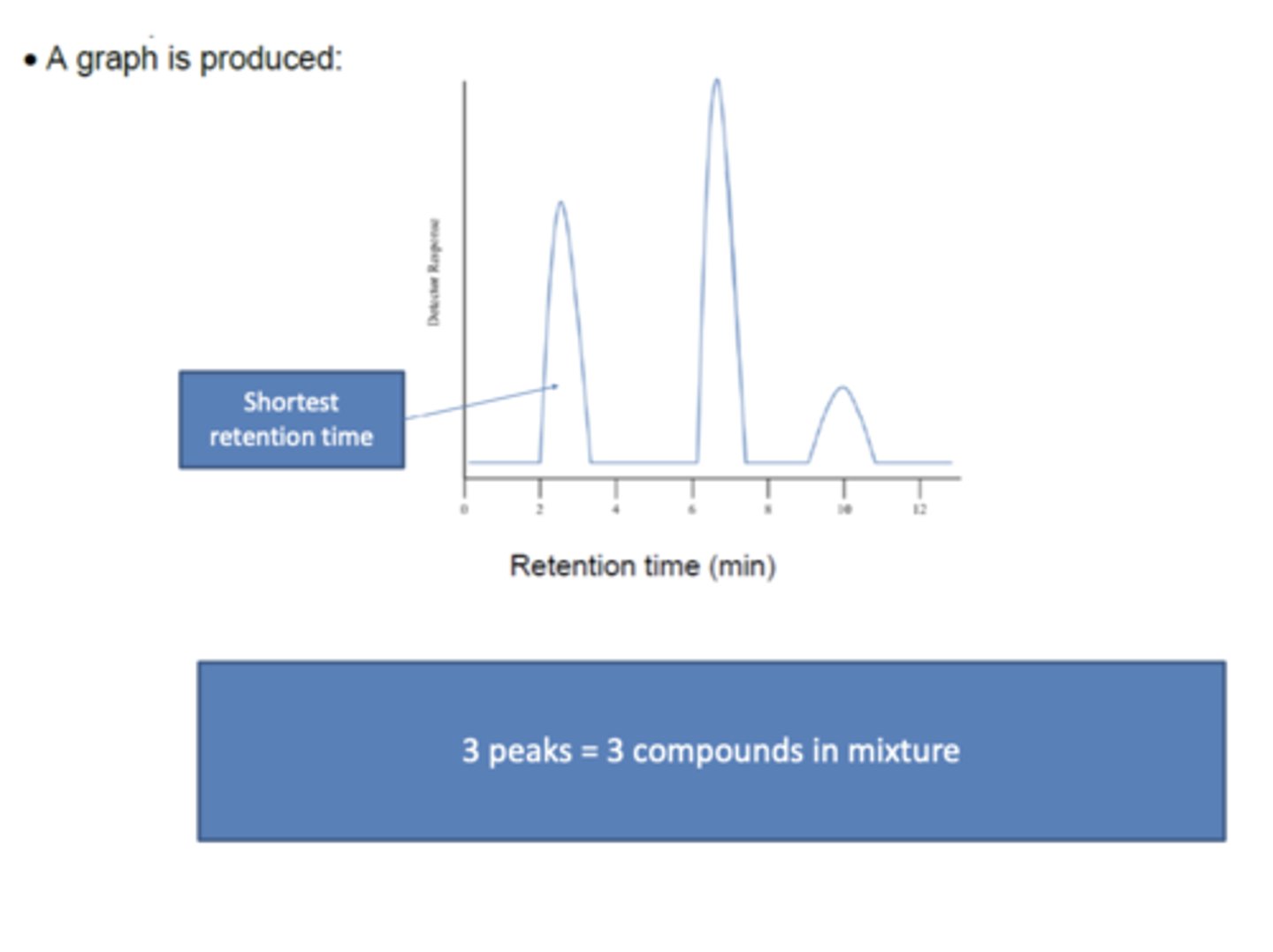
How can the number of compounds in a sample be evaluated using gas chromatography? (1)
Count the number of peaks; each peak corresponds to a single compound.
How can a substance's identity be determined using gas chromatography? (1)
By matching its retention time (tR) with a value in a data book
What does the area under a peak in gas chromatography represent? (1)
The peak area is proportional to the concentration of the substance in the original sample
Why is gas chromatography used in doping tests for athletes? (1)
It is highly accurate and can identify substances in a mixture