chem 101 thermochem
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
what is energy
the capacity to do work (w) or produce heat (q)
is absolute energy measureable
no, only the energy transferred can be measured (change in E) → measured indirectly by heat or work
system vs surroundings
system = part of universe we are studying (often represents reaction taking place)
surrounding = everything else
what is work
energy transfer due to an applied force
heat
energy transfer due to a difference in temperature
OR
energy transfer due to anything other than work
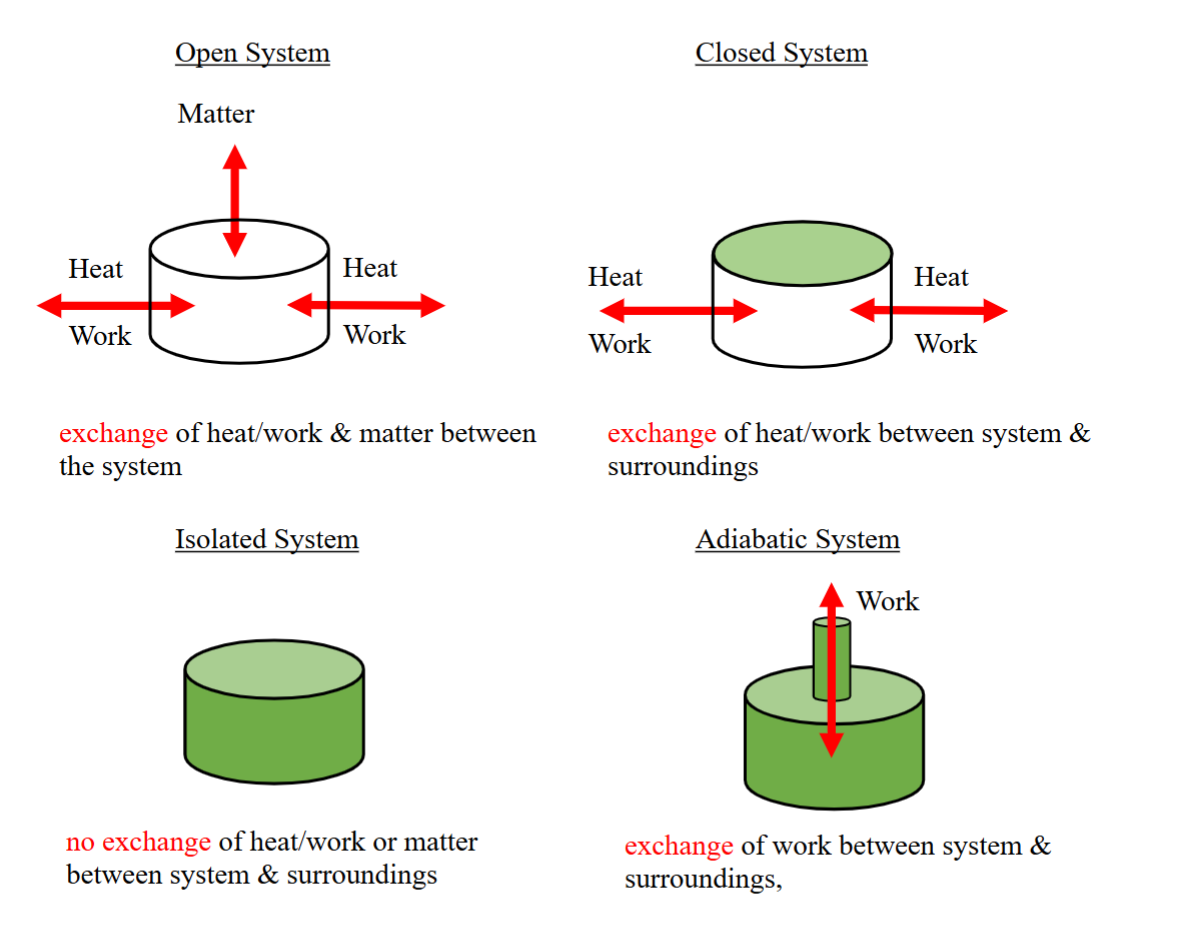
open, closed, isolated and adiabatic system
adiabatic (thermos)
state function
functions or properties that define the present condition of a system (not dependent on path taken)
T or F: heat and work are state functions
false, they are a transfer of E
first law of thermodynamics
the energy of an isolated system is constant
(equal to zero)

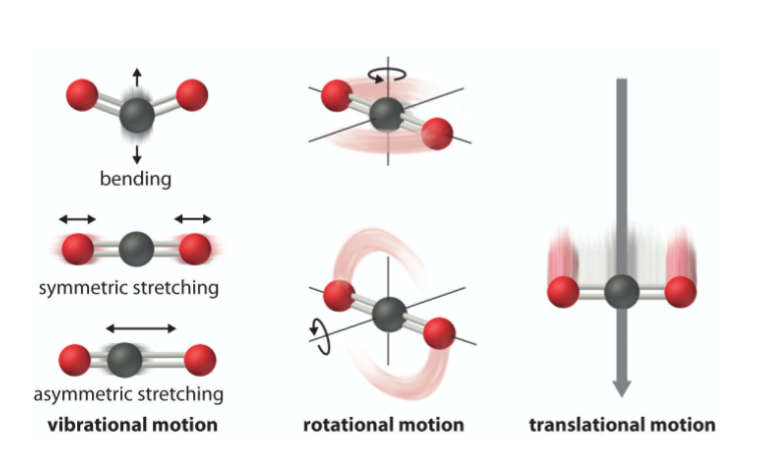
internal energy
just U or E (no change) all the motion inside molecule
consists of translational energy (motion)
energy stored in bonds (rotational, vibrational and electronic)
energy stored in the intermolecular forces
positive delta U, positive q, positive w
energy of system increases
system absorbs heat (endothermic)
work done ON system by surroundings (prespective of system)
negative delta U, negative q, negative w
energy of system decreases, system releases heat, work done BY system on surroundings (perspective of system)
extensive property + example
property that depends on the amount of substance
ex. heat, mass, volume
intensive properties + examples
property that does not depend on amount of substance
ex. temp, density, and melting point
since heat is an extensive property, what does heat depend on
how many degrees is the substance heated or cooled
amount (mass)
nature
heat capacity
amount of heat required to changed the temperature of a system by one degree
extensive property
specific heat capacity c
(J/gK)
amount of heat required to change to temperature of one gram of a system by one degree
intensive
molar heat capacity C
(J/ mol*K)
amount of heat required to change the temperature of a system by one degree per mol
intensive
calorimeter
an isolated system where no energy or matter is exchanged with surroundings
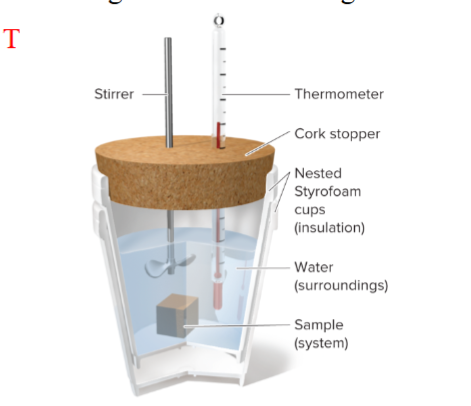
what is calorimeter used to measure
heat released or absorbed during a reaction
bomb calorimetry
exothermic reaction inside bomb will heat up water to measure energy change
determine caloric content of food
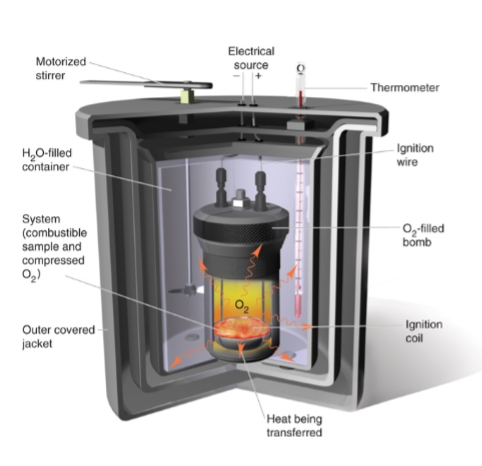
similarities and differences between regular calorimeter vs bomb
similar = both are isolated sytems
differences = bomb is constant volume, regular is constant pressure
when doing bomb calorimeter calculations, what is the most important thing to keep in mind
q(cal)= q(rxn)
what are the most common type of work encountered in chemical processes
work done by a gas the work is negative (expansion)
work done to a gas is positive (compression)
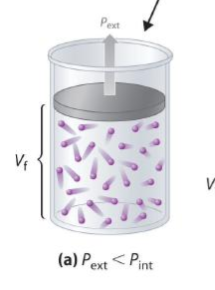
external vs internal pressures of gas expansion
Pext < Pint
Vf > Vi
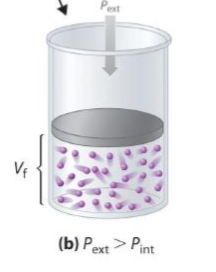
external vs interal pressure of gas compression
Pext > Pint
Vi > Vf
enthalpy
change in systems internal energy + product and energy changes (work and internal energy at constant pressure)
what happens to pressure volume system if volume is constant
work=0, measuring delta U which is equal to enthalpy (delta H)
what happens when pressure volume system if pressure is constant
delta U= qp - PdeltaV (some work will still be done)
qp represents enthalpy
what are the 3 ways to calculate enthalpy
Hess’s Law
Standard enthalpies of formation
Bond Dissociation Enthalpies
Hess’s Law
change in enthalpy of a reaction is the sum of all steps in the reactions
OR
is the sum of enthalpy of any reactions that sum to give the overall reaction
ehthalpy is a ____ and _____ property
state function, extentsive property
standard enthalpies of formation
change in enthalpy that accompanies the formation of 1 mol of substance from its elements (from their standard states)
standard enthalpy of formation of a pure element in its reference form is
ZERO (O2, n2, C, H+)
why is enthalpy of bond dissociation mainly for gas phase
no intermolecular forces