Signaling and Cell Cycle Control
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
During the Eukaryotic cell cycle what occurs in nearly every tissue during devlopment
cell division
what is cell division
specific events required to ensure every cell has eveything it needs to live
After passing M phase and into G1, what 2 things can a cell do?
continues through another division
enters G0
what happens in G0
differentiated cells stop dividing and enter this phase
what is G0 phase also known as
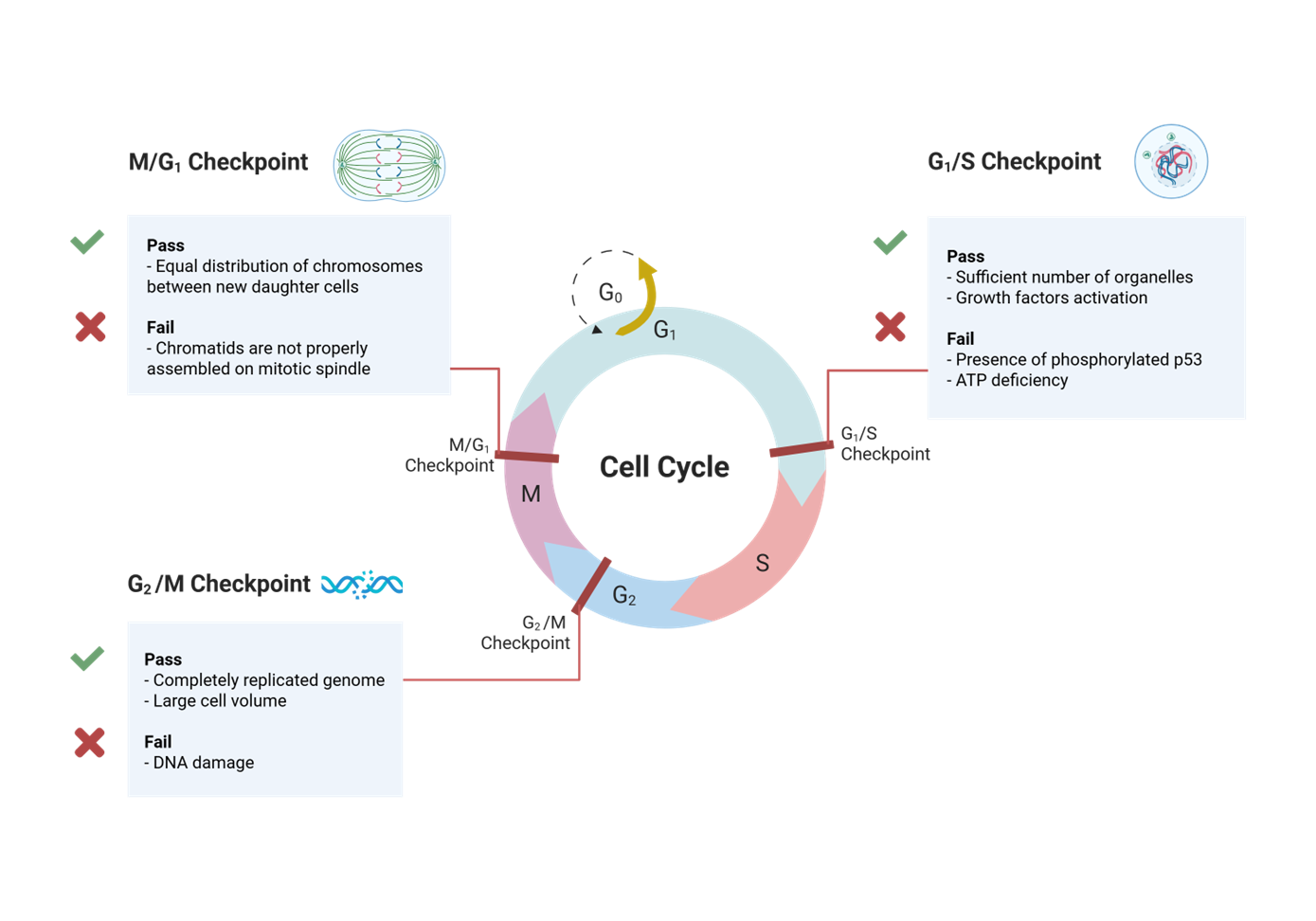
quiescent
what does quiescent mean
being in a state of quiet, inactivity, stillness, or dormancy
what does uinregulated cell division lead to?
cancer development
What type of development is primarily post-natal
mammary gland development
Mammary gland development is primarily post-natal
• The ductal tree develops as an invagination of ectoderm.
• At birth, a rudimentary “ductal tree” structure is already
present.
• Very limited development occurs until the onset of puberty
In relation to signals and receptors in pro-growth cell signals , what type of protein molecules require a cell membrane receptor
protein signaling molecules
What type of signaling molecule use nuclear receptors
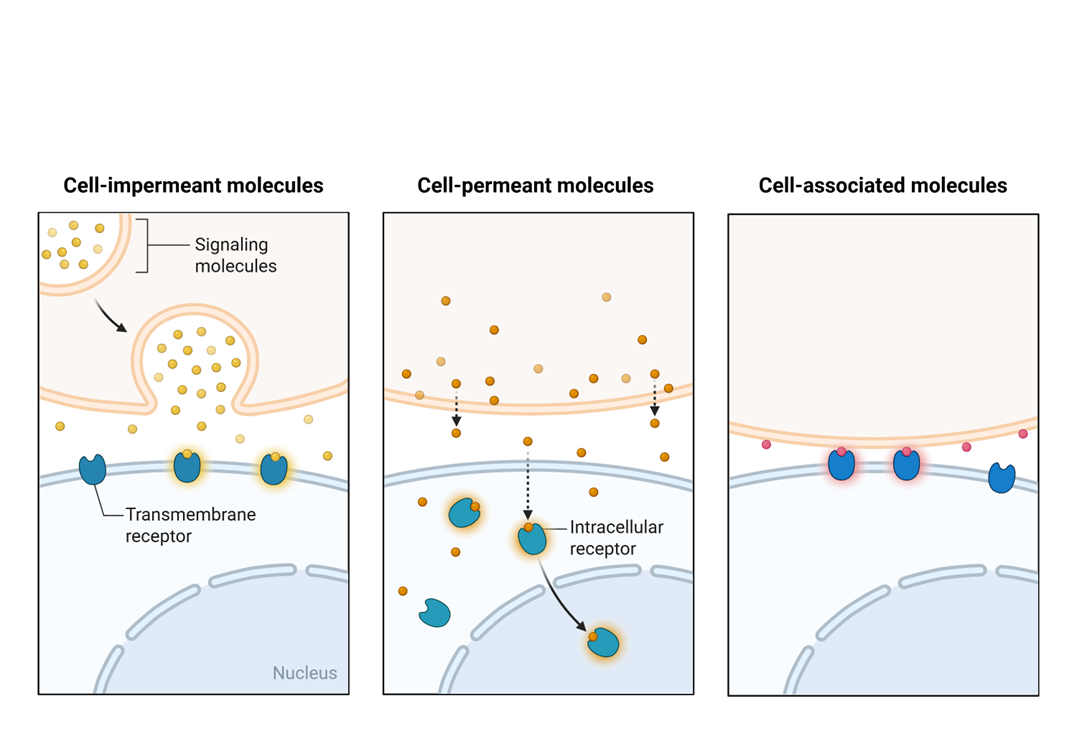
hydrophobic signaling molecules
what is an example of a hydrophobic signaling molecule that uses nuclear receptors
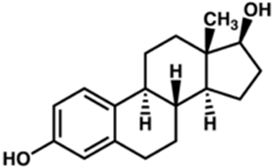
steroid hormones
What is significant about these things:
altered gene expression
increased translation
increased nutrient uptake and metabolic rat e
altered morphology
these are all the possible changes that a cell growing to divide may face
what nuclear hormone receptor binding induces estrogen receptor (ER) dimerization
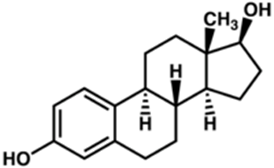
estrogen
As nuclear hormone receptor:
Estrogen binding induces estrogen receptor (ER) dimerization.
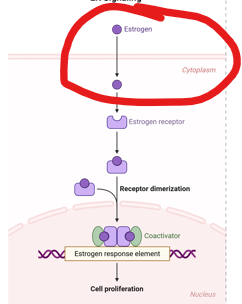
What is the first step in this process
the estrogen receptor is translocated to the nucleus
As nuclear hormone receptor:
Estrogen binding induces estrogen receptor (ER) dimerization.
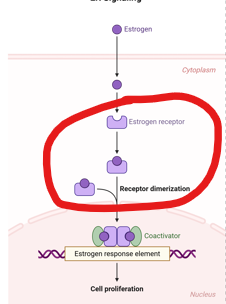
What is the second step in this process?
binds to genes containing an estrogen response element (ERE)
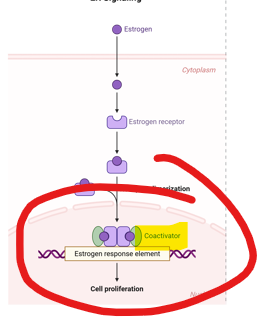
As nuclear hormone receptor:
Estrogen binding induces estrogen receptor (ER) dimerization.
What is the last step in this process
ER interacts with coactivator proteins for the recruitment of transcription machinery
what does ER effects in combination with other transcription factors determine
the level of ecpression for each gene in the cell
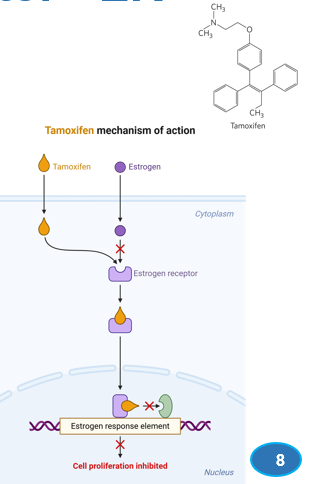
What is an estrogen antagonist that competes with estrogen for binding to ER
Tamoxifen
What type of activation is it when:
peptide binding to RTKs (receptor tyrosine kinases) stimulates autophosphorylation
RAS- Dependent Activation
During RAS- Dependent Pathway Activation
What peptide binding stimulates autophosphorylation
Peptide binding to RTKs (receptor tyrosine kinases)
During RAS- Dependent Pathway Activation
Peptide binding to RTKs (receptor tyrosine kinases) stimulates autophosphorylation
How is this done?
GRB2, an adaptor protein, uses its SH2 domain to bind these phosphotyrosines, recruiting the GEF SOS to the membrane. SOS then activates Ras by catalyzing the exchange of GDP for GTP.
What results in GDP/GTP exchange by RAS
SOS activity
How is RAS signaling turned off
GTPase activating protein (GAP)
The RAS dependent pathway is activated by RTKs for many growth factors, such as IGF-1, EGF, and PDGF
How does RAS-GTP activate its protein kinase activity
RAF-GTP interacts with RAF
RAS-GTP interacts with RAF activating its protein kinase activity
Whats the first thing that happens in this cascade
RAF phosphorylates MEK
RAS-GTP interacts with RAF activating its protein kinase activity:
RAF phosphorylates MEK
Whats the next thing that happens in this cascade
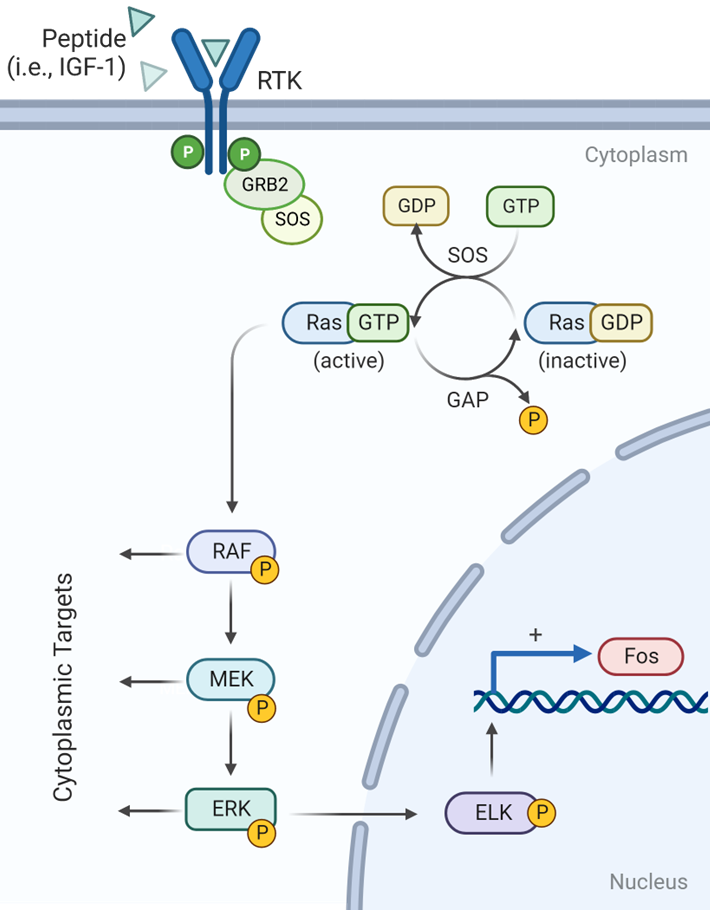
MEK phosphorylates ERK
RAS-GTP interacts with RAF activating its protein kinase activity:
RAF phosphorylates MEK
MEK phosphorylates ERK\
_____________
Whats the next thing that happens in this cascade
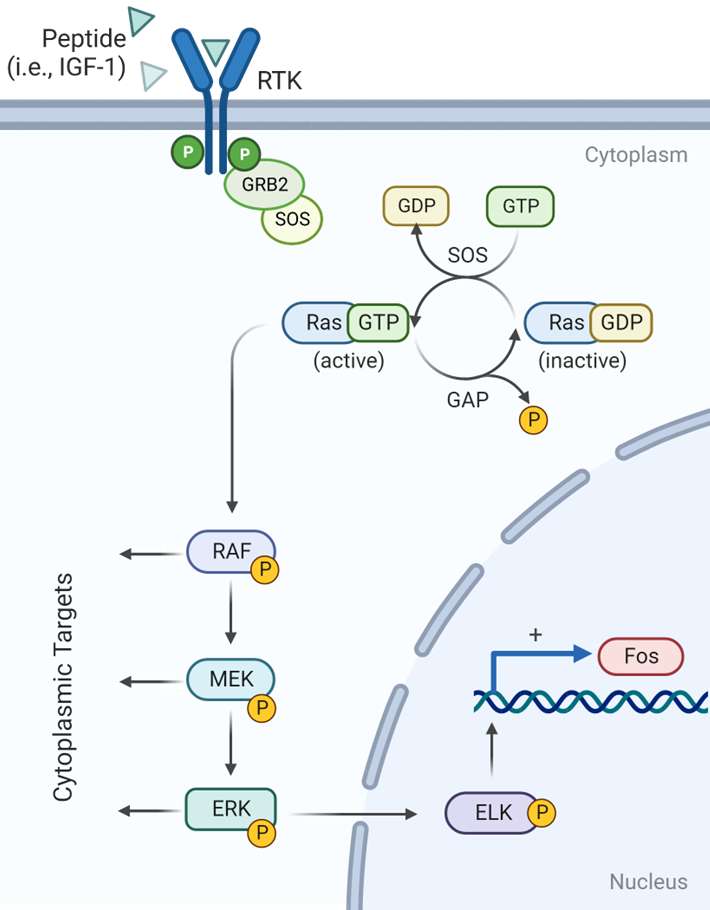
ERK phosphorylates transcription factors in the nucleus
RAS-GTP interacts with RAF activating its protein kinase activity:
RAF phosphorylates MEK
MEK phosphorylates ERK\
ERK phosphorylates transcription factors in the nucleus
Whats the next thing that happens in this cascade
ELK-1 binds Serum Response Element (SRE) located in
pro-growth genes to activate their transcription
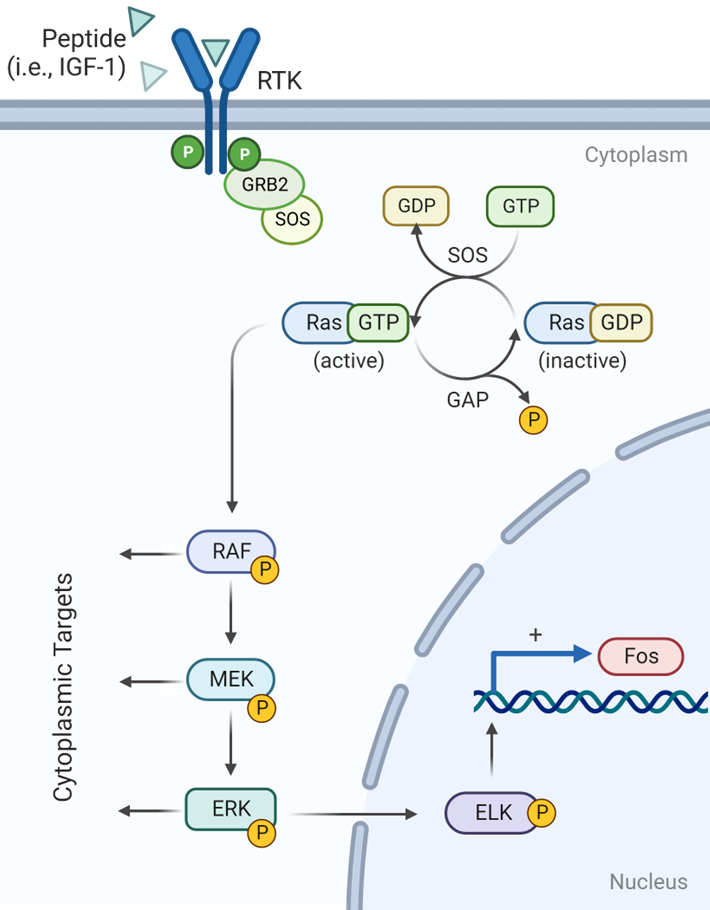
RAS-GTP interacts with RAF activating its protein kinase activity:
RAF phosphorylates MEK
MEK phosphorylates ERK\
ERK phosphorylates transcription factors in the nucleus
ELK-1 binds Serum Response Element (SRE) located in
pro-growth genes to activate their transcription
What is one of these pro-growth genes?
FOS
In thew RAS- Dependent pathway what does this sequential activation of protein kinases result in
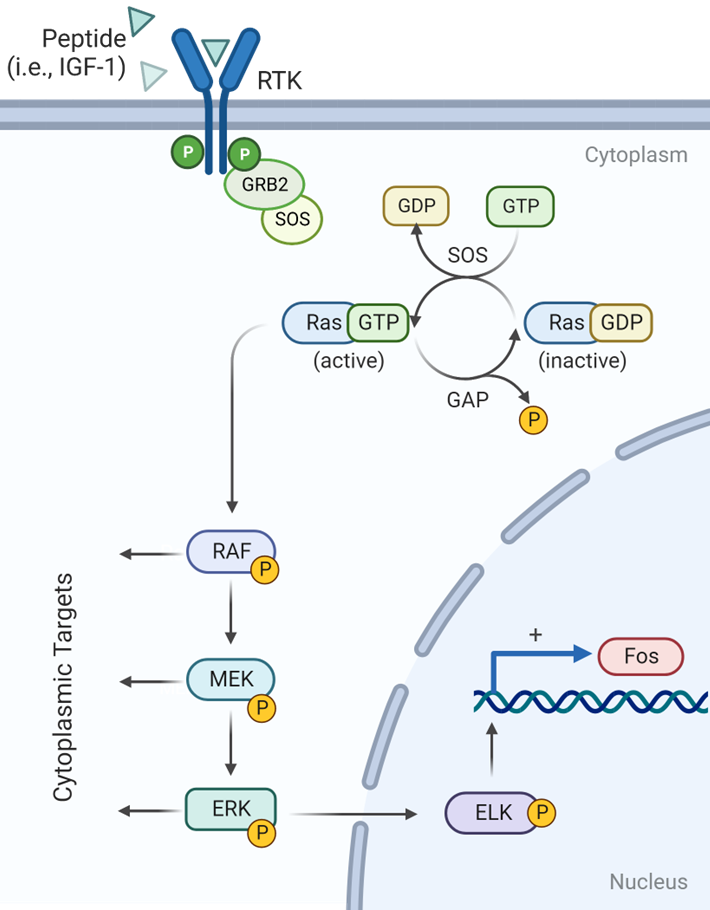
signal amplification and diversification
what is another word for signal amplification and diversification
signaling cascade
In RAS-Dependent pathways how is the phosphorylation and activity of target proteins (signal cascade) reversed
protein phosphatases
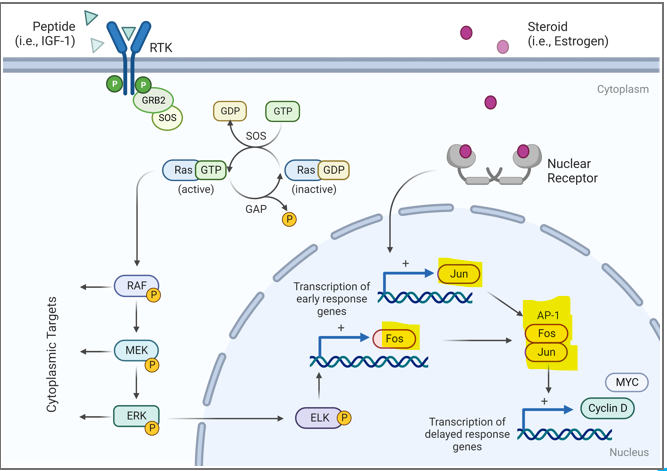
What are JUN and FOS
activating protein-1 (AP-1)
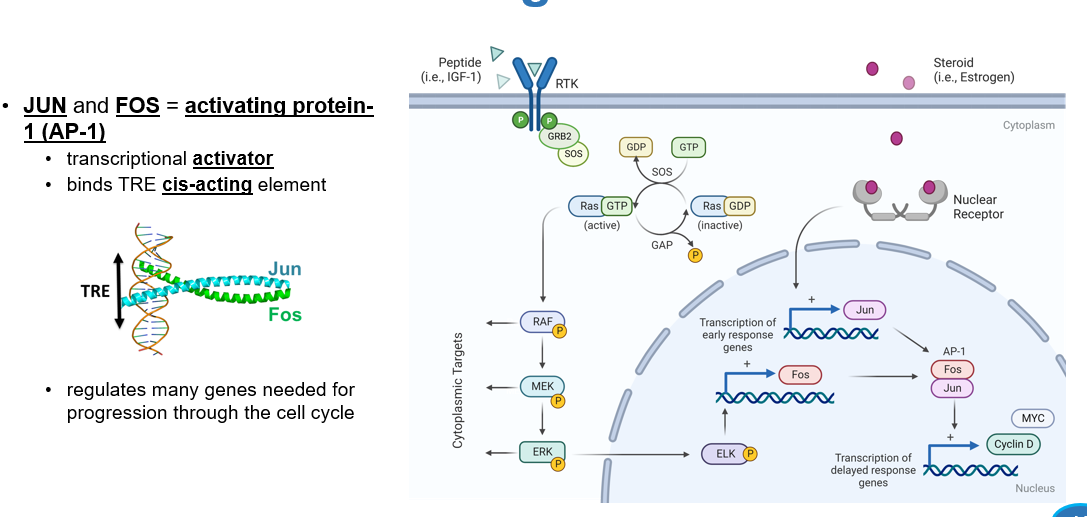
JUN and FOS are activating protein- 1, what is their significance?
binds the TRE cis-acting element, regulating many genes needed for progression through the cell cycle
What is an activatr of proliferation genes
Myc/Max
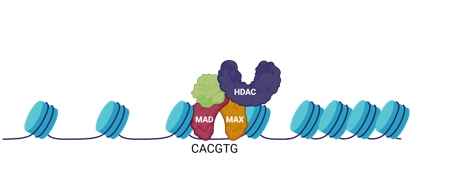
what is a repressor of pro-growth genes
Mad/Max
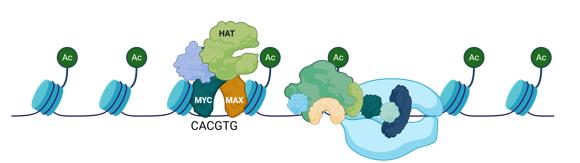
What is a complex that is an activator of many pro-growth genes and stimulates RNA Poll activity
Myc/Max
What kick starts the cell cycel in relation ot the activator of proliferation genes
upregulation of cyclin D (a pro growth gene)
What controls cell cycel timing
family of kinases
A family of kinases controls cell cycle timing. What does each active kinase have?
one catalytic subunit
one regulatory subunit
what is in one catalytic subnit of an active kinase
cyclin dependent kinase (CDK)
what is an example of a regulatory subunit
cyclin
Family of kinases controls cell cycle timing with each active kinase having one catalytic subunit ( cyclin dependent kinase (CDK) and one regulatory subunit (cyclin).
What cyclin- cdk complex is found in early G1
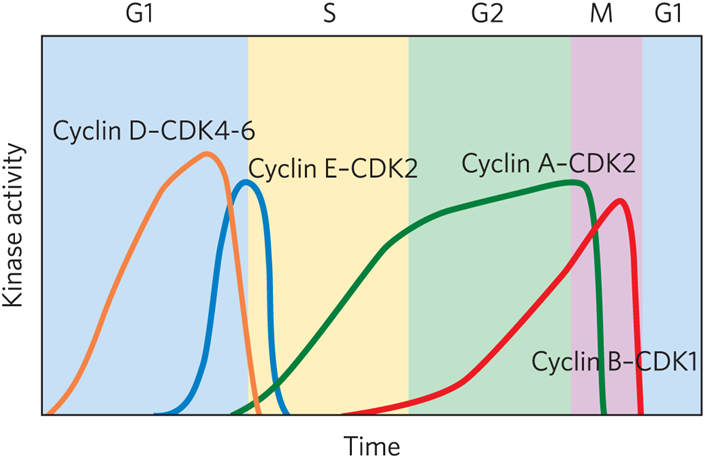
Cyclin D-CDK4-6
Family of kinases controls cell cycle timing with each active kinase having one catalytic subunit ( cyclin dependent kinase (CDK) and one regulatory subunit (cyclin).
What cyclin- cdk complex is a restriction point
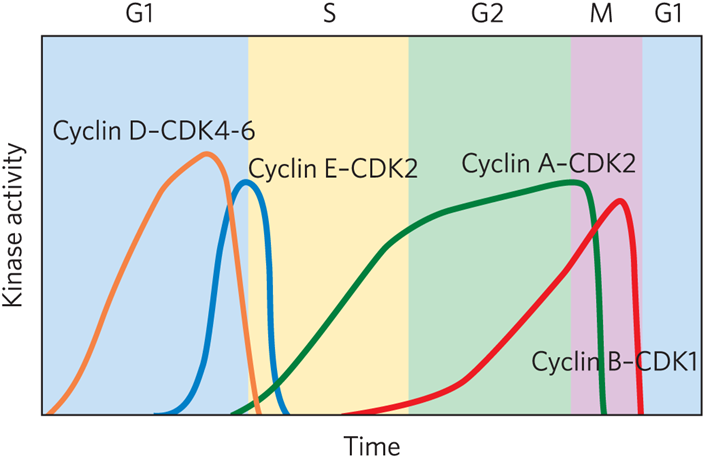
Cyclin E-CDK2
Family of kinases controls cell cycle timing with each active kinase having one catalytic subunit ( cyclin dependent kinase (CDK) and one regulatory subunit (cyclin).
What cyclin- cdk complex is located in S phase?
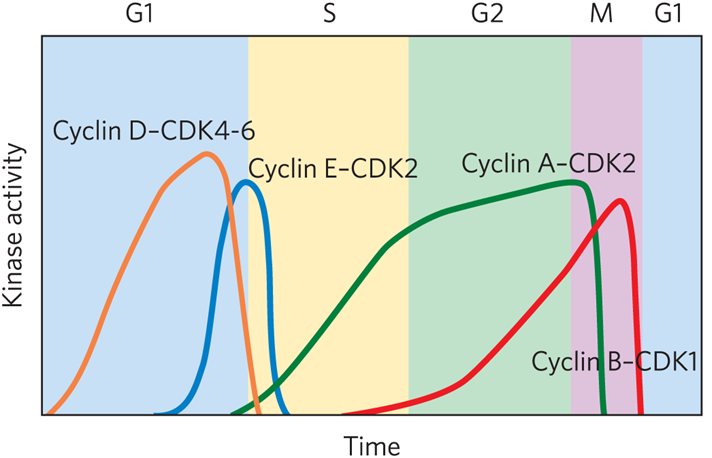
Cyclin A-CDK2
What result in the oscillations of cyclin-CDK activity?
CDK phosphorylation or dephosphorylation
controlled degradation of the cyclin subunit =
ubiquitin
periodic synthesis of CDKs and cyclins
the action of specific CDK-inhibiting proteins
(CIP)
How does CDK get regulated
by phoshorylation and proteolysis
what is a regulatory protein that targets proteins for destruction
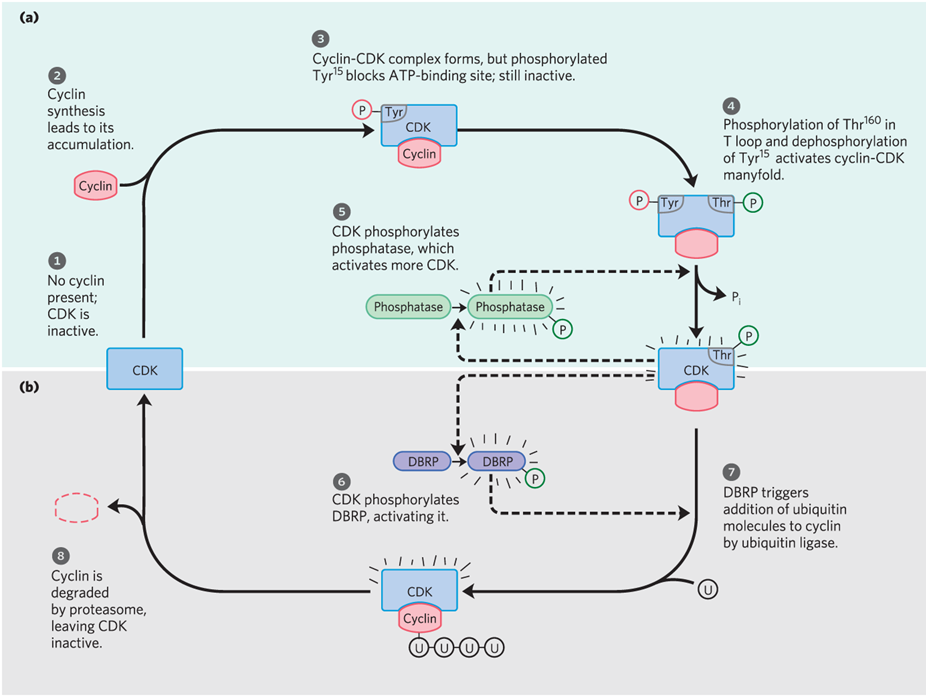
ubiquitin
what are proteolytic enzyme complexes within cdk regulation by phosphorylation and proteolysis?
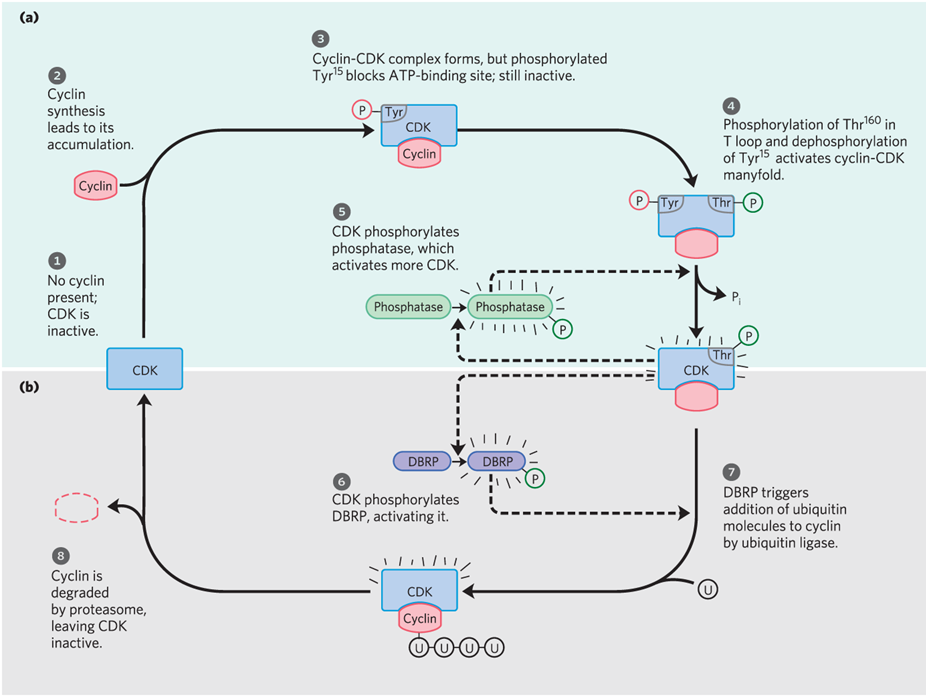
proteasomes
what are destruction box recognizing proteins within cdk regulation by phosphorylation and proteolysis?
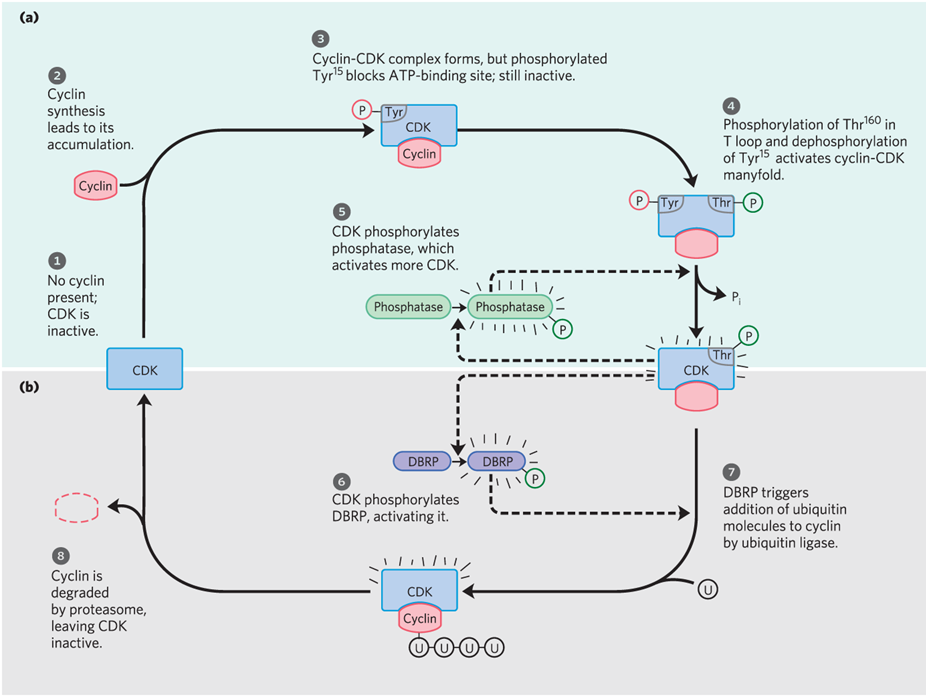
DBRP
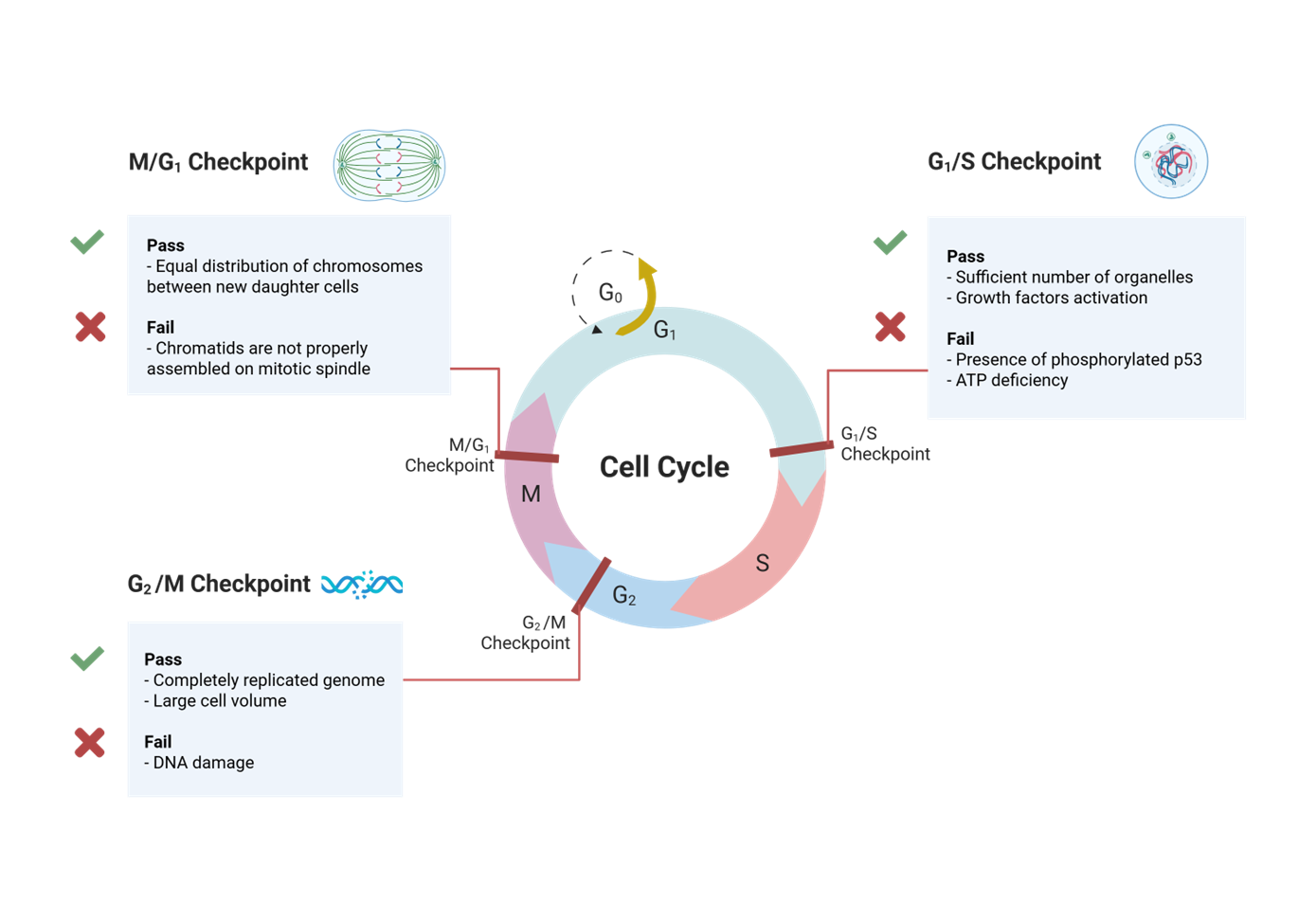
what is a checkpoint before DNA replication
Restriction point
what is a cell considered when it passes the restriction point?
it is committed
How are Cyclin-dependent kinase activity inhibitors are expressed in this checkpoint and how are they termed?
in G1 and they are termed CDK inhibitors (CKI)
Cyclin-dependent kinase activity inhibitors are expressed in G1 termed CDK inhibitors (CKI).
What blocks cyclin D binding
Inhibitor of cdk4 (INK4) proteins
Cyclin-dependent kinase activity inhibitors are expressed in G1 termed CDK inhibitors (CKI).
What protein binds to an inhibits the activites of intact CDK-cyclin complexes
CDK interacting protein (CIP)
what is an example of CDK interacting protein (CIP)?
p21
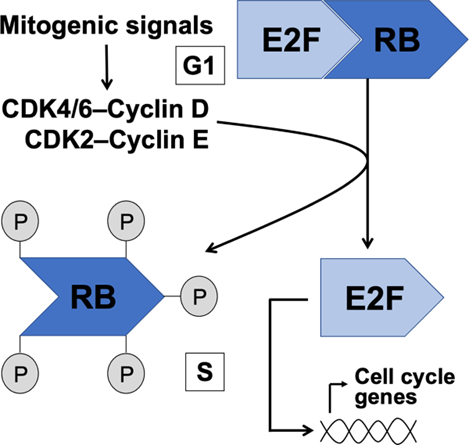
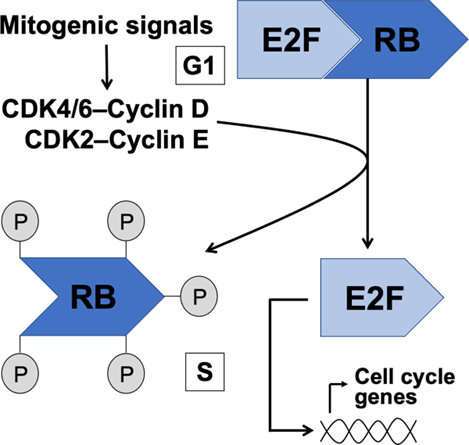
At the restriction point, what is a corepressor that binds the activator E2F to arrest cell division
retinoblastoma protein (pRb)
What happens to cyclin D-CDK4/6 and cyclin E-CDK2 at the restriction point
hyperphosphorylate Rb releasing Rb from E2F
what is an activator of genes needed in S phase
E2F
E2F is an activator of genes needed in S phase
including:
______________________
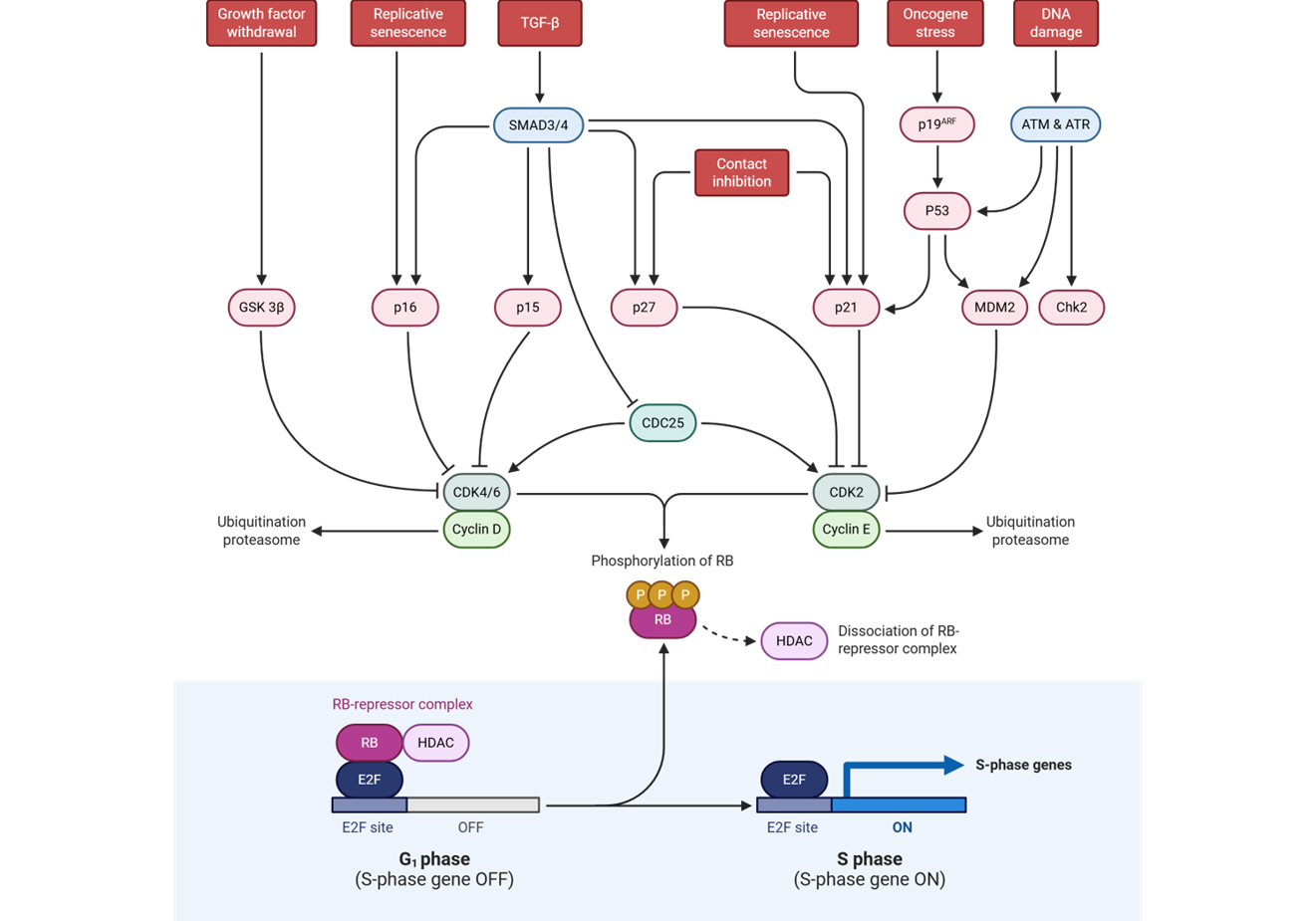
cell cycle machinery genes (cyclins, cyclin-dependent kinases)
E2F is an activator of genes needed in S phase
including:
cell cycle machinery genes (cyclins,
cyclin-dependent kinases
_________
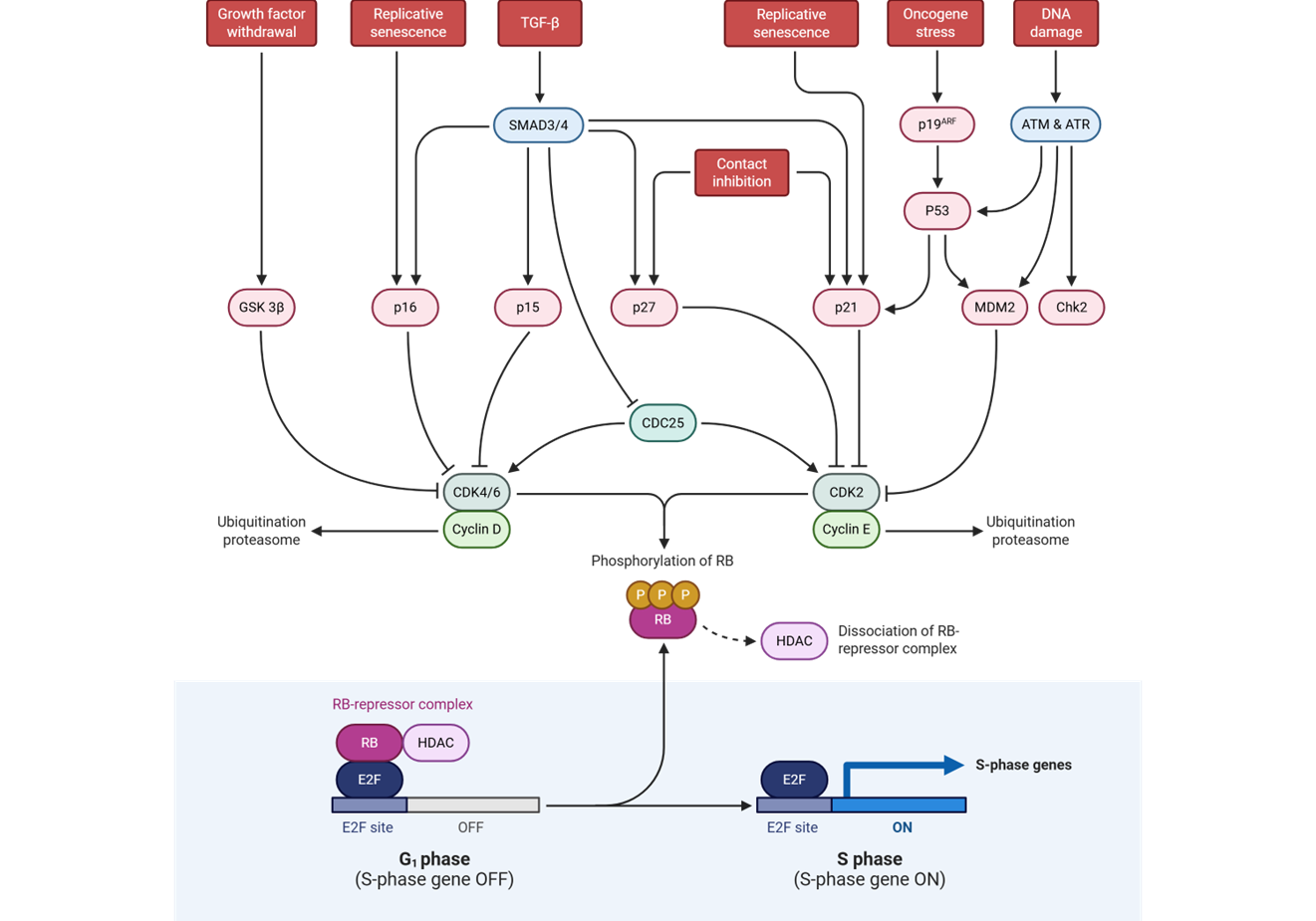
pro-growth transcription factors (Myc, E2F
family)
E2F is an activator of genes needed in S phase
including:
cell cycle machinery genes (cyclins,
cyclin-dependent kinases
pro-growth transcription factors (Myc, E2F
family)
_____________________
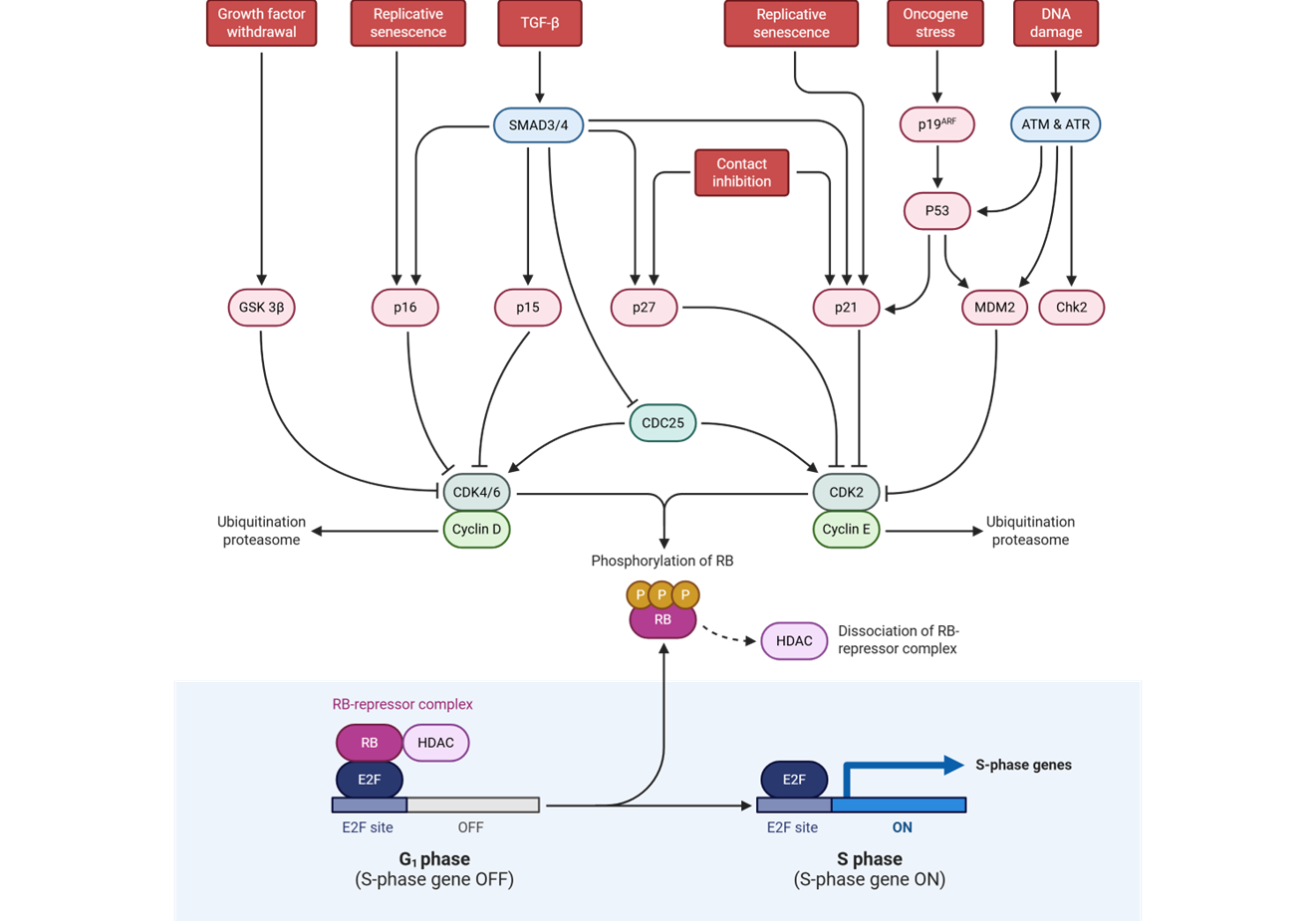
components of the DNA replication
machinery (replisome) and proteins of
chromatin (histones
When does restriction point block progression
if dna damage is present
What happens in regards to the restriction point when there is DNA damage
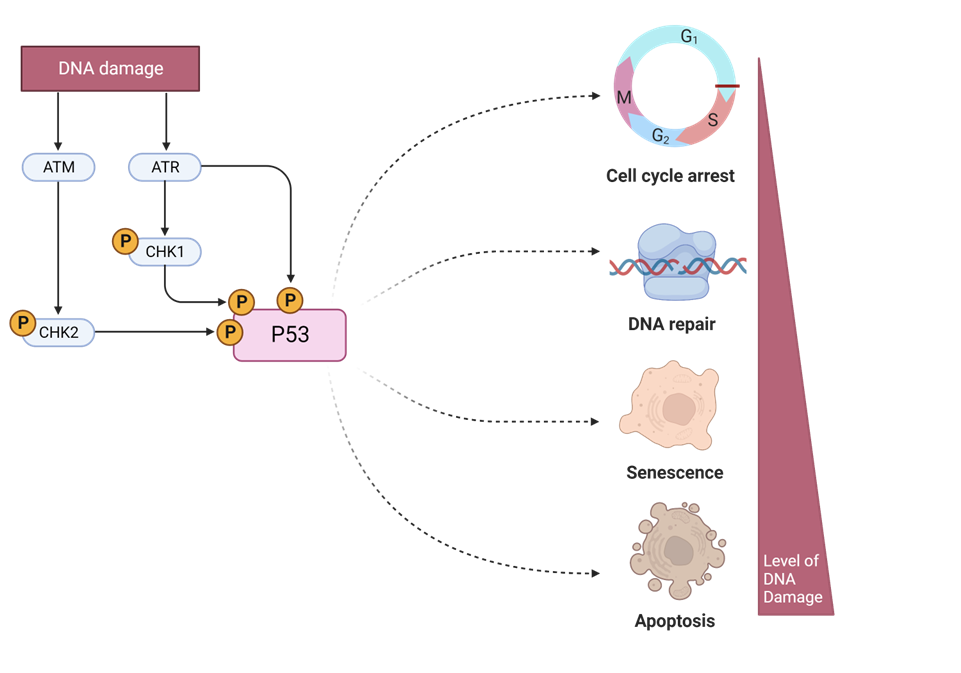
p53 activation upregulates transcription of CKI genes such as p21
How does p53 upregulate transcription of CKI genes such as p21
it blocks CDK from binding ATP
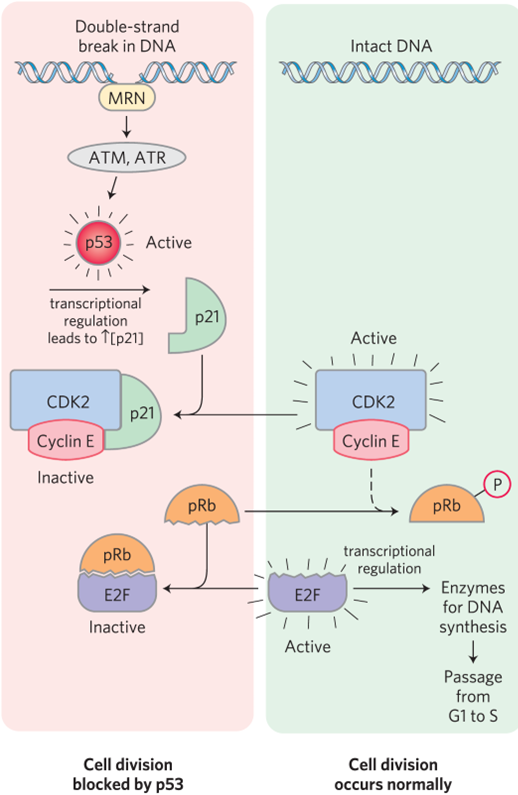
What were the clinical findings in Retinoblastoma and
Cell Cycle Dysregulation
Genetic testing shows a mutation in the RB1 gene, which encodes the retinoblastoma protein (pRb), a key regulator ofthe G1/S checkpoint in the cell cycle
What is the molecular insight of retinoblastoma and cell cycle dysregulation
pRb normally binds and inhibits E2F transcription factors, preventing premature entry into S phase. When RB1 is mutated, pRb cannot restrain E2F, leading to uncontrolled transcription of genes required for DNA replication. This loss of cell cycle control results in excessive proliferation and tumor formation, characteristic of retinoblastoma. Familial cases follow the two-hit hypothesis, where one defective allele is inherited and the second is lost through somatic mutation
What does the transition to S-phase from G1 require
syntheis of cyclin A
what is a transcriptional activator of cyclin A
EF2
aside from the transition to S phase from G1 requires synthesis of cyclin A, what else does it require
sufficient active cyclin E-CDK2 to activate cyclin A-CDK2
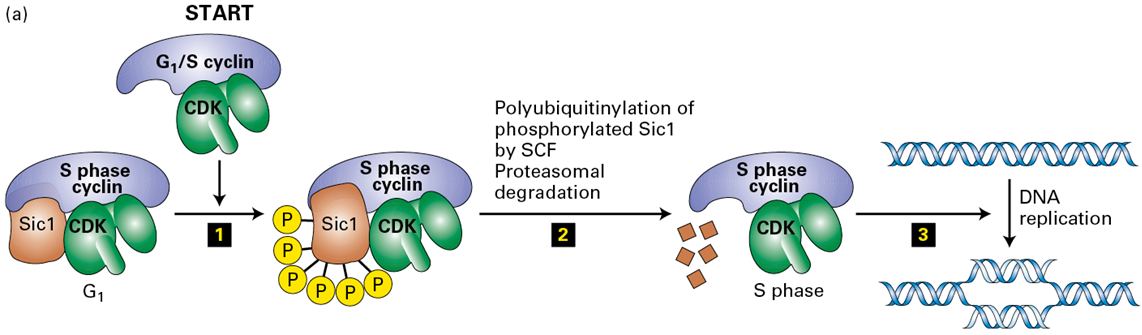
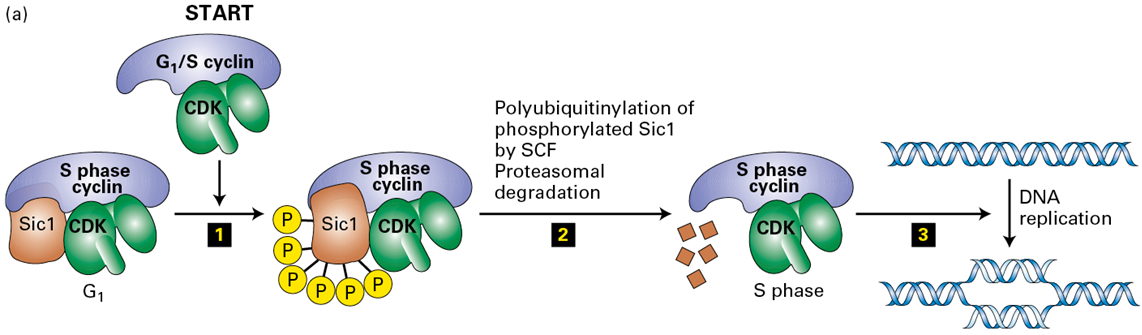
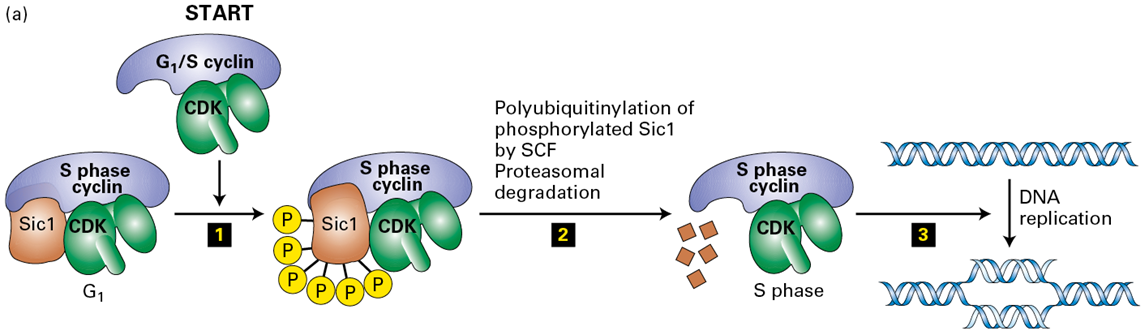
How does Cyclin E-CDK2 drives the transition from G1→S
by phosphorylating the CDK inhibitor Sic1 (or p27 in mammals), targeting it for ubiquitination and subsequent degradation by the SCF ubiquitin ligase complex
aside from the transition to S phase from G1 requires synthesis of cyclin A, there is a requiremrnt of sufficent active cyclin cyclin E-CDK2 to activate cyclin A-CDK2.
What results from the phsophorylation of SIc1
it makes it susceptible to a ubiquitin ligase
what coordinates replication initaition?
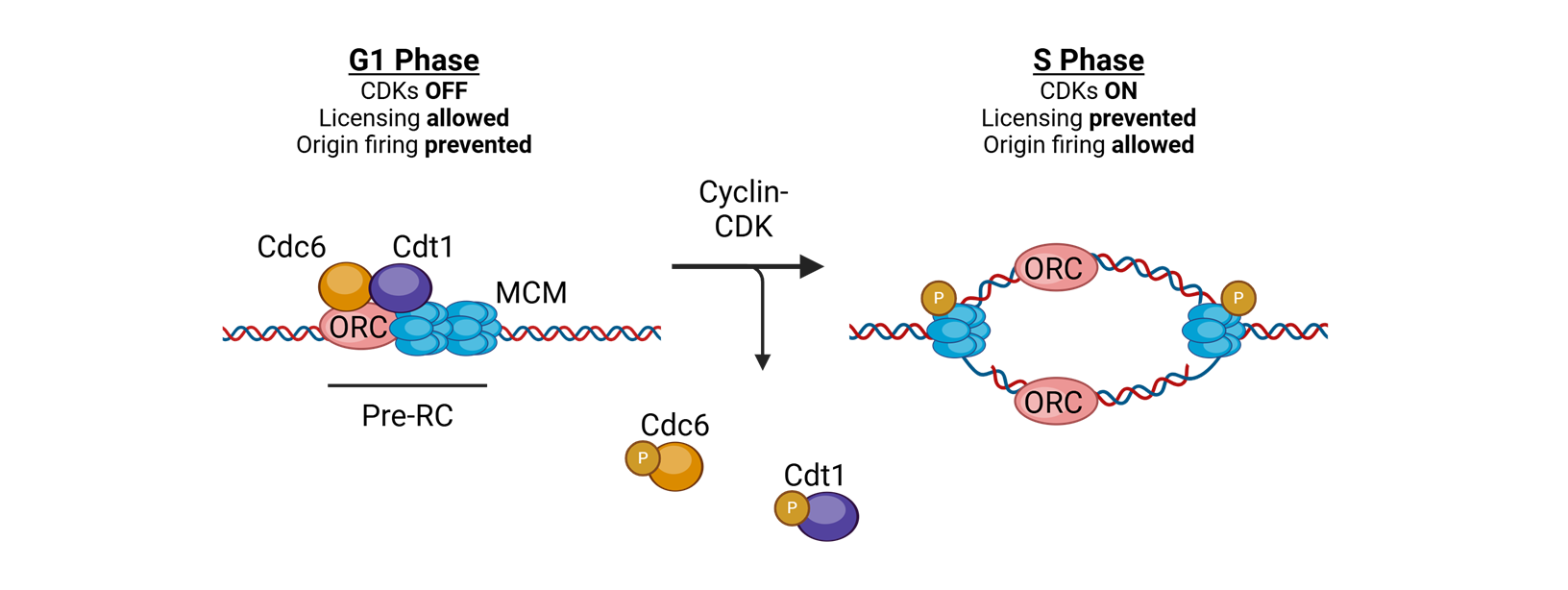
licensing
for licensing to coordinate replication what must bind tightly to DNA in the G1 phase?
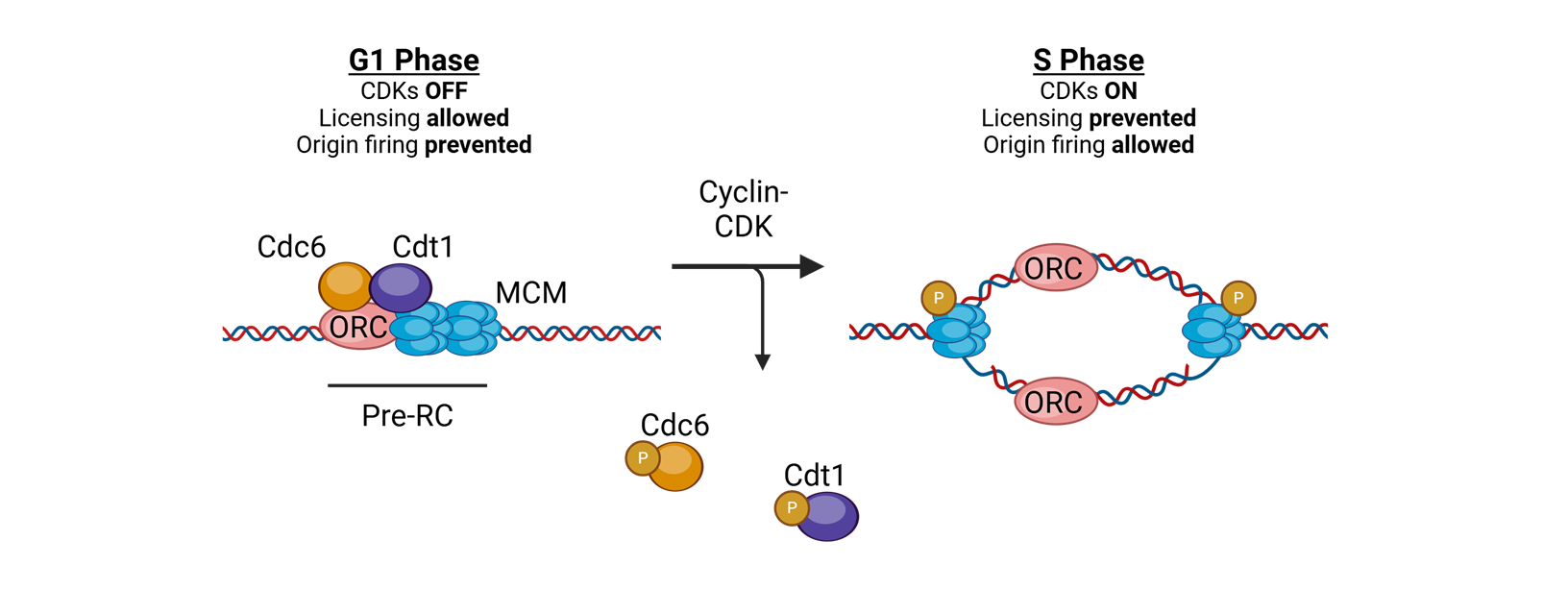
Origin of replication complexes (ORC)
what is the mechanisim after the origin of replication complexes bind tightly to DNA in G1 phase
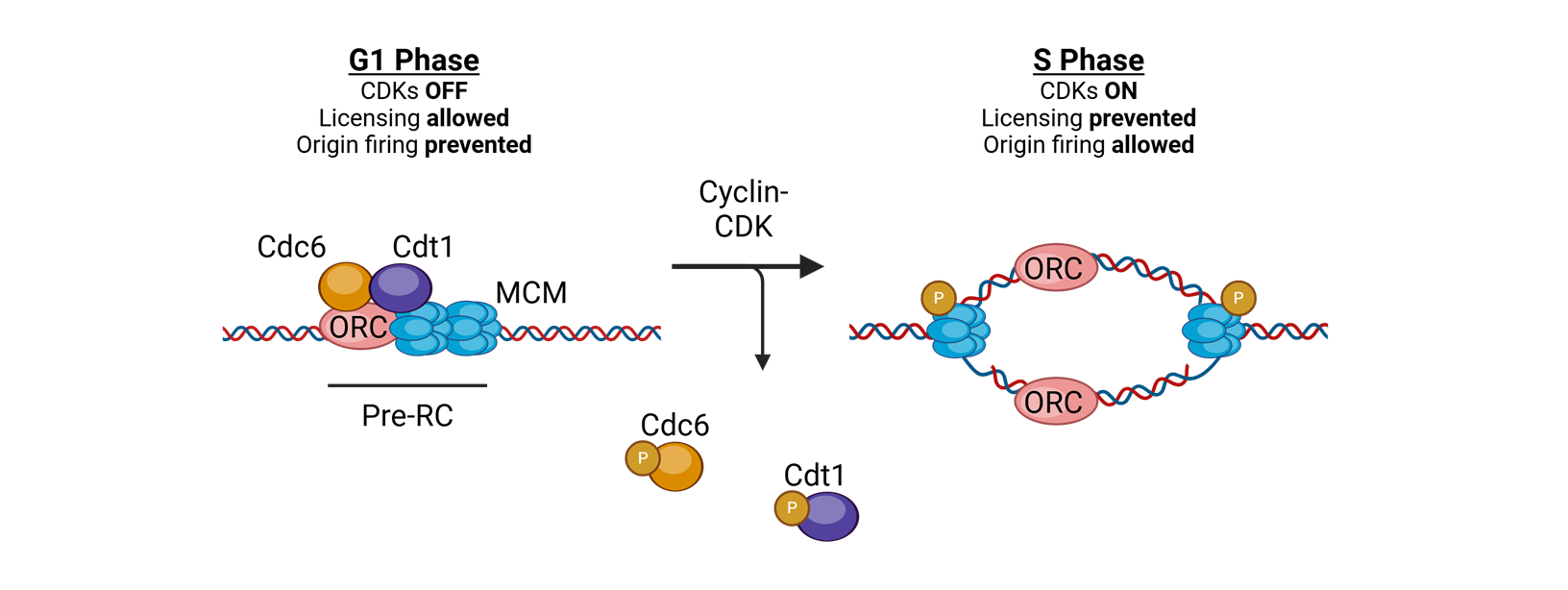
Cell division cycle 6 (CDC6) and CDT1 join and load the helicase Mini Chromosome Maintenance (MCM)
What does MCM do?
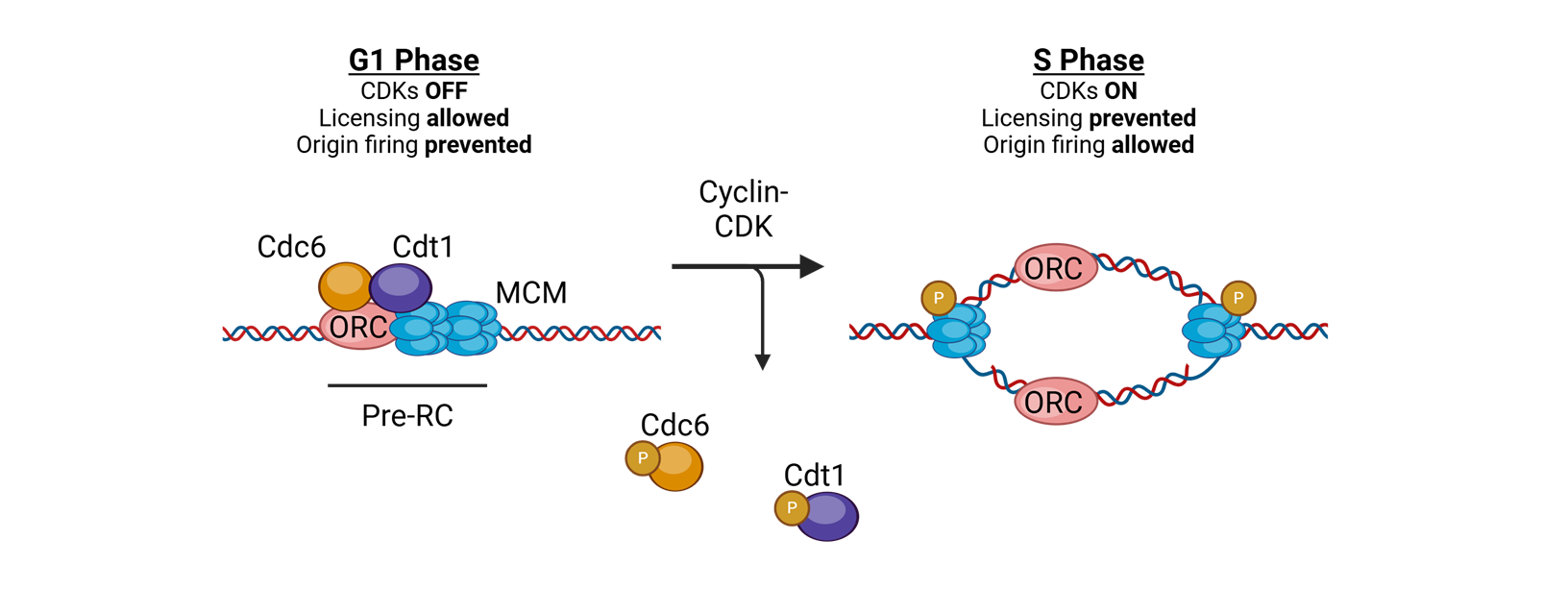
translocates 3′→5′ along the leading strand template using ATP
for licensing to coordinate replication what happens in S-phase
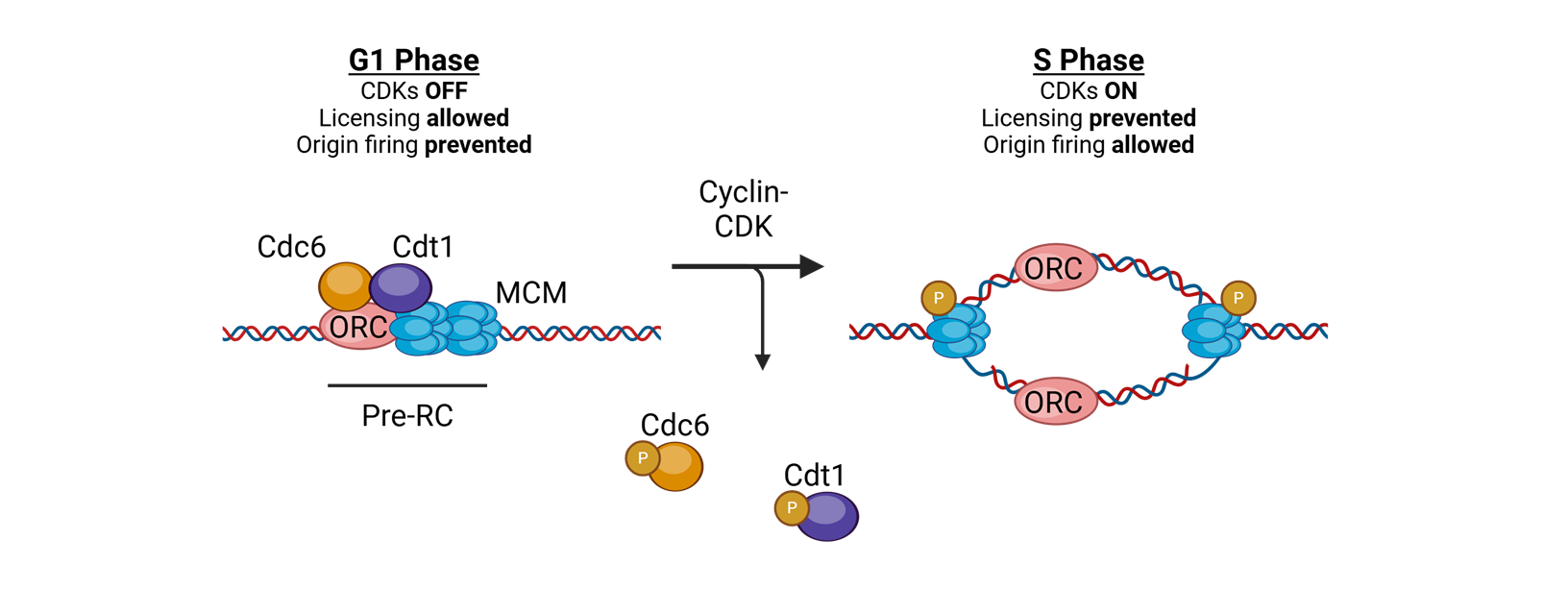
replication is initiated by phosphorylation of complex proteins by cyclin A–CDK2
or licensing to coordinate replication, in the S phase or licensing to coordinate replication.
What happens after this?
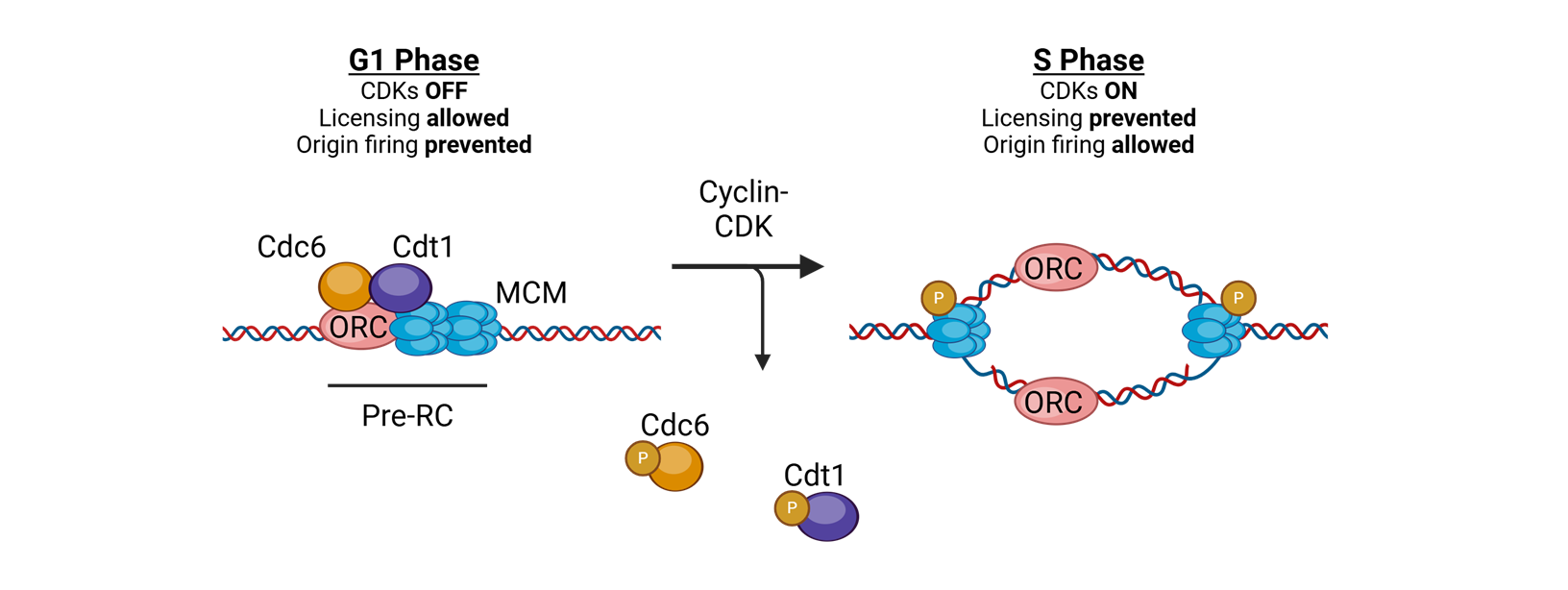
The replisome is assembled, and bidirectional DNA synthesis is initiated