Diffusion and Dilution
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
diffusion
the movement of particles from an area of high concentration to an area of low concentration (move randomly, no path)
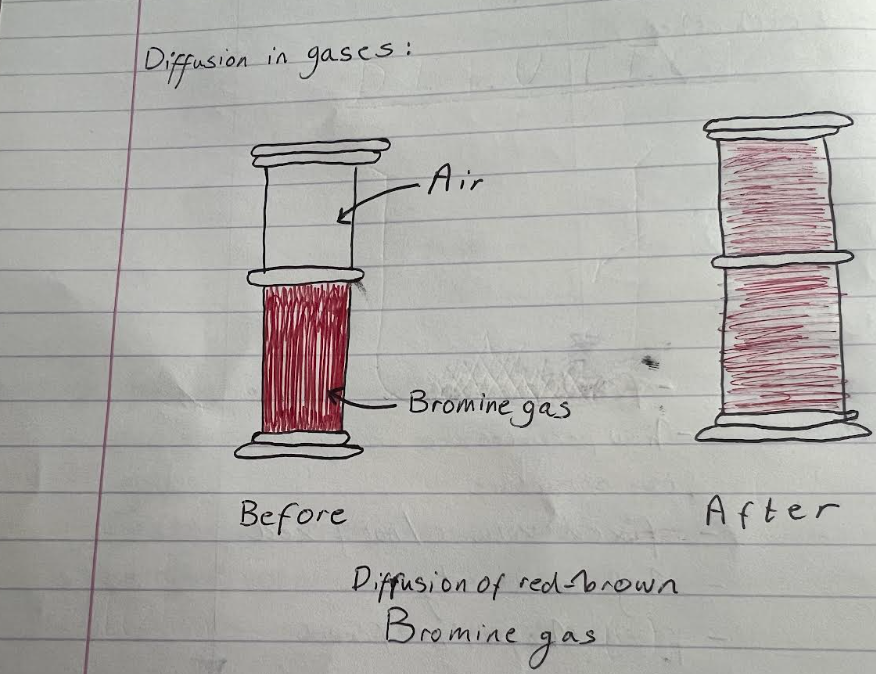
here we see the diffusion of bromine gas from one gas jar to another - after 5 minutes the bromine gas has diffused from the bottom jar to the top jar. the air and bromine particles are moving randomly and there are large gaps between the particles, therefore the particles can easily mix together.
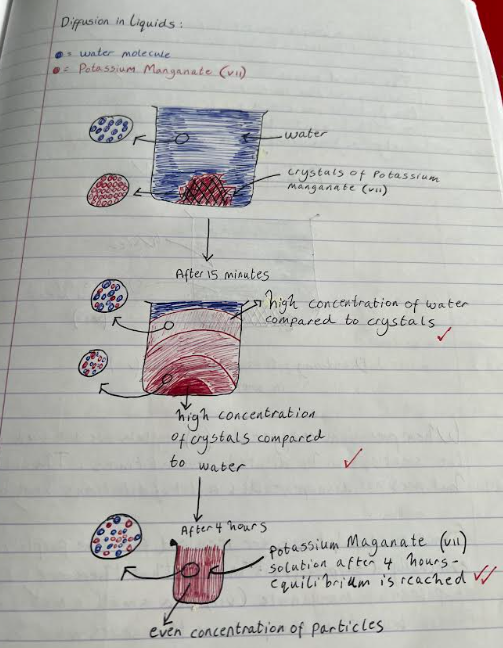
when potassium manganate (VII) crystals are dissolved in water, a purple solution is formed. a small number of crystals produce a highly intense colour. the water and potassium manganate (VII) particles are moving randomly and the particles can slide over each other, therefore the particles can easily mix together.
comparison between diffusion in ls and diffusion in gs
diffusion in liquids is slower than diffusion in gases because the particles in liquids are closely packed together and mover more slowly.
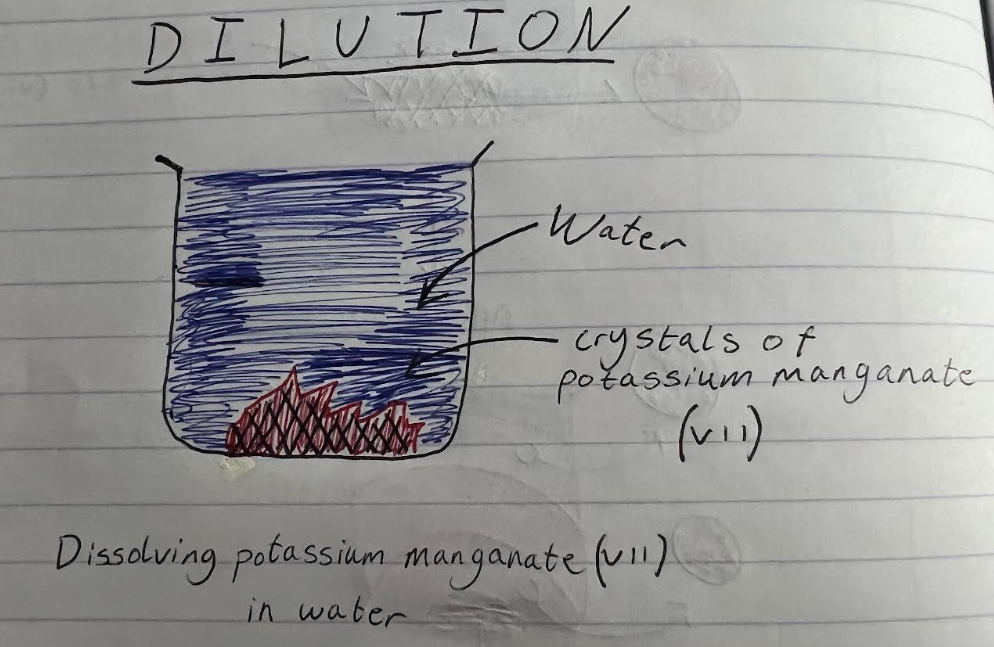
when potassium manganate (VII) crystals are dissolved in water, the solution can be diluted several times. the colour fades but does not disappear until a lot of dilutions have been done. since many dilutions are required for the purple colour to eventually disappear, this implies that there are lots of particles of potassium manganate (VII) in a small amount of solid. If this is true then the particles themselves must be very tiny.
solvent
the liquid in which a solute dissolves
solute
the substance which dissolves in a liquid to form a solution
solution
the mixture formed when a solute is dissolved in a solvent
saturated solution
A solution with the maximum concentration of solute dissolved in the solvent
soluble
describes a substance that will dissolve
insoluble
describes a substance that will not dissolve