1.2 Biological molecules
1/81
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
82 Terms
What is the basic function of DNA and RNA in all living cells?
DNA: Holds genetic information which codes for polypeptides (proteins)
RNA: Transfers genetic information from DNA to ribosomes
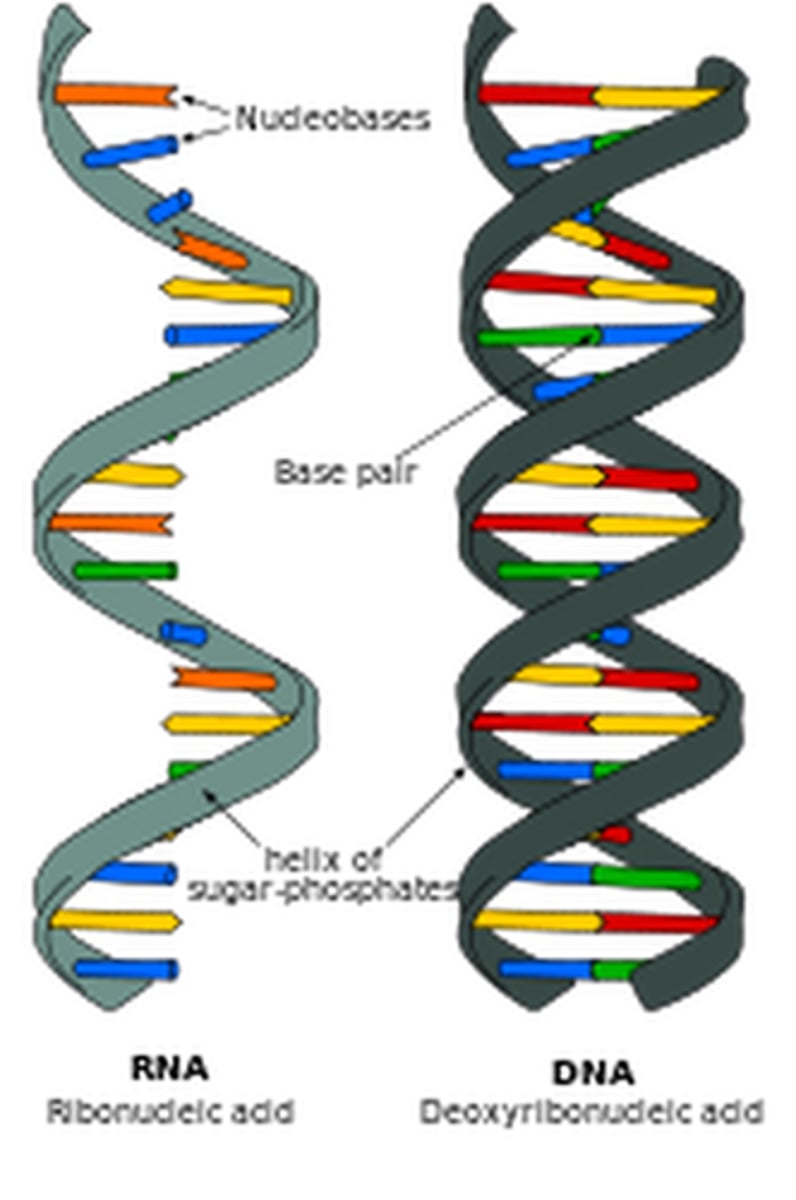
Describe DNA and it's role.
● Codes for the sequence of amino acids in the primary structure of a protein
● Which in turn determines the final 3D structure and function of a protein
What is a nucleotide?
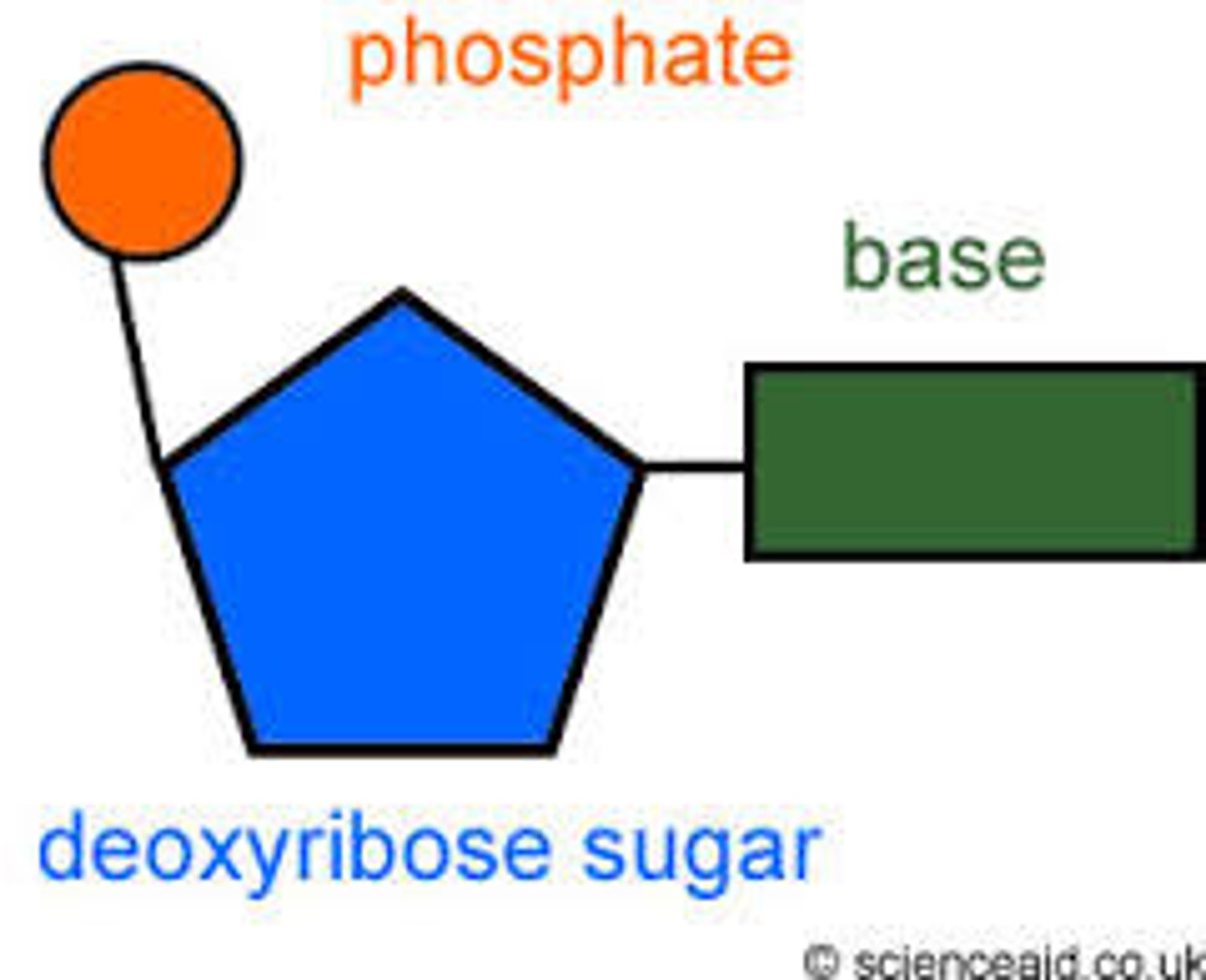
● A monomer consisting of a pentose sugar, a phosphate group and a nitrogenous base.
● Nucleotides can join together to form a polynucleotide (polymer) by condensation reactions
Monomer of DNA
Monomer of RNA
Energy currency (ATP)
What is a nucleic acid?
Molecule made up of 1 or more polynucleotide chains (RNA or DNA)
● Molecule that carries the genetic code for the production of proteins.
● Made of nucleotides.
What is DNA nucleotide made of?
● A phosphate group
● A pentose sugar called deoxyribose
● A nitrogenous base: A, C, G or T
What is RNA nucleotide made of?
● A phosphate group
● A pentose sugar called ribose
● A nitrogenous base: A, C, G or U
What does A, C, G, T and U stand for?
Adenine
Cytosine
Guanine
Thymine
Uracil
What are the differences between the DNA and RNA monomers?
● DNA contains the base thymine but RNA contains uracil instead
● DNA contains deoxyribose but RNA contains ribose
What is the difference between the DNA and RNA polymer?
● DNA is much longer as it includes many nucleotides (approx 23,000 genes)
● whereas RNA is much shorter (single-stranded / single helix) as it is only the length of 1 gene
Describe how nucleotides join together to form polynucleotides.
● Condensation reactions, removing water molecules
● Between phosphate group of one nucleotide and deoxyribose/ribose of another
● Forming phosphodiester bonds (strong covalent bonds)
Diagram of DNA structure
DNA molecule diagram
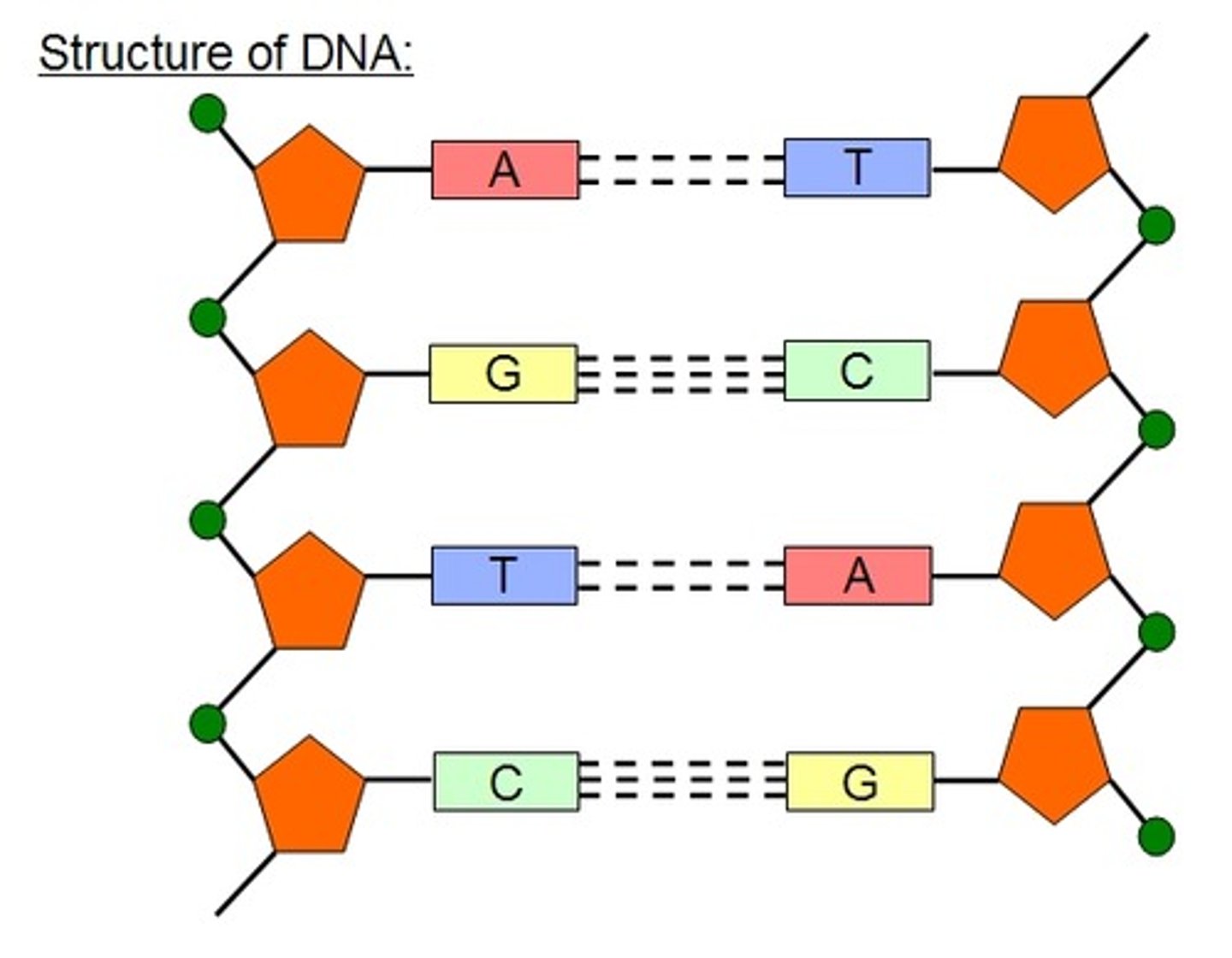
Describe the structure of DNA.
● Polymer of nucleotides (polynucleotide)
● Phosphodiester bonds join adjacent nucleotides
● 2 polynucleotide chains held together by hydrogen bonds
● Between specific complementary base pairs - A + T, C + G
● Double helix
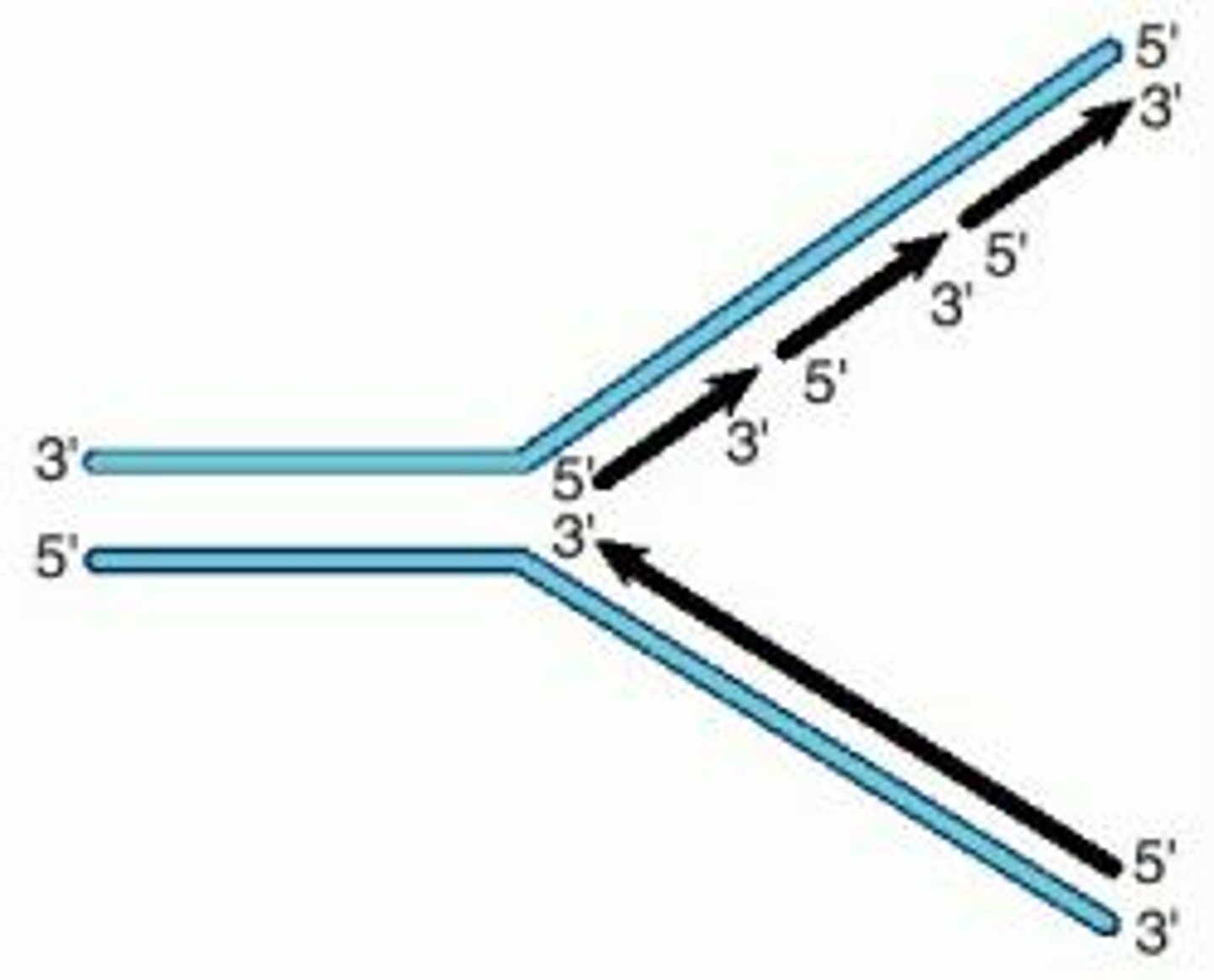
Antiparallel
The 2 strands run in opposite directions
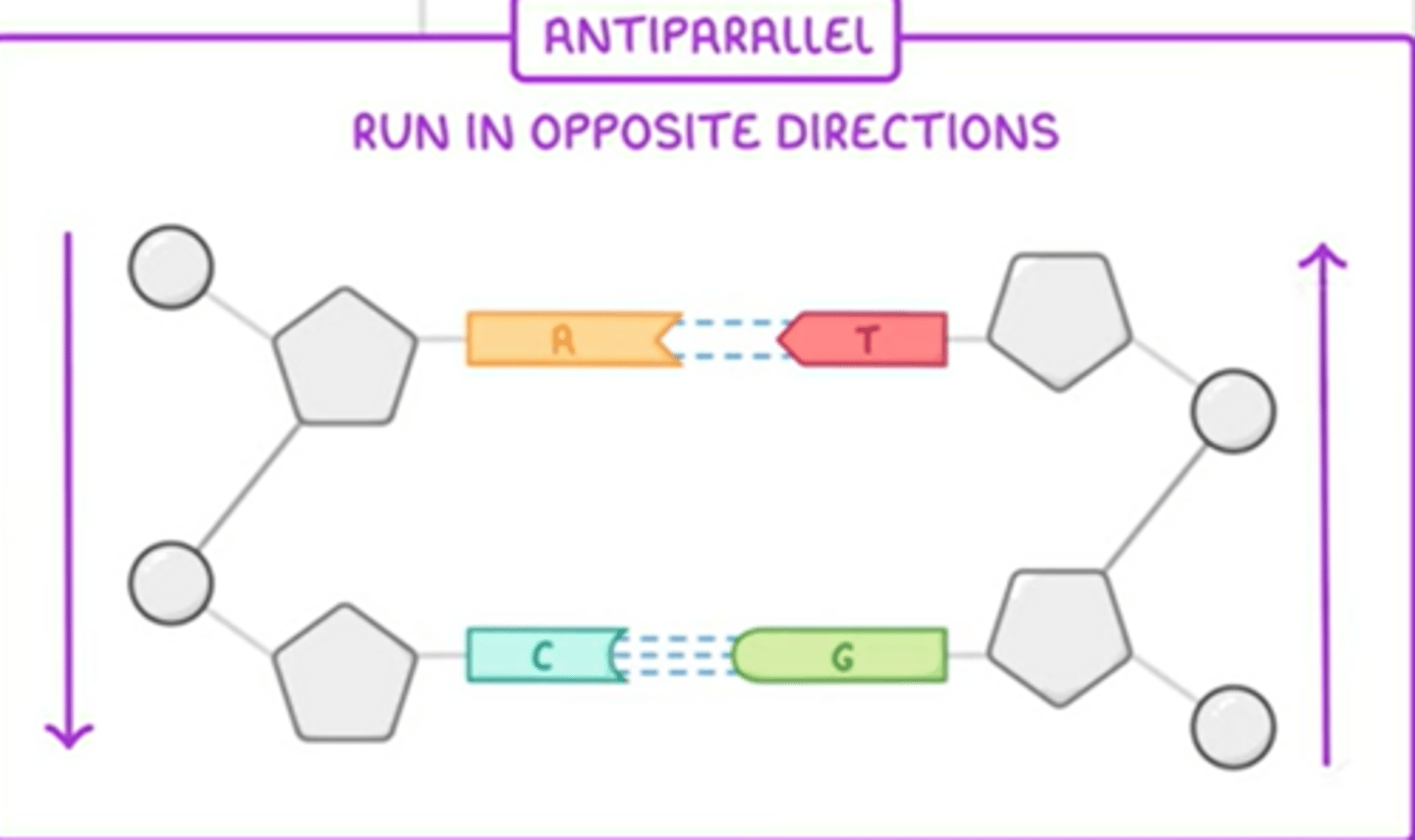
How does the structure of DNA relate to its function?
● Double stranded → replication can occur using each strand as a template
● Weak hydrogen bonds between bases → strands can be separated for replication
● Complementary base pairing → accurate replication
● Many hydrogen bonds between bases. Sugar phosphate backbone → stable/strong molecules
● Double helix with sugar phosphate backbone → protects bases/hydrogen bonds, from mutations
● Double helix → coiled/compact to store lots info
● Long molecule → store lots of genetic information: base sequence code for polypeptides
Why did many scientists initially doubt that DNA carried the genetic code?
The relative simplicity of DNA - chemically simple molecule with few components.
Similarities between eukaryotic and prokaryotic DNA
● Identical nucleotide structures (pentose sugar, phosphate group, nitrogenous base)
● Both nucleotides joined by phosphodiester bonds
Describe the structure of RNA.
● RNA is a polymer of a nucleotide formed of a phosphate group, ribose (pentose sugar) and a nitrogenous base
● Base contains either A, C, G or U
● A relatively short polynucleotide chain, and is single-stranded
What is the role of RNA?
Copy and transfer the genetic code from DNA in the nucleus to the ribosomes.
Give the 3 types of RNA.
● mRNA (messenger RNA)
● tRNA (transfer RNA)
● rRNA
What is mRNA and what does it do?
● mRNA is a copy of a gene from DNA
● Created in the nucleus, then travels from nucleus to a ribosome in the cytoplasm.
● DNA is too large to leave the nucleus. mRNA is much shorter (only the length of one gene) so can leave the nucleus.
Why is mRNA short-lived?
As it's only needed temporarily to help create a protein, therefore by the time enzymes could break it down, it would have already carried out its function
Describe the structure of mRNA.
● mRNA is a copy of a gene from DNA
● Long ribose polynucleotide (but shorter than DNA)
● Single-stranded & linear
● Every 3 bases in the sequence code for a specific amino acid, these 3 bases are therefore called codons.
Relate the structure of messenger RNA (mRNA) to its functions.
● Breaks down quickly so no excess polypeptide forms.
● Ribosome can move along strand & tRNA can bind to exposed bases.
● Can be translated into a specific polypeptide by ribosomes.
Describe the structure of transfer RNA (tRNA).
● Found in cytoplasm only
● Single stranded / single polynucleotide strand. Fewer nucleotides than mRNA.
● Folded into a clover shape;
Hydrogen bonds between specific base pairs hold the molecule in this shape.
● Anticodon on one end: specific sequence of 3 bases
● Amino acid binding site at the other end.
Describe the role of tRNA.
● Anticodon on one end: specific sequence of 3 bases
● Amino acid binding site at the other end.
● Each tRNA is specific to one amino acid and has an anticodon that is specific to that amino acid.
● Anti-codon base pairs with a complementary codon on mRNA
● tRNA involved in translation. Carries the amino acids that are used to make proteins to the ribosomes.
Order DNA, mRNA and tRNA according to increasing length.
1. tRNA
2. mRNA
3. DNA
Which bases are purine and which are pyrimidine?
A & G = 2-ring purine bases
T & C & U = 1-ring pyrimidine bases
Suggest how you can use incomplete information about the frequency of bases on DNA strands to find the frequency of other bases.
1. % of adenine in strand 1 = % of thymine in strand 2 (and vice versa)
2. % of guanine in strand 1 = % of cytosine in strand 2 (and vice versa)
Because of specific complementary
base pairing between 2 strands
Who are Meselson and Stahl?
Meselson and stahl are scientists who proved DNA replicated by semi-conservative replication by using nitrogen isotopes and a centrifuge.
What are isotopes of nitrogen?
Have different masses e.g. N-14 is lighter than N-15
What is a centrifuge?
● A centrifuge is a machine used to separate a mixture of substances
● based on how heavy each substance is
If a test tube contained both N-14 and N-15, then was spun in a centrifuge, what would be observed?
● The test tube would have a band of the N-15 (heavier) at the bottom, and N-14 (lighter) above it.
If we then took some molecules containing N-14 and combined them with some N-15, spun this in a centrifuge, what would be observed?
The test tube would have:
● A band at the bottom of N-15 (heaviest)
● A band above this of the combined N-14 & N-15
● A band above this of N-14 (lightest)
What are the steps of meselson and stahl's investigation?
1. Grow 2 sets of bacteria, one in light nitrogen and one in heavy
2. Light is control, heavy is for investigation
3. DNA will contain the light or heavy nitrogen that it was grown in
4. Extract some DNA from both and spin in centrifuge
5. Heavy (N-15) forms band at the bottom, light (N-14) forms band at the top
6. Then put bacteria that was in heavy nitrogen that now has DNA containing heavy nitrogen into light nitrogen broth to divide once (1st generation)
7. Then centrifuge again and observe the results
8. Then allow bacteria to divide again on 14N (2nd generation)
9. Each strand of double helix acts as new template for next generation
10. Extract and centrifuge again
11. Allow a 3rd division and repeat
What are the findings of Meselson and Stahl?
One band formed, containing both N-14 and N-15, proving semi-conservative (the correct model) but also didn't disprove dispersive model.
Models of DNA replication
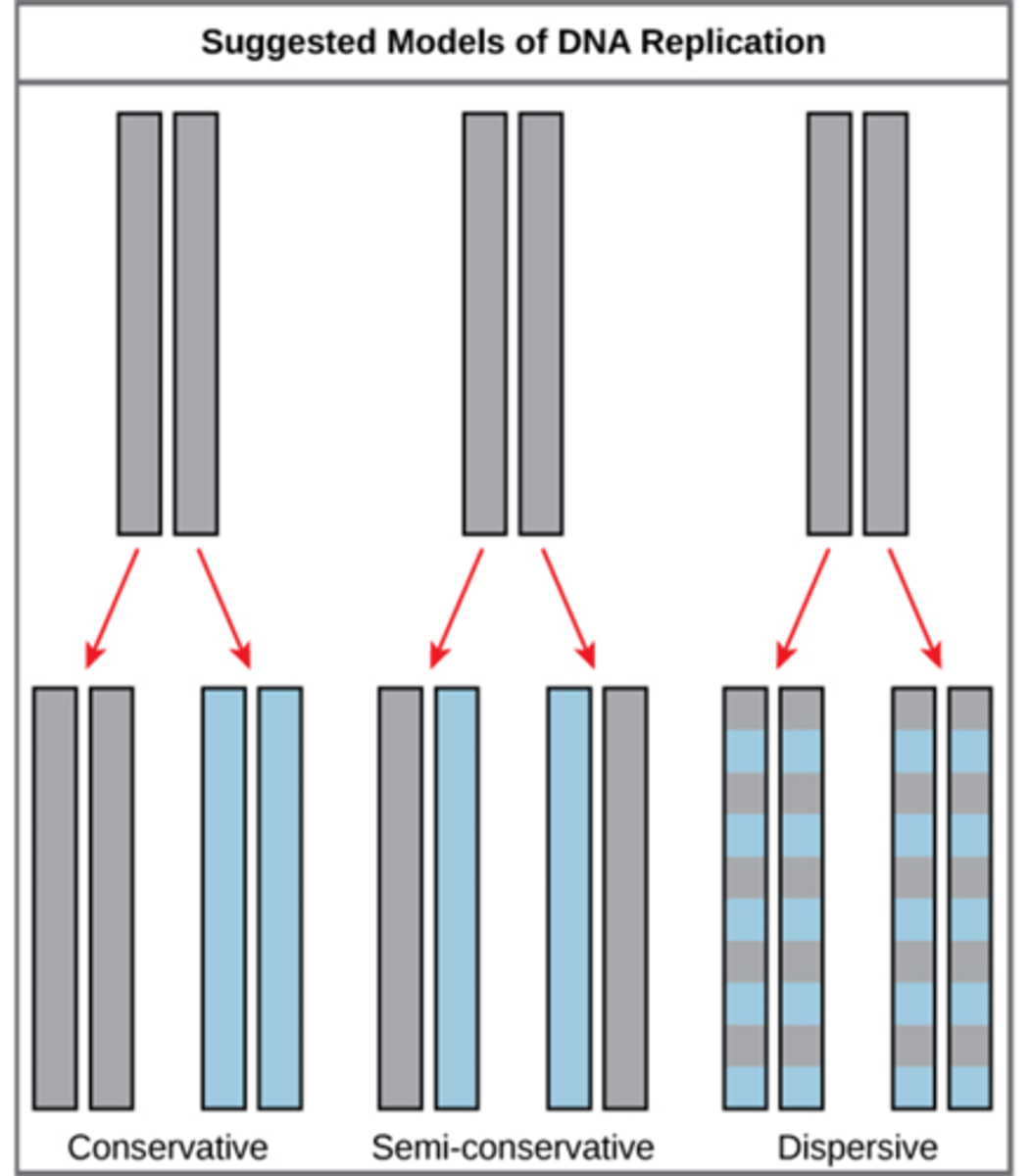
Why is semi-conservative replication important?
Ensures genetic continuity between generations of cells.
What would be seen if it was conservative replication?
● No banding in the middle
● Bottom band
● Top band will increase in thickness after each replication
● Entirely new material for each new double helix
What is the history of DNA?
● Doubted DNA as a carrier of genetic code due to its relatively simplicity
● Originally doubted Watson & Crick's semi-conservative model
● W + C founded double helix
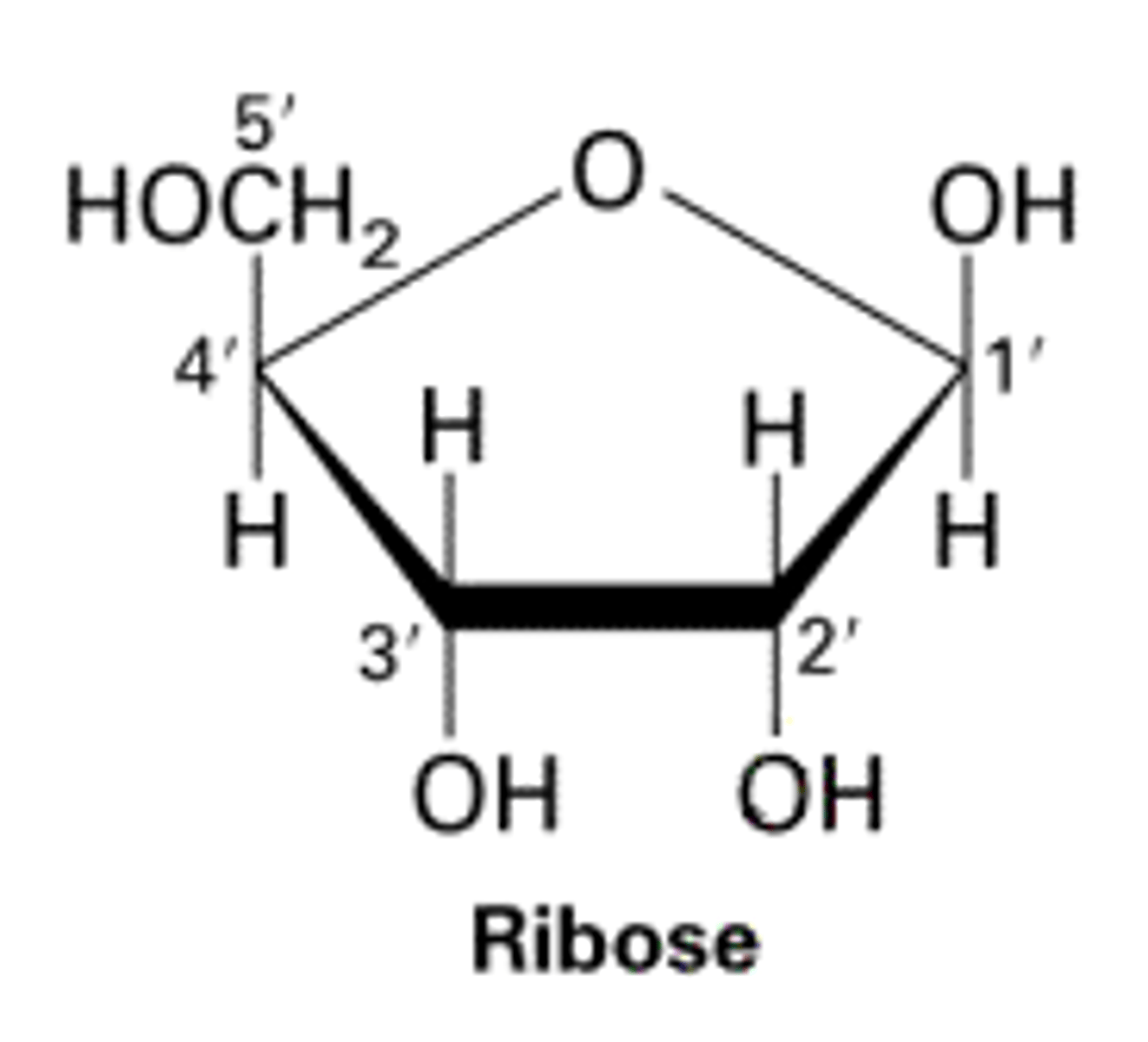
How are carbon atoms arranged in pentose sugar molecules?
C-atoms numbered after oxygen clockwise. Carbon-5 is outside pentose molecule.
What is semi-conservative DNA replication?
● One parental DNA splits to make 2 brand new DNA, each with a parent strand & daughter strand.
● Each of the 2 new molecule has one strand of new material and one strand of original.
Why is DNA replication referred to as semi-conservative?
One strand of the original DNA is conserved in the new DNA molecule after replication.
When and why must DNA replication occur?
Before cells divide (by mitosis or meiosis) all DNA must replicate to provide a copy of the new cell
What are complementary base pairs in DNA?
2 H- bonds between Adenine + Thymine
3 H- bonds between Cytosine + Guanine
What are complementary base pairs in RNA?
2 H- bonds between Adenine + Uracil
3 H- bonds between Cytosine + Guanine
What enzymes are involved in DNA replication?
DNA helicase
DNA polymerase
Describe the steps of semi-conservative DNA replication.
1. DNA helicase breaks the hydrogen bonds between complementary base pairs on parental DNA.
2 strands unwind from each other and separate. Each strand is a template for a new strand.
2. DNA nucleotides floating around in the nucleus are attracted to the their complementary base pairs on the template strand.
3. DNA polymerase attaches to both strands and and joins adjacent nucleotides together, to form phosphodiester bonds, (between phosphate of one and deoxyribose of another) by a condensation reaction, H bonds reform
4. Both enzymes continue to move along the DNA molecule until 2 identical molecule of DNA form that coil to form 2 helixes.
What do the daughter cells contain?
One strand of original DNA
One strand of newly synthesised DNA
Use your knowledge of enzyme action to suggest why DNA polymerase moves in opposite directions along DNA strands.
● DNA has antiparallel strands
● Arrangements of nucleotides on two ends are different
● DNA polymerase is an enzyme with a specific shaped active site
● So can only bind to substrate with complementary shape (phosphate end of developing strand)
During replication, the 2 DNA strands separate and each acts as a template for the production of a new strand. As new DNA strands are produced, nucleotides can only be added in the 5' to 3' direction.
Use the figure in part (a) and your knowledge of enzyme action and DNA replication to explain why new nucleotides can only be added in a 5' to 3' direction.
1. Reference to DNA polymerase;
2. (Which is) specific;
3. Only complementary with / binds to 5' end (of strand);
4. Shapes of 5' end and 3' end are different / description of how different.
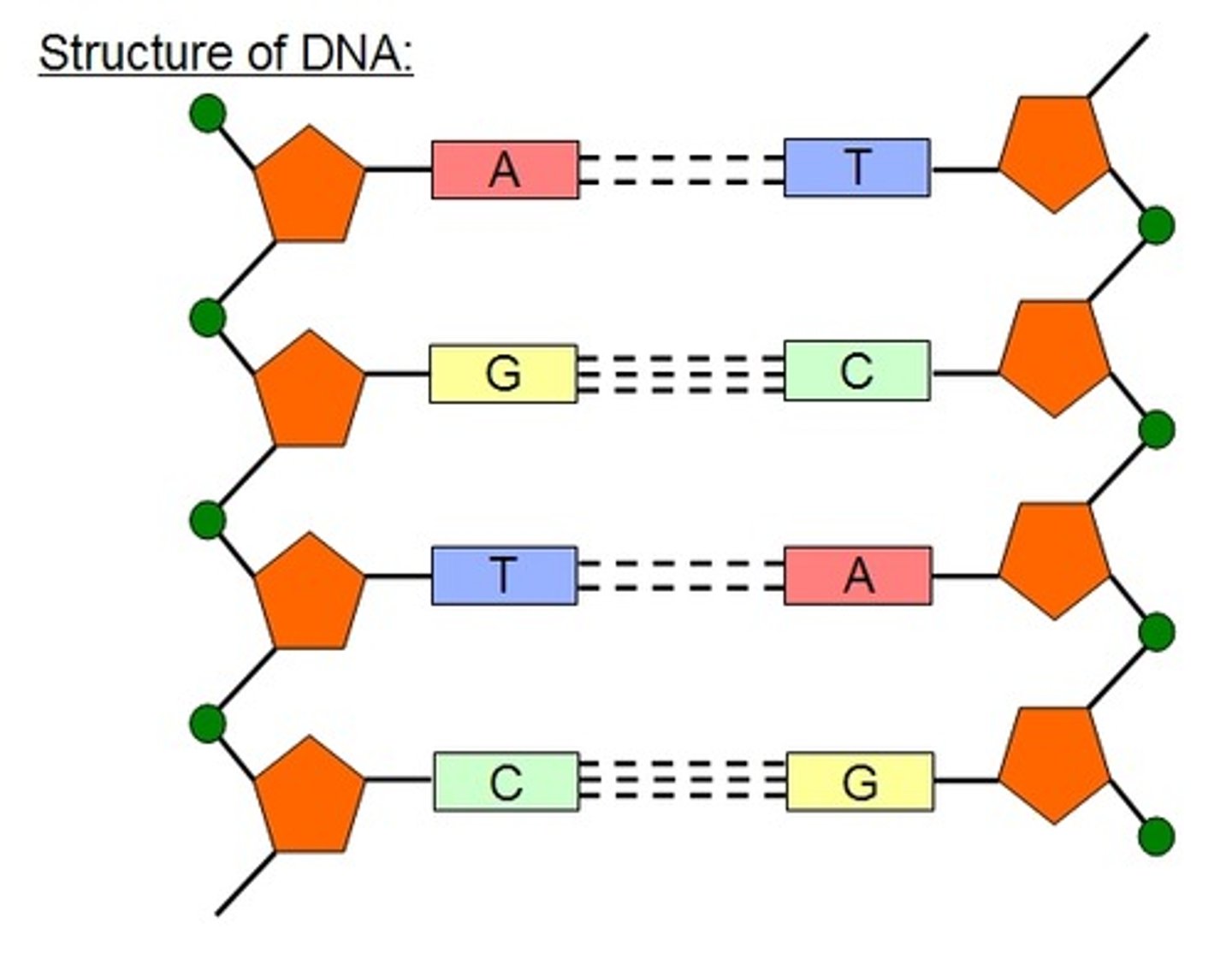
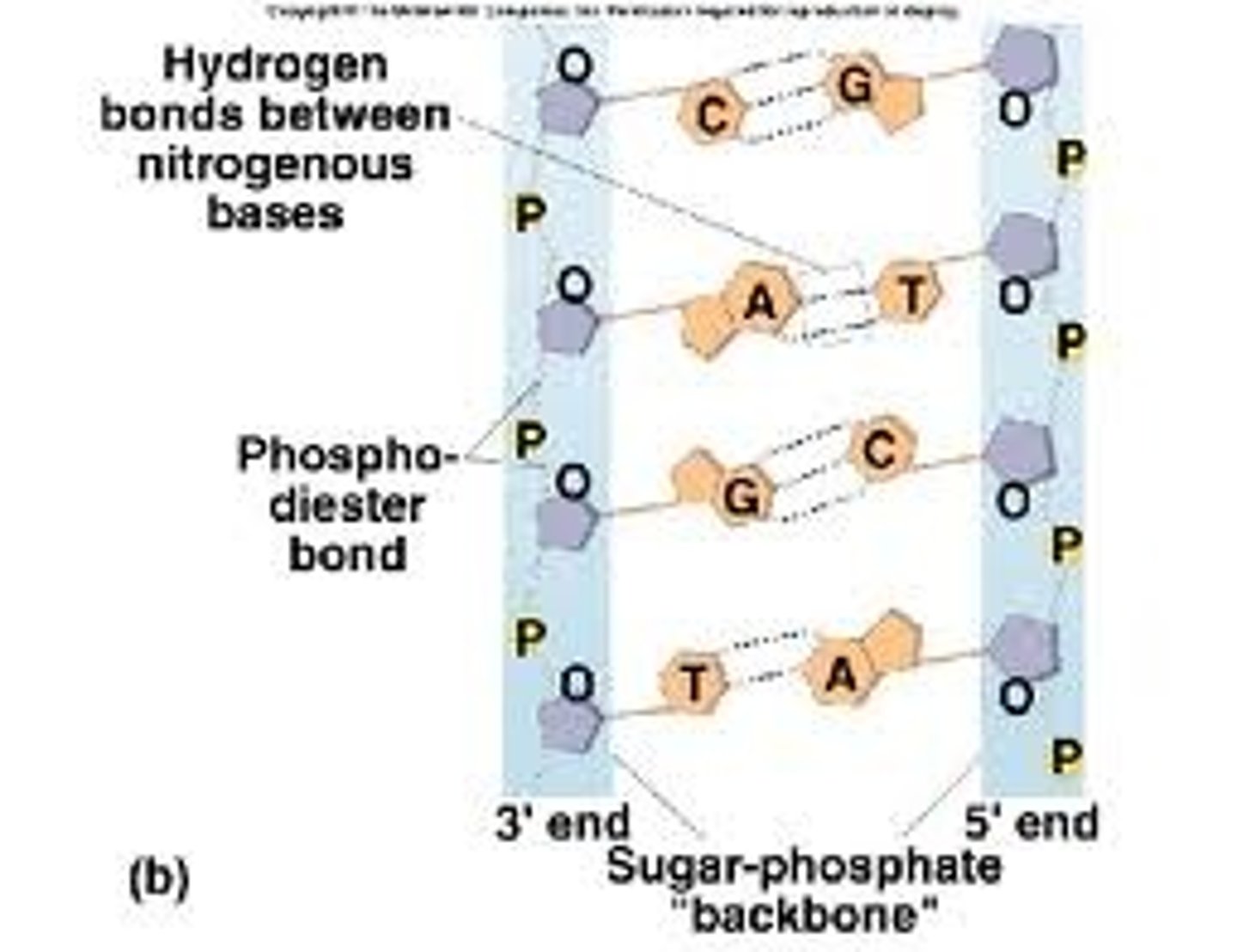
Explain the role of DNA ligase.
● Each strand has a 5' (5 prime) and a 3' end
● The 2 strands run in opposite directions (ie one starts at 5' the other starts at 3')
● Unzipping of strands results in replication fork
● Bases are added from the 5' end to the 3' end
● Leading strand is made continuously by adding bases one by one from 5' to 3' end
● Lagging strand runs in opposite direction so can only add bases in small chunks at a time (okazaki fragments)
● DNA ligase joins the shorter fragments together.
Describe how a phosphodiester bond is formed between two nucleotides within a DNA molecule.
1. Condensation reaction (removing water);
2. (Between) phosphate and deoxyribose;
3. (Catalysed by) DNA polymerase
Name the two scientists who proposed models of the chemical structure of
DNA and of DNA replication.
Watson and Crick
Describe the work of Meselson and Stahl in validating the Watson-Crick
model of semi-conservative DNA replication.
1. Bacteria grown in medium containing heavy nitrogen (¹⁵N) so nitrogen is incorporated into DNA bases
○ DNA extracted & centrifuged → settles near bottom, as all
DNA molecules contain 2 'heavy' strands
2. Bacteria transferred to medium containing light nitrogen (¹⁴N) and allowed to divide once
○ DNA extracted & centrifuged → settles in middle, as all DNA molecules contain 1 original 'heavy' and 1 new 'light' strand
3. Bacteria in light nitrogen (¹⁴N) allowed to divide again
○ DNA extracted & centrifuged → half settles in middle, as
contains 1 original 'heavy' and 1 new 'light' strand; half settles
near top, as contains 2 'light' strands
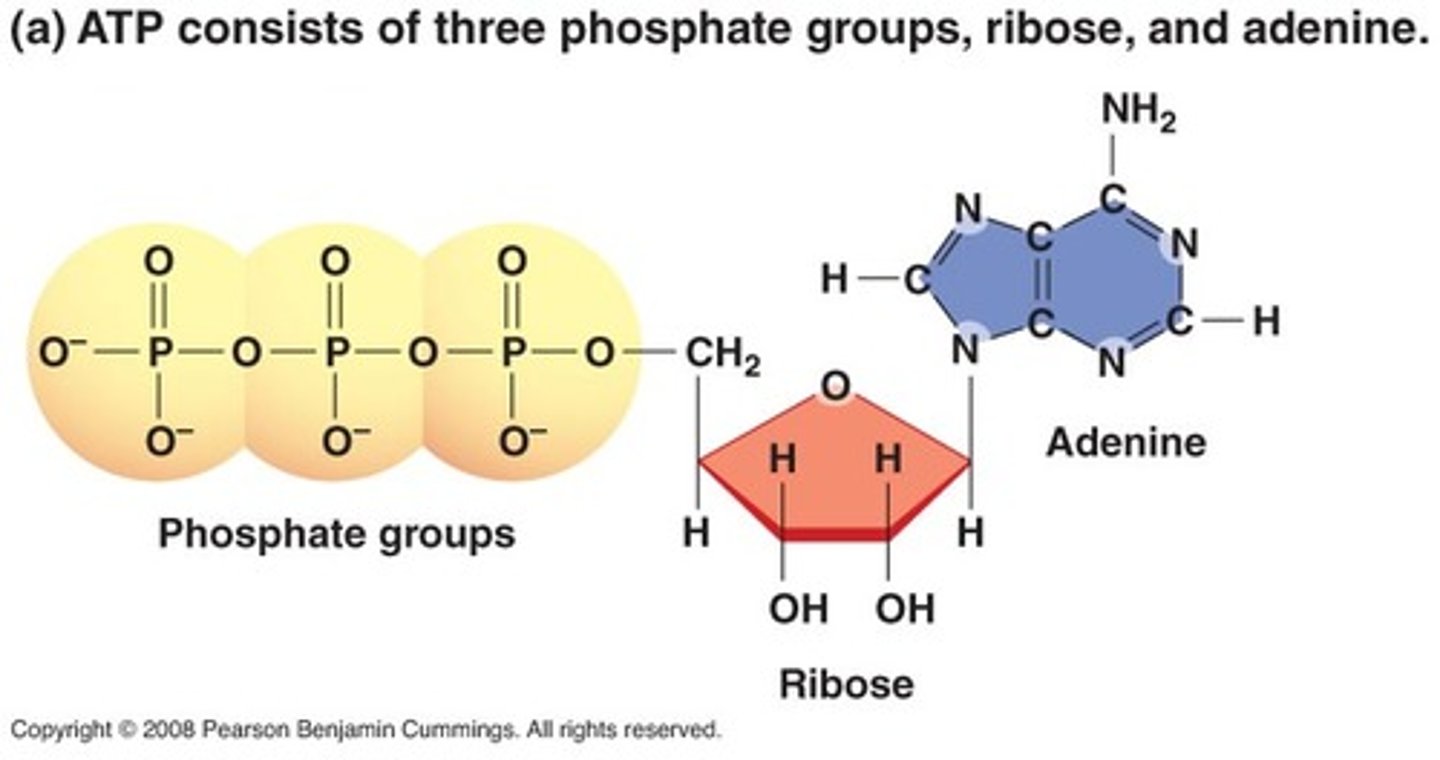
What is ATP?
● Adenosine triphosphate, a nucleotide derivative
● Immediate source of energy for biological processes
● Metabolic reactions require a steady supply of ATP
Describe the structure of ATP.
● Ribose (pentose sugar)
● Triphosphate, 3 inorganic phosphate groups (Pi)
● Adenine- a nitrogenous base
Describe how ATP is broken down
● ATP (+ water) → ADP + Pi (inorganic phosphate)
● Hydrolysis reaction, using a water molecule
● Catalysed by ATP hydrolase (enzyme)
Give 2 ways in which the hydrolysis of ATP is used in cells
● Coupled to energy requiring reactions within cells (releases energy)
○ e.g. active transport, protein synthesis
● Inorganic phosphate released can be used to phosphorylate (add phosphate to) other compounds, making them more reactive
Describe how ATP is resynthesised in cells
● ADP + Pi → ATP (+ water)
● Condensation reaction, removing a water molecule
● Catalysed by ATP synthase (enzyme)
● During respiration and photosynthesis
Explain how ATP is made during respiration.
● ATP is made from ADP (only has 2 phosphate groups) the addition of an inorganic phosphate
● Via a condensation reaction
● Using the enzyme ATP synthase
Explain how ATP is hydrolysed
● ATP can be hydrolysed into ADP + Pi, using enzyme ATP hydrolyse
● By breaking one of the Pi bonds in a hydrolysis reaction, released a small amount of energy to be used in chemical reactions → immediate
What is phosphorylation?
● The inorganic phosphate released during the hydrolysis of ATP
● Addition of a phosphate group to a molecule to make a compound more reactive
Explain how the properties of ATP make it suitable for its function.
● Releases energy in small manageable amounts, → little energy lost as heat
● Lowers activation energy and can be reformed for reuse.
● Small and soluble → easily transported around the cell
● Only one bond hydrolysed to release energy → immediate
● Phosphorylation → transfer energy to another molecule by transferring one of its phosphate groups. Make compounds more reactive
● ATP can't pass out the cell → all cells have a constant supply of ATP or ADP + Pi
Compare how glucose is suited for its function. (Make sure to include points for how ATP is, and compare.)
● Glucose releases large amount of energy, could be wasted
● Glucose small and soluble —> easily transported around the cell
● Several bonds must be broken to release energy
● No phosphorylation in glucose as doesn't contain phosphate groups
● Cells may run out of glucose
Where does ATP synthesis happen?
Plants - chloroplasts
Animals - mitochondria
Explain how hydrogen bonds occur between water molecules.
● Water is polar molecule
● Slightly negatively charged oxygen atoms attract slightly positively charged hydrogen atoms of other water molecules
Why is water a polar molecule?
O is more electronegative than H, so attracts the electron density in the covalent bond more strongly.
Forms O δ- (slight negative charge) & H δ+ (slight positive charge).
State the 5 properties of water that are important in biology.
1. Metabolite
2. Solvent (can dissolve solutes)
3. High specific heat capacity
4. Large latent heat of vaporisation
5. Strong cohesion between water molecules
What is the importance of water being a metabolite?
● A metabolite in condensation / hydrolysis / photosynthesis / respiration
● Blood plasma 90% water, cytoplasm mainly water
What is the importance of water being a solvent?
Can dissolve other substances e.g. inorganic ions, enzymes, urea, etc so...
● Metabolic reactions can occur (faster in solutions)
● Allows transport of substances e.g. nitrates in xylem, urea in blood
Why is water a good solvent?
● Polar molecules dissolve readily in water due to water being dipolar.
● Slight positive charge H δ+ attracts any negative ions. Slight negative charge O δ- attracts any positive ions in solutes
● These polar molecules often describe as as hydrophilic
● Non polar molecules cannot dissolve in water are hydrophobic e.g. lipids
What is the importance of water having a high specific heat capacity?
● Buffers changes in temperature
● As can gain/lose a lot of heat/energy without changing temperature.
1. Good habitat for aquatic organisms as temperature more stable than land.
2. Helps organisms maintain a constant internal body temperature.
- Important so enzymes don't denature
What is the importance of water having a large latent heat of vaporisation?
Requires lots of energy to convert from liquid to gas, to break H bonds.
● Allows effective cooling via evaporation of a small volume (eg. sweat)
● So helps organisms maintain a constant internal body temperature
What's latent heat of vaporisation?
- Thermal energy absorbed by water to change state from liquid to gas
- Without causing an increase in temperature
What is the importance of strong cohesion between water molecules?
1. Supports columns of water in tube-like transport cells of plants eg. transpiration stream through xylem in plants
2. Produces surface tension where water meets air, supporting small organisms (to walk on water)
Where are inorganic ions found in the body?
In solution in cytoplasm and body fluid, some in high concentrations and others in very low concentrations.
What are inorganic ions and where are they found in the body?
● Ions that do not contain carbon atoms.
● Found in cytoplasm & extracellular fluid.
● May be in high or very low concentrations.
Describe the role of hydrogen ions H⁺
● Maintain pH levels in the body → high concentration = acidic / low pH
● Affects enzyme rate of reaction as interact with H-bonds & ionic bonds in tertiary structure of proteins, which can cause them to denature.
Describe the role of iron ions Fe²⁺
● Component of haem group of haemoglobin
● Allowing oxygen to bind / associate for transport as oxyhaemoglobin
● 4 haem groups per haemoglobin molecule
Describe the role of sodium ions Na⁺
1. Involved in co-transport of glucose / amino acids into cells (2.3)
2. Involved in action potentials in neurons (6.2)
3. Affects water potential of cells / osmosis (2.3)
Describe the role of phosphate ions PO₄³⁻
1. Component of nucleotides, allowing phosphodiester bonds to form in DNA / RNA
2. Component of ATP, allowing energy release
3. Phosphorylates other compounds making them more reactive
4. Hydrophilic part of phospholipids, allowing a bilayer to form