aldehyde, ketones, carboxylic acids
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
carbonyl
adds polarity HC’s → only an H-bond acceptor
carbonyl middle of chain → ketone | on end → aldehyde
ketone
end of parent chain drop “e”, replace to -one, otherwise use -oxo if other functional groups have higher priority (eg. 3-oxobutanal)
primary alcohol oxidized to secondary alc which becomes a ketone
aldehyde
end of parent chain drop “e”, replace to -al, otherwise use -oxo if other functional groups have higher priority (eg. 3-oxobutanal)
aldehyde > ketone
primary alcohol is aldehyde after primary is oxidized
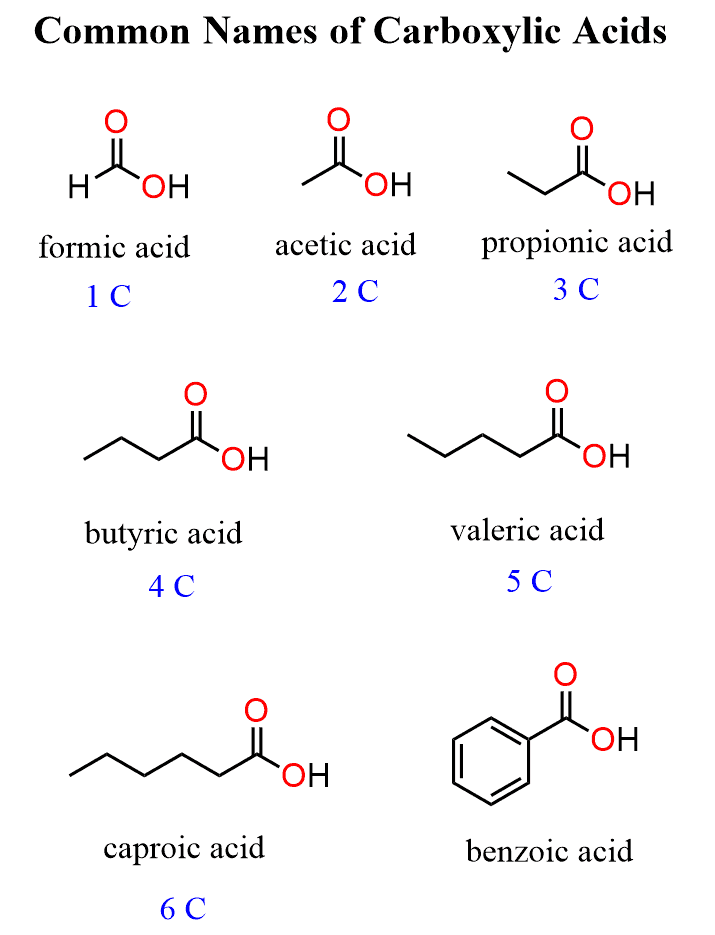
carboxyl properties
H-bonding (OH) → increases melting/boiling points and miscibility in water
H in the OH can be partially donated, giving HC acid properties
carboxyl reactions
R-COOH made by oxidizing an aldehyde ONLY (NOT KETONE)