introduction to polymer AND acrylic resin
1/116
Earn XP
Description and Tags
lecture given 3/2/2026
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
117 Terms
what are the parts of the material properties pyramid?
properties, processing, composition, structure
what kinds of things are made of polymers?
plastics, rubber, elastomer, fiber, foam, composite, adhesive, glue, paint
a polymer has a molecular weight of over…
5000
what was the first use of polymer in dentistry, and what is the most recent advancement?
1901- study on polymethylmethacrylate denture
2023- first 3DP high ceramic filled composite
methane
CH4
ethane
CH3CH3
butane
CH3CH2CH2CH3
hexane
C3(CH2)8CH3
addition
R* + C=C → R-C-C*
condensation
a-R-a + b-R’-b → a-R-R’-b + a-b
a-b is usually an acid or water
what is the structure of a polymer?
chain
primary level of a polymer
configuration (sequence)
secondary level of polymer structure
conformation- a helix and b sheets
tertiary level of polymer structure
3D structure with multiple polymers
linear polymer chain structure
like spaghetti, linear structure with no networking
crosslinked polymer chain structure
like a net, has crosslinked chains
no melting point
amorphous polymers
have molecular chains arranged randomly with no long term order
they soften rather than melt suddenly
polystyrene
crystalline polymers
have a regular order or pattern of molecular arrangement
have a sharp melting point
HDPE is semi-crystalline
the glass transition appears to be a…
second order transition as the heat capacity and thermal expansion coefficient of the polymer undergo finite changes
the glass transition temperature depends on…
the rate of measurement
t/f we have a complete theoretical understanding of glass transition
false
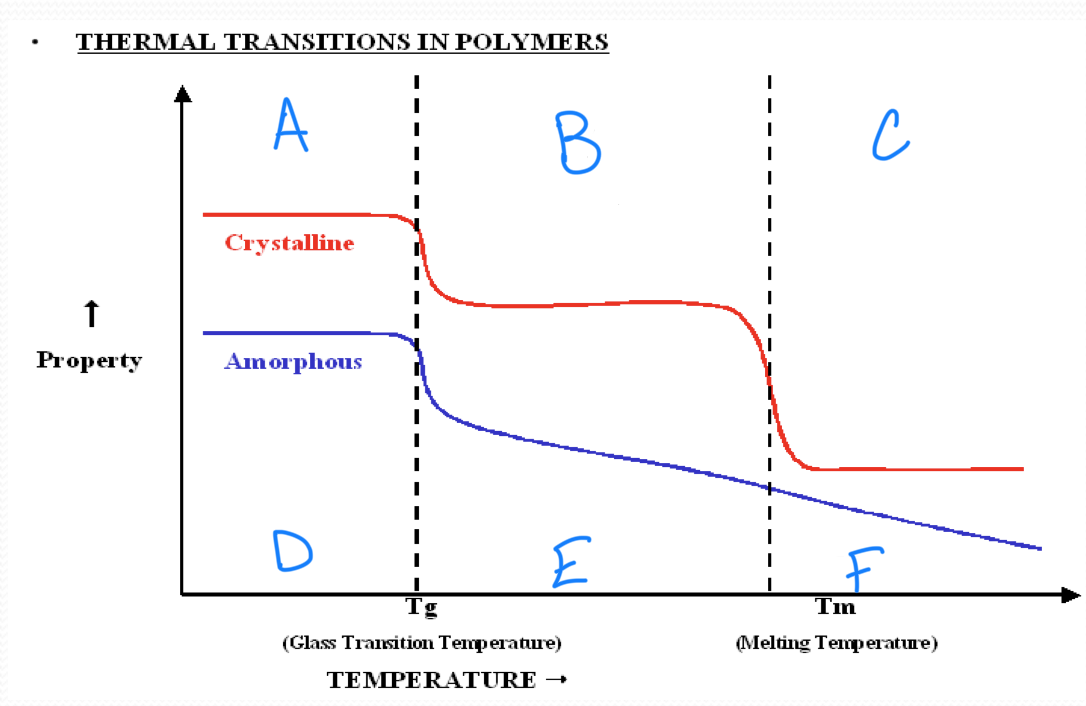
a
glassy state
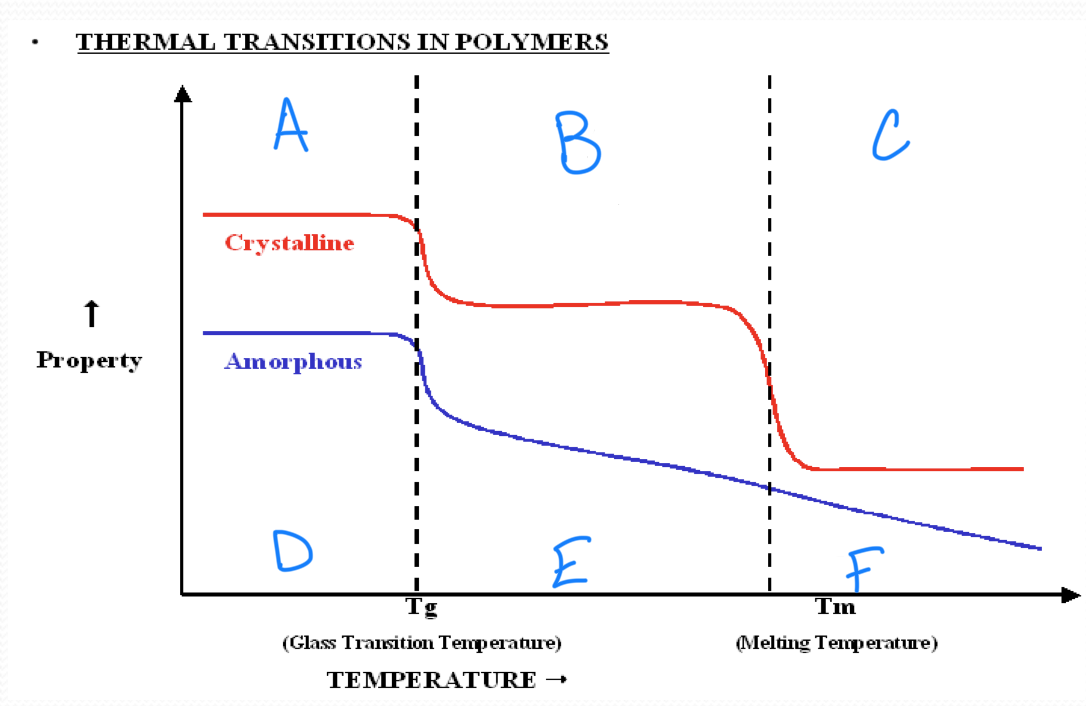
b
rubbery state or viscoelastic state
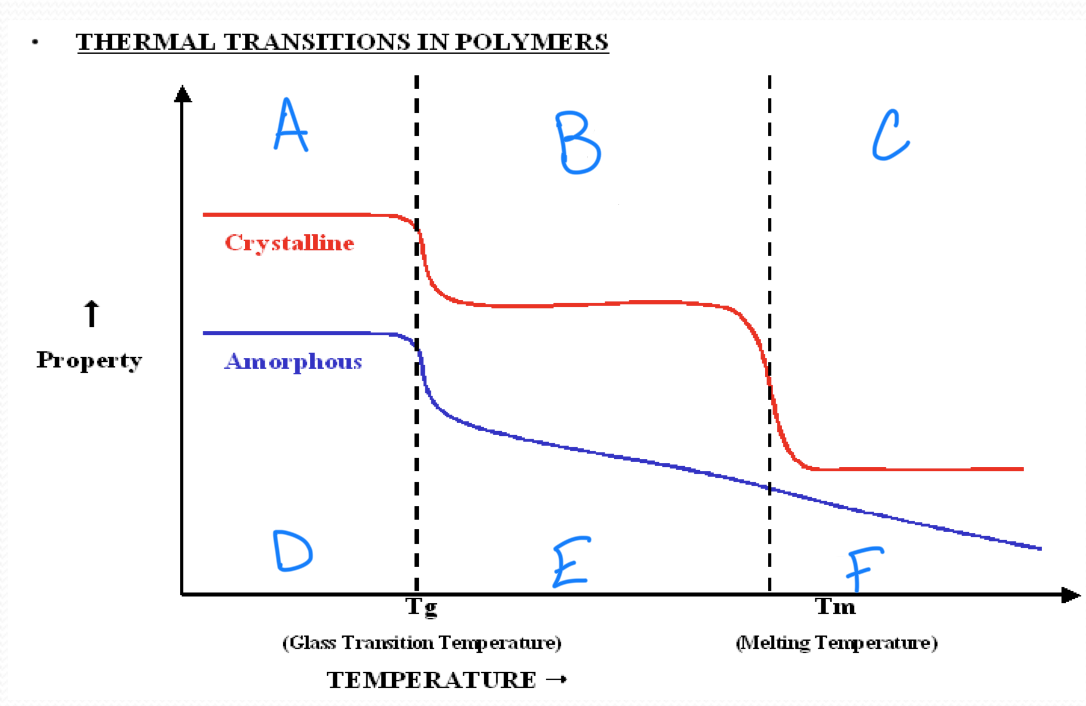
c
melt state
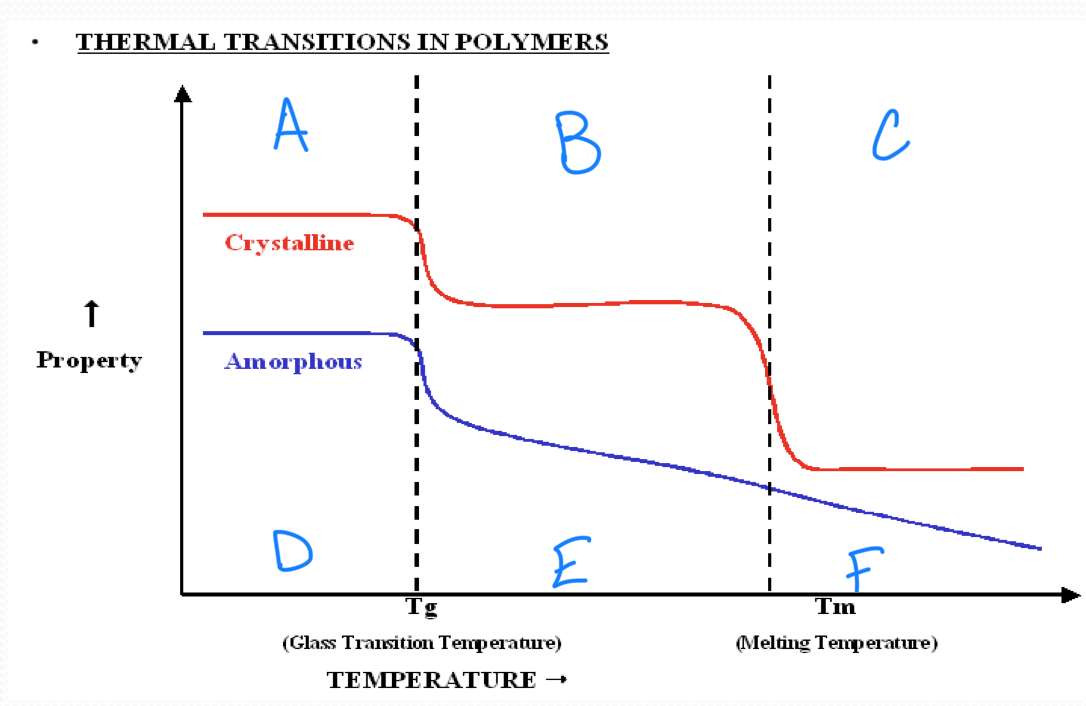
d
brittle
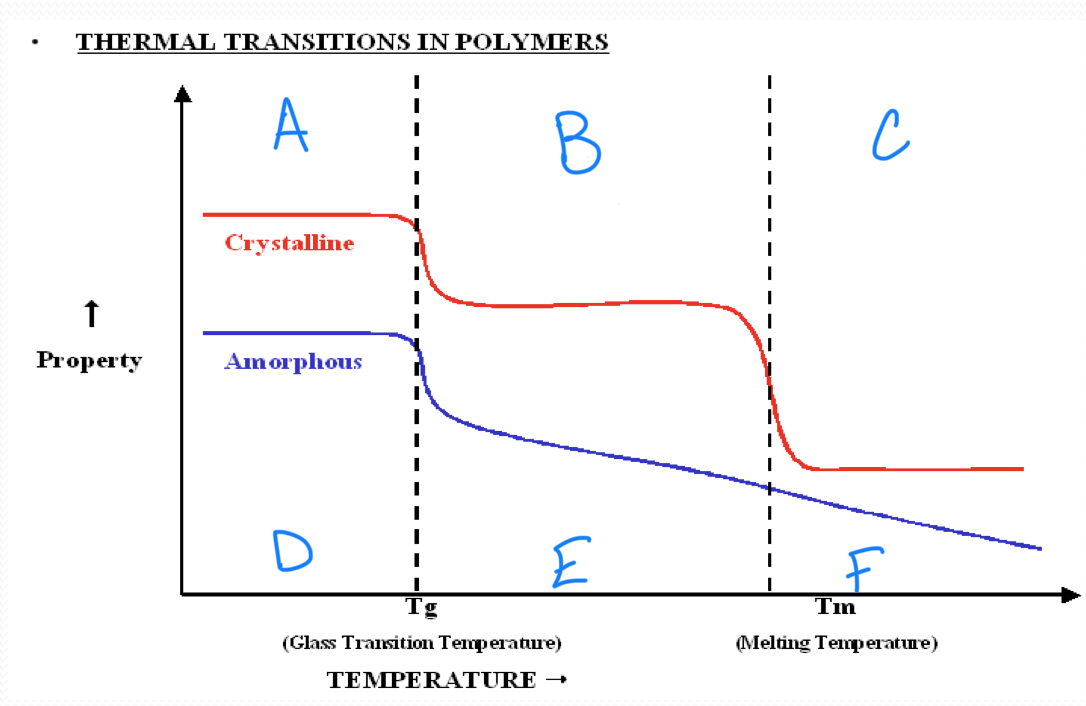
e
tough/soft
f
melt
Tg ___ Tm
<
in crystallizable polymer, Tg and Tm have a ____ correlation
rough
a melting temperature range is observed in all ________ polymers
semicrystalline
stronger intermolecular forces result in a(n) ______ in tm
increase
rigid chains have much higher…
tm
plastic working temperature ___ Tg
rubber working temperature ___ Tg
<, >
copolymer
polymer chain made by 2 or more monomers
linear homopolymer
base unit = repeating unit
statistical copolymer
base unit =/= repeating unit
alternating copolymer
2 base units = repearting unit
what type of copolymer are dental materials usually made of?
statistical copolymer
t/f polymer is typically a mixture of different sized molecules
true
Mn
number average molecular weight
Mw
weight average molecular weight
Mn < ____ < Mw < ___
Mv, Mz
polydispersity index (PDI)
Mw/Mn
threshold molecular weight value
TMWV- lowest molecular weight where the desired property value is achieved
the higher the molecular weight…
higher Tm, crystallinity, Tg, density, viscosity, wear resistance, strength
lower solubility
materials with higher PDI for same average Mn…
wider Tg range, lower surface hardness, lower Tm or soft point (Tg), lower tensile/tear/impact strength, lower brittleness, lower aging resistant, lower viscosity, higher creep
radical addition polymerization
R-C-C-R’ → R-C* + *C-R’ or R-C- + +C-R’ or R-C+ + -C-R’
R-C* + C=C → R-C-C-C*
R-C-C-C* + C=C → R-C-C-C-C-C*
what are the mechanisms of free radical polymerization?
induction (activation, initiation), propagation, termination chain transferring
induction
activation- fast, R-R’ → R. + R.’
initiation- slow, R. + MH → R-H + M.
propagation
fast, produces heat
termination
M. + M. → M-M
chain transferring
M. + M’H → M’. + MH
activation
free radicals can be generated by activation of radical producing molecules, ie initiator, using heat, redox reaction, and light
heat
commonly employed initiator is benzoyl peroxide (BPO) which is activated rapidly between 50-100C to release 2 free radicals per BPO molecule
redox chemical reaction
consists of 2 reactants, when mixed undergo reaction
eg tertiary amine (the activator) and benzoyl peroxide (the initiator)
visible light or ultraviolet light
camphorquinone (CQ) and an organic amine (dimethylaminoethyl-methacrylate) generate free radicals when irradiated by light in the blue or violet region
light is a wavelength of about 470mn is needed
initiation
free radicals formed by initiator open double or triple carbon bonds in the monomer
new free radicals with monomer structure form
slow reaction
propagation stage
rapid addition of monomer molecules to free radical- free electrons shifted to end of chain
the resulting free radical monomer complex acts as a new free radical center which is approached by another monomer to form a dimer, which also becomes a free radical
very fast reaction, exothermal
chain transfer stage
the active free radical of a growing chain is transferred to another molecule and a new free radical for further growth is created termination occurs in the latter
termination stage
reaction terminates when insufficient free radicals exist, reaction also may be stopped by any material that will react with free radical
can occur from chain transfer
addition polymerization reaction is terminated by: direct coupling of 2 free radical chain ends, exchange of hydrogen atom from one growing chain to another
this relates to the oxygen inhibited layer- terminated early because of oxygen
photo polymerization
the use of light source (light energy) in the preparation of polymers
photoinitiator
UV- irgacure 2959— 3D print, post curing
blue visible light: CQ, lucirin (TPO), PPD,
content- 05.-1% initiator in most commercial dental composite
shade issue- CQ is a yellowish compound
t/f european commission is banning TPO use
true
inhibitors
used to completely stop the conversion of monomer to polymer produced by accidental initiation during storage
phenols, quinones, or hydroxyquinones, or even molecular oxygen are employed to inhibit the polymerization
butylated hydroxytoluene is often used in dental composite
inhibitor effect
used in 3DP to prevent undesired polymerization of resin fluid
oxygen permeable sheet in carbon 3D
thermoplastic polymer has _____ structure
linear chain
polymer network density
a mechanical property
material property changes in resin polymerization
exothermal reaction
temperature increase
material property changes in resin polymerization
light absorption change
low translucent, shade change
material property changes in resin polymerization
volume shrinkage
shrinkage stress
material property changes in resin polymerization
viscosity increase
determine working time
material property changes in resin polymerization
modulus increase…
hardness and strength increase
what is the working time (s, minimum) and setting time (min, maximum) of restorative materials, self cure?
90, 5
what is the working time (s, minimum) and setting time (min, maximum) of restorative materials, dual cure?
90, 10
what is the working time (s, minimum) and setting time (min, maximum) of luting materials, self ans dual cure?
60, 10
what is the working time (s, minimum) of light cure materials?
none because you control the light
degree of conversion
the fraction of monomer’s functional group has been reacted in polymerization
auto acceleration effect / gel effect / trommsforff-norrish effect
a dangerous reaction behavior that can occur in free radical polymn system- high temp
a broadening of the molecular weight distribution (MWD) simultaneously occurs
pre-made CAD/CAM polymer/Composite
higher defree of conversion from pre-polymerization (done by manufacturing)
need to form a shape for final use by milling
not a ceramic even though there is a ceramic content (as filler)
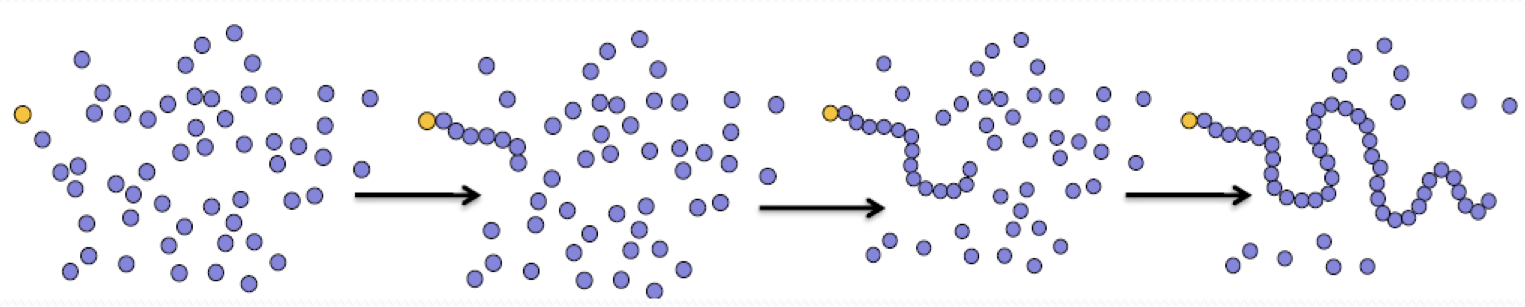
what is this?
chain growth
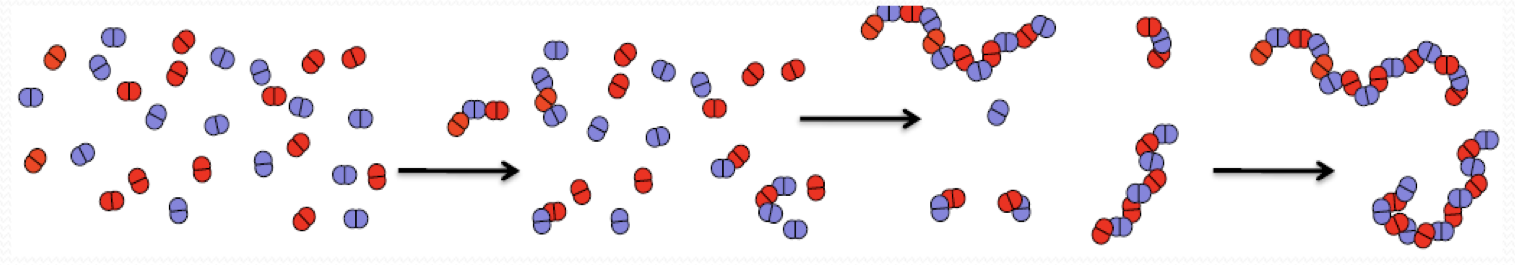
what is this?
step growth
what type of materials go through step growth?
impression material
what type of materials go through chain growth?
adhesive cement, composite material
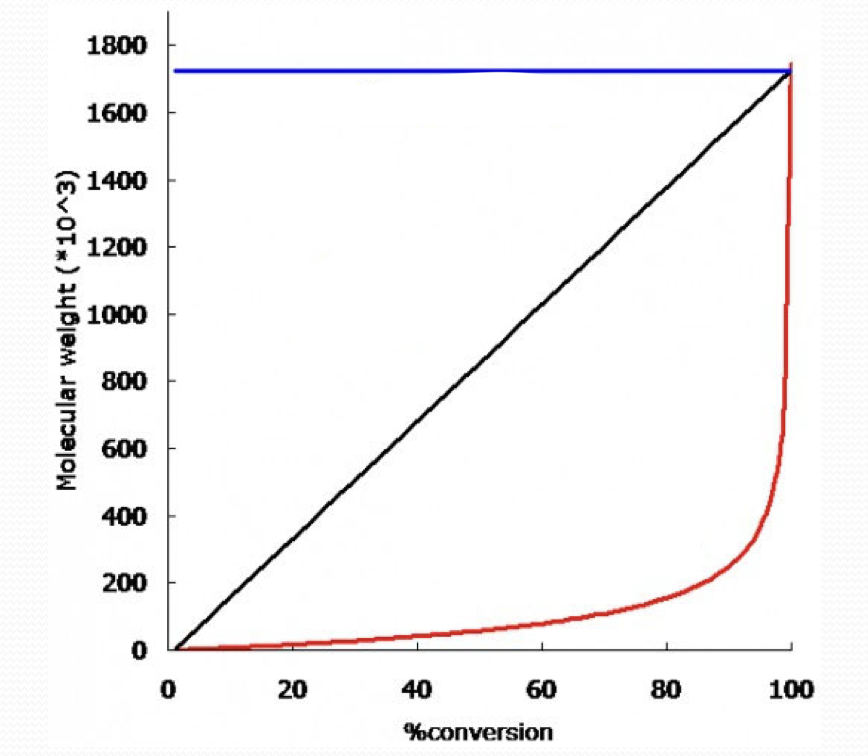
what type of growth is the blue line?
chain growth
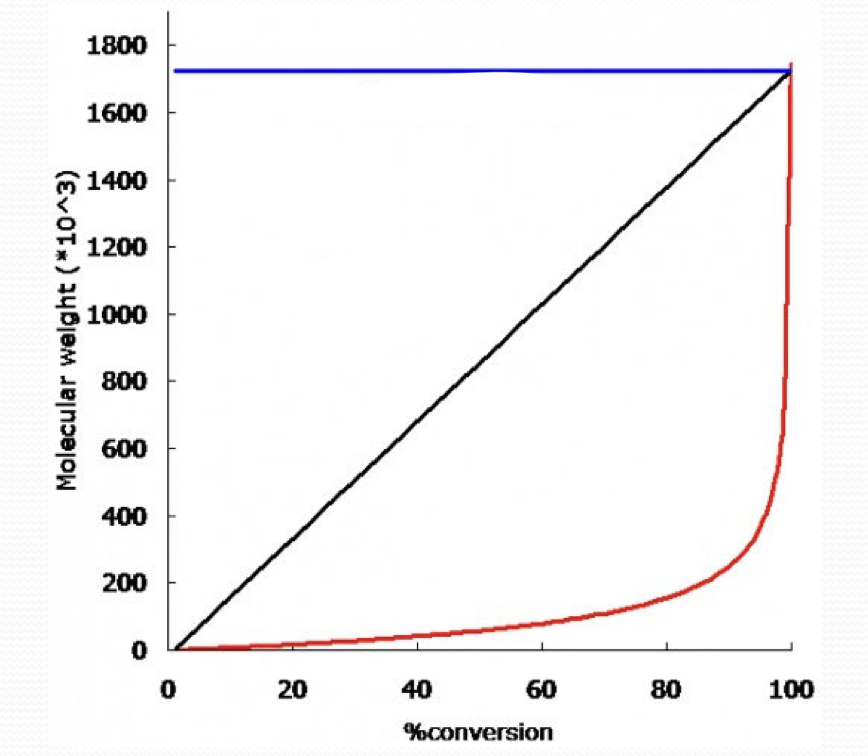
what type of growth is the black line?
living chain growth
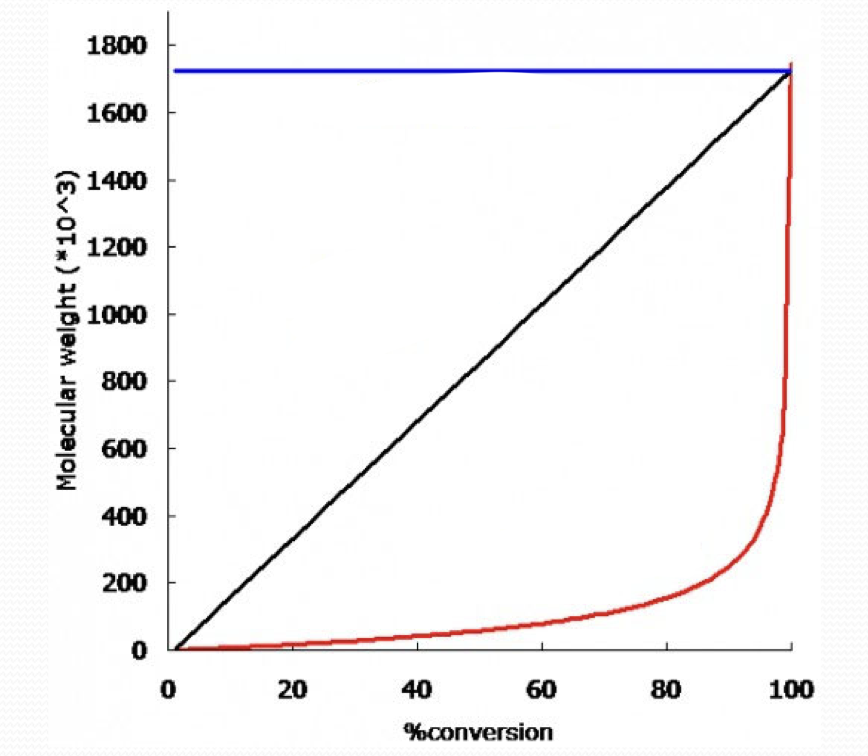
what type of growth is the red line?
step growth
what are examples of step growth polymerization?
polycarbonate chemistry, polyurethane chemistry, epoxy chemistry (endo materials)
what happens during silicone polymer condensation?
alcohol releases from the reaction mixture and contributes to the contraction/shrinkage of the impression
what happens during additive silicone setting reaction, and what is an example of additive silicone?
polyaddition- step growth polymerization
not polycondensation, no small molecules released, not free radical shrinkage, less shrinkage
PVS
photopolymer
a polymer that can be selectively cross-linked by radiation (UV, blue light)
normally composted by large molecular weight oligomer or monomers
from soluble to insoluble polymer, or the opposite way
used for 3D printing, electric circuit print, lithography, plate printing
vat polymerization
what post processing of 3D printed material is necessary?
cleaning/washing- remove adhered monomer resin
trimming- clean off the extra support structures
post curing- increase the degree of conversion
creep of polymer materials
the long term deformation of a polymer under a sustained or intermittent load at stresses below the yield point
may be partially or fully recovered with extended time
time dependent!
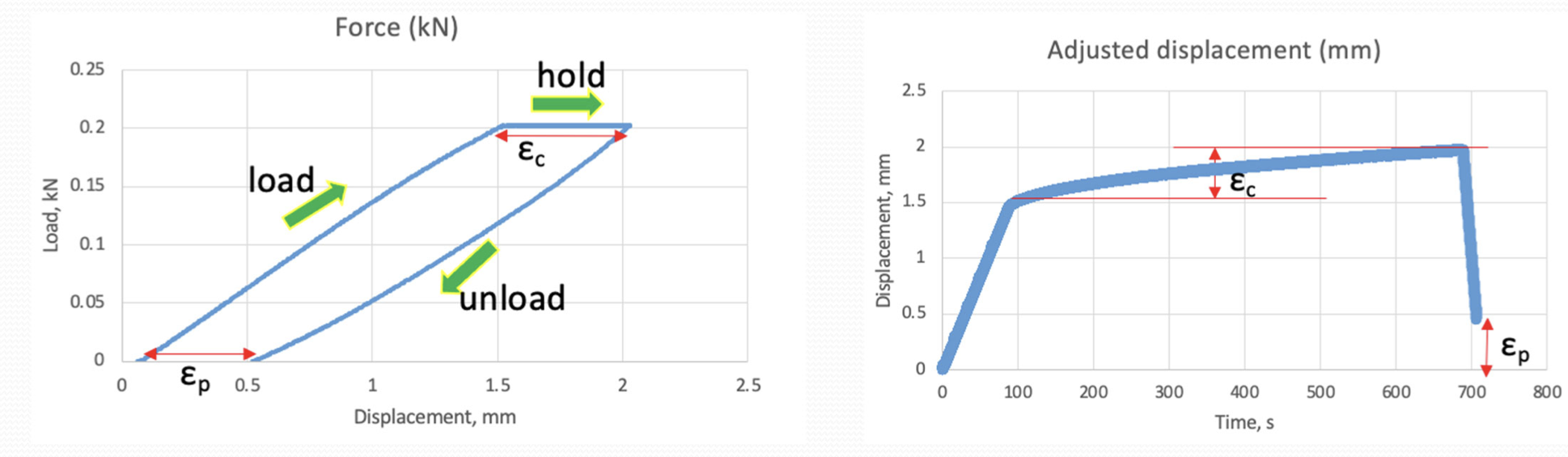
what is this?
creep compliance in stress strain curves
what do plasticizers do?
separate polymer chains
lower Tg, E, H, and increase toughness
polymer early stage degradation
polymer long chain may break down while the material is in the same dimension but deteriorate property
may release small molecules
higher water absorption
when engineering polymer, what is the goal?
improved mechanical and thermal properties, improved resistance to degradation, improved chemical resistance
polyaryletherketon (PAEK)
commercialized for industry in 1980s
high performance thermoplastic polymers
use for dental implants, framework, restoration
polyaryletheretherketone (PEEK) is one form
what are characteristics of PEEK?
elastic modulus 3.8 GPa, similar to bone
may be processed via injection molding, 3DP
can be used in metal free RPD
radiolucent- reduce artifact in CT
cannot be repaired, must be made once, bonding issue
what are acrylic polymer uses in dentistry?
denture bases and artificial teeth, denture liners and tissue conditioners, composite restorative and pit/fissure sealant, custom tray for impressions, temporary restoratives, mouth guards, maxillofacial prosthesis, veneers, cements and adhesives
what are acrylic acids made of?
acrylic acid (propenoic acid) and methacrylic acid