3.3.2.4 chlorination of alkanes + free-radical substitution
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
in what conditions does alkanes react with halogens to form what products?
in the presence of ultraviolet (UV) light
to form halogenoalkanes + a hydrogen halide
give the word equation for the reaction between a halogen + alkane
halogen + alkane → halogenoalkane + hydrogen halide
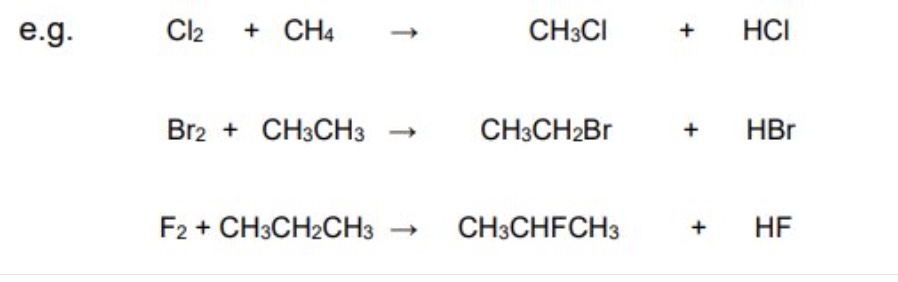
what type of reaction is the reaction between a halogen + alkane? how?
a substitution reaction → a hydrogen in the alkane is replaced ny the halogen
what happens to the hydrogen lost from the alkane?
it will join with the second half of the diatomic halogen molecule to form HX (HF, HCl, or HBr or HI)
once the reaction between a halogen + alkane, what may happen?
the process may continue until all the hydrogen atoms in the alkane are replaced by halogen
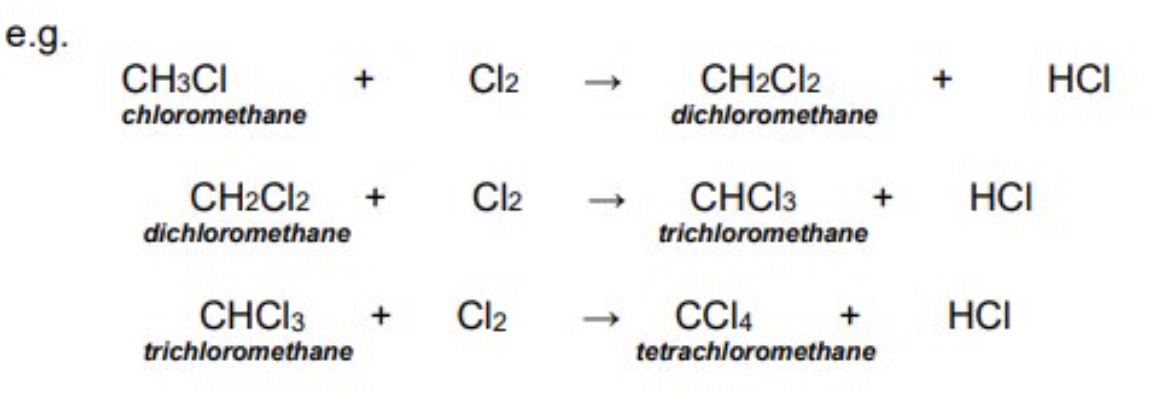
when writing balanced equations for these reactions, what’s a trick to balance the hydrogen?
the number of hydrogen halides (HX) can be deduced from the number of hydrogen atoms removed from the alkane → one HX will be formed for every H that is removed from the alkane
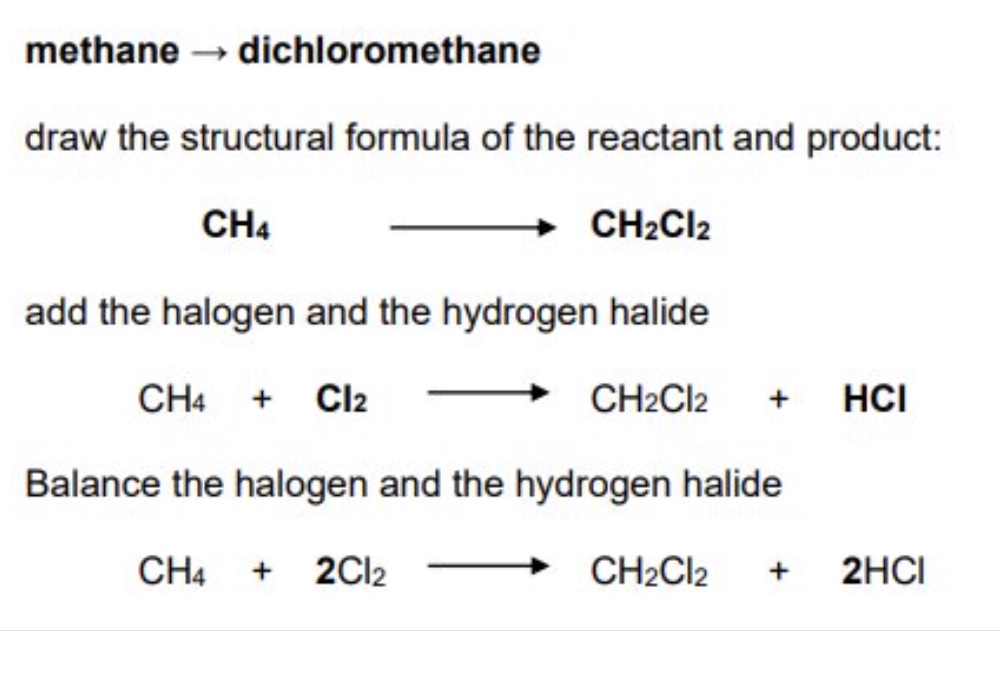
eg write the balanced symbol equation for the reaction between methane + chlorine to form dichloromethane
what is a free radical?
relative species due to the presence of an unpaired electron
what are the three stages free radical substitution mechanism consists of?
stage 1 → initiation
stage 2 → propagation (1st + 2nd)
stage 3 → termination
describe the initiation step + give the equation of this stage
this step creates the free radicals
ultra-violet light causes the covalent bond between the halogen atoms to break + creates 2 halogen radicals
→ the X-X bond in halogen molecules is weaker than the C-H bonds in alkanes + is therefore broken first)
X₂ → 2X•
define propagation
where a molecule + a radical create a molecule + radical
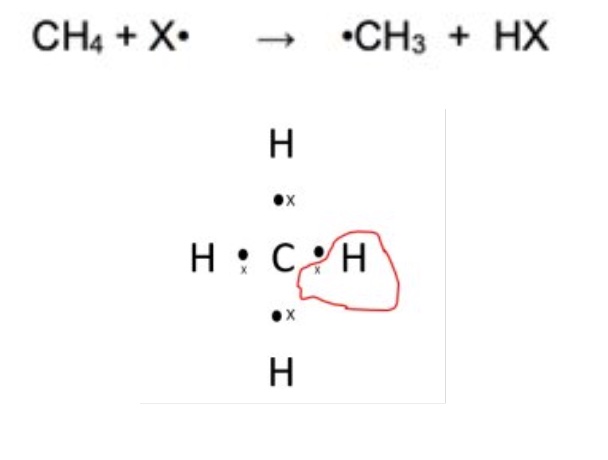
describe the 1st propagation + give the equation for this step
the halogen radical reacts with the alkane
the radical removes a hydrogen from the alkane creating a hydrogen halide molecule
the alkane is left as an alkyl radical
CH₄ + X• → •CH₃ + HX
describe the 2nd propagation + give the equation for this step
the alkyl radical reacts with a halogen molecule + creates a halogenoalkane
the remaining halogen atom from the molecule is left as a halogen radical
•CH₃ + X₂ → CH₃X + X•
what will the halogen radical created at the end of second propagation go on to do?
it goes on to react with another alkane molecule + 1st propagation is repeated → there will be a chain reaction of first + second propagations until you run out of halogen molecules or alkanes
define termination
when 2 radicals react to form a molecule
describe the termination step + give the equations for this step
if two radicals collide, they combine to make neutral molecules
the reaction is terminated as no further free radicals are generated
there are three possible termination reactions
two halogen radicals combine
a halogen radical combines with an alkyl radical
two alkyl radicals combine

in this process what is the role of the halogen radical?
it acts as a catalyst as its unchanged
what might happen if an excess of the halogen is used?
further substitution is possible in the propagation step, which leads to the formation of CH₂X₂, CHX₃ or CCl₄
what can the chance of further substitution be reduced by?
by using an excess of the alkanes