NEURO 1020: MEMBRANE POTENTIAL MT1
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Membrane Hypothesis
RMP
Julius Bernstein, 1902/
1. RESTING MEM POTENTIAL arises from high resting selective permeability to K+ and a concentration gradient for K+ ions across membrane
MAIN IDEA:
CHANGES IN K+ CNTRN SHOULD ALTER VM
Membrane Hypothesis debunking
Hodgkin and Huxley deproved
- giant squid axon
- varied K+ extracellular cntrn and measured Vm-> more depolarized than if just cell was j K+ permeable
Model Cell CRITERIA
1. ELECTRONEUTRAL:
number of - and + charges equal on both sides of mem
2. OSMOTICALLY BALANCED: same number of TOTAL ions on either side (no water movement)
3. No NET ION MOVEMENT-> Cell is at equilibrium
- makes it easy to change membrane potential
Relationship between K+ and Vm
high K+ OUT
low K+ OUT
high K+ OUT:
K+ and Vm has a linear relationship- high resting permeability to K+
- Vm is more positive
LOW K+ OUT:
K+ and Vm has EXPONENTIAL relationship (against membrane hypothesis)
- Na permeability has more weight in determining Vm (MORE DRIVING FORCE)
-Vm more negative
GHK permeability conclusions
if ions permeability is higher, Vm is closer to the equilibrium potential of ion
if two ions are equally permeable, Vm will be in the middle of the Eions
GHK relationship to nernst
GHK is weighted average of nernst potentials
- relative permeability changes weight
E(K) and E(Na)
-75 mV and +55 mV
GHK at 37 degrees Celsius
58 * log (pK[Kout] + .../ pK[Kin] + ... + pCl[Cl OUT]
Vm for Cl-
NEED TO INVERT OUT AND IN CNTRN IN GHK (in / out)
- charge is negative, z = -1
- P(Cl-) has very small effect on Vm because ECl is close to Vm already
- Cl- prevents other ions from taking over GHK, stabilizes
Na-K ATPase
exchanges 3Na OUT to 2K IN
- maintains concentration gradient
-contributes -5 mV to Vm, so INa (current) is 1.5x greater than IK
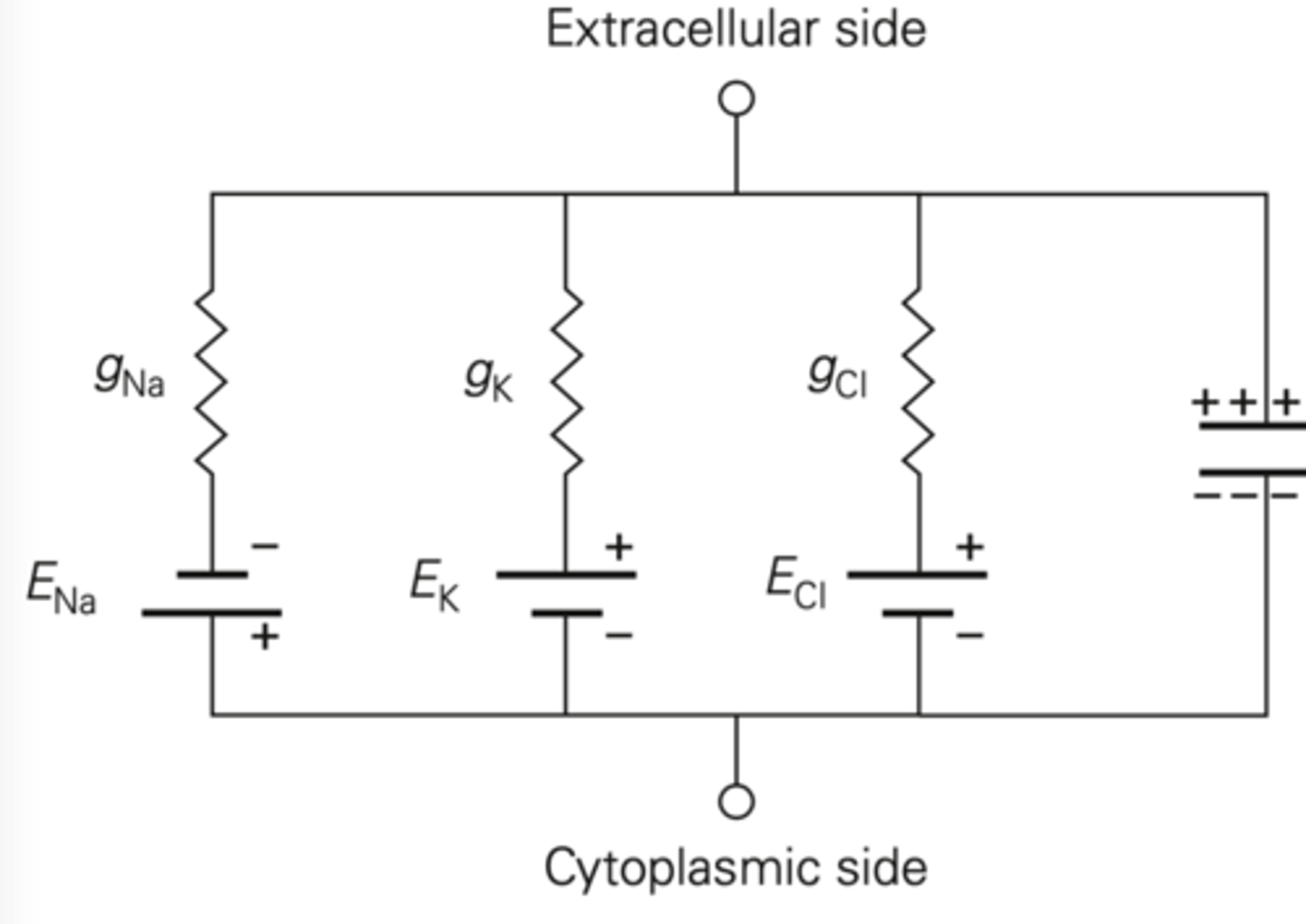
Membrane electrical circuit modeling
Resistor: ion channel
- K has high conductance, so small resistor
Battery: equilibrium potential determined by Na and K reversal potentials
Hyper or depolarize?
increase K IN
increase K OUT
increase NA IN
increase NA OUT
hyperpolarize
depolarize
hyperpolarize
depolarize
* THINK OF OUT/IN
RMP is determined by
selective membrane permeability to specific ions
- K+, Na+, Cl- are main ions, maintained by pumps
Leigh syndrome
neurodegenerative mitochondrial disorder
- membrane potential channel issues lower ATP/energy production
- hypotonia, deafness, weak immune, hypoglycemia
at rest, cell is most permeable to
K+
low K+ driving force
HIGH NA+ driving force
Vm = -75 mV
Membrane Hypothesis(bernstein)
Action potential
2. ACTION POTENTIAL produced by membrane becoming permeable to all ions (not just K+)
MAIN IDEA:
during an AP, membrane conductance should increase and Vm should = 0 mV
Mem Hypothesis AP debunking
Hodgkin and Huxley
squid axon
- at AP peak, transient reversal of membrane potential inside cell / OVERSHOOT
- due to Na+ -> decreases in extracellular Na+ decreases AP peak
- IF WAS JUST K+, THERE WOULD BE NO OVERSHOOT
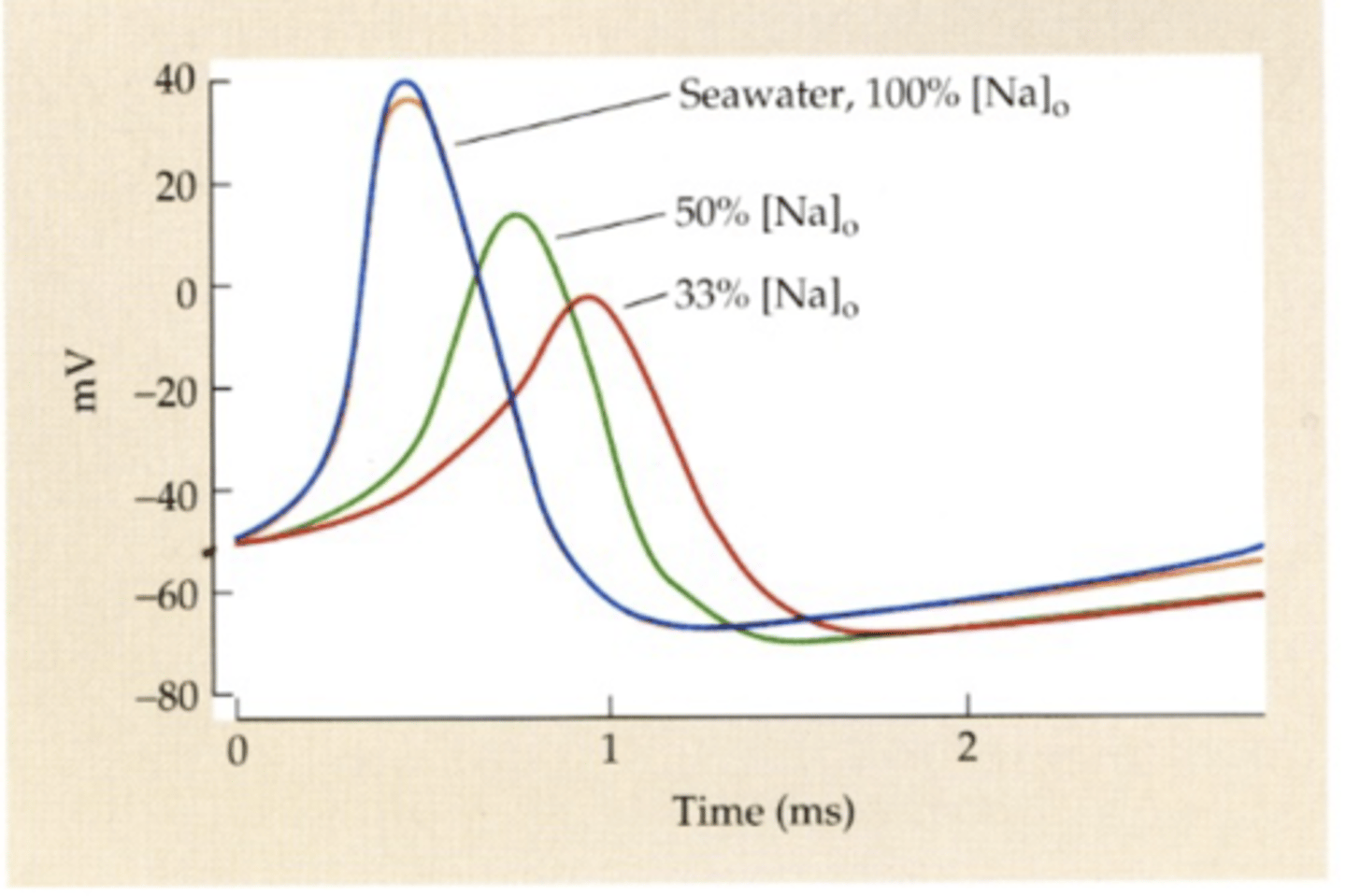
overshoot reasoning
- Na+ is necessary for nerves to produce aps
- Na+ reversal potential/ENA is positive -> high Na+ driving force
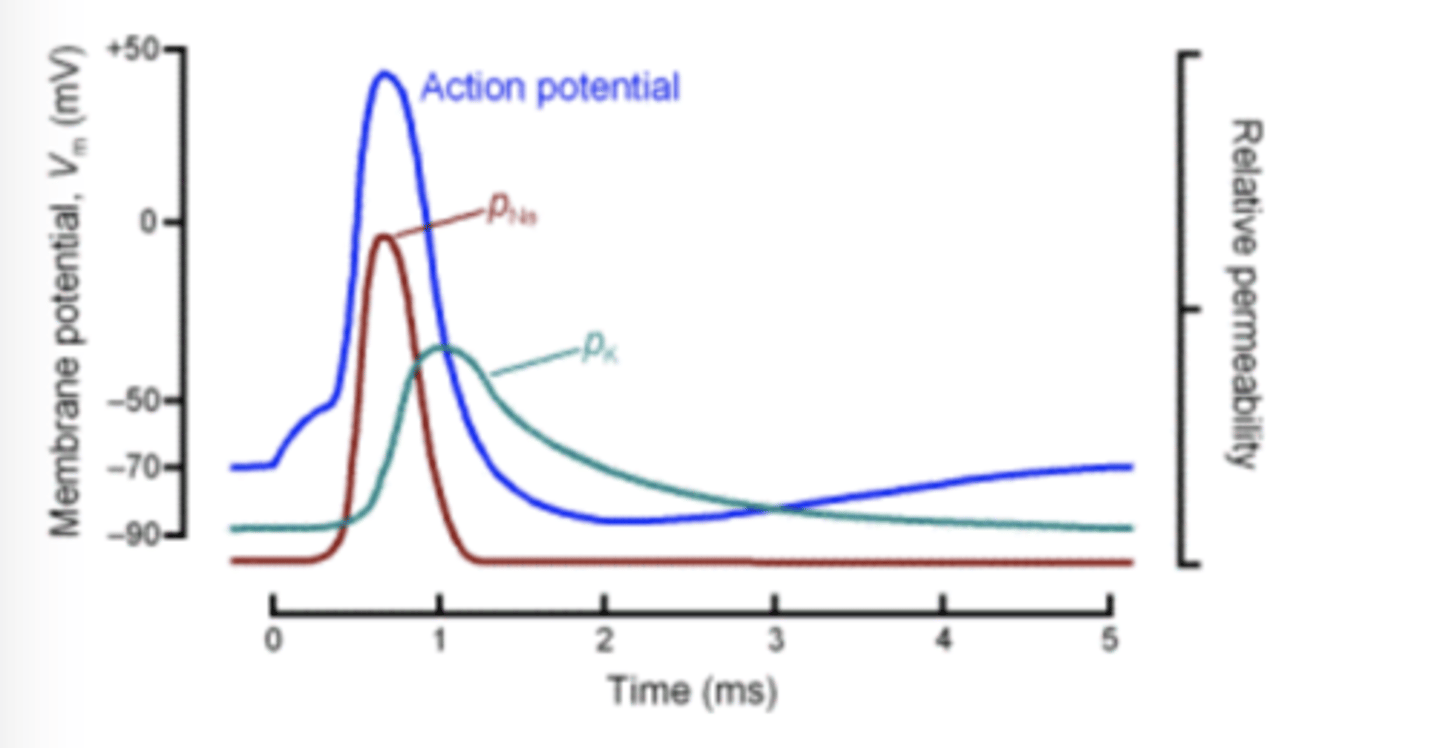
Ionic basis of AP
1. cell depolarized past threshold
2. Na conductance increase and Na+ influx depolarize
3. overshoot until Vm close to ENa, low driving force
4. Na channels inactivate, K+ conductance increase(VG K )
5. K+ rush out of cell (negative current), cell can hyperpolarize because long lasting increase in g(K) and EK is low
Voltage clamp
"clamp" membrane potential of a cell to measure current
- amplifier compares COMMAND POTENTIAL with measured membrane potential
- current is injected by amplifier until command potential = actual membrane potential/voltage difference
- CANCEL OUT ANY VG CHANNELS by injecting current equal and opposite in sign = no net ion flow
- measure current generated by Vg channels and analy ze
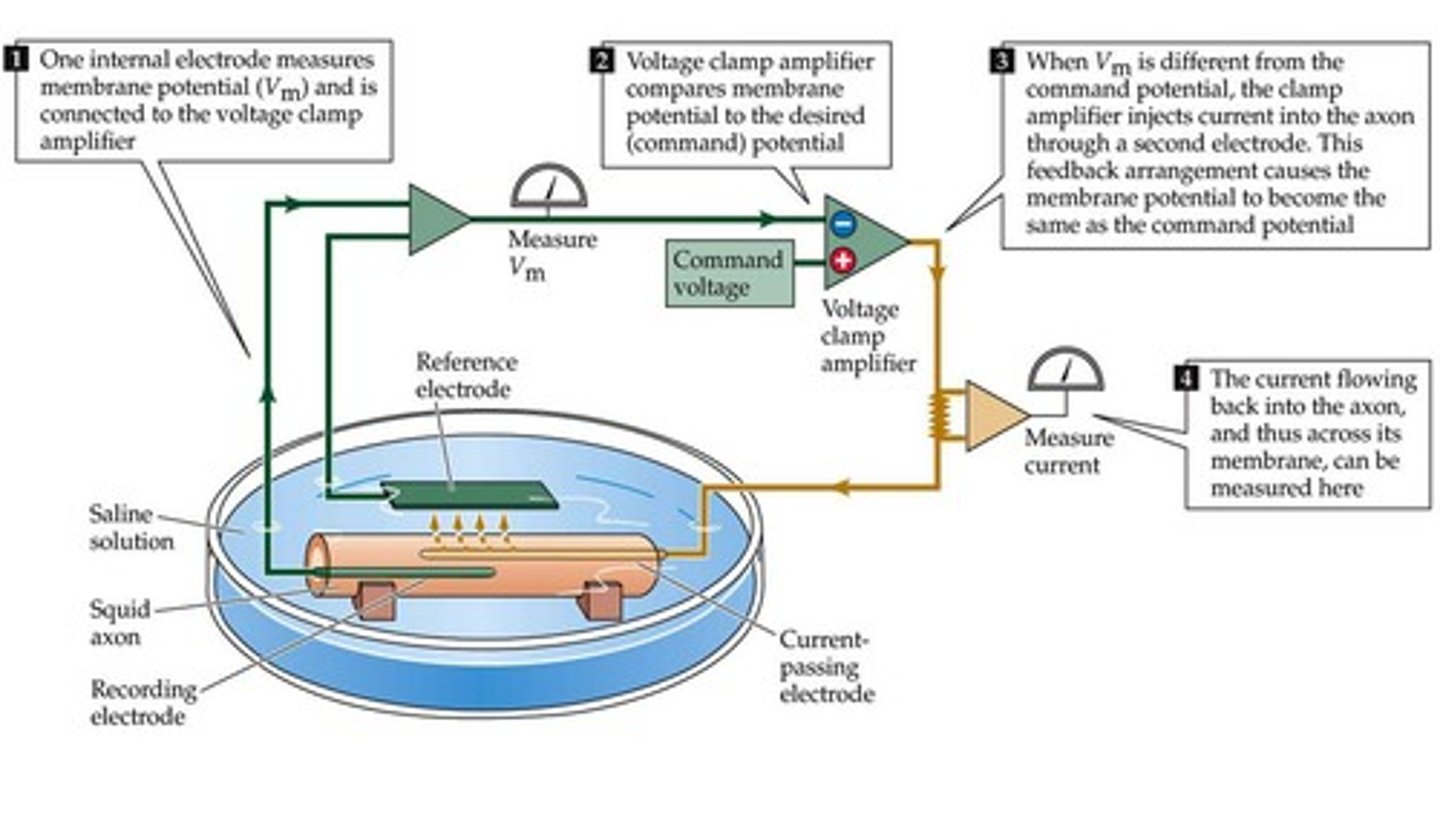
command potential
GOAL potential difference set by experimenter
membrane capacitance
ability of membrane to store charge
- why membrane potential doesn't change immediately with current injection
capacitative current
- a transient current that flows across a cell membrane in response to a voltage change
- can be seen in voltage clamp trace
action potential threshold
point at which net Na and net K currents are equal
- any depolarization after this point = ap
- specific channel composition determines- hillock has higher Na density channels than soma
Na channel blockers
TTX and STX
used to isolate and study individual channel behavior
K channel blockers
TEA
axon hillock ion channel density
highest in the cell
highest probability of ap in axon hillock
further from hillock, less probability of an ap
action potentials are result of
temporally coordinated changes in selective permeability of membrane
squid vs human axons
humans have more diverse channels
- diverse threshold, function, etc
- cells are all plastic- functions can change
channel diversity in humans
- type (ion, speed, threshold, delayed rectifier)
- combination in cell
-geometry of neruon
-alternative splicing
-plasticity
alternative splicing ion channels
- exons included can slightly change how same channel is expressed from cell to celll
- diff splice variants = diff sensitivity
inflammatory hyperalgesia
- depol at soma further down- much stronger
-isopotential (Vm is sam at every part of the cell)-> not true once signals fired
-inflammation increases distance of axon initial segment to soma, causes pain sensitization: inhibitory neuron AP threshold increases
more excitation than inhibition