anastasia bundle
1/123
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
124 Terms
Describe the changes a tumour cell goes through before metastasis.
- Mutation occurs to form cancer cells.
- Primary tumour then develops and survives on normal vascular system.
- Neoangiogenis of tumour occurs as it grows.
- Part of the tumour detaches allowing intravasation to occur.
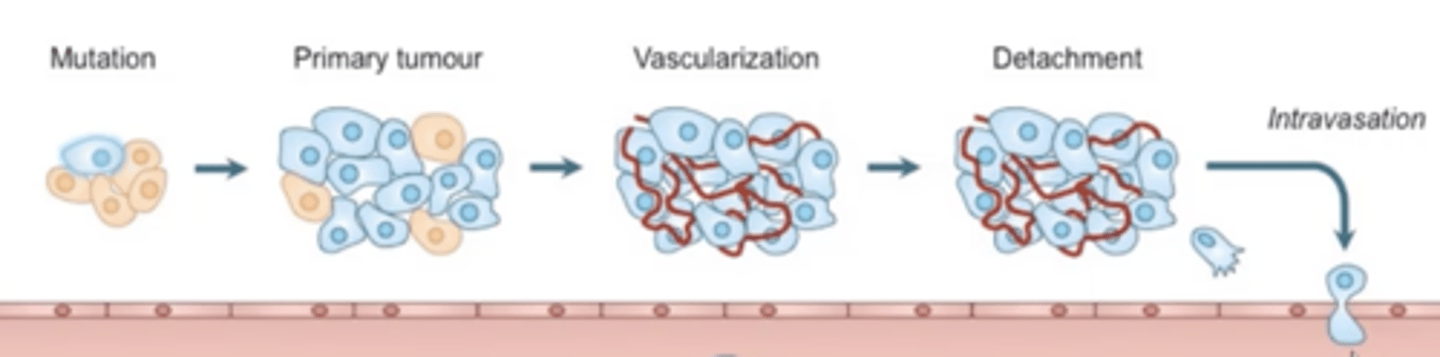
What is intravasation?
- Cancer cells enter circulation by passing through. endothelial cells that line blood vessels.
- Occurs through proteolytic degradation.
What is proteolytic degradation?
The breakdown of tissue using enzymes that allow cancer cell to enter circulation and metastasise (intravasation).
Describe proteolytic degradation.
- Macrophages secrete MMP which cleaves E-cadherin, causing the release of tumor cells.
- MMP also cleaves the extracellular matrix (ECM), facilitating tumor cell release.
- Changes in the ECM alter integrin expression on tumor cells.
- MMP also activates cytokines and growth factors in the tumor microenvironment.
What is E-cadherin?
A protein that binds cells together, including cancer cells.
How do changes in integrin expression affect tumour cells?
MMP cleaving the extracellular matrix also causes changes in integrin expression which means that integrins then bind to different extracellular matrix components.
What is epithelial to mesenchymal transition (EMT) and how does this benefit tumour cells?
- The changing of shape of a tumour cell as it is separated from other tumour cells.
- Happens after MMP cleaves E-cadherin during proteolytic degradation.
- Allows increased mobility for cancer cells so they can invade surrounding tissue and spread (metastasise).
How does intravasion occur?
- Growth factors and chemokines are secreted by macrophages to cause tumour cell migration.
- Tumour cells secrete factors that affect macrophage function in return.
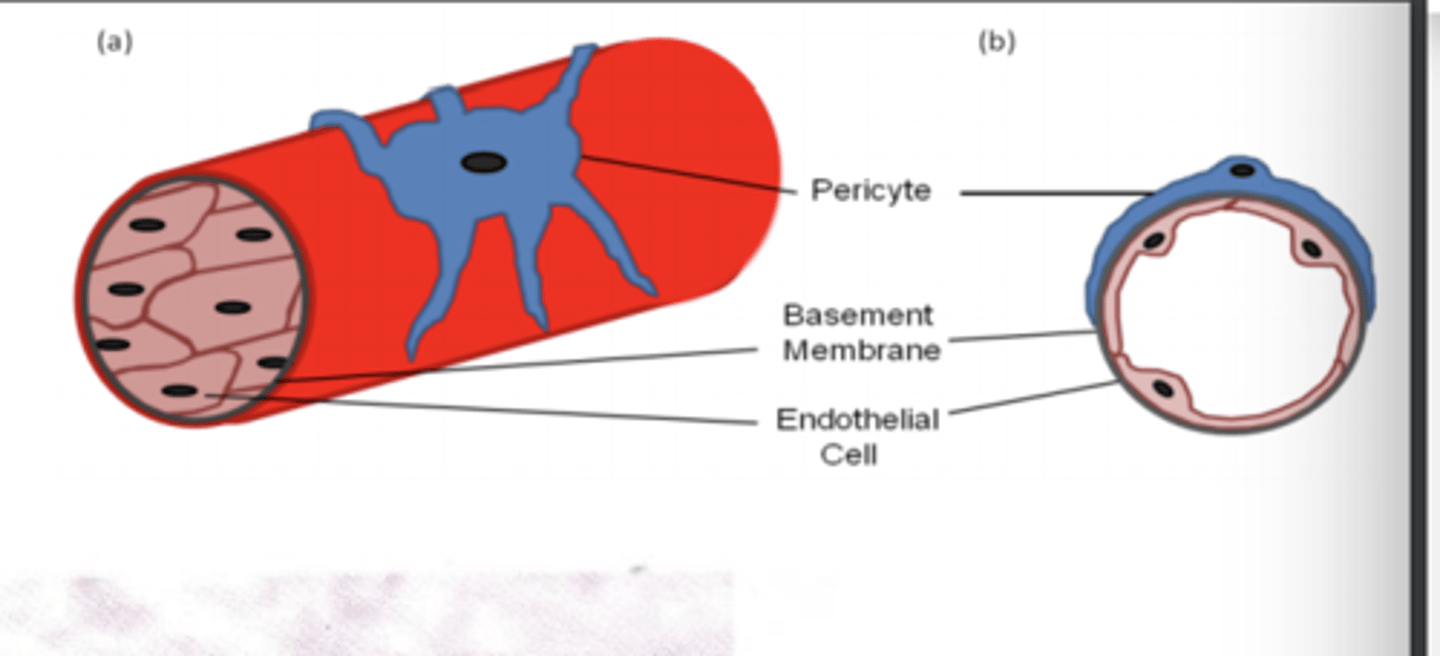
- Pericytes are gatekeepers of blood vessels and secrete CXCL12 which attach to CXCR4 on tumour cells and therefore allowing the cancer cell to migrate into blood vessels.
What are pericytes and how do they enhance tumour metastasis?
They are gatekeepers of blood vessels and
secrete CXCL12 which causes tumour cell migration through CXCR4R.
Describe generally what happens after intravasian has occured.
- Tumour cell travels through blood vessel and extravasian occurs.
- It then invades a group of cells and forms a secondary tumour.
- Continues to grow allowing vascularisation
What can happen to tumour cells as they travel through the blood?
- Undergo apoptosis or necrosis due to stress of blood flow.
- Killed by NK cells.
- Platelets can aggregate on surface preventing death from NK cells
What can happen to tumour cells after extravasian?
- Can die in tissue as they cannot grow in that specific environment.
- Can remain dormant as environment only allows them to live but not grow, happens next to a quiescent stromal cell.
- Can survive and reproduce, happens next to activated stromal cells.
How can secondary tumours form after extravasian?
- Environment must allow growth for cancer cells.
- Must be next to an activated stromal cell.
- After cancer cell divides (micrometastasis), it recruits bone marrow derived cells that become altered by tumour microenvironment which allows angiogenesis of secondary tumour and therefore macrometastasis.
How does metabolism of normal cells differ from cancer cells?
- Cancer cells obtain energy from alternate sources whilst normal cells don't.
- Allows normal cell to use metabolites for other processes.
What is the Warburg effect?
- Cancer cells will undergo anaerobic glycolysis even in the presence of oxygen.
- This metabolic adaptation causes increased glucose uptake and lactate production.
- This provides cancer cells with the energy required for rapid proliferation.
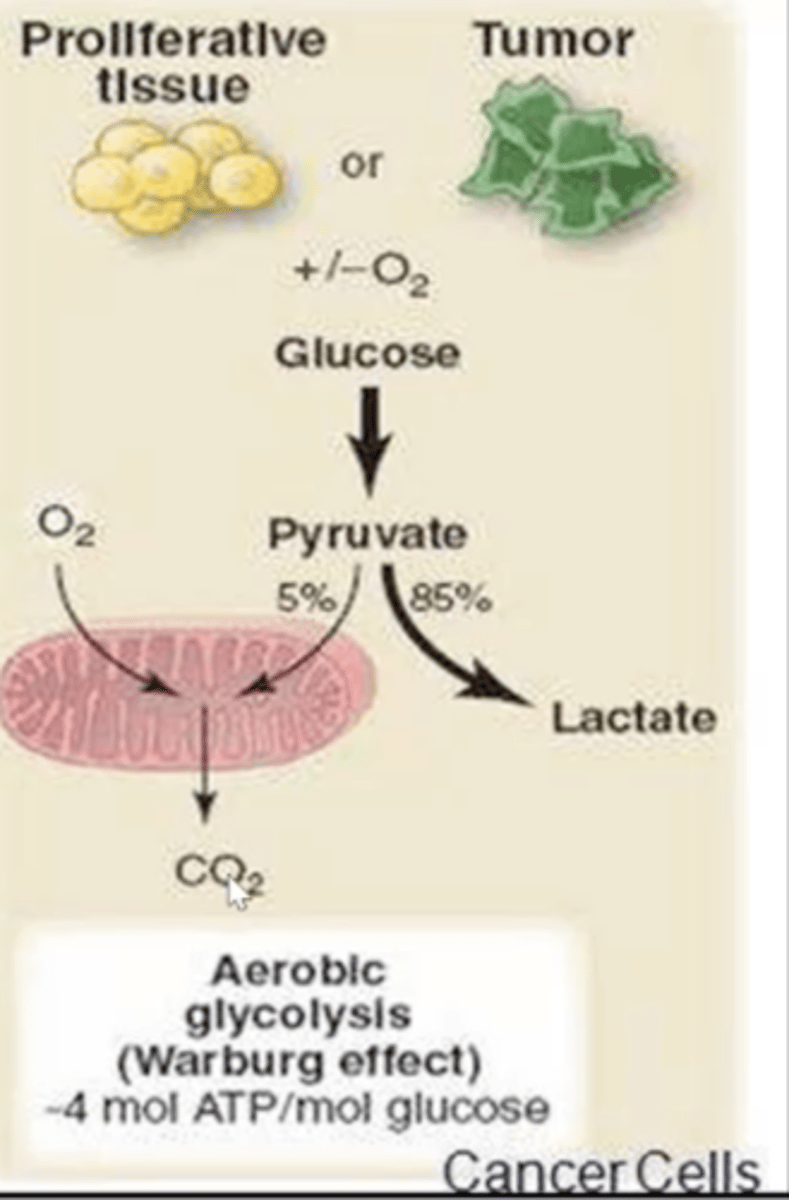
Why do cancer cells use the Warburg effect even though it less efficient than aerobic?
Generates metabolic intermediates for biosynthesis and maintaining a more acidic microenvironment for tumor progression.
How do NK cells kill cancer cells?
- Detect downregulation of MHC-1 in tumour cells.
- Can signal directly to T cells to activate CD8 cells.
- CD8 cells recognise peptides derived from cytoplasmic proteins that are bound to MHC1 to kill cancer cells.
How do CD8 T cells kill tumour cells?
Secretory and non-secretory method
What is the secretory mechanism of CD8+ T-cells to kill tumour cells?
They release perforin and Granzymes which pierce holes into the plasma membrane which kill the pathogens.
What is the non-secretory mechanism of CD8+ T-cells to kill tumour cells?
Once the T-cell Receptor has bound to the MHC-1 receptor the CD8+ cell also has a Fas ligand (FasL) which can bind to the Fas receptor on the pathogen which initiates apoptosis in the target cell.
What are tumour specific antigens?
An antigen that causes the tumour e.g., mutation of TSGs or oncogenes that lead to altered protein production.
What is a tumour antigen?
A protein with an abnormal structure caused by a mutation.
What is a tumour associated antigen?
Present on tumour cells and some normal cells and is a mutation in a gene unrelated to cancer formation but causes synthesis of abnormal proteins.
How do tumour cells stop T-cell killing?
- Cancer cells can stop the MHC antigen-processing complex and prevent loading of antigenic peptides onto MHC molecules, preventing the recognition of tumour antigens.
- Tumour cells can upregulate expression of PDL1 that can bind to PD1 on T-cells and deactivate them.
- Cancer cells can produce cytokines like IL-10 and VEGF that cause immunosuppression by recruiting T-regs and myeloid derived suppressor cells.
How can tumour cells take energy away from immune cells?
They compete for glucose with them.
How can cancer cells induce an immunosuppressive environment?
- Cancer cells have a lower pH environment due to their metabolism which can affect T-cell maturation.
- This also affects recruitment of MDSCs.
- This causes immunosuppression.
What are anti-tumour and pro-tumour cells?
Cells of the immune system can have both tumour antagonising and tumour-promoting effects.
Describe pro-tumour cells.
- Found in hypoxic environments.
- Release pro-angiogenic cytokines and enzymes.
- Produce reactive oxygen and nitrogen species which cause more mutations.
- Secrete a wide range of metalloproteinases which breakdown extracellular matrix which promotes metastasis.
Describe anti-tumour cells.
- Activated by LPS or IFN-γ.
- Produce cytokines and chemokines which stimulate immune system e.g., increase lymphocyte numbers.
- Cause tumour cell lysis.
- Secrete specific metalloproteinases that are required.
How do macrophages contribute to tumourigenesis?
- Release chemotactic factors that attract monocytes.
- Monocytes differentiate in macrophages.
- Tumour associated macrophages (TAMs) then work causing tumourigenesis.
How do tumour associated macrophages (TAMs) cause tumourigenesis?
- They release angiogenic factors e.g., VEGF and IL-8.
- Also release metalloproteases that digest the extracellular matrix.
- Also release mitogenic factors that promote tumour cell proliferation.
Describe tumour stem cells.
- Refer to any stem cell within a tumor e.g., normal stem cell.
- Control on number of stem cells that can be produced.
- Can divide and differentiate to generate all functional elements of a specific tissue.
What's the difference between normal stem cells and cancer stem cells?
Cancer stem cells can form tumours when transplanted into another animal but normal stem cells cannot.
Describe cancer stem cells.
- Arise from normal stem cells and progenitor cells.
- Require multiple mutations for this to occur.
- Can produce an unlimited amount of cells.
- Make up a small percentage of a tumour.
- Thought to be responsible for growth of other tumour cells.
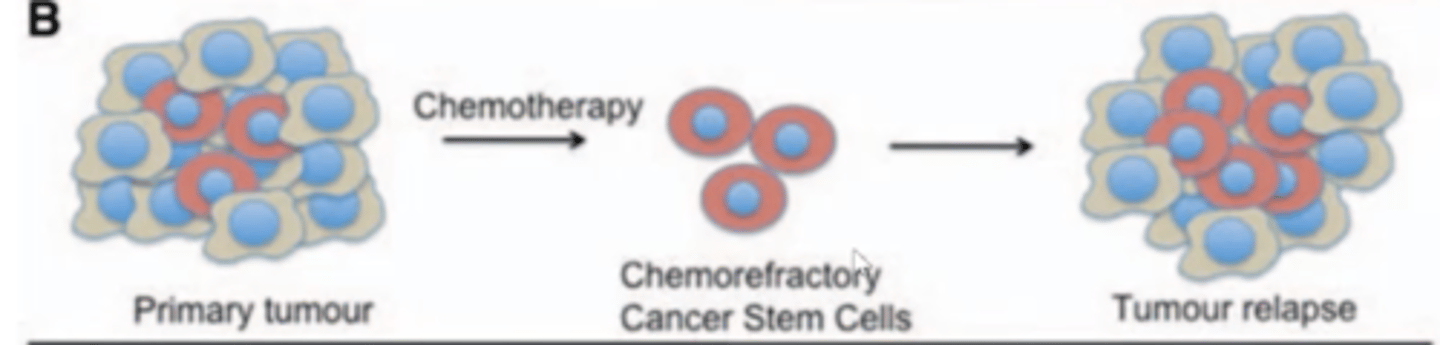
Why does chemotherapy not always work?
- Cancer stem cells are more resistant to chemotherapy.
- Chemotherapy may destroy normal tumour cells however cannot kill cancer stem cells.
- This allows a tumour relapse to occur.
How can cancer stem cells be targeted?
Through the use of:
- Enzyme specific markers e.g., CD44 and CD24.
- Enzymes e.g., ALDH.
- Transcription factors e.g., OCT4
- Drug efflux pumps
- Activated signalling pathways
What should be considered in a tumour microenvironment?
O2 conc
Describe the tumour microenvironment.
- Dynamic as can exhibit an oxygen gradient.
- Some regions can be well-oxygenated (normoxic) and others are poorly oxygenated (hypoxic).
- Hypoxic regions can make cells dormant and form layers in a tumour microenvironment.
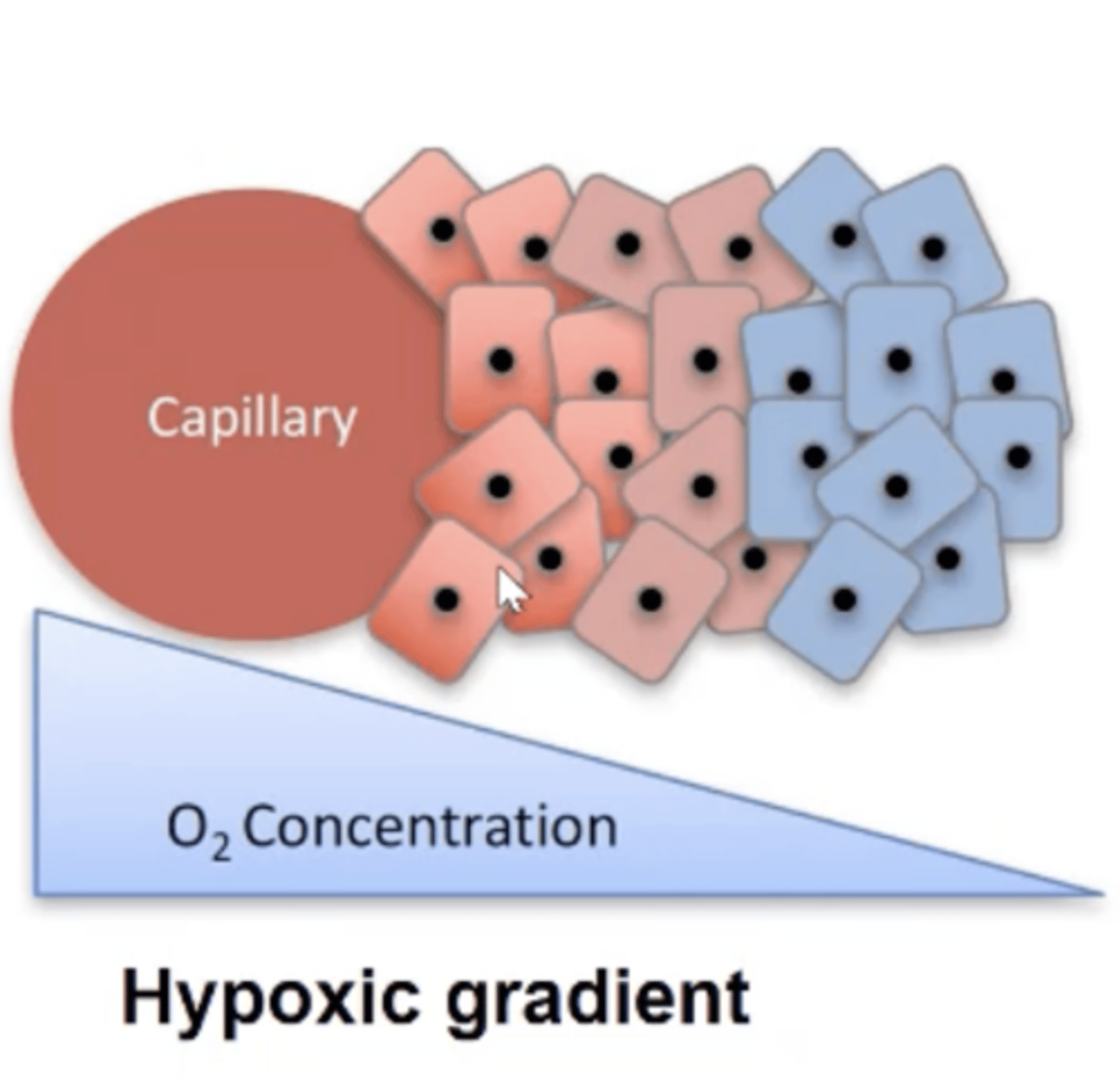
Describe normoxic (normal oxygen levels) cancer cells.
- Near a blood vessel.
- Low HIF-1 alpha expression.
- More susceptible to chemo and radiation therapy.
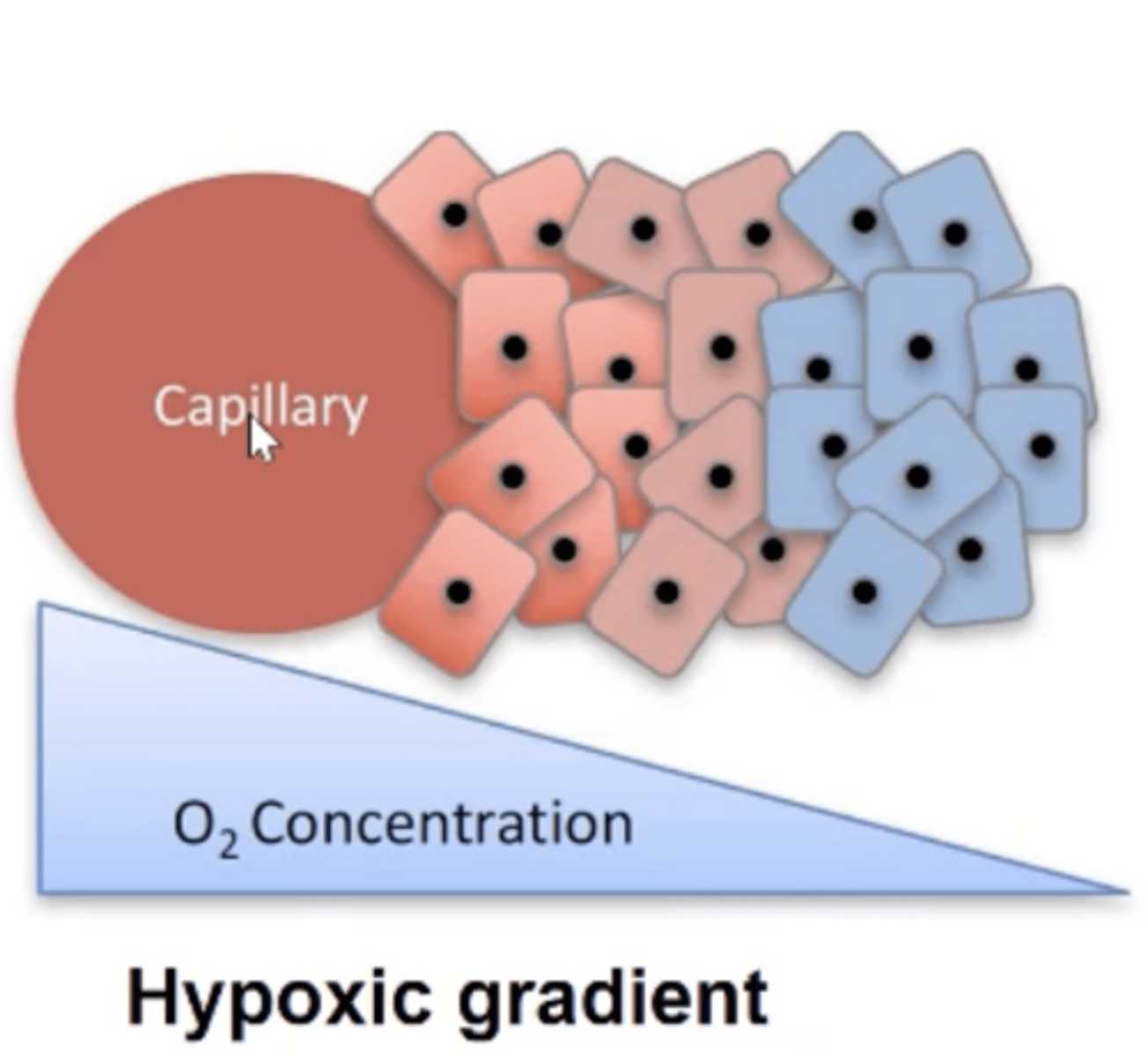
Describe hypoxic cancer cells.
- Increased HIF1-alpha expression which causes altered angiogenesis.
- Can be dormant due to lack of O2.
- Increased genetic instability.
- Poor immune response
- Less susceptible to chemo and radiation therapy.
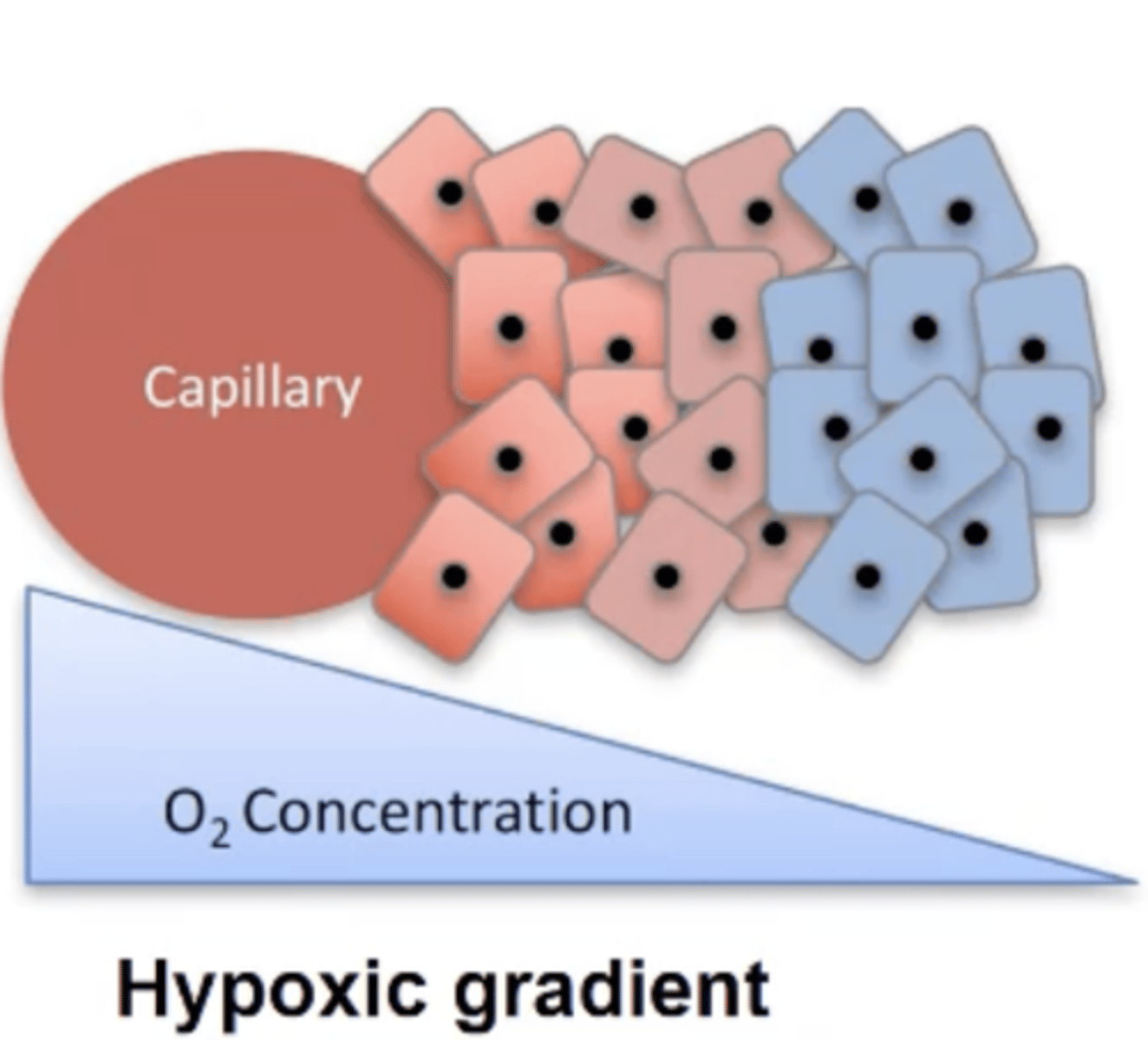
Why do hypoxic cancer cells have increased genetic instability?
- Lack of oxygen increases stress on cancer cells causing errors in DNA replication therefore causing mutations.
- Hypoxic cancer cells can also release reactive oxygen and nitrogen species which cause more mutations.
Describe the structure of a tumour in a microenvironment.
- Normal growth happens closes to blood vessel as more access to oxygen.
- The further away from blood vessel, the less oxygen for cells.
- This explains why necrotic regions are furthest away from blood vessel, then hypoxic.
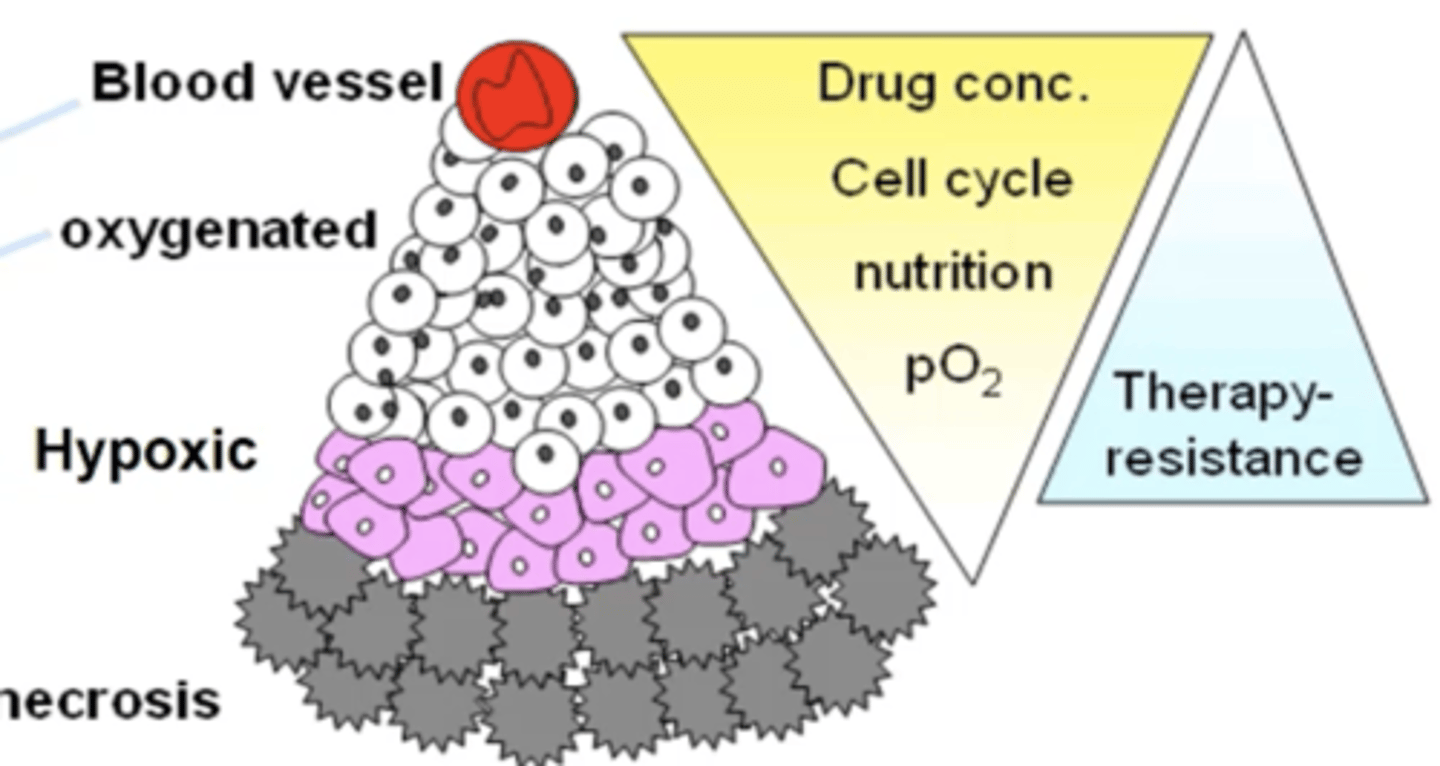
How is necrosis caused?
- A lack of oxygen delivery to cells.
- Impaired waste removal.
How can necrosis affect other cells?
- Causes spilling of cellular contents.
- This causes inflammation and injury to nearby cells.
- Produces an inflammatory response.
What is neoangiogenesis?
Formation of new blood vessels in order for cancer cells to collect enough O2 and remove waste.
What is the angiogenic switch?
- When tumours continue to grow, new blood vessels need to form in order for the tumour to get enough oxygen and remove waste.
- Allows rapid growth of tumour and facilitates metastasis.
- Does not happen when tumour is small.
Switch happens once tumour becomes too big to rely solely on normal blood vessels.
Describe the main difference between normal blood vessel organisation and tumour organisation.
Tumour vascular system is highly disorganised due to pro and anti angiogenic signalling.
Describe the stages of inducing angiogensis.
- Endothelial cells secrete MMP which digest the extracellular matrix.
- Tumours then release proangiogenic factors e.g., HIF1-α. which allows endothelial cells to form new blood vessels towards the tumour cells.
- This supports tumour growth and metastasis.
What is the difference between blood vessel organisation in normal cells vs tumour cells?
- Tumour vasculature is disorganised and irregular, the opposite in normal cells.
- Therefore it is less efficient in oxygen and nutrient supply.
Why do tumour cells produce a disorganised vascular network?
Due to the imbalance of pro- and anti-angiogenic factors within different parts of the tumour.
How can the disorganised vascular network affect anti-cancer drugs?
Abnormal blood vessels in tumors cause uneven blood flow, leading to irregular distribution of drugs within the tumor.
What are tumour suppressor genes (TSGs)?
Genes that regulate mitosis and prevent cells dividing too quickly.
What are the three main TSGs?
- Cyclin/Cyclin-dependent kinases/Cyclin dependent kinase inhibitors.
- Retinoblastoma protein
- p53
How do cyclins control the cell cycle?
- They bind to CDKs which then become active and phosphorylate target proteins in the cell.
- This triggers proteins to become active and carry out task specific to that phase of the cell cycle.
How do retinoblastomas control the cell cycle?
- Prevent cell cycle leaving the G1 phase.
- Inhibits the genes necessary for progression into S phase.
- Phosphorylation of retinoblastoma deactivates it allowing cell cycle to continue.
Describe how mutated retinoblastoma causes cancer.
- Activated G1-CDK phosphorylates and inactivates Rb.
- Phosphorylated Rb releases E2F transcription factor which activates transcription of genes that encode proteins required for S phase like S-cyclins.
- Cell enters S phase and continues to proliferate.
How can retinoblastoma act as a tumour suppressor gene?
- RB can bind to E2F to inhibit it.
- This suppresses expression of genes necessary for DNA replication.
- This stops the cell cycle preventing the cell cycle continuing.
- Therefore mutant RB can cause cancer.
How can mutant Rb cause cancer even though it is a recessive gene?
- People can inherit one mutated gene and the second 'normal' gene can later mutate.
- This provides two mutant RB genes which can then cause cancer.
What are sporadic cancers?
- Cancer with no known cause and is by chance.
- No relatives with a similar cancer.
What are familial cancers?
- Cancer caused by a combination of genetic and environmental factors.
- Relatives showing the same type of cancer.
- No specific pattern of inheritance e.g., not passed from parent to child.
What are hereditary cancers?
- An altered gene is passed down from parent to child.
- People with hereditary cancers are more likely to have relatives with the same type of cancer.
- May develop more than one cancer and at an earlier age.
Describe p53.
- Acts as a brake for the cell cycle if DNA damage is detected.
- Can increase the expression of genes involved in DNA repair pathways.
- Can induce apoptosis of cells is damage is too great.
How is p53 activated?
- DNA damage
- Cell cycle abnormalities
- Low oxygen (hypoxia)
Describe the action of p53 when there is no DNA damage.
- MDM2 (E3 ubiquitin ligase) can bind to p53 and tag it with ubiquitin.
- This allows the proteasome to break down p53.
- This allows cell cycle to continue as p53 no longer present to keep cell cycle in G1.
Describe the action of p53 when there is DNA damage.
- p53 is phosphorylated activating it.
- Activated p53 acts as a transcription factor to increase production of p21.
- p21 is a CDK inhibitor which prevents CDK's binding to cyclins inhibiting their activity.
- This prevents the cell leaving the G1 phase and allows the DNA to be repaired.
How does p53 induce apoptosis?
- It initially inhibits BCL-2 which is a pro-survival protein.
- It then activates BAX causing mitochondrial outer membrane permeabilisation (MOMP) and release of cytochrome C into the cytosol.
- Cytochrome C triggers caspase-3 activation through formation of the cytochrome c/Apaf-1/caspase-9 containing apoptosome complex that dismantles the cell.
How do cells control their growth?
- Through the cell cycle
- Growth factor receptor signalling.
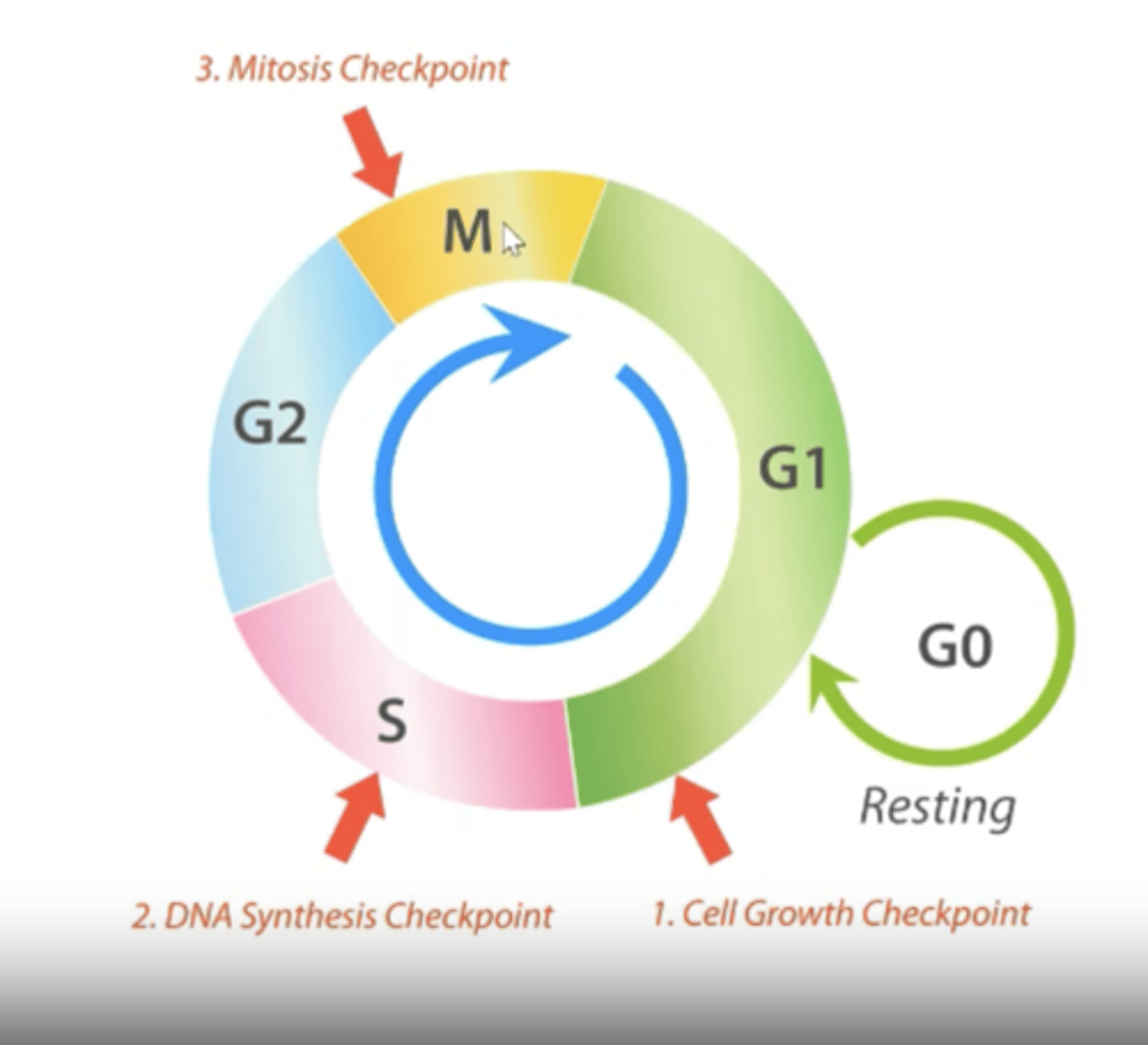
What are the stages of the cell cycle?
G1 (+G0), S, G2, M
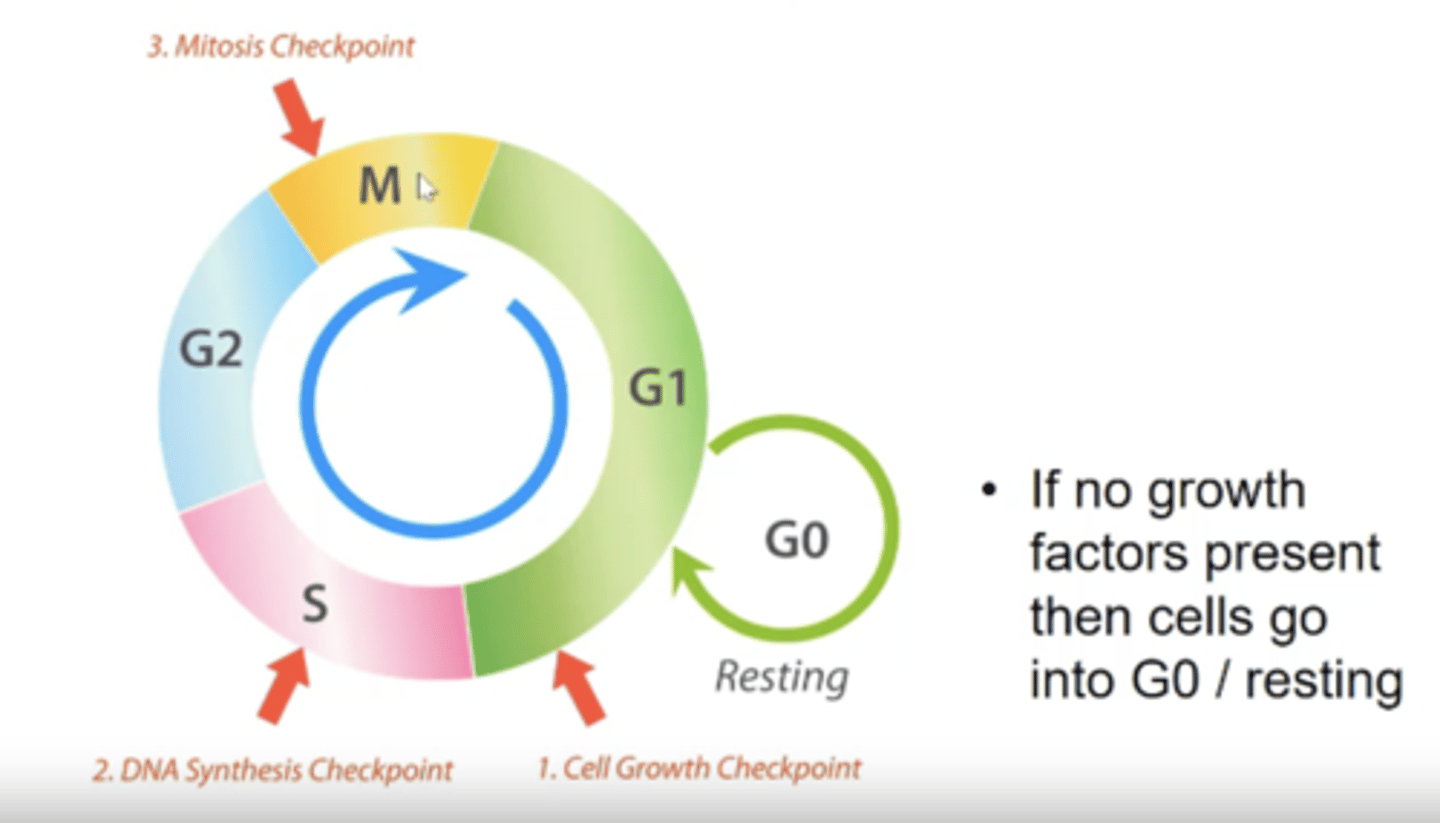
Describe each stage of cell cycle.
- Cell remains in G0/resting stage until a growth factor is present and then enters G1.
- G1 is first and the cell grows in size.
- Then moves to S phase where DNA is synthesised.
- Then moves in G2 phase where it undergoes more growth.
- Then moves into M phase where mitosis takes place.
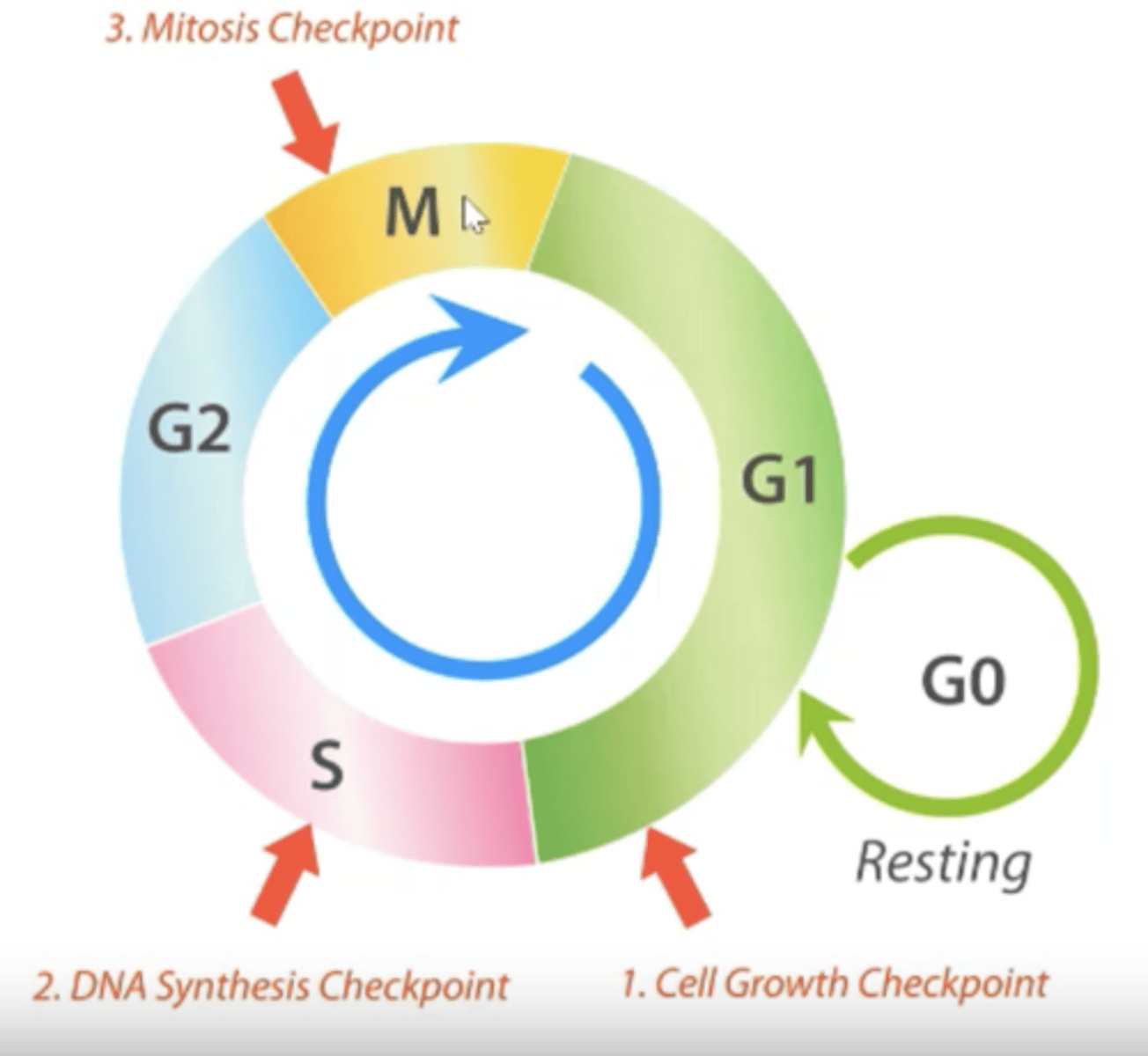
What are examples of growth factors and what receptors do they work on?
- EGF works on EGFR.
- HGF works on HGFR.
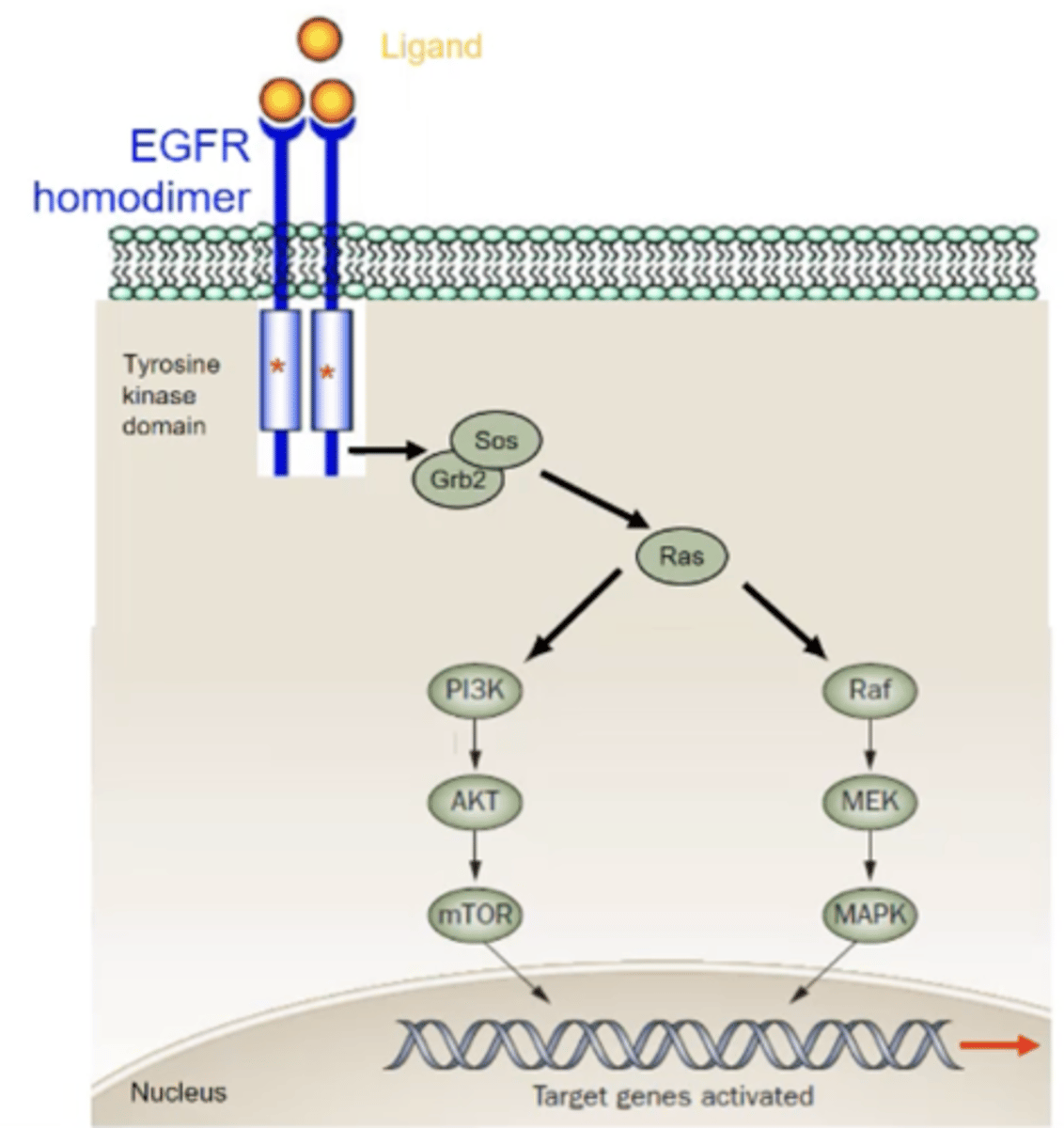
Describe EGFR signalling.
- EGF binds to EGFR which causes a homodimer to form with another EGFR receptor.
- The homodimers then phosphorylate each other in the intracellular tyrosine kinase domain .
- Adapter molecules e.g., SOS and SHC, activate RAS which then activates Raf, MEK and MAPK
- This causes target genes to activate and therefore cell proliferation
What is receptor endocytosis?
Receptor endocytosis is when cell surface receptors, along with their bound ligands, are brought inside the cell through the cell membrane, helping regulate signaling and activating target genes.
What are proto-oncogenes?
Genes that regulate normal cell growth e.g., RAS.
Cells remain in the G0 phase until proto-oncogene is present.
Describe the RAS proto-oncogene
- A small G-protein involved in GTPase reaction cycles.
- Relays a growth signal from a growth factor receptor e.g., EGFR, on the cell membrane to a cascade of tyrosine kinases e.g., RAF, MEK and MAPK.
- This causes target genes to activate and therefore cell proliferation.
What is an oncogene?
A mutated proto-oncogene that causes a cell to divide uncontrollably.
What happens if the Ras gene is mutated and becomes an oncogene?
- The Ras protein will activate without the growth factor present.
- This will activate target genes leading to over expression of protein.
- Cell will undergo constitutive signalling.
What is constitutive signalling?
- Cell will never enter G0 phase and will continuously go through cell cycle.
-
What mutations can happen to a proto-oncogene?
- Gene amplification
- Gene rearrangements
- Large structural deletions
- Subtle mutations
How does gene amplification produce an oncogene?
Produces too many copies of a gene and therefore causes cell to keep proliferating.
e.g., too much growth factor production causes cell to keep reproducing.
How does gene rearrangement produce an oncogene?
The promoter gene can be in the wrong place so a gene that is expressed weakly can be expressed at high levels which can then stimulate the cell cycle over and over again.
How does large structural deletions produce an oncogene?
Deletions in receptor sequences can cause cell to proliferate.
How do subtle mutations produce an oncogene?
A single nucleotide change can cause gene to remain active at all times which causes cell to keep replicating.
e.g., if Ras is mutated, cell will continue to replicate without needing a growth factor.
What is gene translocation?
Where a segment of one chromosome breaks off and becomes attached to another chromosome.
What are fusion proteins?
Caused by translocation which are special proteins that result in potent transcription activation e.g., BCR-ABL
How are fusion proteins formed?
- Chromosomes break at specific regions known as break-point cluster regions (BCRs).
- During translocation, genes may lose their regulatory domains.
- This loss of regulatory domain enables the fusion of the BCR with the gene, resulting in the formation of a fusion protein.
- This can contribute to the development of tumors due to the absence of regulatory domains.
Describe the BCR-ABL oncogene.
- Produced when parts of the chromosome 9 and 22 switch places.
- Promotes uncontrolled cell growth.
- Target for tyrosine-kinase inhibitors
How does BCR-ABL oncogene cause cancer?
- It can activate signalling pathways from GRB2 to PIP2 and PIP3.
- This can then activate PDK1 and AKT which can then influence cell growth and p53 action.
- BCR-ABL can also produce IL3 and G-CSF allowing it to activate JAK and STAT5 pathways.
- These all lead to cell proliferation.
How does understanding mutations of proto-oncogenes help develop treatment for cancer?
- Helps identify new targets for cancer treatment.
- Can target oncogene rather than DNA of cancer.
- For example, the HER2 gene is amplified in 25% of BRCA patients and therefore drugs (trastuzumab) can target HER2 and prevent overexpression to prevent tumour developing.
What is cancer?
Abnormal growth of cells in an uncontrolled way that can spread or metastasise into other tissues.
What are the types of tumours?
Benign and malignant.
Describe benign tumours.
Abnormal growths that are no longer under normal regulation.
Describe malignant tumours.
- Poorly differentiated cells growing rapidly in a disorganised manner.
- Can invade surrounding tissues and become metastatic which cause the growth of similar tumours in other organs.
How can cancers be classified and what are the main classes?
Based on cell origin:
- Carcinomas
- Sarcomas
- Lymphomas
- Leukaemias
Describe carcinomas.
- Most common type (85%).
- Arise from cells that cover external and internal body surfaces.
Describe sarcomas.
- Around 12% of cancers are sarcomas.
- Originate from cells found in the supporting tissues of the body e.g., bone, muscle and fat.
- Highly malignant
Describe lymphomas.
- Cancers which originate from lymph nodes and tissues of the body's immune system.
- Can spread to intestine, spinal cord, bone and brain.
Describe leukaemias.
Cancers of immature white blood cells that proliferate in the bone marrow and accumulate in the bloodstream.
What is staging of cancer?
It determines the exact location of the cancer and its degree of metastasis at diagnosis.
What is staging of cancer based on?
- The site of primary tumour.
- The size of tumour.
- How far it has invaded into local tissues and structures.
- Whether it has spread to regional lymph nodes.
- Whether it has metastasized to other regions of the body.
What is cancer grading?
- Graded 1 to 4 to show how advanced the tumour is (1 is the least advanced).
- Based on number of cell abnormalities, the more abnormalities the more advanced the tumour.