Geochemistry Exam 2
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
Isotopes
an element with a different number of neutrons than expected; varying atomic mass, isotopes always have more neutrons than protons
Relative Abundance
comparison of the light and heavy stable isotopes in a sample to a known standard
light isotope
less neutrons
heavy isotope
more neutrons
VSMOW
vienna standard mean ocean water; pure water with no salt or other chemicals in it, used as a standard comparison for hydrogen and oxygen isotopes
PDB
Pee Dee Belenmite; geologic formation in North/South Carolina consisting of marine deposit fossils of belenmitella americana, used as a standard comparison for carbon isotopes
Fractionation
heavy/light, rare/abundant, = R, always comparing to fixed standard (VSMOW + PDB)
Depleted
lighter, has more of the lighter isotope than standard
Enriched
heavier, has more of the heavy isotope than standard
Change per mil
fractionization; (Rsample/Rstandard - 1) x 1000
Causes of Fractionation
covalent bonds, ionic bonds, oxidation states, crystallization, temperature
How do covalent bonds affect fractionation?
heavier bonds form more stable bonds and lighter isotope’s bonds are easier to break; more apparent at low temps
How do ionic bonds affect fractionation?
minimal effect
How do oxidation states affect fractionation?
heavier isotopes prefer to bond to higher oxidation state elements
How does crystallization affect fractionation
heavier isotopes form a solid first, leaving liquid behind that is depleted (more light isotopes)
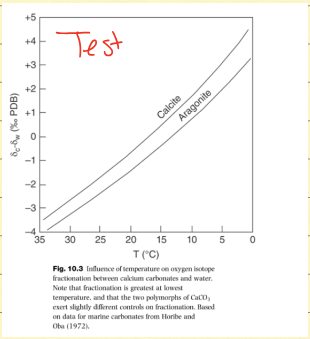
How does temperature affect fractionation?
For calcite, as temps decrease more calcite/aragonite precipitates. Heavier isotopes precipitate out first, leaving behind depleted solution so as temps decrease, relative abundance increases
Kinetic fractionation
when an isotope reacts, diffuses, or evaporates faster than another due to the process and/or catalyst; results in lighter isotope accumulating in the product
What reacts faster?
lighter molecules
What carbon do plants prefer?
prefer lighter 12C because it reacts faster. CO2 is converted to lighter C in C6H12O2 (sugar) during photosynthesis
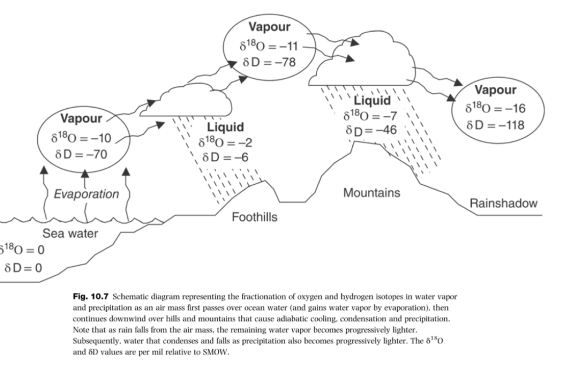
Rayleigh Fractionation
global scale evaporation, condensation, precipitation; isotopes fractionate when material phase changes
Uses for isotopes
can help us date things, see how climate is changing, see what an organism’s diet was like, find out origin of an organism
Daughter isotopes
called radiogenic because they form from radioactive decay. what the original isotope breaks down into
Alpha radiation radioactive decay
lose a particle with 2 protons, 2 neutrons, and a +2 charge. U→Th + He (a) + j + E

Beta radioactive decay
lose one electron in a series. happens in series with alpha decay. Th→Pa + -ie-(B) + j + v

Positron radioactive decay
emission of a positively charged electron (formed when a proton becomes neutral + releases a positive particle) Al→ MG + j + v

Gamma radiation
released in alpha, beta, and positron decay. stabilizes the nucleus to a lower energy/more stable state
Fission radioactive decay
nucleus splits into 2 or more nuclei. produces an alpha particle
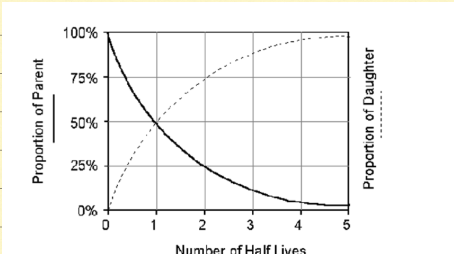
Decay curve
exponential growth of daughter isotope, exponential decay of parent isotope
half-life
amount of time it takes for a substance to decay to half its original mass. Pt = Po x e^-kt. t1/2 = -0.69/y

Zero order rate
linear decay of substance. constant rate, typically seen in dissolution of some salts. y=mx+b
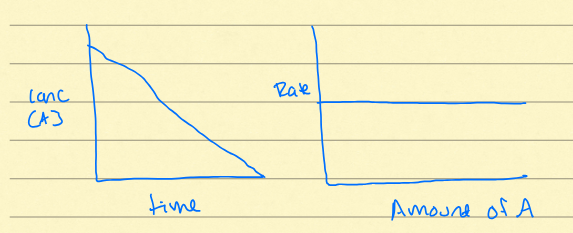
First Order Rate
exponential decay of isotope, rate increases over time (linear). typically seen in population growth, radioactive decay, hillslope. Pt = Po x e^-kt
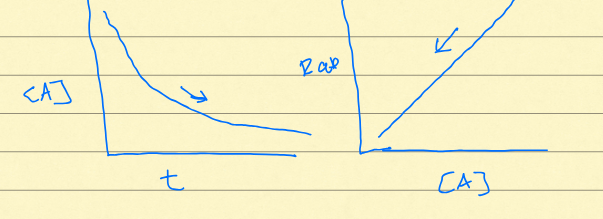
Daughter isotope accumulation
D = Po(e^yt - 1)

Isotopes and their uses
Carbon 14 = 100-30,000 years (clothing, more modern stuff)
Potassium 40 = 100,000-4.5 billion (rocks)
Rubidium 87 = 10 million - 4.5 billion (rocks)
Uranium 238/235 = 10 million - 4.6 billion (rocks)

Ka
dissociation constant or equilibrium constant of each H+ dissociation reaction. Ka = [A] [H+] / [HA]
![<p>dissociation constant or equilibrium constant of each H+ dissociation reaction. Ka = [A] [H+] / [HA]</p>](https://knowt-user-attachments.s3.amazonaws.com/bb395ae3-934a-47e6-88d8-0720b6c8ff57.png)
pKa
pKa = -log (Ka). quantitative measure of the strength of an acid
strong vs weak acid
strong acids have smaller pKa/Ka while weak acids have larger ones
why is the pH of water 1-14?
Because kw= [H+] [OH-] = 1×10^-14 or the concentration of each is 10^-7. when [H+] = [OH-] then pH is 7
![<p>Because kw= [H+] [OH-] = 1×10^-14 or the concentration of each is 10^-7. when [H+] = [OH-] then pH is 7</p>](https://knowt-user-attachments.s3.amazonaws.com/9ecb09bd-67f6-4479-8eaf-51b7f28840bf.png)
why are some polyprotic acids (multiple hydrogens) weak?
they don’t completely dissociate immediately, well-buffered
dissociation of strong vs weak acid
strong acids completely dissociate while weak ones don’t
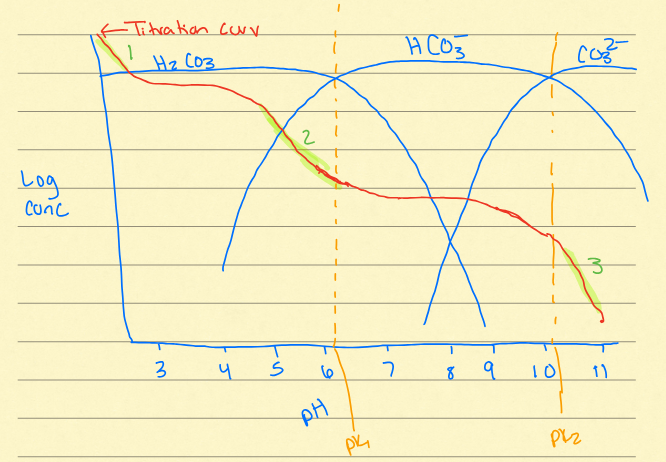
Dissociation of polyprotic acids
at different pHs, different species of the acids are dominant in solution. Bjerrum plot
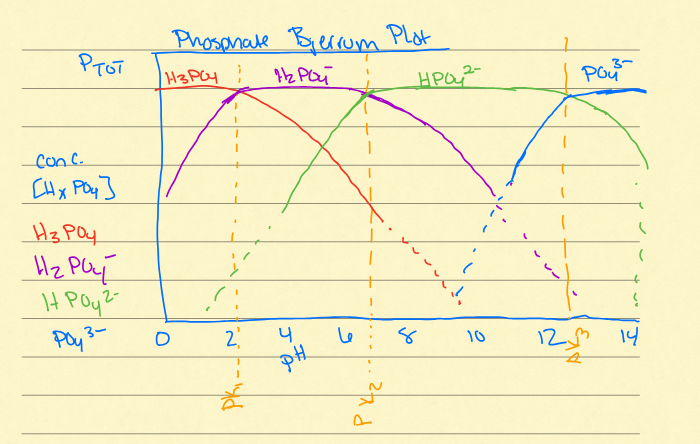
what is H2CO3*
it encompasses all of the reactions that determine how much CO2 is gaseous and how much is aqueous
Alkalinity definitions
the potential of a liquid to neutralize acid
ability of a solution to accept acidity and not change pH
amount of acid needed to convert anions in solution into uncharged species
Amount of acid needed to titrate a solution to the pH of the CO2 equivalence point
Alkalinity lab definition
defined by titration, a process that adds acid slowly to a solution while pH is measured after each addition, discussed in meq
Calculating alkalinity
put data into USGS calculate and get alkalinity in mg/L HCO3 -
In karst waters, alkalinity roughly equals [HCO3-] so amount of acid = alkalinity
![<p>put data into USGS calculate and get alkalinity in mg/L HCO3 - </p><p>In karst waters, alkalinity roughly equals [HCO3-] so amount of acid = alkalinity</p>](https://knowt-user-attachments.s3.amazonaws.com/064c2e1c-e8d7-438a-9e38-bd3198e703b8.png)
buffer capacity
measure of buffering in a solution (resistance to change)
buffer zones on bjerrum
right before H2CO3
second downward curve/hump
final downward curve/hump
How do we get stuff in water?
chemical reactions add stuff to H2O. can be from mineral dissolution+precipitation, mineral weathering, and reactions within solution between species or solids
Big Picture Controls on Water Chemistry
water cycle
dissolved vs particulate ratio
mineral solubility and reaction types
water measurements/parameters (ionic strength, likeliness to react, etc)
stability of species in water
TDS - Total Dissolved Solids
measure of all dissolved components in water (organics, inorganics, ions, colloids, etc)
NAPLS - non acqueous phase liquids
things in water that aren’t dissolved
DNAPLS
dense non acqueous phase liquids, sink to the bottom of the H2O column (diesel fuel)
LNAPLS
light non acqueous phase liquids, float on top of water, common organic contaminants (gasoline)
colloids
small solids that are in water but not dissolved, have surface chemistry reactions
sizes of undissolved stuff in water
nanostructures (atoms, ions, proteins, viruses) → colloids (atmospheric dust, spore, soot, latex paint) → particulates (pollen, fog, sand, bacteria, red blood cells)
Ionic Potential
z/r; ration of electric charge to radius of the ion, tells us how strongly the ion will be attracted to ions of opposite charge or repelled by ions of the same charge.
high z/r
associated with strong bonding, high charge, small radius. strong bonds with oxygen and repels other cations (PNS), less soluble
low z/r
associated with weaker bonding, more soluble
Mineral Solubility
depends on pH, not all minerals have same solubility patterns
DIC (dissolved inorganic carbon)
DIC = CO2 (g) + H2CO3* + HCO3- + CO32-
Important Equations
CO2 + H2O → H2CO3*
H2CO3* →H+ + HCO3-
HCO3- → H+ + CO32-
H2O → H+ + OH-
CaCO3 → CA2+ + CO32-
CaMG(CO3)2 →Ca2+ + MG2+ + 2 CO32-
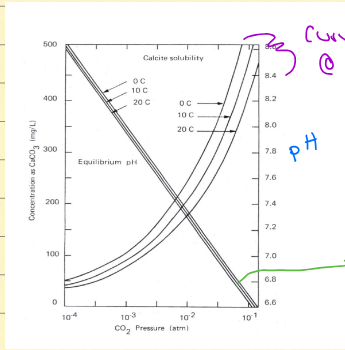
pH/solubility of CaCO3 graph
above curve/line = precipitation occurs (oversaturation), on curve/line = equilibrium, below curve/line = dissolution (undersaturated)
Saturation index
represents saturation (how full water is) numerically; what is measured in solution vs what we expect to be there
Partial Pressure
Px = n (gas A)/ n (total) x 1 atm
Saturation Index + Equilibrium
at equilibrium SI = 0
oversaturated (precipitate) SI = positive
undersaturated (dissolved) SI = negative
How do you form a cave?
Need dissolvable rock (limestone or dolostone) and weak acid (carbonic acid formed from CO2 dissolved in water)
Calcite solubility
more soluble in cold water and at higher pH
Ionic Strength
element/charge of z; higher charged elements have a strong impact on IS
Electrical Conductivity
related to TDS and IS; more ions in water means better conductivity
Hardness
“soap test”= ions prevent lathering of soap; soft water = lots of suds, less ions; hard water = no suds, more ions. 50,000 x ([CA] + [Mg])
Karst
a special type of aquifer with very large pore spaces
How does structure affect cave development?
more synclines/anticlines allows for more aggressive water runoff into rock, causing dissolution; narrower/inconsistent pathways
How does water control cave development?
aggressive recharge of water through sinkholes creates large, tubular passages