topic 17 organic chem 2
1/91
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
92 Terms
define an SN1 reaction
- 2 step reaction: slow step (RDS) and instant step
- due to lack of direct route for nucleophile to attack
- consists of a carbocation intermediate
- rate of reaction (RDS) depends on concentration of halogenoalkane only
- for all tertiary halogenoalkanes (+ some secondary)

define a SN2 reaction
- one step reaction
- consists of a temporary transitional intermediate phase
- rate of reaction (RDS) depends on concentration of halogenoalkane and nucleophile
- for all primary halogenoalkanes (+ some secondary)

compare and contrast the mechanism of hydrolysis, using aqueous KOH, of the primary halogenoalkane RCH₂X with that of the tertiary halogenoalkane R₃CX.
include diagrams of any intermediate or transition states. (6)
- both are nucleophilic substitution
- primary halogenoalkane undergo SN2 reaction via a transition state
- tertiary halogenoalkane undergo SN1 reaction via a carbocation intermediate
- both the concentration of RCH₂X and OH⁻ are in the RDS
- only the concentration of R₃CX is in the RDS
state what optical isomerism is.
- a form of stereoisomerism
- optical isomers are non-superimposable mirror images of each other
what prerequisites are required for optical isomerism to arise?
1. a chiral centre (carbon atom, sometimes nitrogen, with 4 different groups/atoms attached to it)
2. mirror images are non superimposable
3. no internal plane of symmetry (due to chirality)
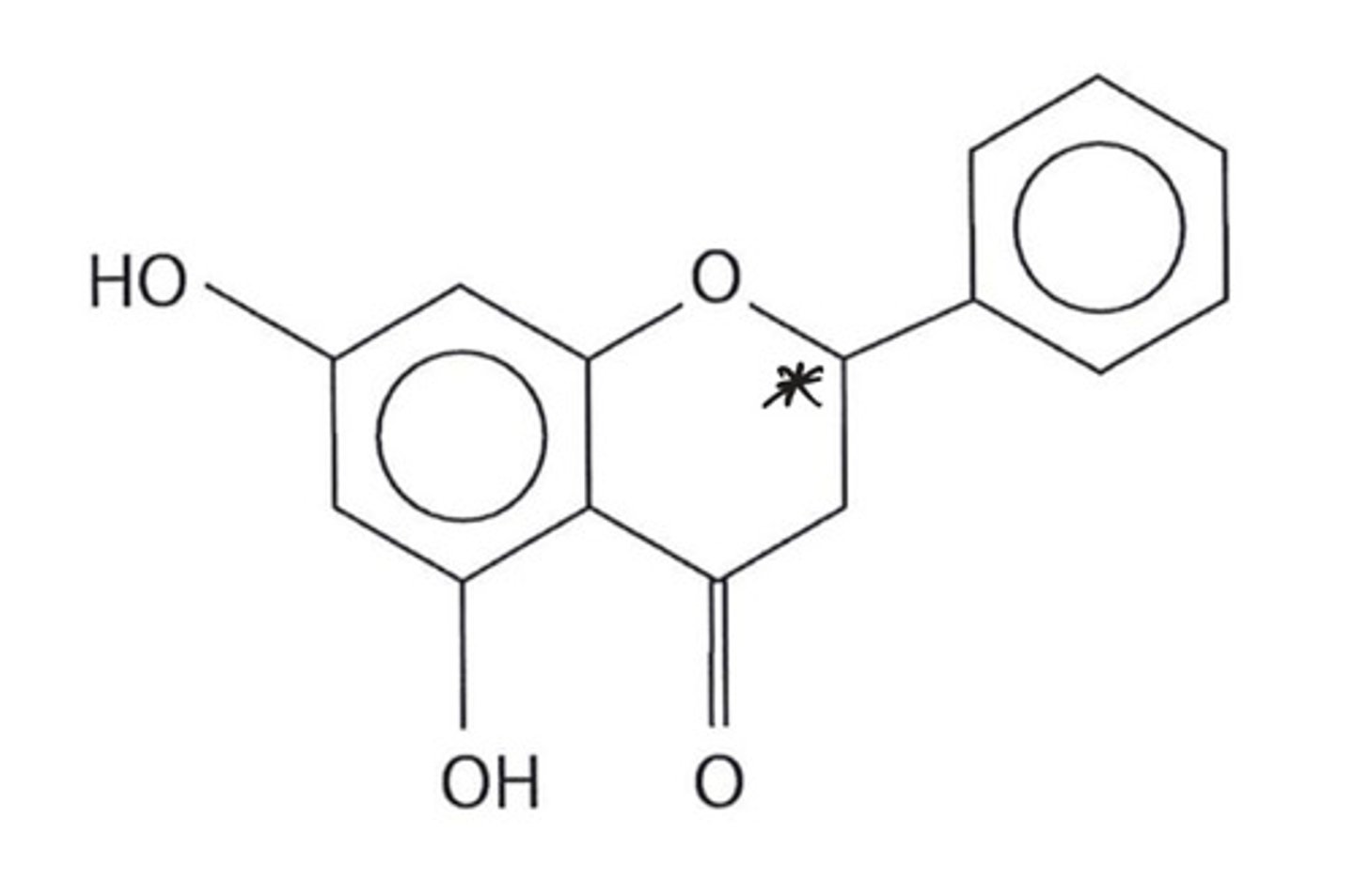
suggest how you can determine the chiral carbon in pinocembrin (photo attached)
there are 4 different groups attached to the carbon atom marked with an asterisk
define the term enantiomer isomer.
- isomers that are mirror images of each other
- another term for optical isomers
describe the similarities and differences between two optical isomers
- same chemical properties
- similar physical properties
- completely different biological properties (eg thalidomide)
define plane polarized light
light where waves all vibrate parallel to a single plane in a single direction
define the term chiral
form 2 isomers which are non-superimposable
explain how optical isomers rotate polarized light
- one optical isomer rotates polarized light anticlockwise
- whereas the other isomer rotates polarized light clockwise
when plane polarised light is passed through an optical isomer, the plane of polarisation is...
rotated
state the bond angle of optical isomers
- tetrahedral structure
- so bond angle of 109.5
define racemic mixture
- a mixture of equal amounts of 2 enantiomer (50:50)
- does not rotate plane polarized light, since the 2 enantiomers cancel each other's light rotating effect
★ can be produced by nucleophilic substitution (SN1 / SN2) or addition
explain why its possible to react two achiral compounds together and form a racemix mixture of a chiral compound
- when 2 achiral compounds react, there's often an equal chance of forming each of the enantiomers
- eg butane and chlorine; equal chances of each H atom to be replaced by the Cl
explain the SN1 and SN2 mechanisms in relation to their optical activity
SN1 (3°) - optically inactive
SN2 (1°) - optically active
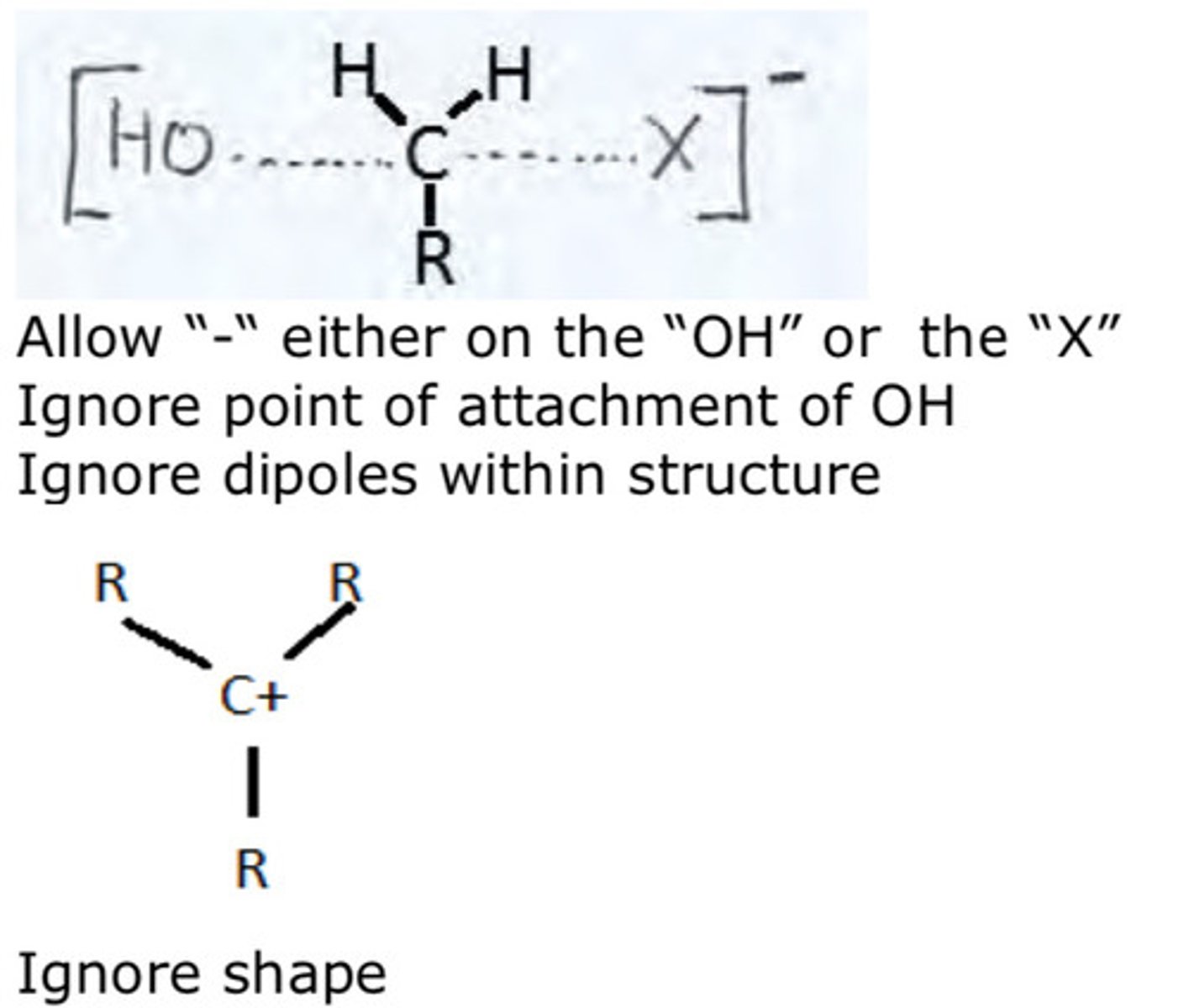
draw the nucleophilic substitution mechanism of a SN1 (3°) reaction with regards to its optical activity
★ start with one enantiomer, end up with a racemic mixture
[occurs in 2 steps]
1) SLOW STEP: loss of leaving group (X-) , resulting in a planar carbocation intermediate around reaction site
2) incoming nucleophile can attack via either face of the compound
3) results in 2 optically active isomers (50% S and 50% R) so no optical activity
![<p>★ start with one enantiomer, end up with a racemic mixture</p><p>[occurs in 2 steps]</p><p>1) SLOW STEP: loss of leaving group (X-) , resulting in a planar carbocation intermediate around reaction site</p><p>2) incoming nucleophile can attack via either face of the compound</p><p>3) results in 2 optically active isomers (50% S and 50% R) so no optical activity</p>](https://knowt-user-attachments.s3.amazonaws.com/05e54587-6190-48ed-9d0b-8d559f6648fa.jpg)
draw the nucleophilic substitution mechanism of a SN2 (1°) reaction with regards to its optical activity
★ start with one enantiomer, end up with the opposite enantiomer
[occurs in 1 step only]
1) nucleophile attack from the opposite side of the leaving group
2) only one single possible direction of attack, hence an enantiomer that rotates plane polarised light in the opposite direction is formed
![<p>★ start with one enantiomer, end up with the opposite enantiomer</p><p>[occurs in 1 step only]</p><p>1) nucleophile attack from the opposite side of the leaving group</p><p>2) only one single possible direction of attack, hence an enantiomer that rotates plane polarised light in the opposite direction is formed</p>](https://knowt-user-attachments.s3.amazonaws.com/c7423c7f-b3c5-408b-bd2a-37b859557870.jpg)
2-bromobutane can react with aqueous OH- by an SN1 reaction.
explain why the butan-2-ol produced from a single optical isomer of 2-bromobutane, using the mechanism, is not optically active.
- a racemic mixture is formed (50% R 50% S)
- since the intermediate C+ is trigonal planar around the reaction site
- so incoming nucleophile can attack from either side of the plane
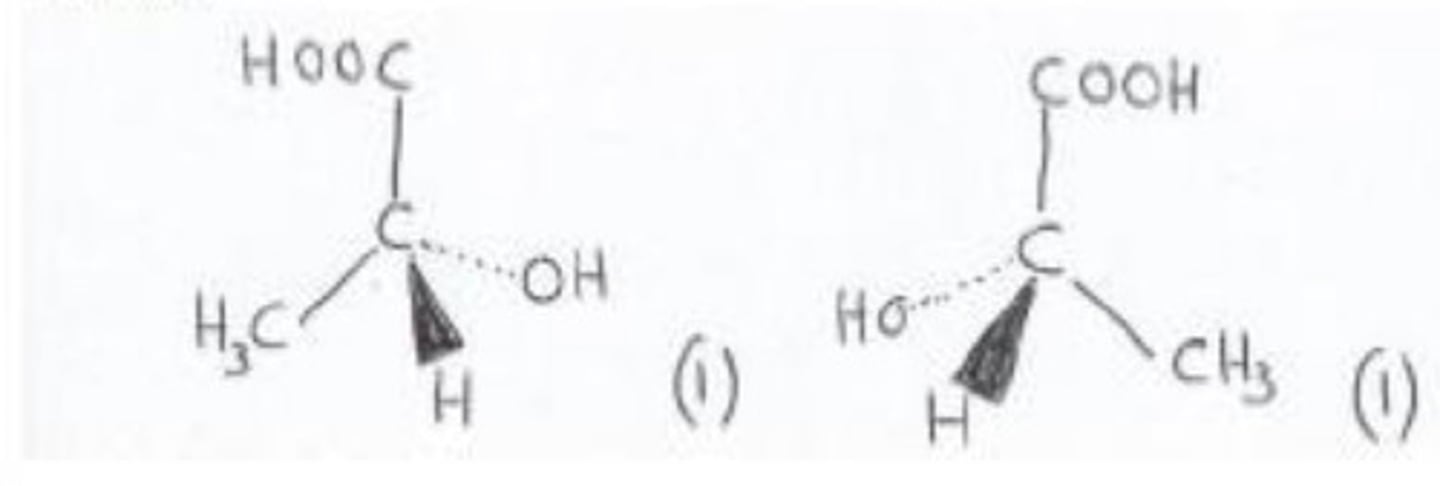
PLA is a biodegradable polyester made from 2-hydroxypropanoic acid, CH3CH(OH)COOH.
draw the two enantiomers of 2-hydroxypropanoic acid.
a molecule has 2 chiral centres. deduce the number of optical isomers that can exist.
4
state how separate samples of the 2-hydroxypropanoic acid enantiomers could be distinguished in a lab.
rotate plane polarised light equally in opposite directions
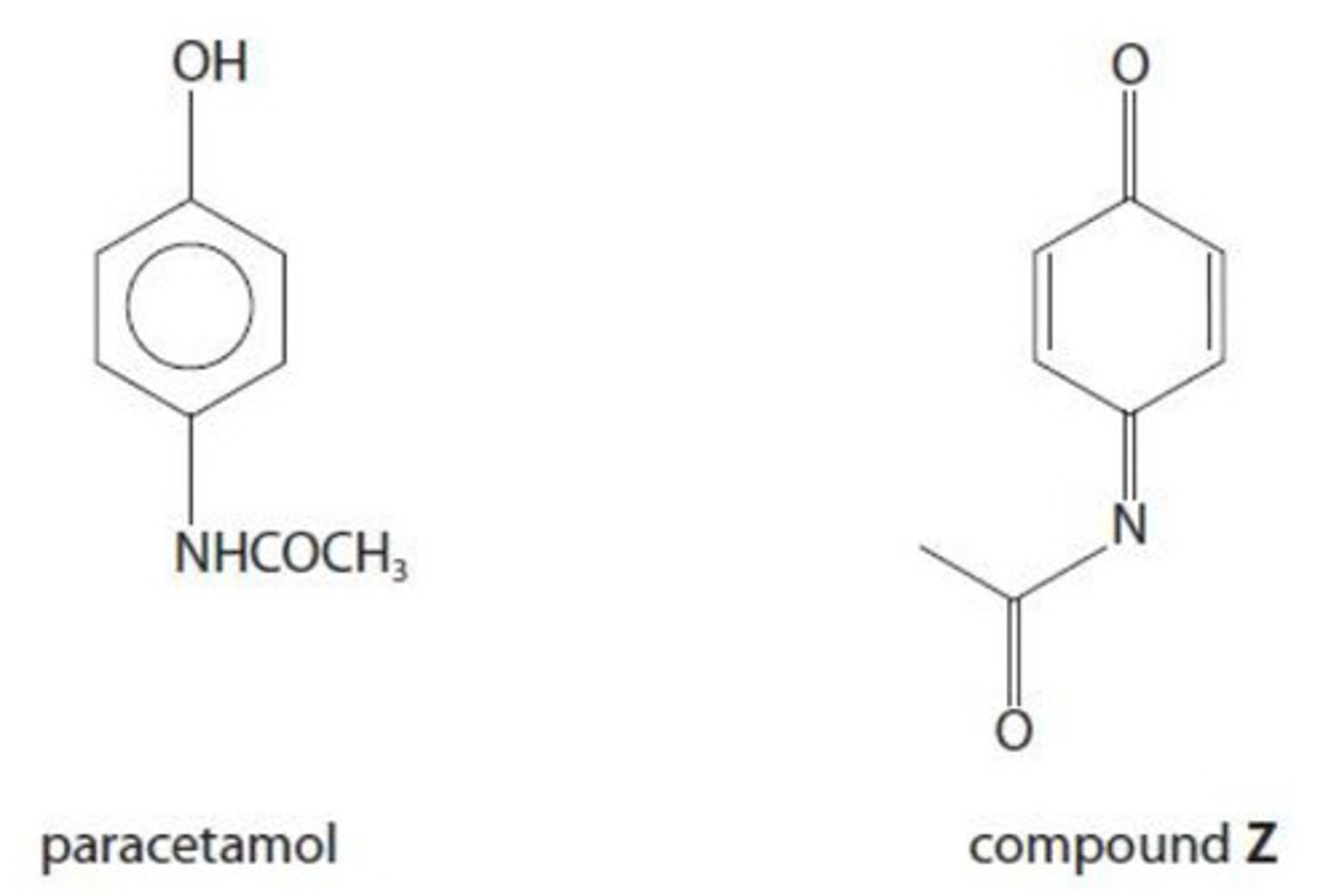
the conversion of paracetamol into compound Z is:
A. addition
B. hydrolysis
C. oxidation
D. reduction
D. since both the -NH and -OH group have lost hydrogen atoms (OIL RIG)
biodegradable polyesters break down naturally.
state why this is an advantage.
does not accumulate in the environment / does not occupy landfill
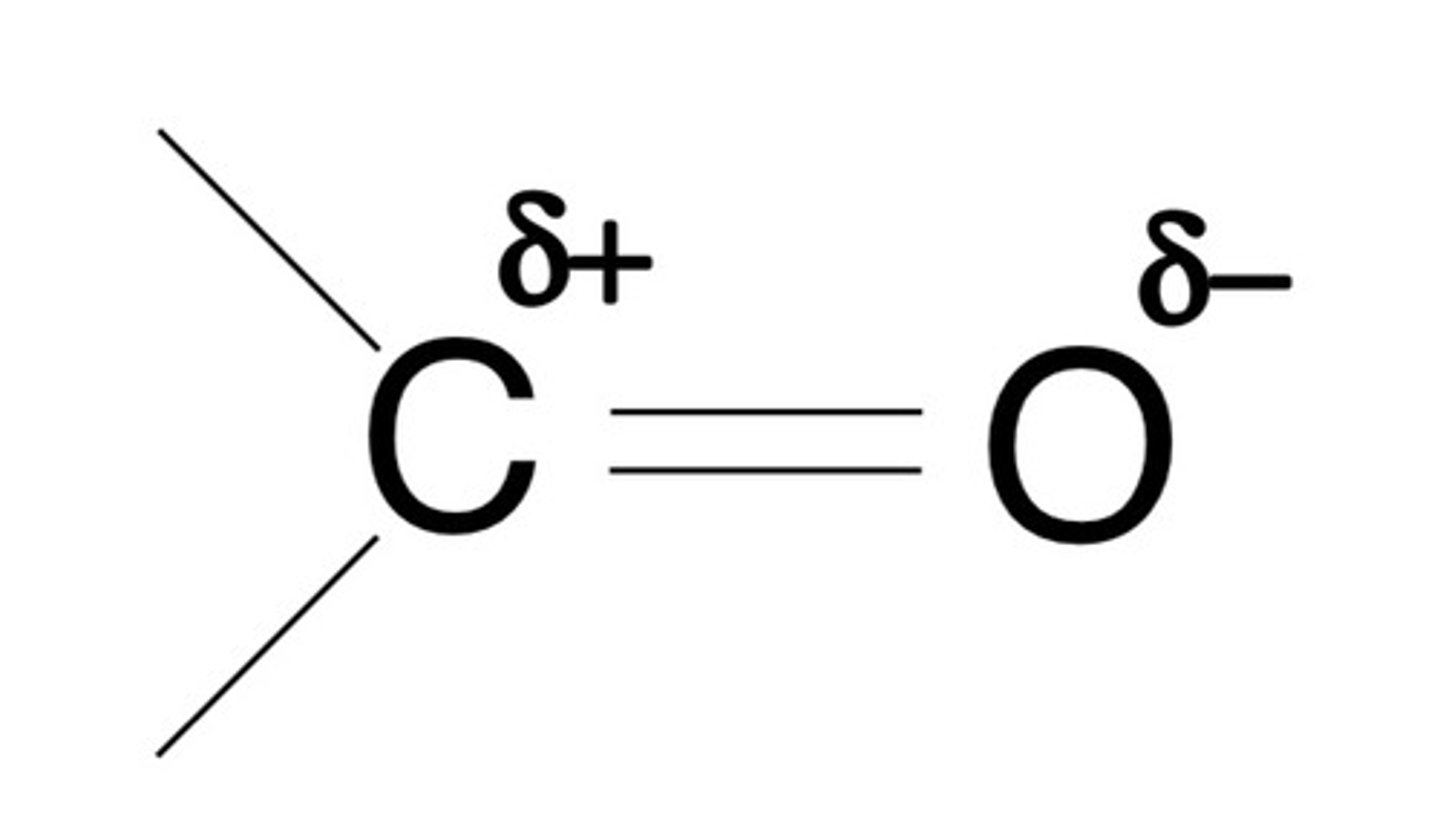
define what is meant by a carbonyl group
- compounds with a C=O bond
- unsaturated polar bonds
give 2 methods of identifying a carbonyl compound
- formation of orange crystalline ppt. with 2-4dinitrophenylhydrazine
- peak at 1400-1600 cm-1 in the infrared spectrum
(to determine whether it is an aldehyde or ketone, further tests must be done)
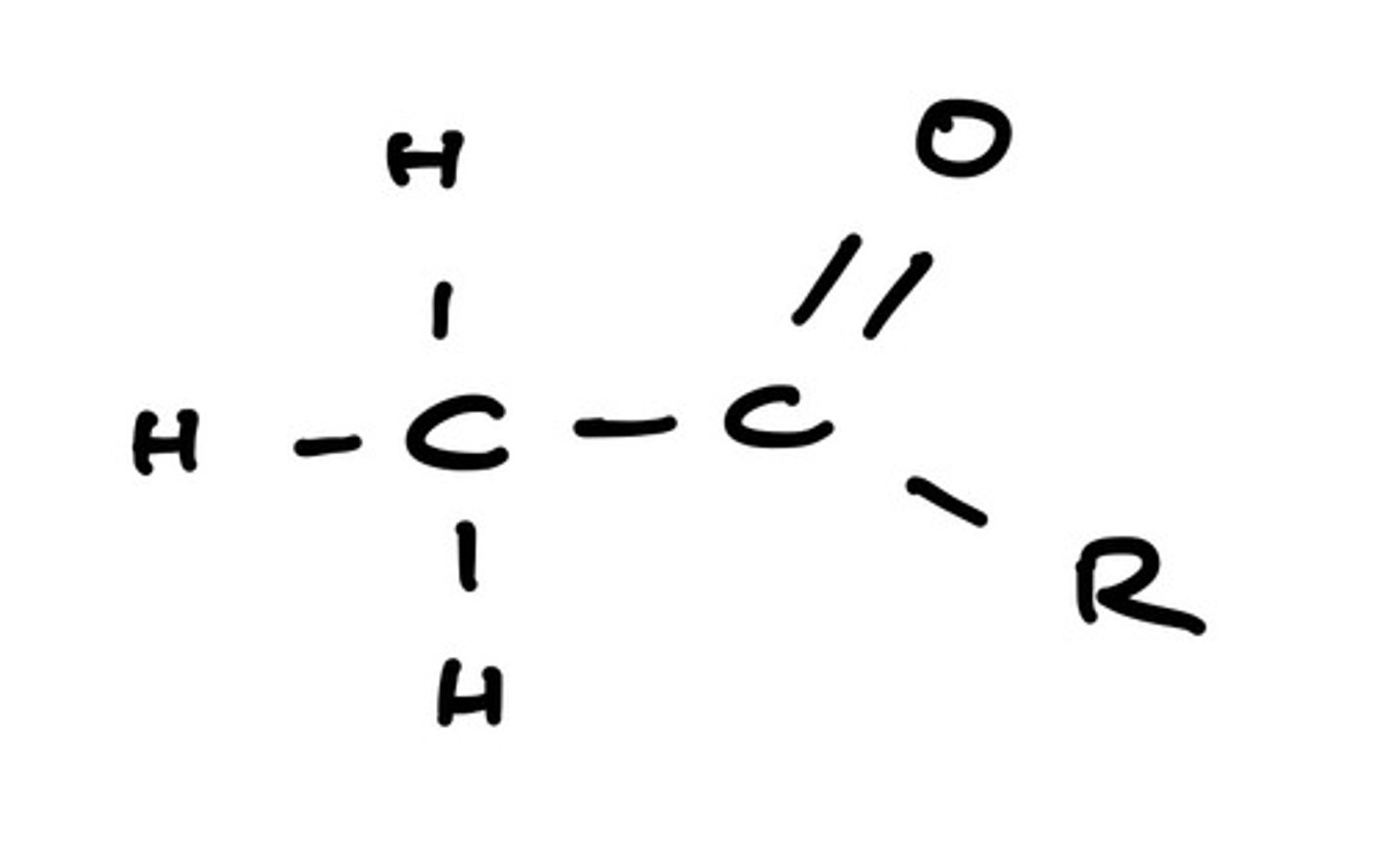
explain why small ketones and aldehydes are completely miscible in water. draw a diagram to aid your explanation.
- short carbon chain = less hydrophilic
- space for hydrogen bonding with water
- able to form hydrogen bonds
explain why the boiling points of aldehydes and ketones is greater than that of a similar sized alkane, but lower than that of a similar sized alcohol.
- alkanes only have LDF
- aldehyde have LDF and permanent dipole-dipole
- alcohol have LDF, permanent dipole-dipole, and hydrogen bonding
what reagents would react with aldehydes and not ketones?
- Fehling's solution (copper ions in NaOH)
- Tollen's reagent (aqueous ammonia and silver nitrate)
explain the colour change of Fehling's solution in the presence of an aldehyde
- blue copper (II) ions are reduced to red copper (I)
- aldehyde is oxidised into carboxylic acid
- upon heating, form insoluble brick red precipitate of copper (I) oxide
★ blue solution → brick red precipitate
explain why Fehling's solution stays blue in the presence of a ketone
- when heated gently with a ketone
- solution stays blue as it cannot be oxidised
state the observation of Tollen's reagent in the presence of an aldehyde (silver mirror test)
- silver (I) ions reduced to silver metal
- any aldehyde is oxidised into carboxylic acid
★ colourless solution → silver mirror precipitate
state the observation of potassium dichromate (VI), K₂Cr₂O₇, in the presence of a primary or secondary alcohol and state what the colour change is due to
★ orange Cr₂O₇²⁻ -> green Cr³⁺
what functional groups does the iodoform test specifically test for?
★ the CH3C=O group
- secondary alcohols, methyl ketones
- ethanal is the only aldehyde that reacts
state the observation of the iodoform test in the presence of methyl carbonyl
- pale yellow precipitate formed (CHI₃ — iodoform)
- antiseptic smell
what reagent would react with phenylethanone but not with phenylethanal?
triiodomethane (iodoform) reaction using alkaline (NaOH) iodine solution
describe a chemical test, and its result, to distinguish between pentan-2-one, CH₃CH₂CH₂COCH₃, and pentan-3-one, CH₃CH₂COCH₂CH₃. (2)
- add a solution of iodine and alkali (NaOH) and warm
- only penta-2-one gives a yellow precipitate since its a methyl ketone
give the steps to show how 2,4-dinitrophenylhydrazine could be used to distinguish between phenylethanone and phenylethanal.
- formation of orange crystalline ppt (due to C=O)
- filter then recrystallisation of products
- determination of melting temperature
- comparison and hence identification from use of database/known values
identify 3 steps required to prepare a sample of pure, dry derivative of a carbonyl compound which has reacted with 2,4-DNPH to form a solid. (3)
1. filter out the precipitate
2. recrystallise
3. leave overnight to dry
state the conditions and reagents required for a carbonyl compound (-C=O) to turn into an alcohol
- nucleophilic addition (H- nucleophile)
- with lithium aluminium hydride (LiAlH4) in dry ether as the reducing agent
- dry ether required as LiAlH4 is extremely reactive, so it may react with water instead
write an equation for the reaction between ethanal and lithium aluminium hydride, using [H] as the reducing agent
CH₃COH + 2[H] → CH₃CH₂OH
write an equation for the reaction between propanone and lithium aluminium hydride, using [H] as the reducing agent
CH₃COCH₃ + 2[H] → CH₃C(OH)HCH₃
write an equation for the reaction between ethanoic acid and lithium aluminium hydride, using [H] as the reducing agent
CH₃COOH + 4[H] → CH₃CH₂OH + H₂O
state the reagents and conditions required for the nucleophilic addition of HCN to carbonyl compounds
reagents: KCN and H2SO4 (we want to obtain HCN but pure HCN is very poisonous)
this reaction lengthens the carbon chain and forms hydroxynitriles (contains both CN and OH groups)
complete the equation:
CH₃CHO + HCN → ___
CH₃CH(OH)CN
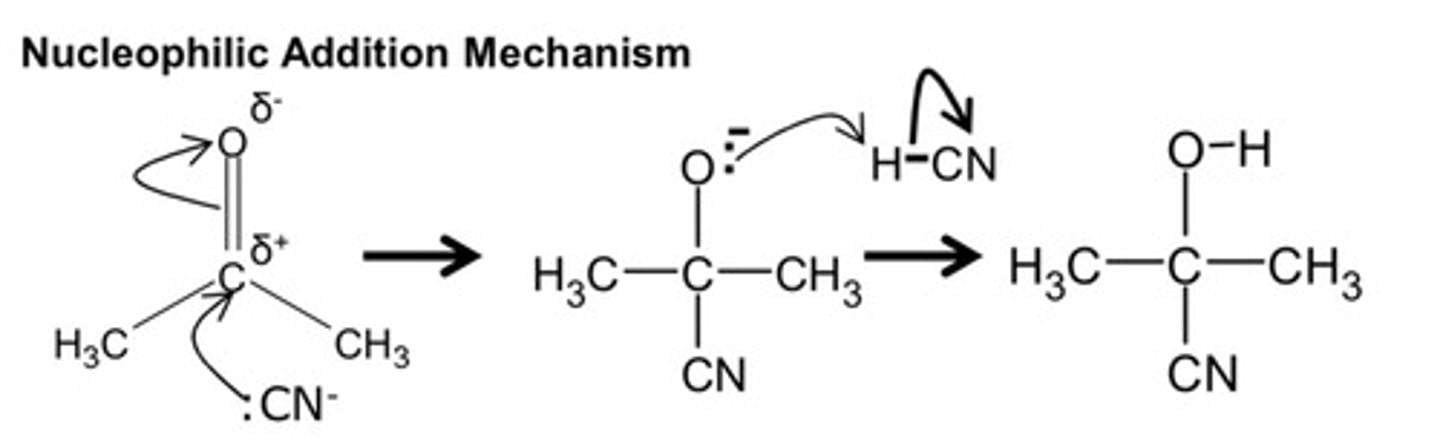
draw the mechanism for the nucleophilic addition reaction of HCN to carbonyl compounds (hydroxynitrile)
MP1 - negative on C̄N
MP2 - δ+ and δ- on the C and O respectively
MP3 - negative on Ō: and arrow onto H
MP4 - arrow from H-C bond to C
MP5 - H attaches to O
draw the mechanism for the reaction between penta-2-one and hydrogen cyanide in the presence of cyanide ions. include curly arrows and any relevant lone pairs.
MP1 - curly arrows from lone pair on C of CN- to C of ketone group
MP2 - curly arrow from C=O to O
MP3 - intermediate step
MP4 - curly arrow from lone pair on O- to H and curly arrow from H-CN bond to anywhere on CN
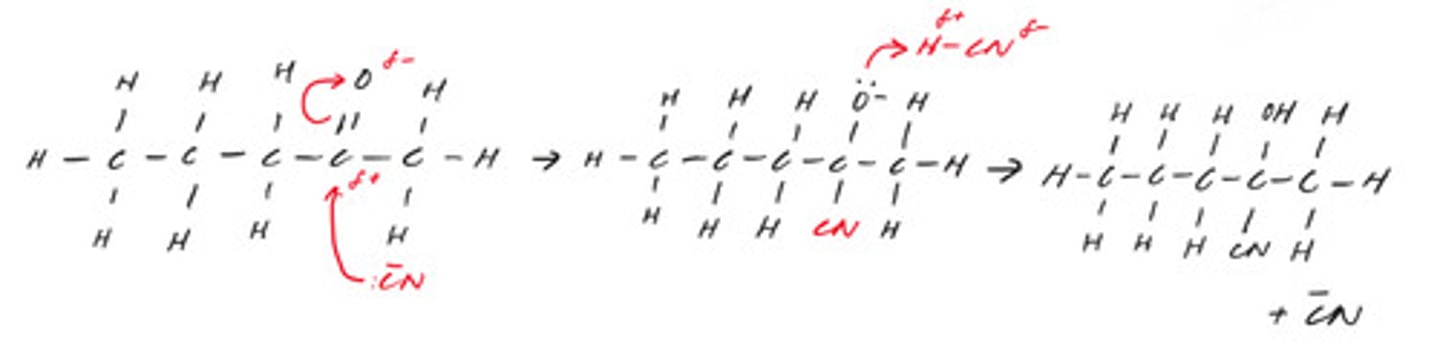
the product of this reaction, 2-hydroxy-2-methylpentanitrile, has a chiral centre. explain why a racemic mixture of 2-hydroxy-2-methylpentanitrile is formed in this reaction. (2)
- planar about carbonyl carbon
- so CN nucleophile attacks from either side of the plane
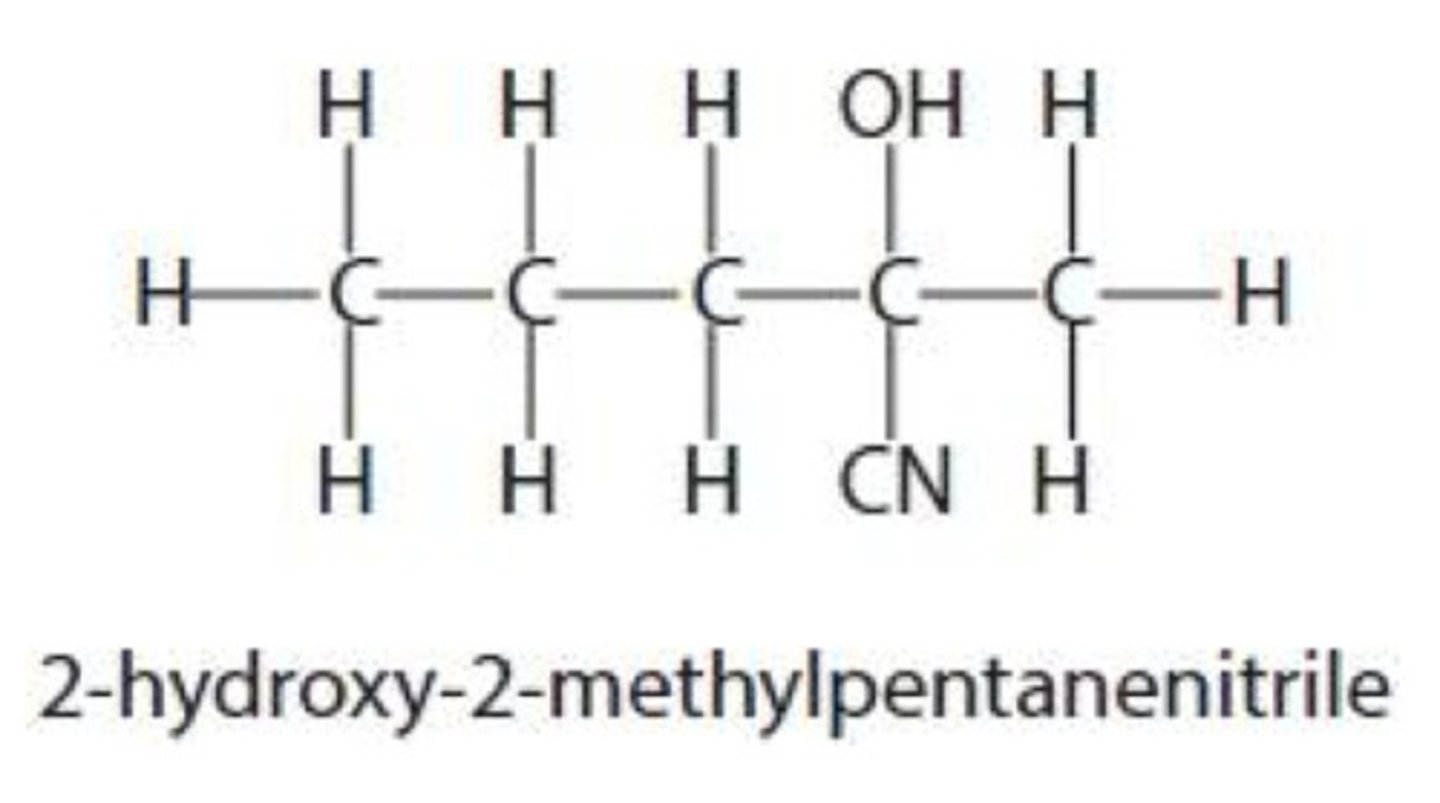
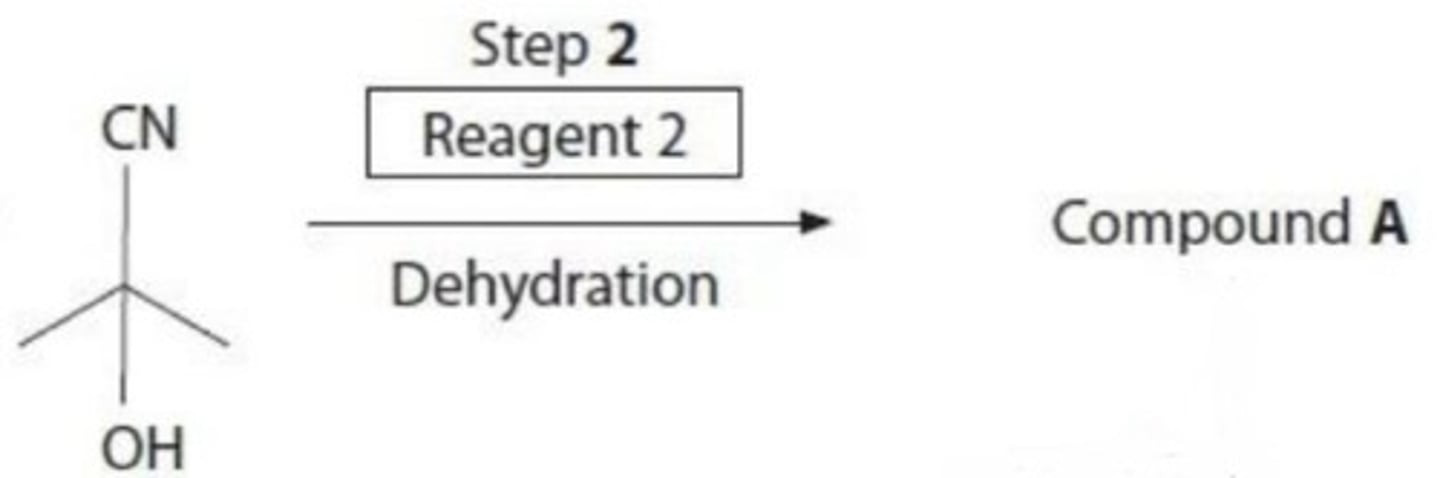
name the reagent required for the dehydration of 2-hydroxypropanenitrile
concentrated phosphoric acid
describe the oxidation and reduction reactions of pentanal and pentan-3-one. Include the reagents and the structures of the organic products formed in any reactions that take place. (6)
IP1 Reagents and conditions for oxidation
- acidified potassium dichromate (VI)
- Fehling's solution / Tollens' reagent
IP2 Oxidation of aldehyde
- pentanoic acid: CH₃CH₂CH₂CH₂COOH
IP3 Oxidation of ketone
- pentan-3-one is not easily oxidised
IP4 Reagents and conditions for reduction
- LiAlH₄ and dry ether
IP5 Reduction of aldehyde
- pentan-1-ol: CH₃CH₂CH₂CH₂CH₂OH
IP6 Reduction of ketone
- pentan-3-ol: CH₃CH₂CH(OH)CH₂CH₃
explain why the boiling point of ethanoic acid is higher than that of propan-1-ol.
- both propan-1-ol and ethanoic acid have LDF, permanent dipole-dipole and hydrogen bonding
- but carboxylic acid can form dimers (dimerise)
- this increases their LDF and their permanent dipole-dipole forces
- delta +ve and -ve signs on H and O respectively
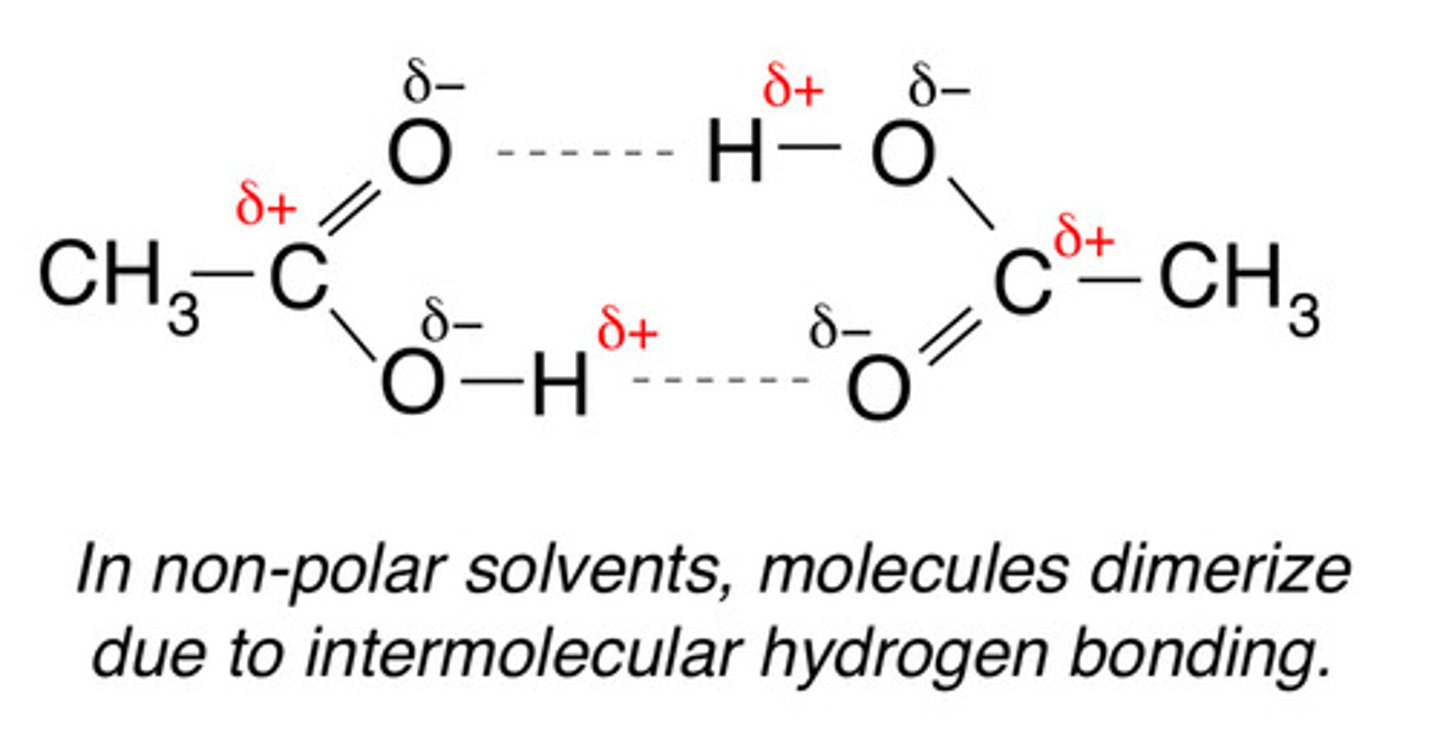
state the conditions and reagents required to oxidise alcohols into carboxylic acids
- excess acidified potassium dichromate (oxidising agent)
- primary alcohol under reflux
- CH₃CH₂OH + 2[O] → CH₃COOH + H₂O
state the reagent required for the acid hydrolysis of ethanenitrile
any diluted strong acid that can provide H+ (eg HCl)
write an equation for the acid hydrolysis of ethanenitrile into ethanoic acid
CH₃CN + 2H₂O + HCl → CH₃COOH (acid) + NH₄Cl
heat under reflux
state the reagent require for the alkaline hydrolysis of ethanenitrile
any diluted strong alkaline that can provide OH- (eg NaOH)
write an equation for the alkaline hydrolysis of ethanenitrile into sodium carboxylate (salt)
CH₃CN + H₂O + NaOH → CH₃COONa (salt) + NH₃ (g)
heat under reflux
suggest how you can obtain a carboxylic acid from alkaline hydrolysis of nitriles
acidification of carboxylate salt
write an equation for the reaction between magnesium and methanoic acid
Mg + 2HCOOH → (HCOO)₂Mg + H₂
write an equation for the reaction between lithium oxide and ethanoic acid
Li₂O + 2CH₃COOH → CH₃COOLi + H₂O
state an observation of the reaction between PCl₅ and ethanoic acid
- steamy white fumes (HCl gas given off)
- turns damp blue litmus paper red (acidic)
state, through evidence from a test, that carboxylic acids cannot be further oxidised.
- remains blue in Fehling's solution
- suggests it cannot be oxidised
state the properties of acyl chlorides
- derivatives of carboxylic acids
- fuming, colourless liquids
- very reactive
- able to absorb water
write an equation for the reaction between PCl₅ and ethanoic acid (formation of ethanoyl chloride)
CH₃COOH + PCl₅ → CH₃COCl + HCl (g) + POCl₃
★ CH₃COCl is known as ethanoyl chloride (acyl chloride)
draw the functional group of acyl chlorides
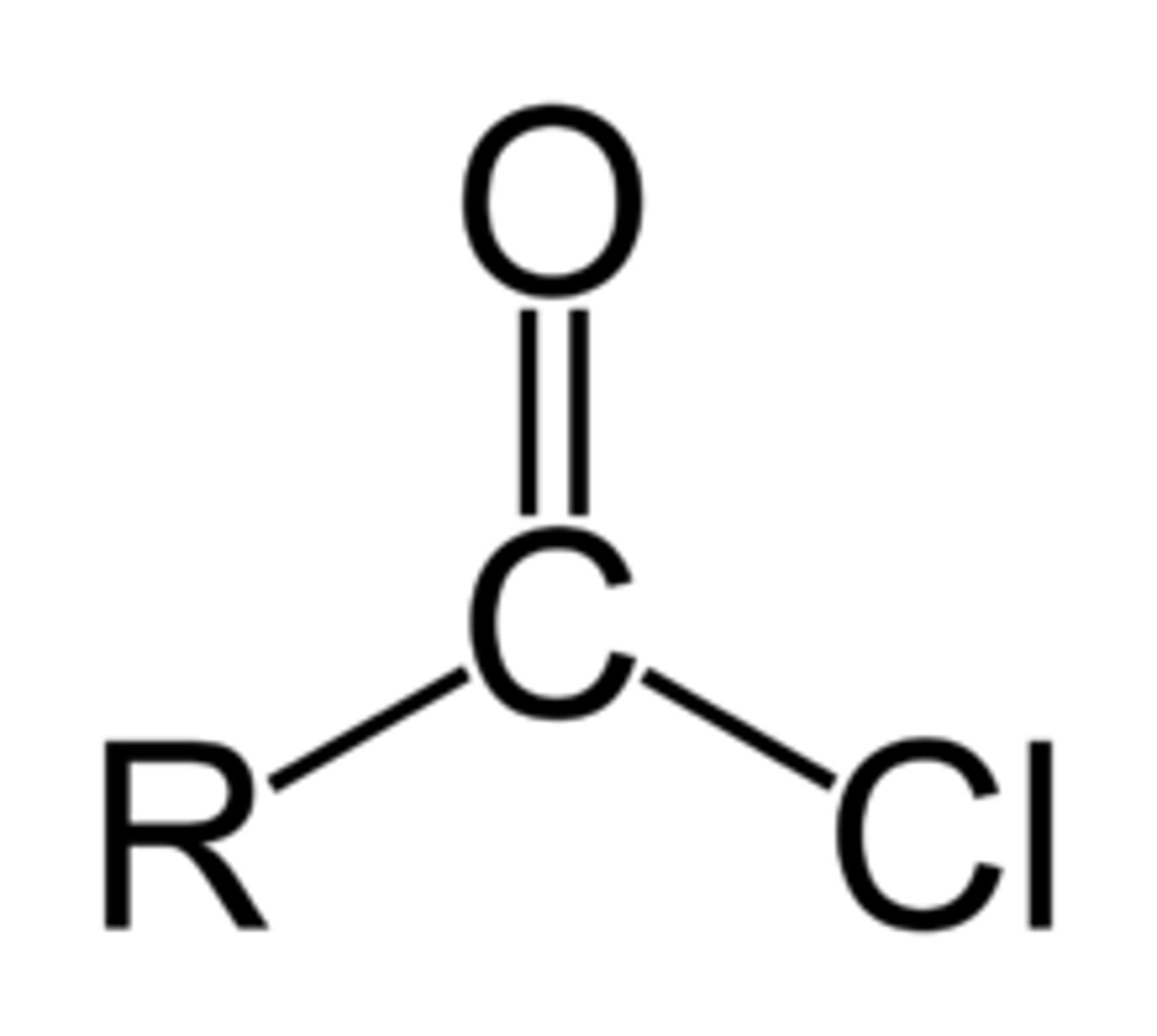
write an equation for the reaction between ethanoyl chloride and concentrated ammonia
1. CH₃C=OCl + NH₃ → CH₃C=ONH₂ + HCl
2. excess NH₃ → NH₄Cl
OVERALL EQUATION: CH₃C=OCl + 2NH₃ → CH₃C=ONH₂ + NH₄Cl
★ any HCl formed will immediately react with excess ammonia to give ammonium chloride
state the observation you would see for the reaction between ethanoyl chloride and concentrated ammonia
- reacts violently with cold, concentrated ammonia
- white solid formed (mixture of ethanamide and ammonium chloride)
- white smoke arise (ammonium chloride)
write an equation for the reaction between ethanoyl chloride and methyl amine (N-substituted amine)
CH₃C=OCl + NH₃CH₃ → CH₃C=ONHCH₃ + HCl
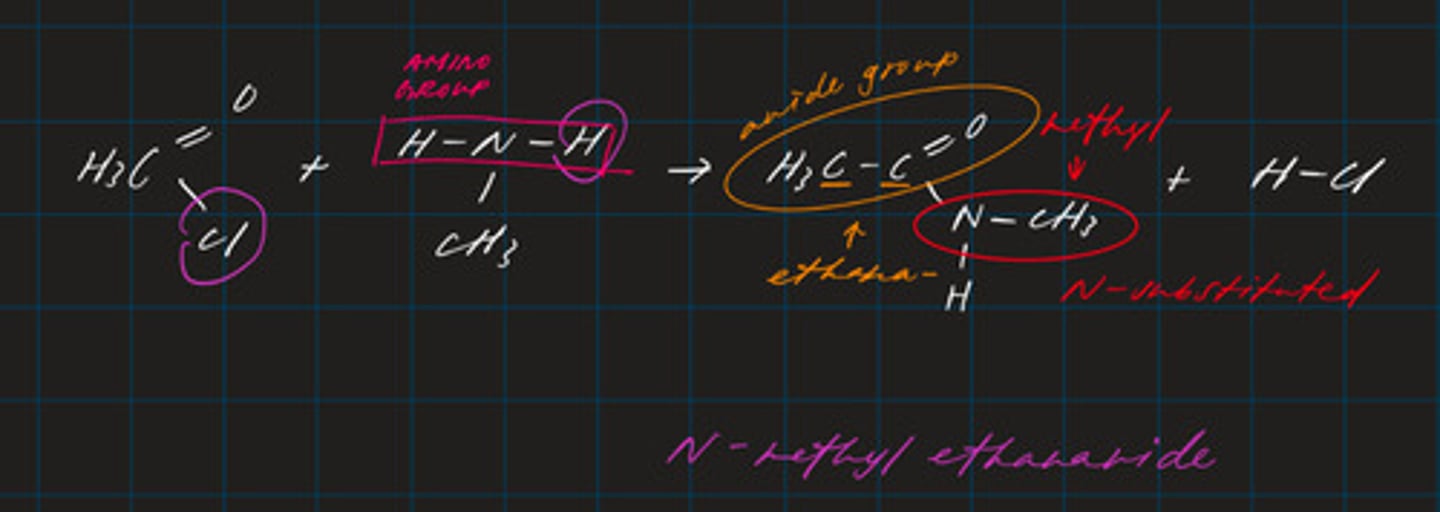
draw the mechanism for the reaction of CH3CH2CH2Cl with an excess of ammonia to form the primary amine. (topic 6)
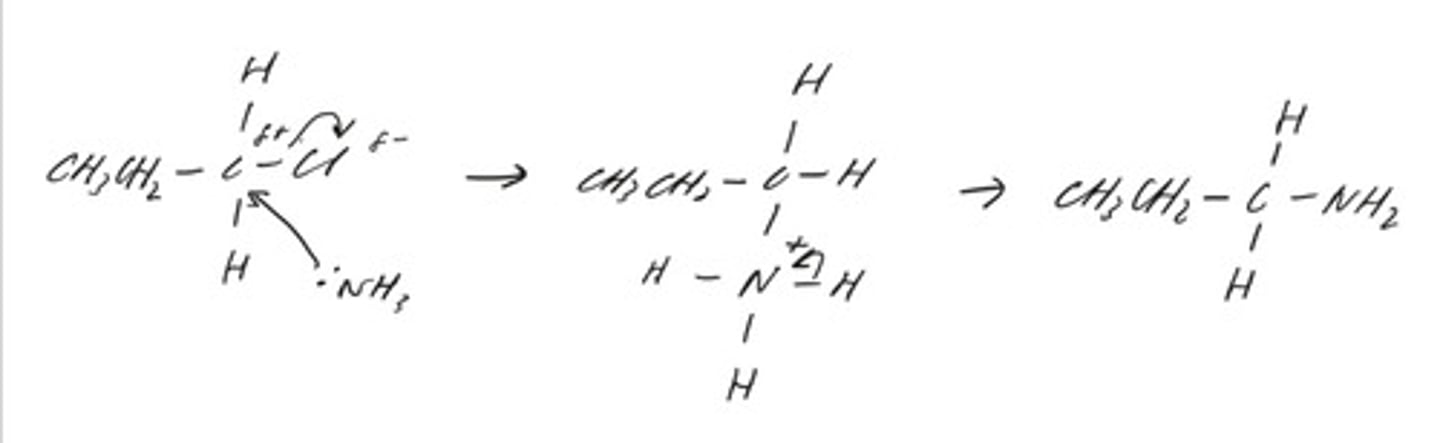
predict the mechanism for the reaction of CH3CH2COCl with ammonia.
include curly arrows and relevant lone pairs. (3)
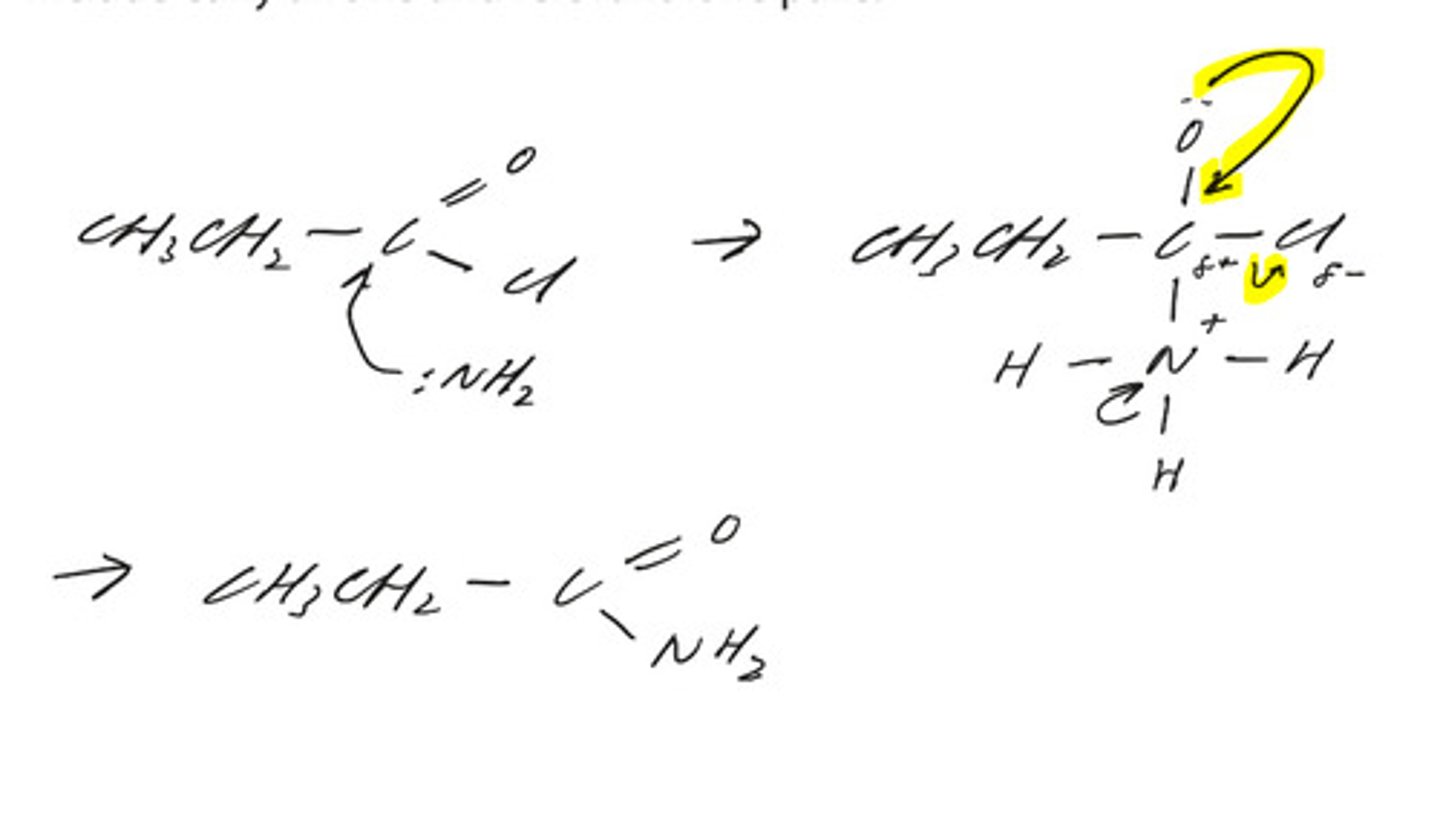
write an equation for the reaction between ethanoyl chloride and ethanol (ethyl ethanoate)
CH₃COCl + CH₃CH₂OH → CH₃COOCH₂CH₃ + HCl (g)
★ ester and hydrogen chloride gas produced
state the observation you would see of the reaction between acyl chloride and water
- reacts violently with water at RT
- does not dissolve
how are esters named?
the prefix of the alcohol with -yl as a suffix and the prefix of the acid with-oate as the suffix
write an equation for the reaction between ethanol and ethanoic acid. name the products formed.
★ -H from the alcohol, -OH from the carboxylic acid to form water
★ the reaction is reversible
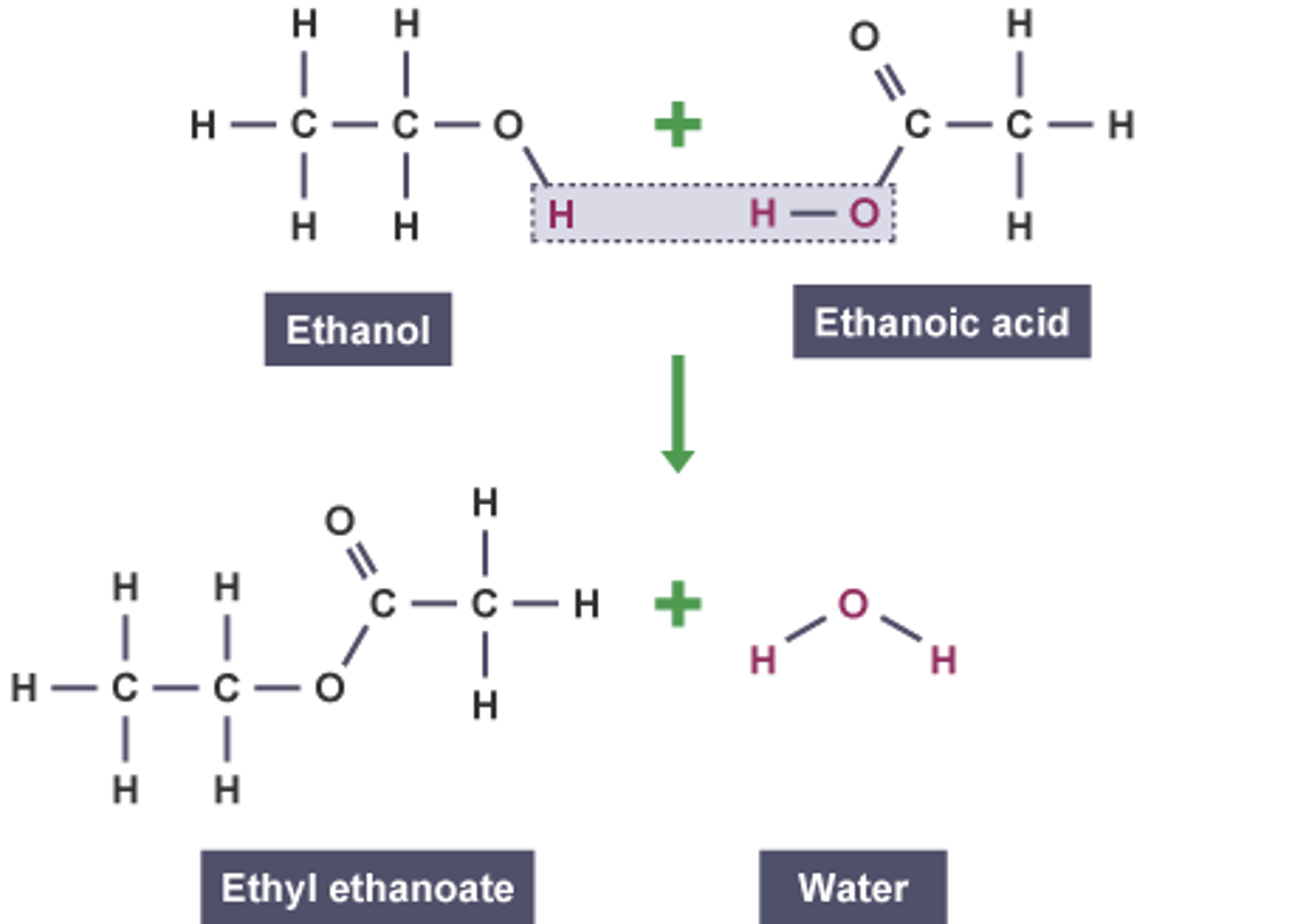
draw an ester link
condensation reaction
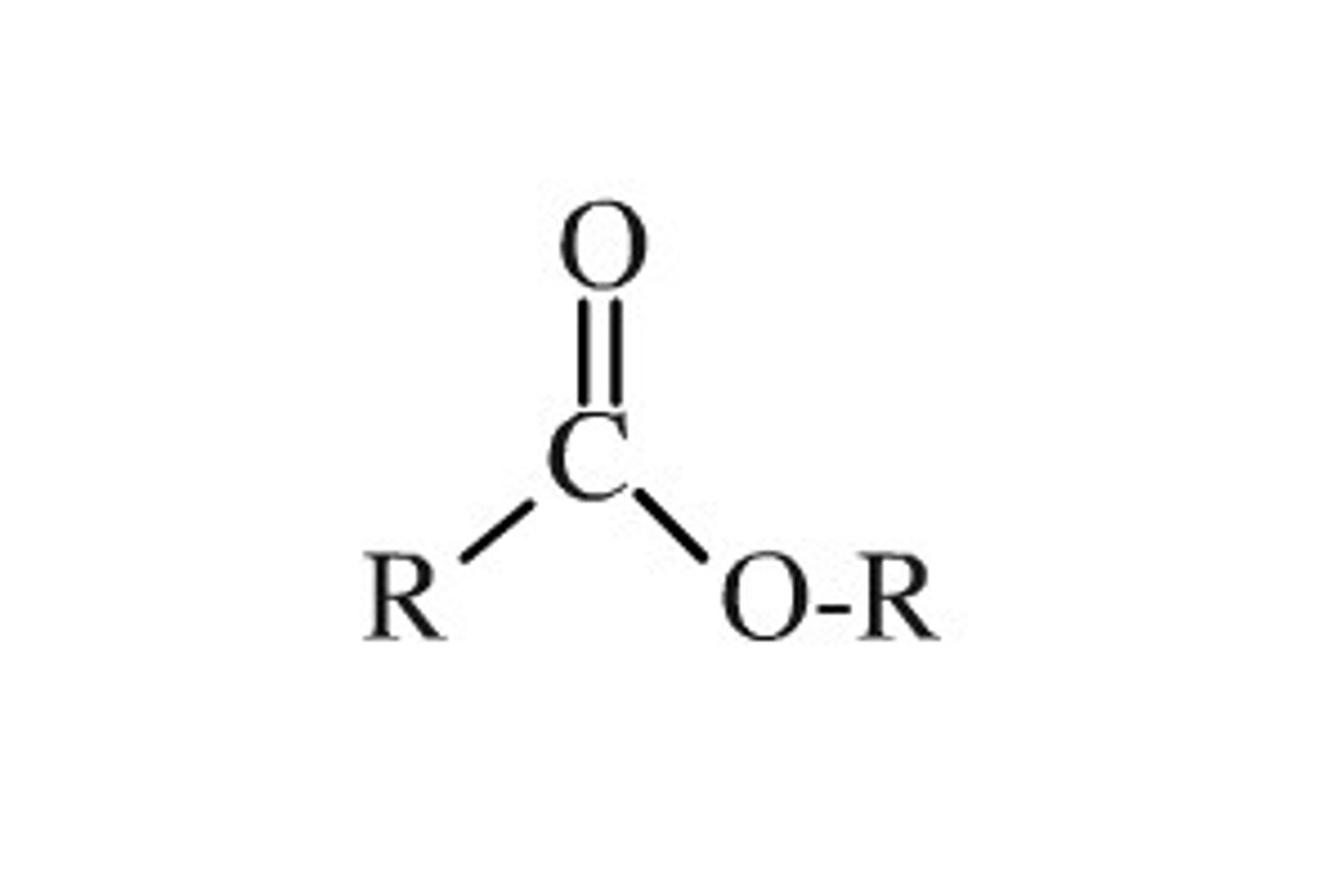
state the purpose of concentrated sulphuric acid in the reaction between carboxylic acids and alcohols to form esters
- a catalyst for the reaction
- removes water produced which moves the equilibrium to the right = bigger yield of esters
compare the two different methods for synthesising esters
carboxylic acids + alcohol:
- slow, reversible reaction (equilibrium)
- low yield
acyl chlorides + alcohol:
- fast, vigorous
- steamy HCl fumes produced (toxic)
suggest why pharmaceutical companies would prefer to use acyl chloride to produce esters instead of reacting carboxylic acids with alcohol
- reaction between acyl chloride and ethanol is non-reversible
- can take place at RT
- does not require a catalyst
write an equation for the reaction between an ester and water (reflux + acidic solution)
CH₃COOCH₃ + H₂O ⇌ CH₃COOH + CH₃OH
write an equation for the reaction between an ester and NaOH (reflux + alkali)
CH₃COOCH₃ + NaOH → CH₃COO⁻Na⁺ (very soluble) + CH₃OH
esters can be hydrolysed by heating under reflux with aqueous acid or alkali.
compare and contrast these 2 methods of hydrolysis. (4)
- both make the same alcohol
- acid hydrolysis is reversible, alkaline hydrolysis is not
- acid hydrolysis produces the carboxylic acid, alkaline hydrolysis produces the carboxylate salt
- acid is a catalyst, alkali is a reactant
the detergent, RC₆H₄SO₃Na, is soluble in water. explain how the bonding in this compound allows it to dissolve in water. (2)
- detergent contains an ionic group (SO₃⁻ & Na⁺)
- energy released when ions are hydrated compensate for energy needed to break intermolecular bonds of components in solution
state the conditions and reagents required to form esters from a carboxylic acid and an alcohol
- heat under reflux
- catalyst (concentrated H₂SO₄)
- reversible reaction with a low yield
- reflux
write an equation for the hydrolysis of ethyl ethanoate with dilute acid
CH₃C=OOCH₂CH₃ + H₂O (H+) ⇌ CH₃COOH + CH₃CH₂OH
★ reflux ester and water with H+ can reform carboxylic acid and alcohol
write an equation for the hydrolysis of ethyl ethanoate with sodium hydroxide (dilute alkali)
CH₃C=OOCH₂CH₃ + NaOH → CH₃COONa + CH₃CH₂OH
★ reflux ester and dilute alkali can form carboxylate salt and alcohol
compare the hydrolysis of ethyl ethanoate with dilute acid or alkali
- H+ acts as an electrophile in an acidic hydrolysis
- OH- acts as a nucleophile in a alkaline hydrolysis
- acid (H+) acts as a catalyst while OH- is a reagent
- acidic reaction reforms carboxylic acid + alcohol vs alkaline reaction reforms carboxylate salt and alcohol
- yield is higher with alkaline hydrolysis
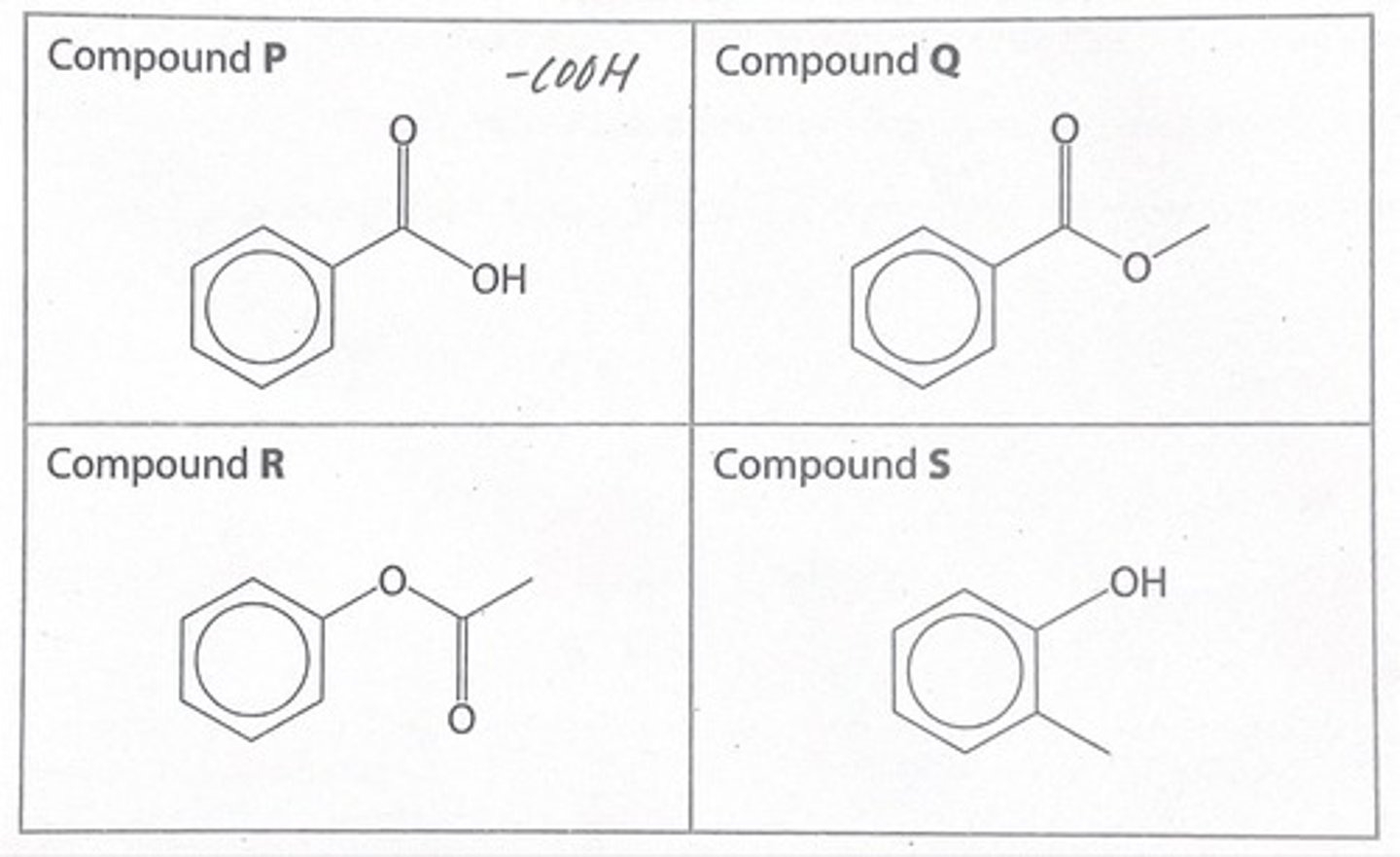
the skeletal formulae of 4 organic compounds are shown/
which of these compounds can be hydrolysed to form methanol as one of the products?
compound Q, which is an ester. it can form a methanol when it is hydrolysed at the O-CH3
C₆H₅R is not soluble in water. explain this fact by identifying
the intermolecular forces involved and comparing their relative strengths. (2)
- strong LDR between molecules in C₆H₅R and strong hydrogen bonds between water molecules
- formation of LDR between C₆H₅R and water do not compensate for energy needed to break bonds
state the reagent needed to convert glucose into sorbitol (photo attached)
LiAlH4 + dry ether (carbonyl → hydroxyl)
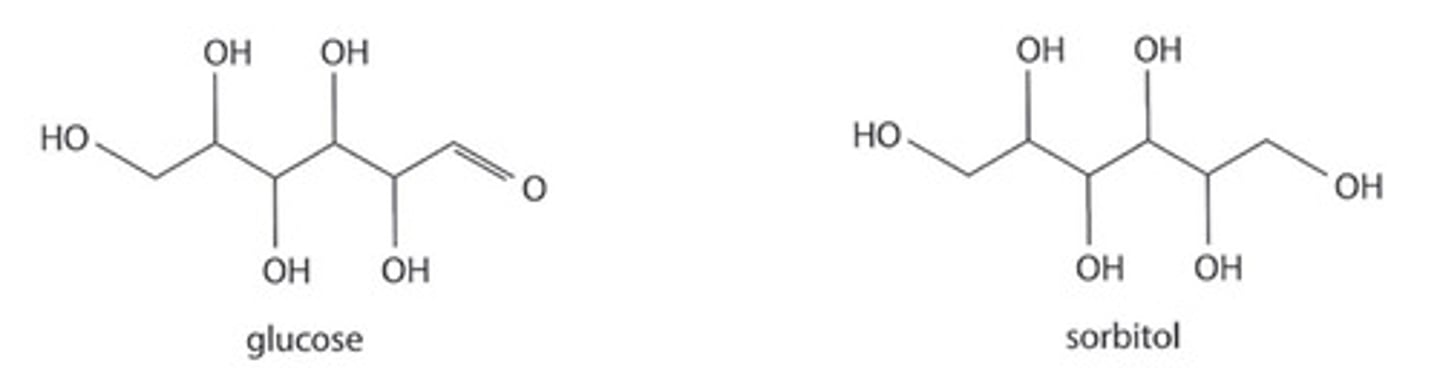
describe a chemical test which will distinguish between glucose and sorbitol. give a result for each substance.
- Fehling's solution + heat = red ppt in glucose; stays blue in sorbitol
- Tollens' + warm = silver mirror in glucose; no ppt in sorbitol
- 2,4 DNPH = orange ppt in glucose; no ppt in sorbitol
write an equation for the reaction between ethanoyl chloride and water (ethanoic acid)
CH₃COCl + H₂O → CH₃COOH + HCl (g)
★ carboxylic acid and hydrogen chloride gas produced
state a safety precaution when handling acidified potassium cyanide
- HCN is an extremely toxic gas
- use a fume cupboard
state the difference between addition and condensation polymerisation
- addition polymerisation: monomers bond to each other without the loss of atoms (double bonds are broken to form repeat units)
- condensation polymerisation: monomers will lose an atom (eg H or O) while forming the polymer, effectively losing a water molecule