OChem lab final
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
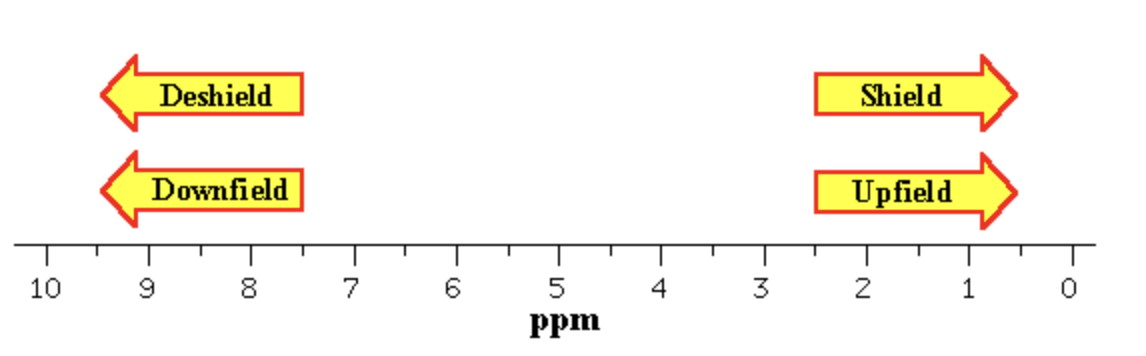
Electron donating groups on an aromatic ring shift protons_____?
upfield
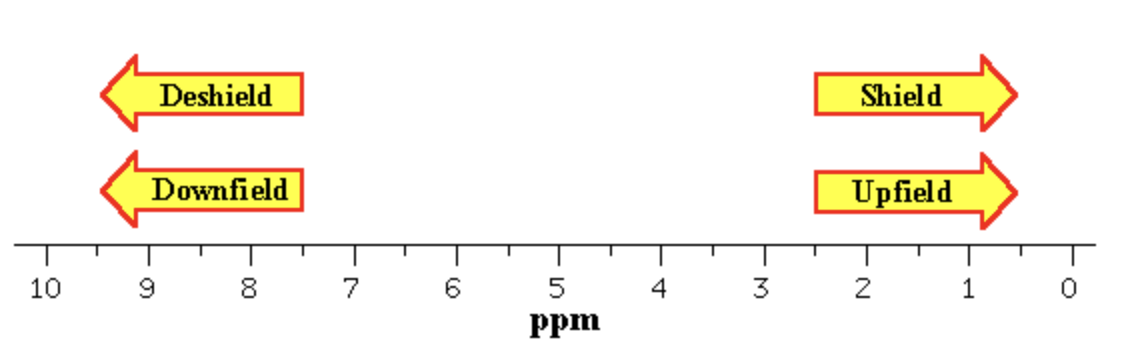
Electron withdrawing groups shift protons_____?
downfield
Protons on a carbon with a___________ are more _________?
positive formal charge
deshielded and move downfield
Protons on a carbon with a ________ are more __________?
negative formal charge
shielded and move upfield
Tetramethylsilane (TMS) appears as a single, sharp, intense peak defined at
0 ppm
Chloroform (CDCl3) solvent
7.26 ppm
Dimethyl Sulfoxide (DMSO)
2.5 ppm
Acetone (CD3COCD3)
2.05–2.17 ppm
Polar molecules have _______in silica-based TLC
Lower Rf values
To determine if a molecule is polar or nonpolar
analyze its symmetry and bond polarity
(e.g., linear, tetrahedral, trigonal planar) with identical surrounding atoms are usually non-polar. Asymmetrical shapes (e.g., bent, trigonal pyramidal) are usually polar
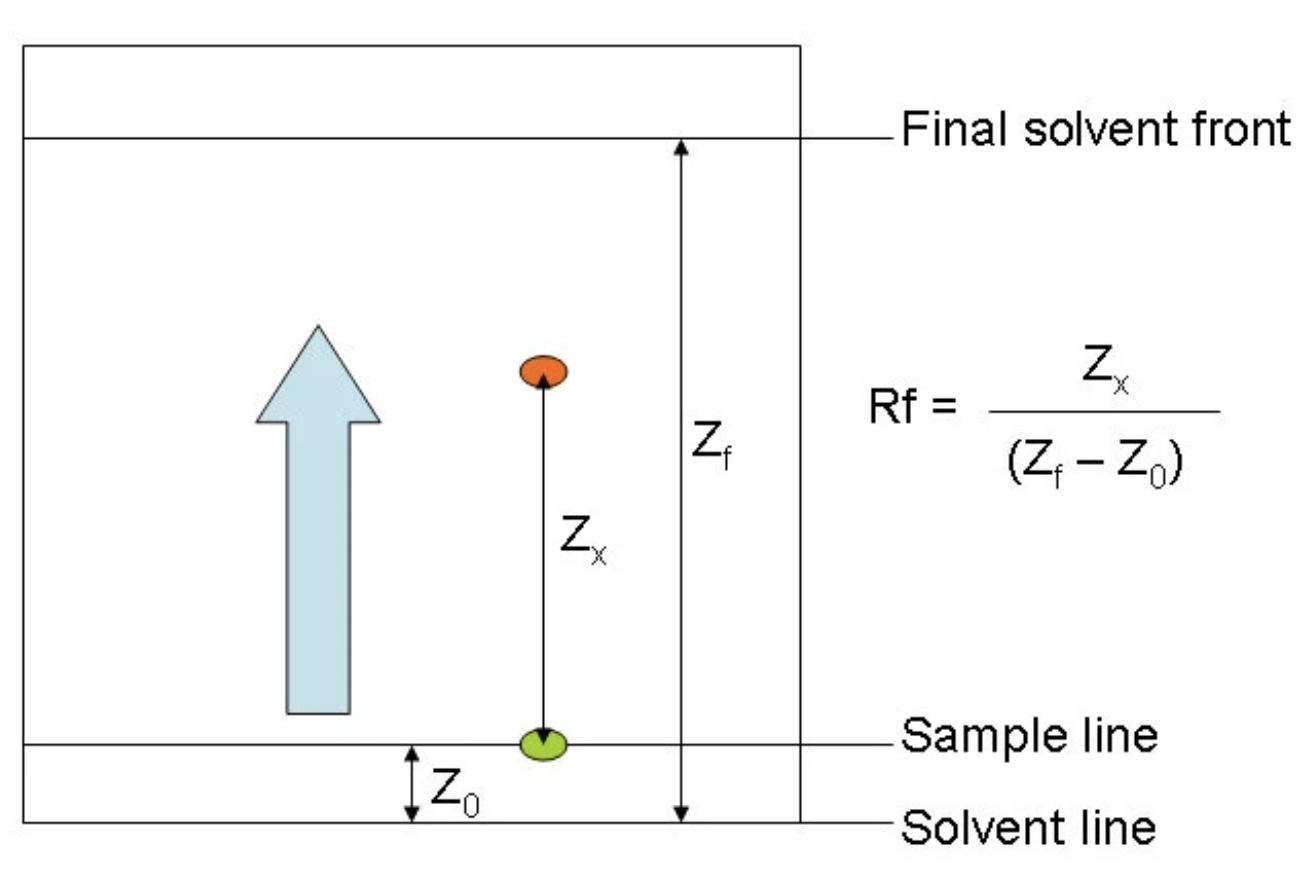
How to calculate Rf
divide the distance traveled by the compound by the distance traveled by the solvent front, both measured from the baseline
How do you “dry” a solution
Drying an organic solution involves removing trace water from an organic solvent after extraction, typically by adding anhydrous inorganic salts that bond water molecules.
Common agents:
Magnesium Sulfate (MgSO4)
Sodium Sulfate: (Na2SO4)
How do you use the rotovap?
Using a rotary evaporator (rotovap) involves evaporating solvents from a sample under reduced pressure, typically with heat, to efficiently concentrate or purify mixtures. Key steps include attaching the sample flask, imitating oration and vacuum, lowering it into a heated bath, and monitoring condensation.
Column Chromatography
This technique is performed by packing a glass tube with an adsorbent. There are many different types of adsorbents (solid phase) that are used.
The most common adsorbents used are silica gel and alumina.
Cotton is added to prevent the sand and silica from running out the bottom. The sand is added on top to create a level surface for the silica to rest on. Next, the silica is added, followed by another layer of sand. This layer of sand protects the silica underneath from being jostled by the addition of sample and solvent later on.
It is important to keep silica and all adsorbents both level and moist while running a column. You want your adsorbent to be as uniform as possible all the way around. If the surface is not level, compounds traveling down one side of the adsorbent will get through faster than those traveling down the other side, which can lead to overlapping bands (think of the side with less silica as giving its compounds an unfair head start, which might allow slower-moving, more polar components to travel as fast as faster-moving, less polar components traveling down the other side). Letting the column go dry will create cracks in the adsorbent, which act as similar shortcuts.