13- Nutrient Cycles
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Why is it important that elements such as carbon, nitrogen, and phosphorus are recycled?
There is limited availability of nutrients in a usable form
Why do organisms require nitrogen?
Amino acids
Nucleic acids
Why is there a need for the nitrogen cycle, despite 78% of the atmosphere being made up of nitrogen?
Very few organisms can use nitrogen gas directly
What form do plants take up nitrogen in?
Nitrate ions
What is the chemical formula of a nitrate ion?
NO3-
Plants take up nitrate ions by which process?
Active transport (by the roots)
How do animals obtain nitrogen-containing compounds?
By eating and digesting plants
How are nitrate concentrations restored in natural ecosystems?
Through the nitrogen cycle
Other than through the nitrogen cycle, how can nitrate concentrations be increased in agricultural ecosystems?
Nitrogen-containing fertilisers
What are the four main stages in the nitrogen cycle?
Ammonification
Nitrification
Nitrogen fixation
Denitrification
Draw the nitrogen cycle from memory.
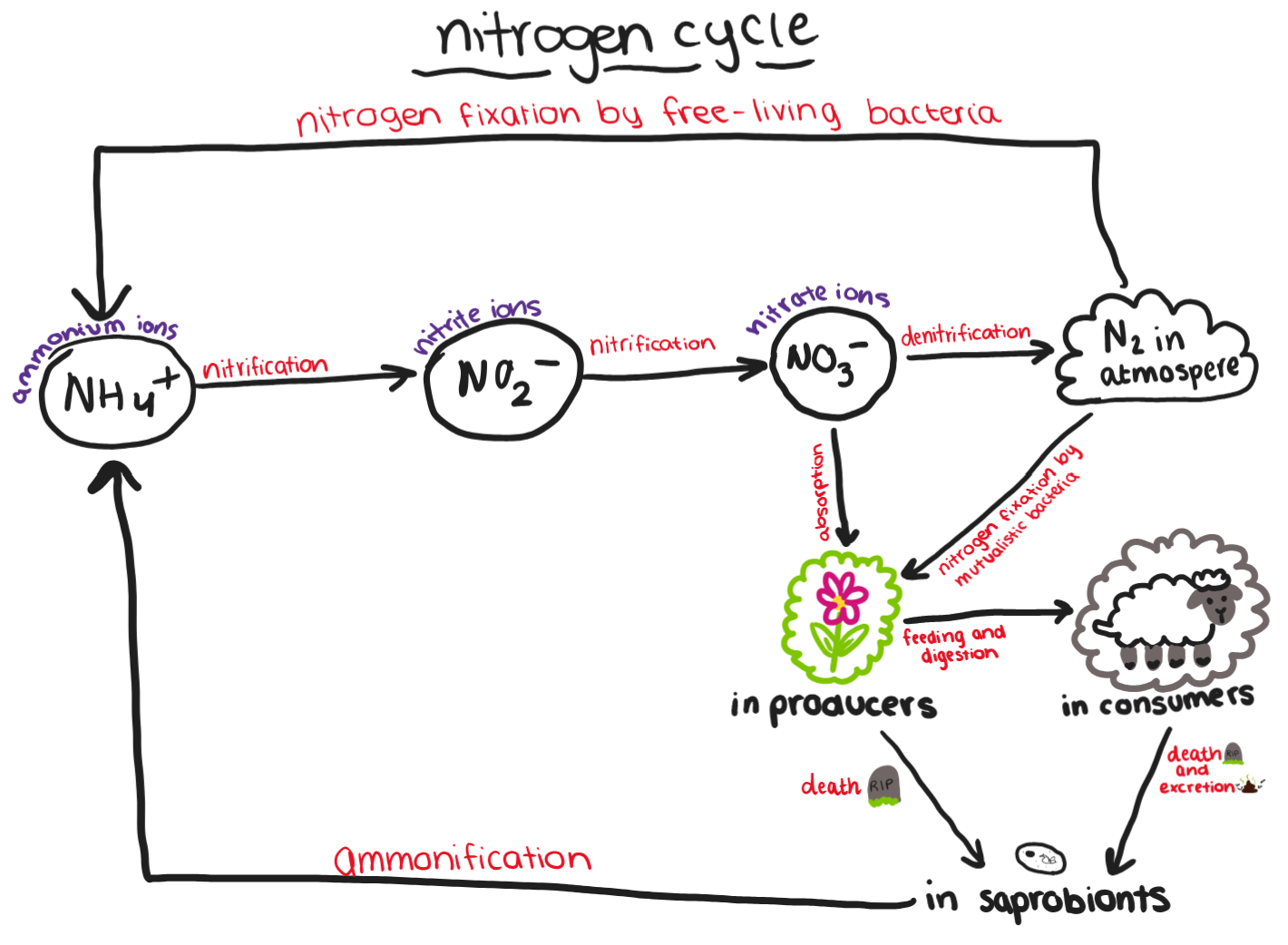
Describe the process of ammonification.
The production of ammonia from organic nitrogen-containing compounds
Saprobionts decompose proteins in faeces and dead organisms, releasing ammonia
Ammonia forms ammonium ions in the soil
Describe the process of nitrification.
The conversion of ammonium ions to nitrate ions
Carried out by nitrifying bacteria in the soil
First, ammonium ions are oxidised to nitrite ions
Then, nitrite ions are oxidised to nitrate ions
How do farmers increase the rate of nitrification and decrease the rate of denitrification?
Nitrifying bacteria are aerobic, denitrifying bacteria are anaerobic
Farmers keep soil well aerated by ploughing (produces air spaces)
Farmers drain soil to prevent air spaces filling with water
Which two types of microorganisms carry out nitrogen fixation?
Free-living nitrogen-fixing bacteria
Mutualistic nitrogen-fixing bacteria
Describe the process of nitrogen fixation by free-living nitrogen-fixing bacteria.
Bacteria in the soil reduce nitrogen gas to ammonia
Ammonia forms ammonium ions in the soil
Describe the process of nitrogen fixation by mutualistic nitrogen-fixing bacteria.
Bacteria live in nodules on the roots of plants such as peas and beans
Reduce nitrogen gas to ammonium compounds and convert these to nitrates
Bacteria obtain carbohydrates from the plant
Plant obtains amino acids from the bacteria
Describe the process of denitrification.
Denitrifying bacteria convert nitrates in the soil to nitrogen gas
Reduces the availability of nitrates for plants
Draw the phosphorus cycle from memory.
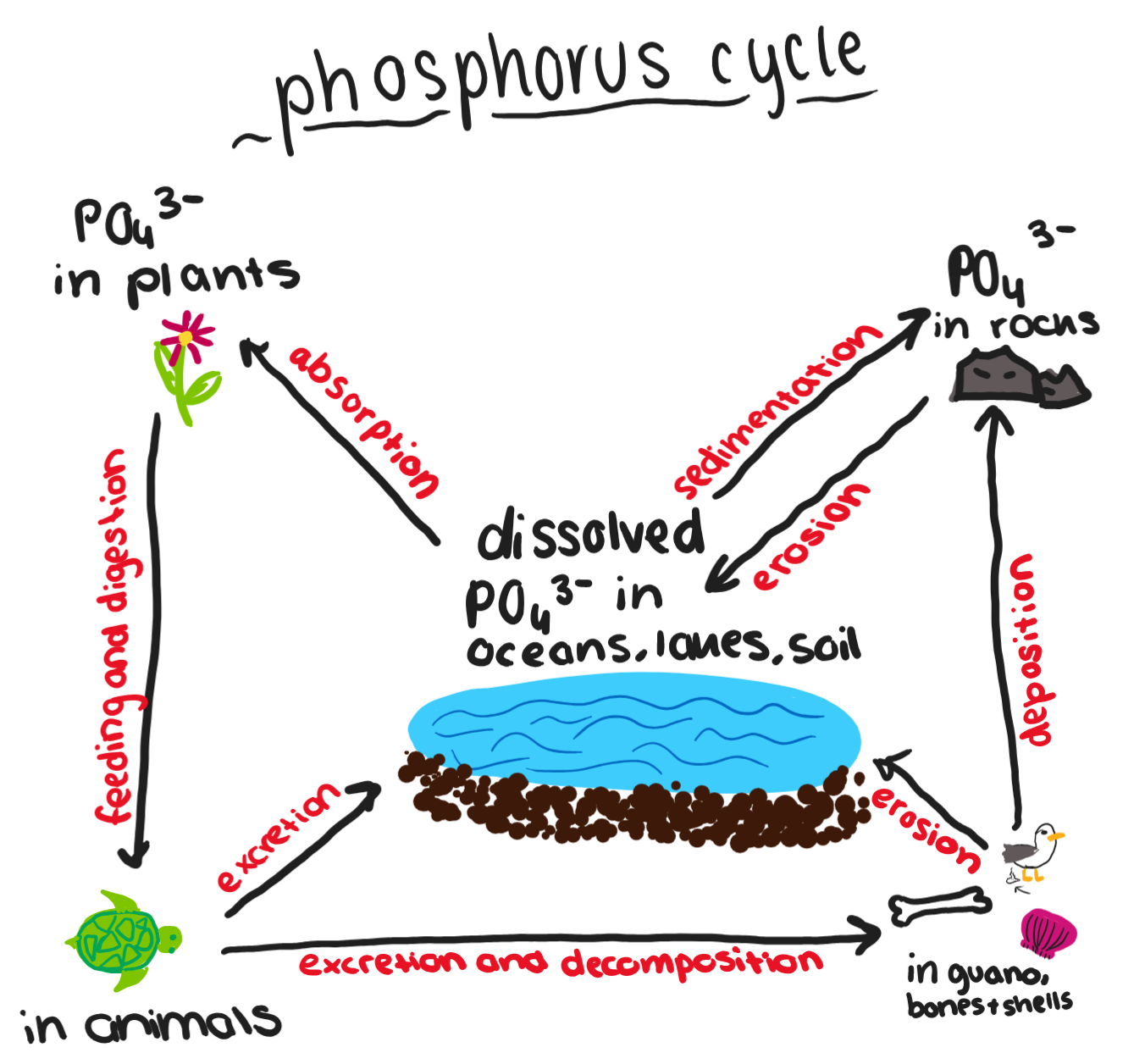
What do organisms need phosphorus for?
ATP
Phospholipids
Nucleic acids
What is guano?
A substance made up of excretory products of some sea birds
What can mycorrhizae between fungi and plants be described as?
Mutualistic
Why is the mycorrhizae between fungi and plants described as mutualistic?
Plant benefits from improved water and inorganic ion uptake provided by the fungi, enabling it to better resist drought
Fungi receives sugars and amino acids from the plant
What is mycorrhizae?
The mutualistic relationship between fungi and the roots of plants
In which the fungi act as extensions of the plant’s root system
Increases surface area of the plant’s roots so that it absorbs water and minerals more readily
Why are fertilisers needed in agricultural systems?
Mineral ions are removed from the soil as they are taken up by crops
Crop is harvested and transported away for consumption
Mineral ions do not return to the soil through decomposition or excretion in same area of land
Fertilisers need to be added to soil to combat loss of mineral ions
What are the two types of fertiliser?
Artificial fertilisers
Natural fertilisers
What are natural fertilisers?
Fertilisers that contain the dead and decaying remains of plants and animals
As well as animal waste such as manure, slurry and bone meal
What are artificial fertilisers?
Fertilisers that are mined from rocks and then converted into different forms
Almost always contain nitrogen, phosphorus and potassium
What are the three main harmful consequences of using nitrogen-containing fertilisers?
Reduced species diversity
Leaching
Eutrophication
How do nitrogen-containing fertilisers reduce species diversity?
Nitrogen-rich soils favour the growth of grasses, nettles and other rapidly growing species
These outcompete many other species, which die as a result
What is leaching?
Rainwater dissolves any soluble nutrients such as nitrate ions
Removes nutrients from the soil and carries them to watercourses such as streams and rivers
What are potential harmful consequences of leaching?
Could prevent efficient oxygen transport in babies (if the watercourses are a source of drinking water)
Could cause stomach cancer in humans (if the watercourses are a source of drinking water)
Can cause eutrophication
Describe the process of eutrophication. (10 marks)
Nitrate ions are leached into watercourses such as streams and rivers
As nitrate ion concentration increases, population size of algae increases
Upper layers of water become densely populated with algae- this is called an ‘algal bloom’
Algae absorbs light and prevents it from reaching water below
Plants at lower depths of water die as they cannot photosynthesise
Population size of saprobionts increases, as they use dead plants as food
Saprobionts respire, decreasing the concentration of oxygen in water
Aerobic organisms like fish die, as there is no oxygen available
Now less competition for anaerobic organisms, so population sizes increase
Anaerobic organisms decompose dead material further, releasing more nitrates and some toxic wastes like hydrogen sulfide