National 5 Chemistry
1/132
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
133 Terms
pH
It is a measure of the number of hydrogen ions that a substance contains- and therefore how acidic or alkaline it is- and runs on a scale of 1 to 14.

Acid
Substance with a pH of less than 7, formed when a non-metal oxide reacts with water. It contains a higher proportion of hydrogen ions than hydroxide ions. Examples include vinegar, hydrochloric acid and nitric acid.
Base
Substance that can neutralise an acid. They come in three forms: metal hydroxide, metal oxide, and metal carbonate. Examples include calcium carbonate, sodium hydroxide and oven cleaner.
Alkali
Substance with a pH above 7, formed when a metal oxide reacts with water. It contains a greater proportion of hydroxide ions than hydrogen ions. Examples include sodium hydroxide and potassium hydroxide.
Neutral
A _____ substance has a pH of 7, and has an equal balance of hydrogen and hydroxide ions. Examples include water and sodium chloride.
Salt
Product of a neutralisation reaction. It is named after the parent acid and base, e.g, sodium chloride, potassium nitrate. Insoluble versions can also be created in precipitation reactions.
Neutralisation
Chemical reaction between an acid and base. Products always include water and a salt, but can also include carbon dioxide in the case of the base being a metal carbonate.
Precipitation
A reaction where a solid is formed. Two soluble salts can react with one another in such a reaction to produce an insoluble salt and a soluble salt, e.g, magnesium iodide + silver nitrate= magnesium nitrate (aq) + silver iodide (s).

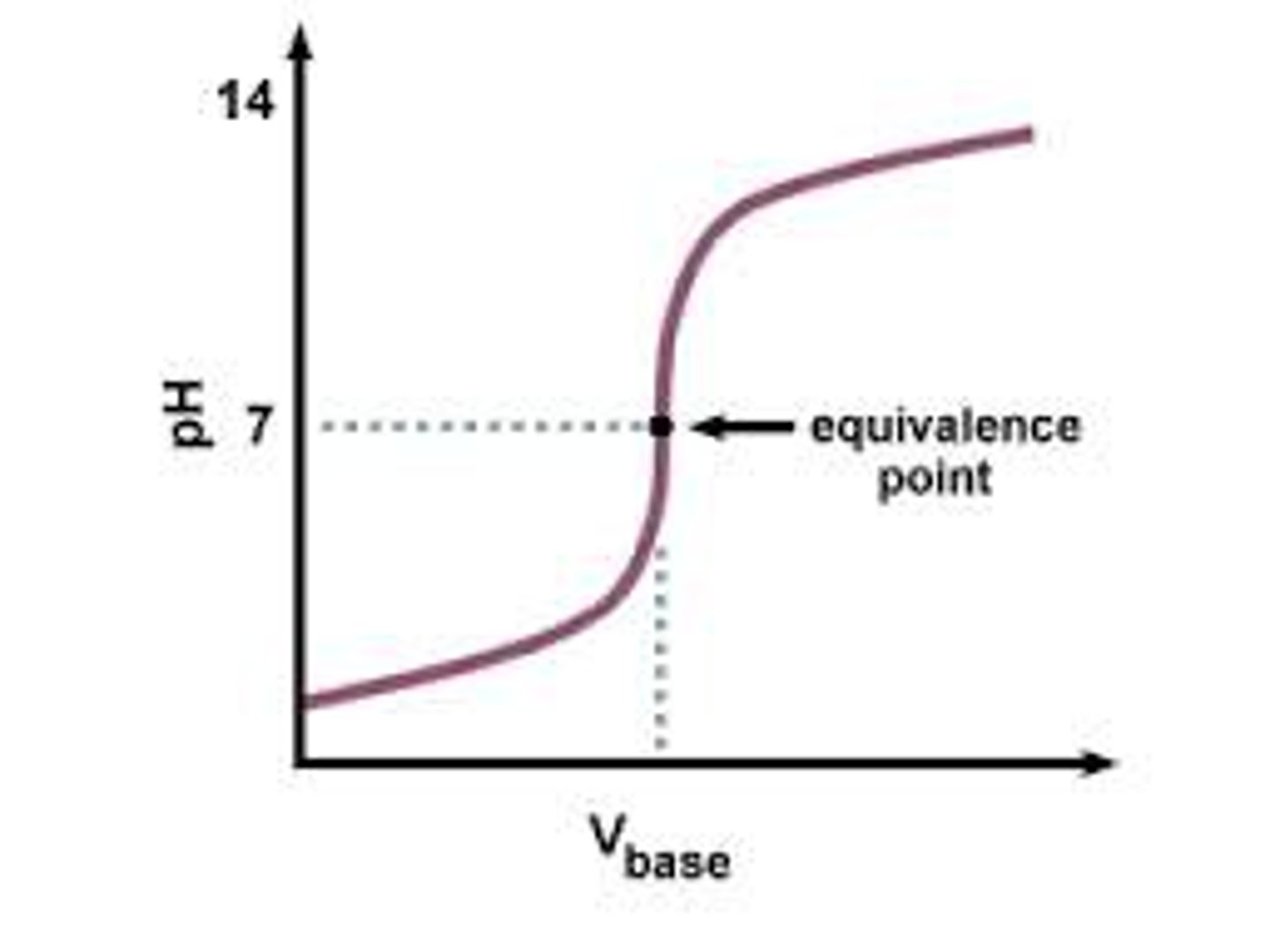
Titration
Accurate method of measuring volumes of acids and alkalis necessary to neutralise one another. A practical experimentation technique, it uses a burette, pipette and conical flask, with known volumes of acid and alkali.
Concordant
Titration results are _______ if they are within 0.2 cm3 of one another.
2 mol l-
Calculation based on results of a titration, e.g, 20 cm3 of nitric acid was used to neutralise 20cm3 of 2 mol l-1 sodium hydroxide. Assuming that one mole of the acid is equal to one mole of the alkali, what is the concentration of the nitric acid?
Spectator ion
An ion that does not undergo chemical change, but is present for the duration of a chemical reaction.
Hydroxide ion
Ion found in higher ratios compared to hydrogen ions in bases.

Hydrogen ion
Ion found in higher ratios compared to hydroxide ions in acids.
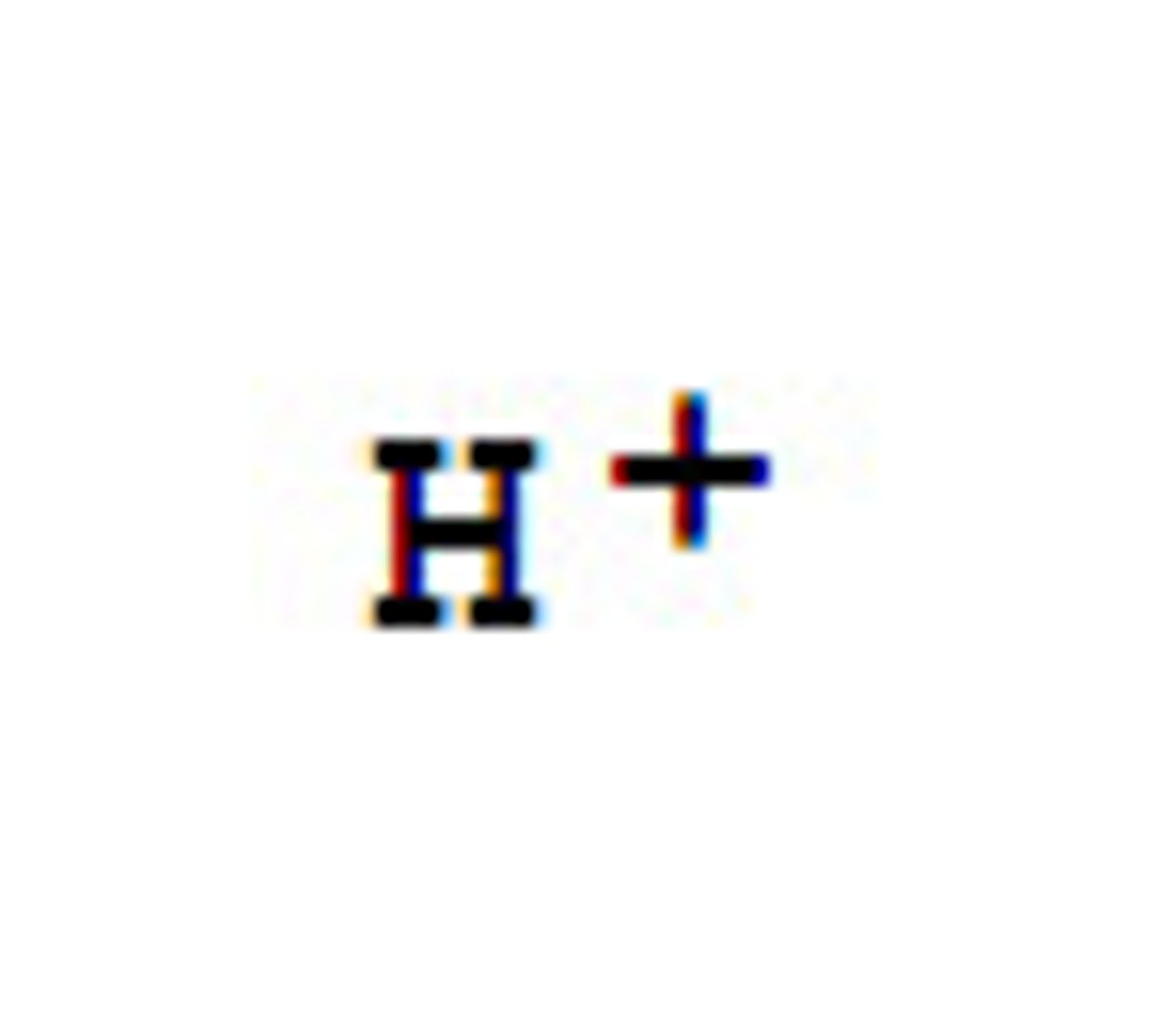
Metal carbonate
A type of base that produces water, carbon dioxide and a salt in a neutralisation reaction with an acid.
Metal oxide
Compound that produces an alkali when it reacts with water.
Non-metal oxide
Compound that produces an acid when it reacts with water oxide
Indicator
Substance used to tell us when neutralisation has taken place in a titration experiment.
End-point
The finishing point of a chemical reaction.
Precipitate
General term used for a solid substance created in a precipitation reaction.
Soluble
Term used to describe a substance if it dissolves in water.

Insoluble
Term used to describe a substance if it is incapable of dissolving in water.

Burette
Piece of apparatus used during a titration experiment. It contains a volume of acid or alkali that is slowly added to a conical flask containing a known volume of acid or alkali, until neutralisation takes place.

Pipette filler
Device used to safely fill a pipette for a titration reaction.

Pipette
Piece of equipment used during titration. It is used to measure a known volume of acid or alkali, which can then be transferred to a conical flask.

Rough titre
A titration 'test-run' which can tell us roughly how much acid and alkali are required to neutralise each other. It is never used as part of results or calculations.
Collision Theory
Theory based upon the idea that atoms react with one another when they crash into one another with adequate energy source.
Surface area
One of several factors that can increase reaction rate. The reactant has more surfaces exposed so ________ is greater, and therefore is a greater surface on which molecules are exposed—and so collide and react more frequently.
High temperature
One of several factors that can increase reaction rate. The reactant molecules are supplied with more energy in the form of heat, and so move more erratically and at greater speed - so collide with more ease.
Higher concentration
One of several factors that can increase reaction rate. In a solution, there are more molecules contained in the solvent, and so collisions are more likely to occur.
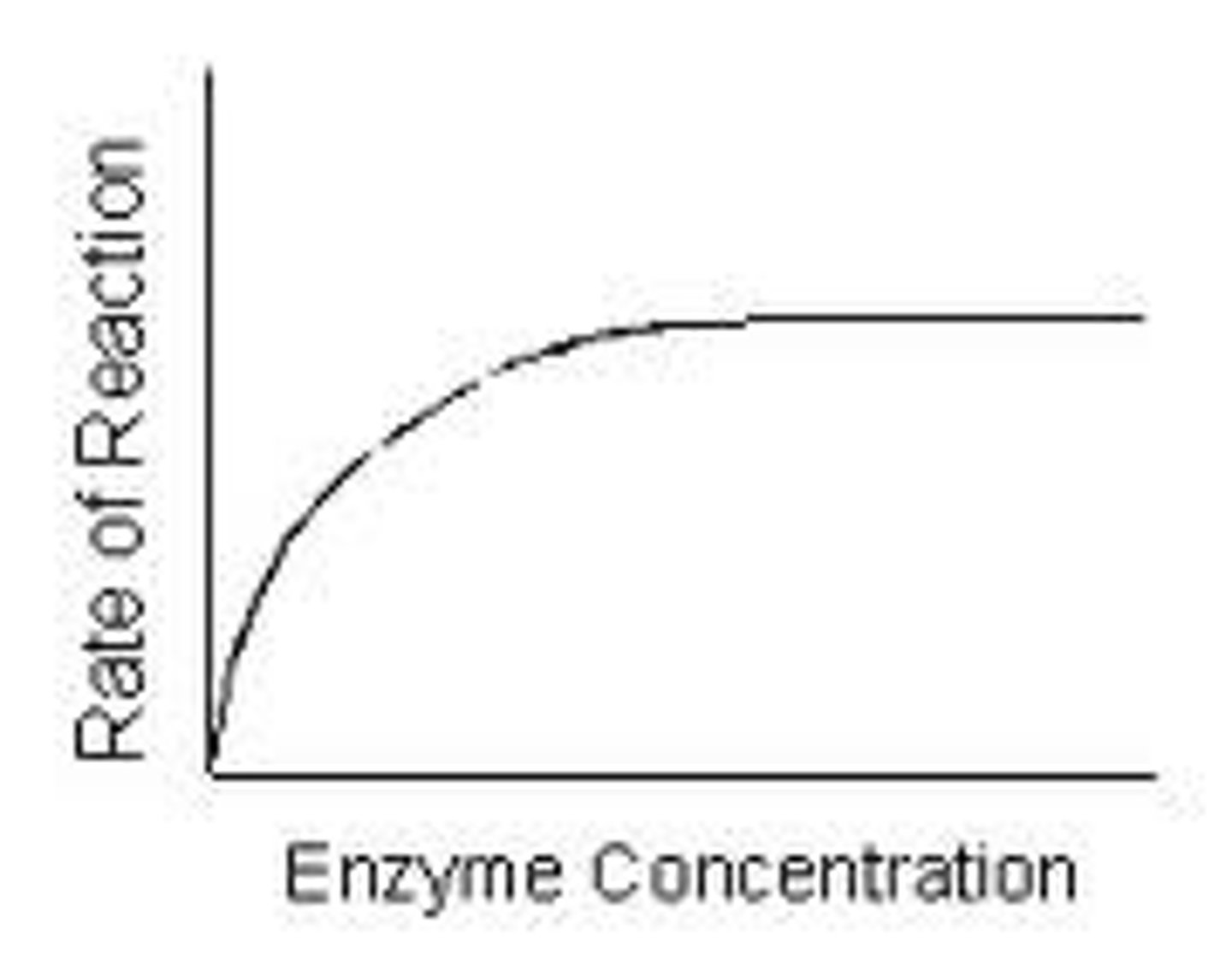
Catalyst
A substance that, when used in a chemical reaction, increases reaction rate without being used up itself. These substances create an alternative path for the molecules to react with one another. Examples include the use of platinum in the Ostwald process.
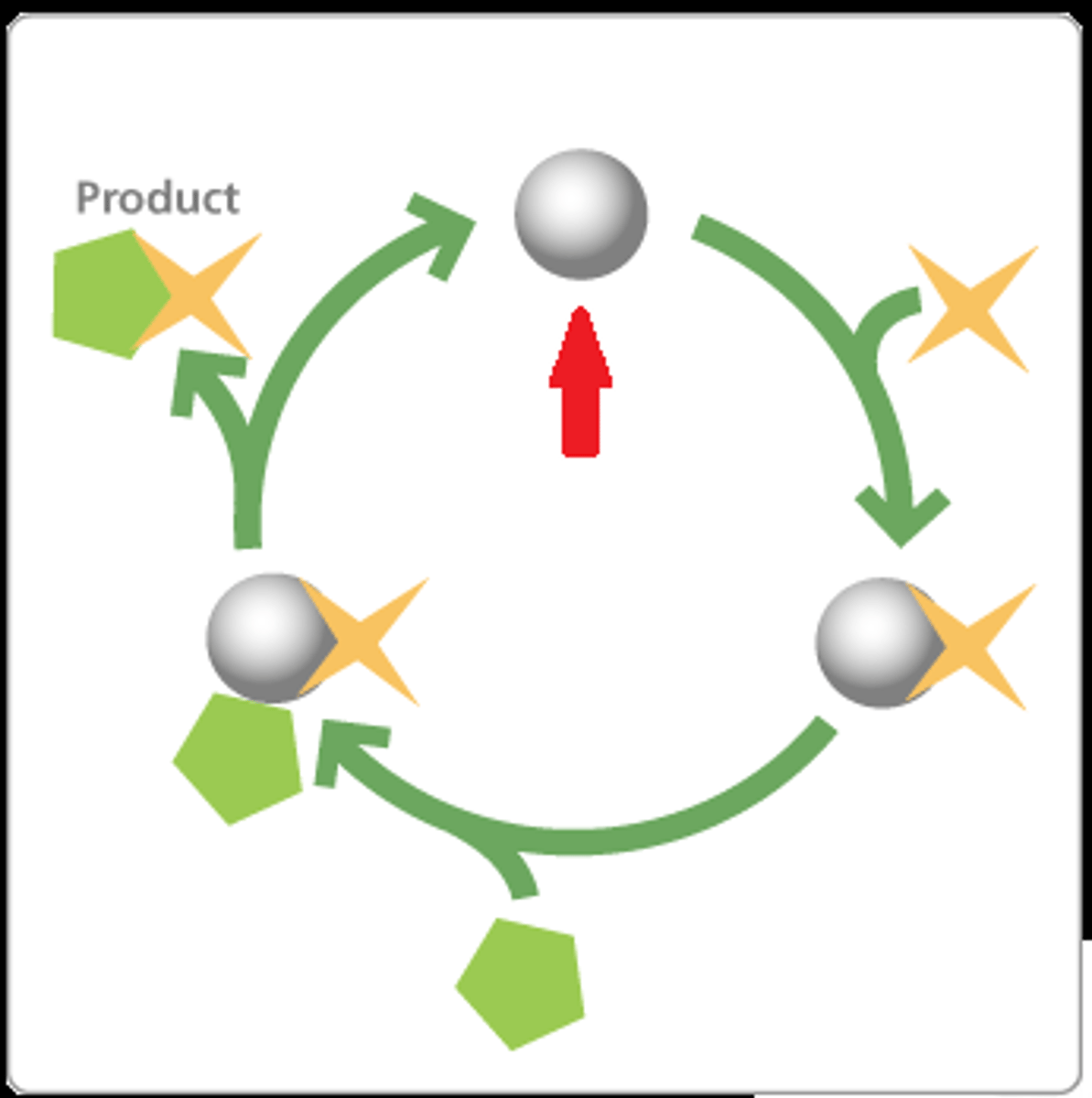
Reaction rate
The speed at which a chemical reaction occurs.
Atom
The smallest particle into which matter can easily be broken into. They contain smaller particles: protons and neutrons in the nucleus, and electrons orbiting the nucleus. They contain an equal ratio of electrons to protons, and so have no charge.
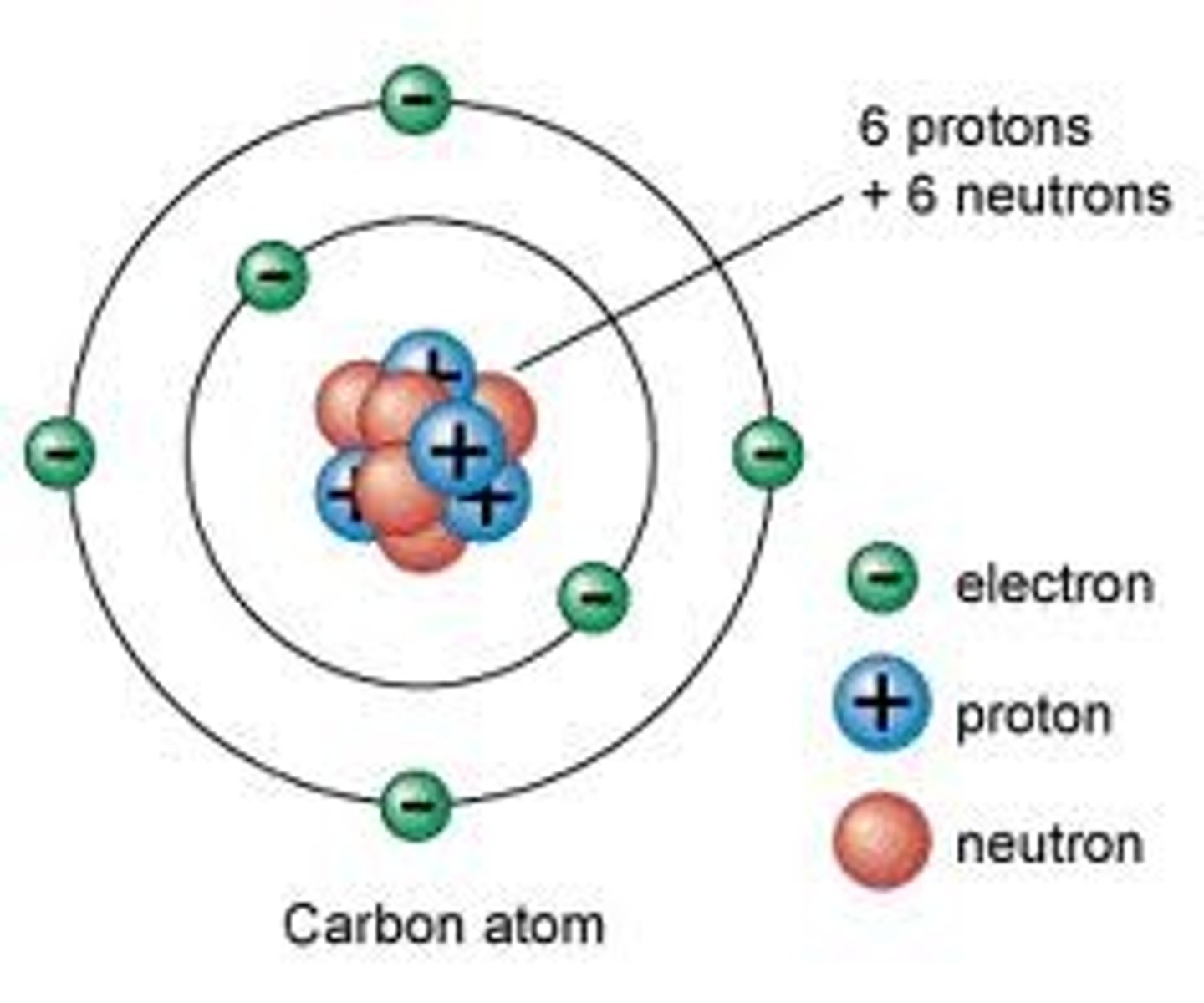
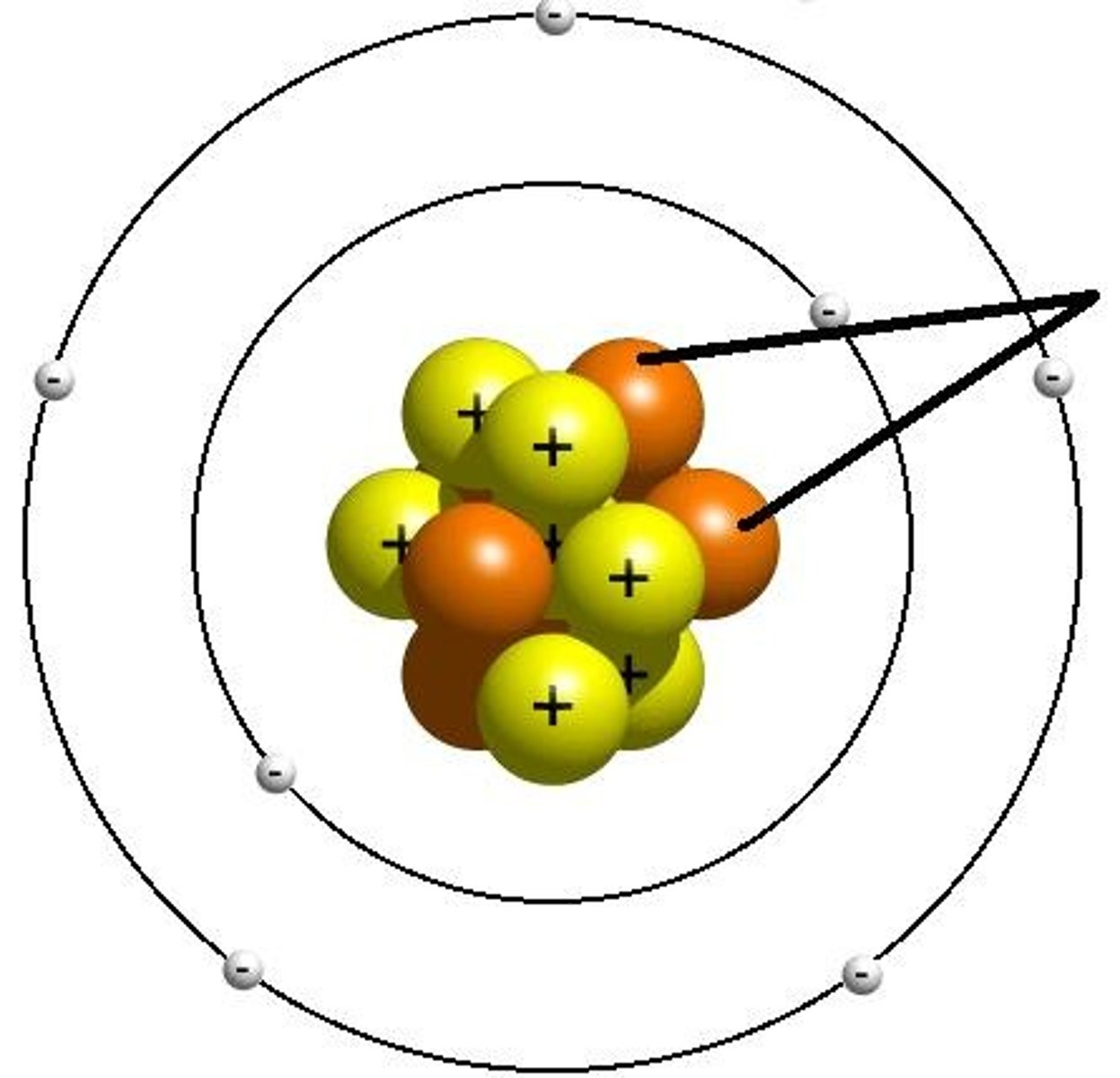
Proton
Sub-atomic particle with a charge of 1+ and a mass of 1 amu. Found in the nucleus.
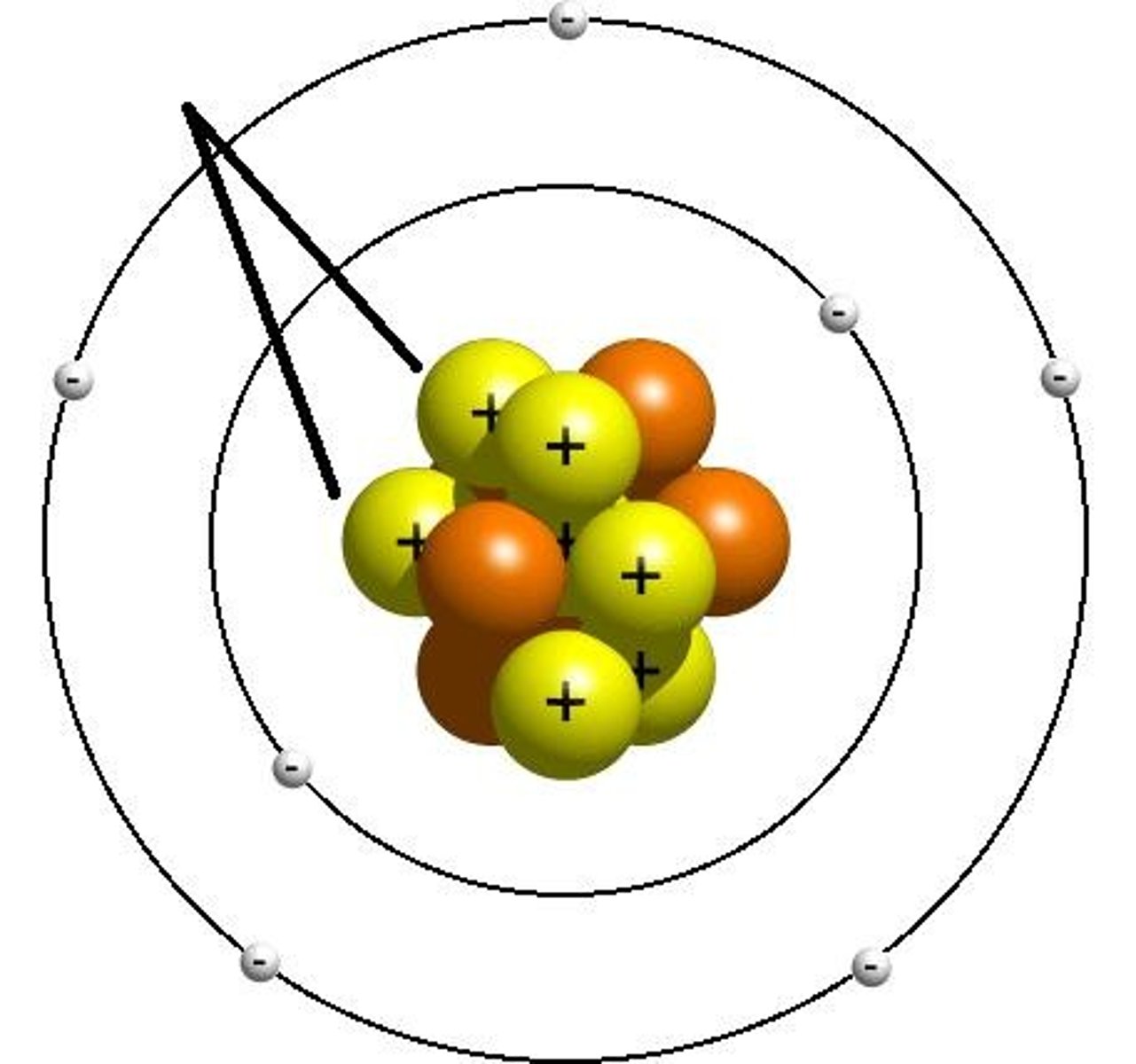
Electron
Sub-atomic particle with a charge of 1- and a mass of nearly nothing. Orbits the nucleus and groups itself into energy levels.
Neutron
Sub-atomic particle with no charge and a mass of 1 amu. Found in the nucleus.
Electron arrangement
The way in which electrons group themselves into shells, or energy levels.
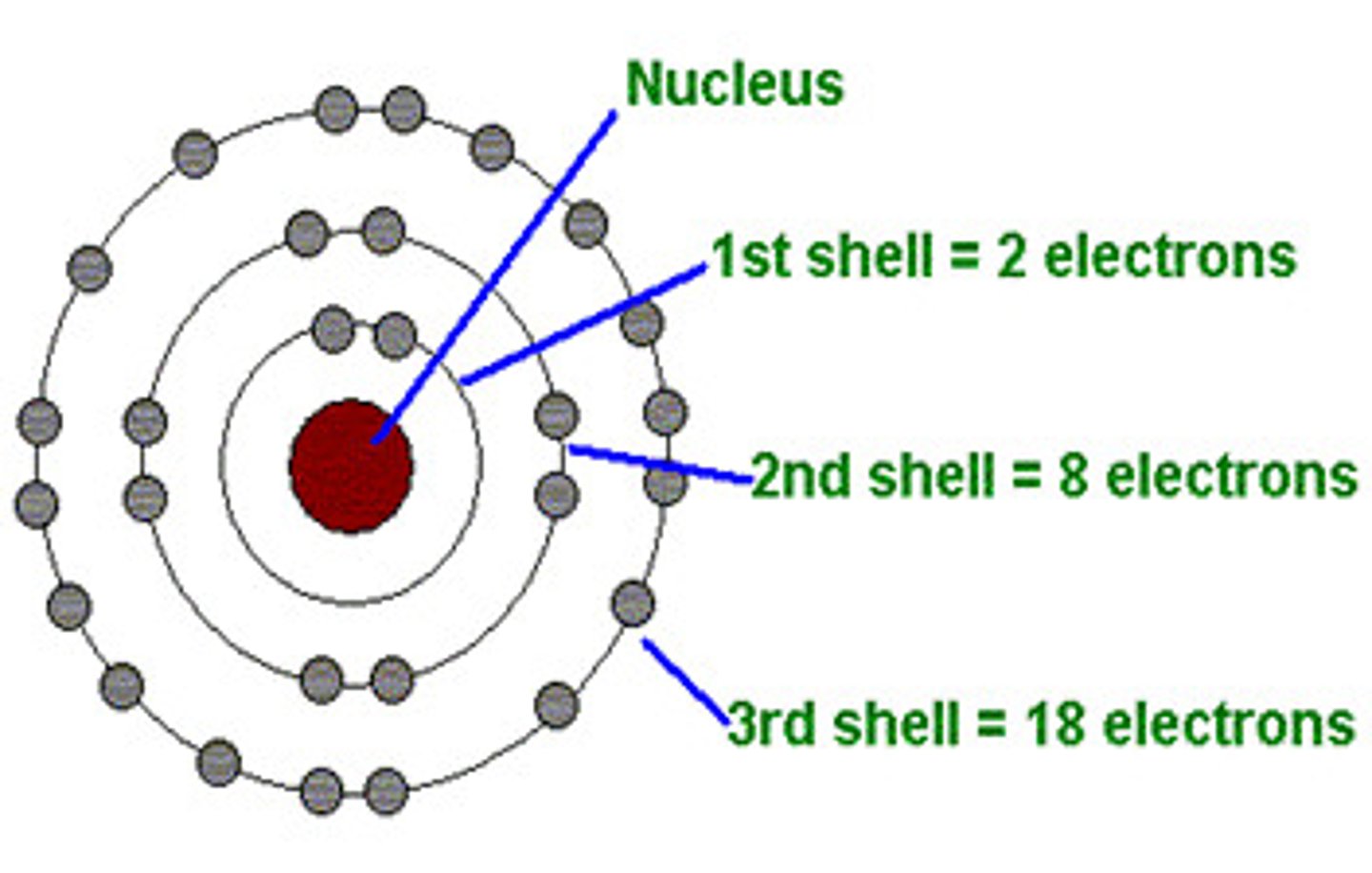
Energy level
Term used to describe a 'layer' of electrons inside the atom. They can only hold a certain quantity of electrons, and are also known as electron shells.
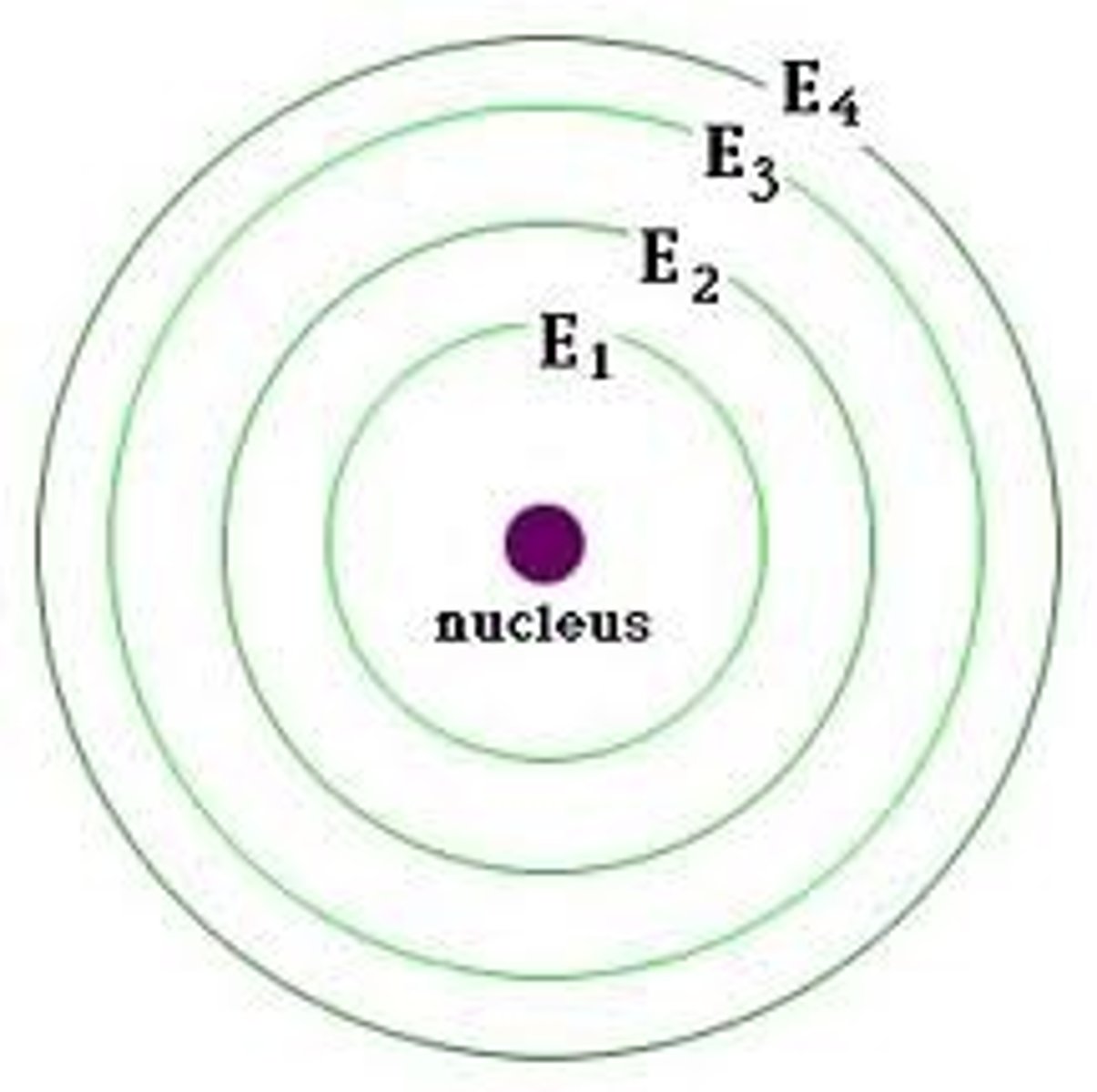
Orbital
Sub-sections of electron shells. Each holds two electrons.
Ion
An atom that has lost or gained electrons and so now carries a charge.
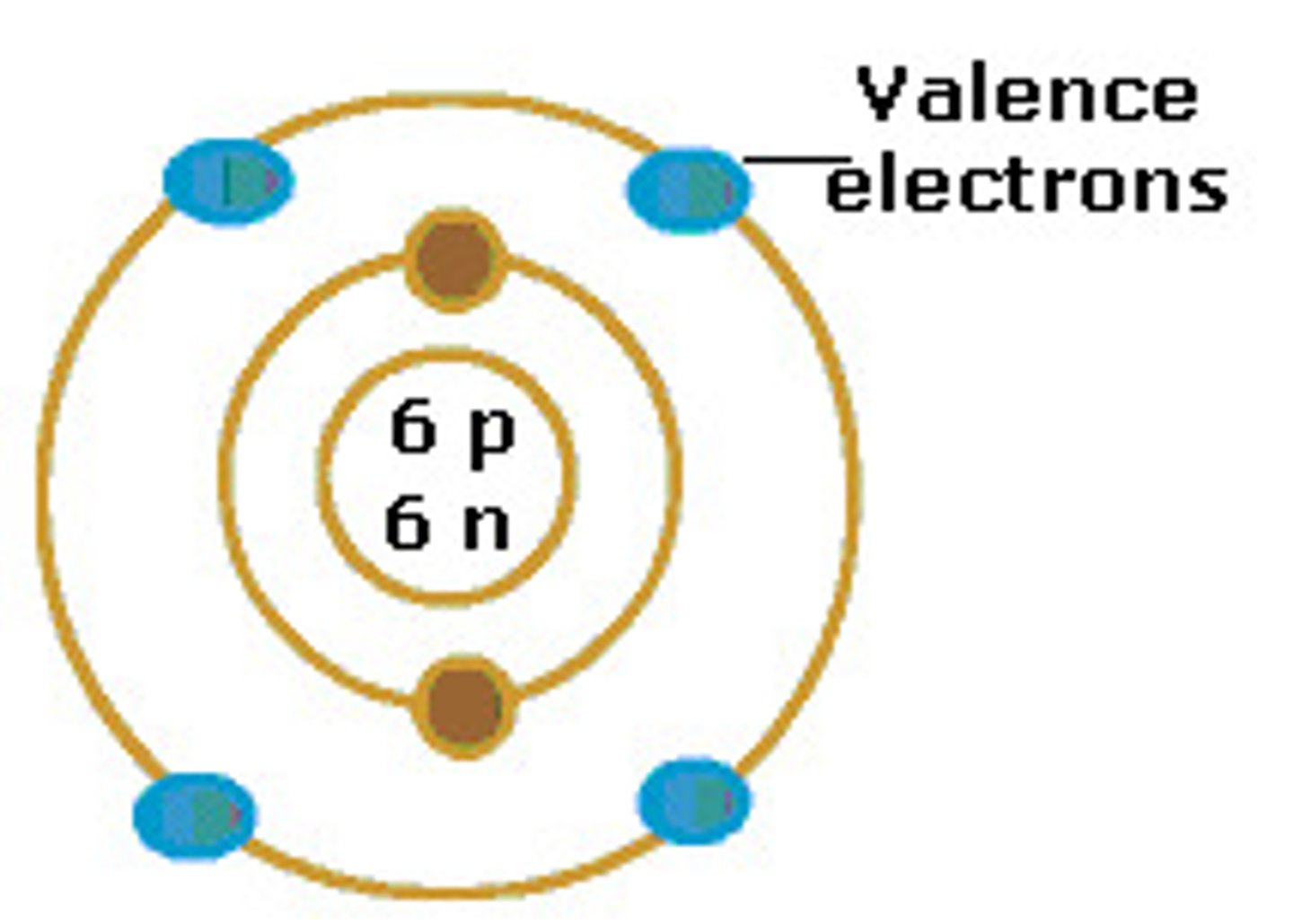
Atomic number
Number representative of the quantity of electrons and protons that an atom has. Corresponds to an element's place on the periodic table.

Mass number
Number representative of the collective number of protons and neutrons in an atom.
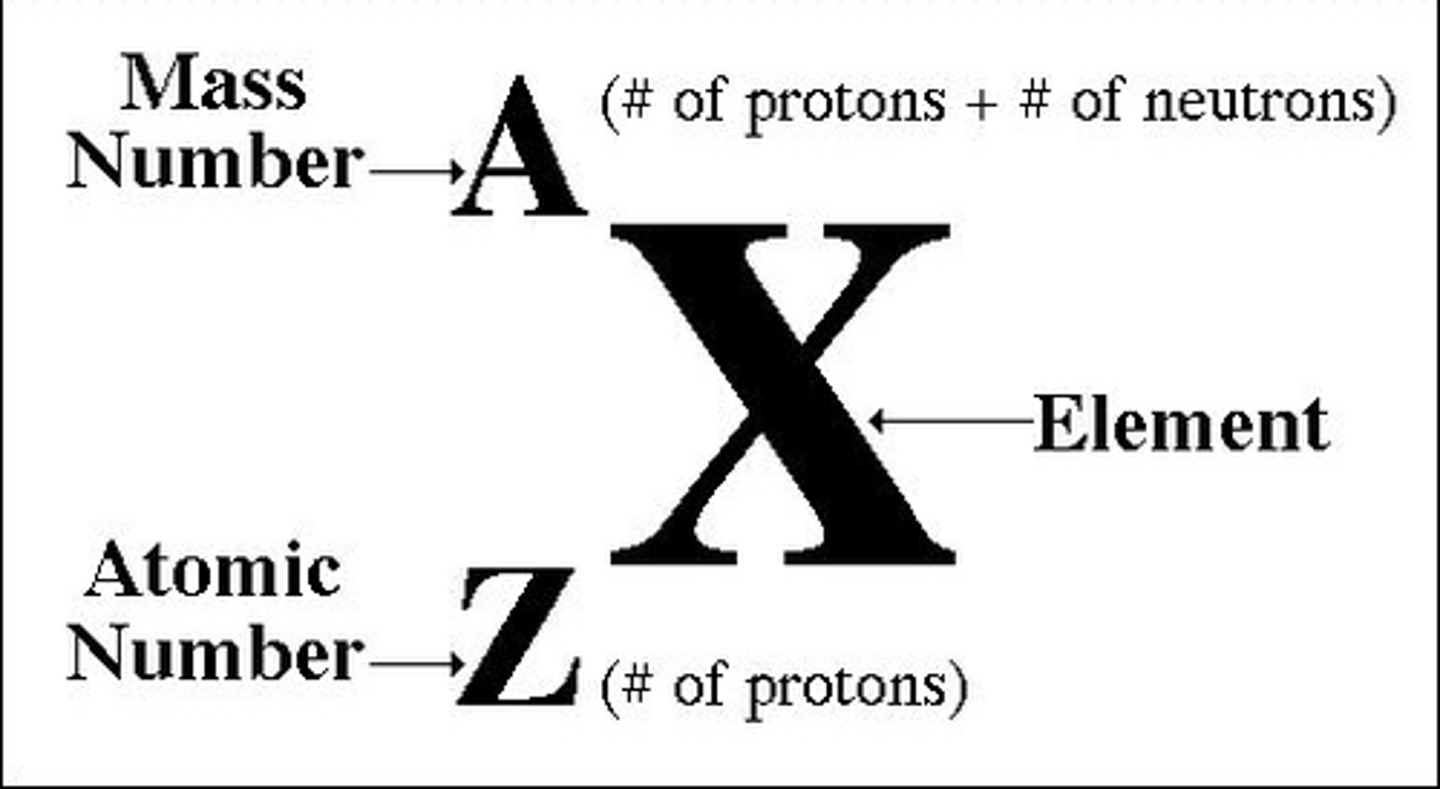
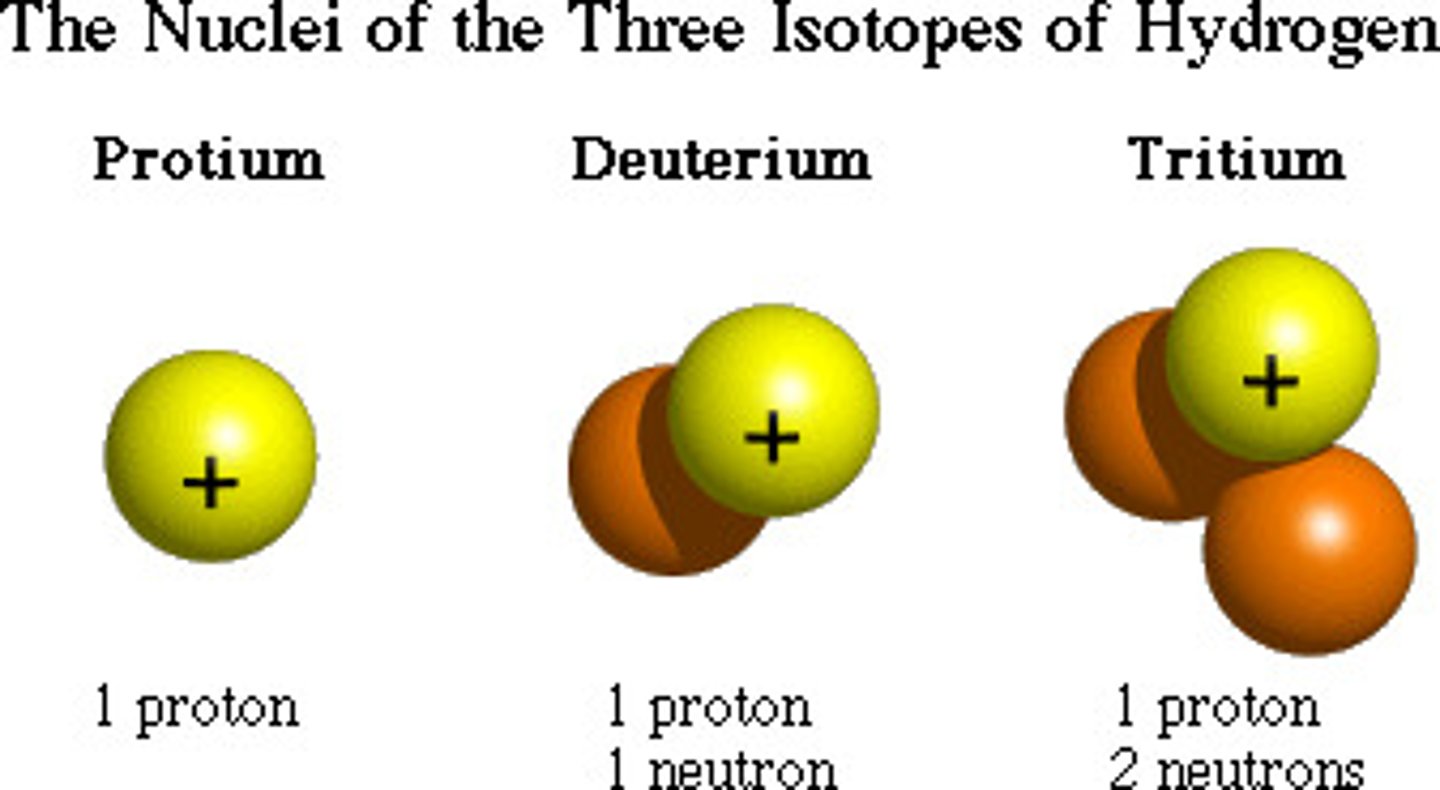
Isotopes
Two atoms of the same element with different mass numbers.
Nuclide notation
A way we can show the charge, mass and atomic numbers of an atom or ion in one diagram. I like to remember it anticlockwise from the top-left like the make-up company MAC: Mass number, Atomic number, then Charge.
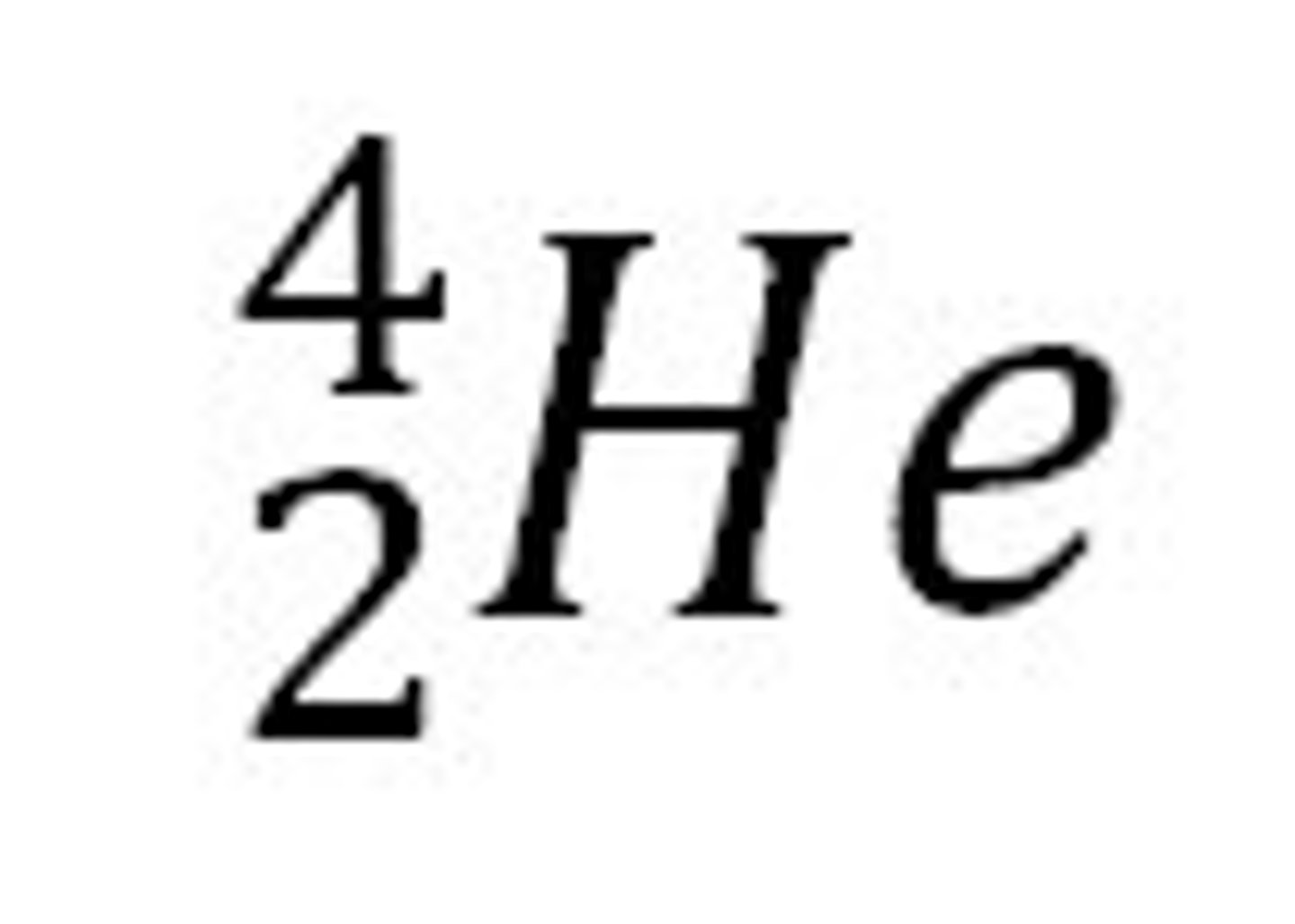
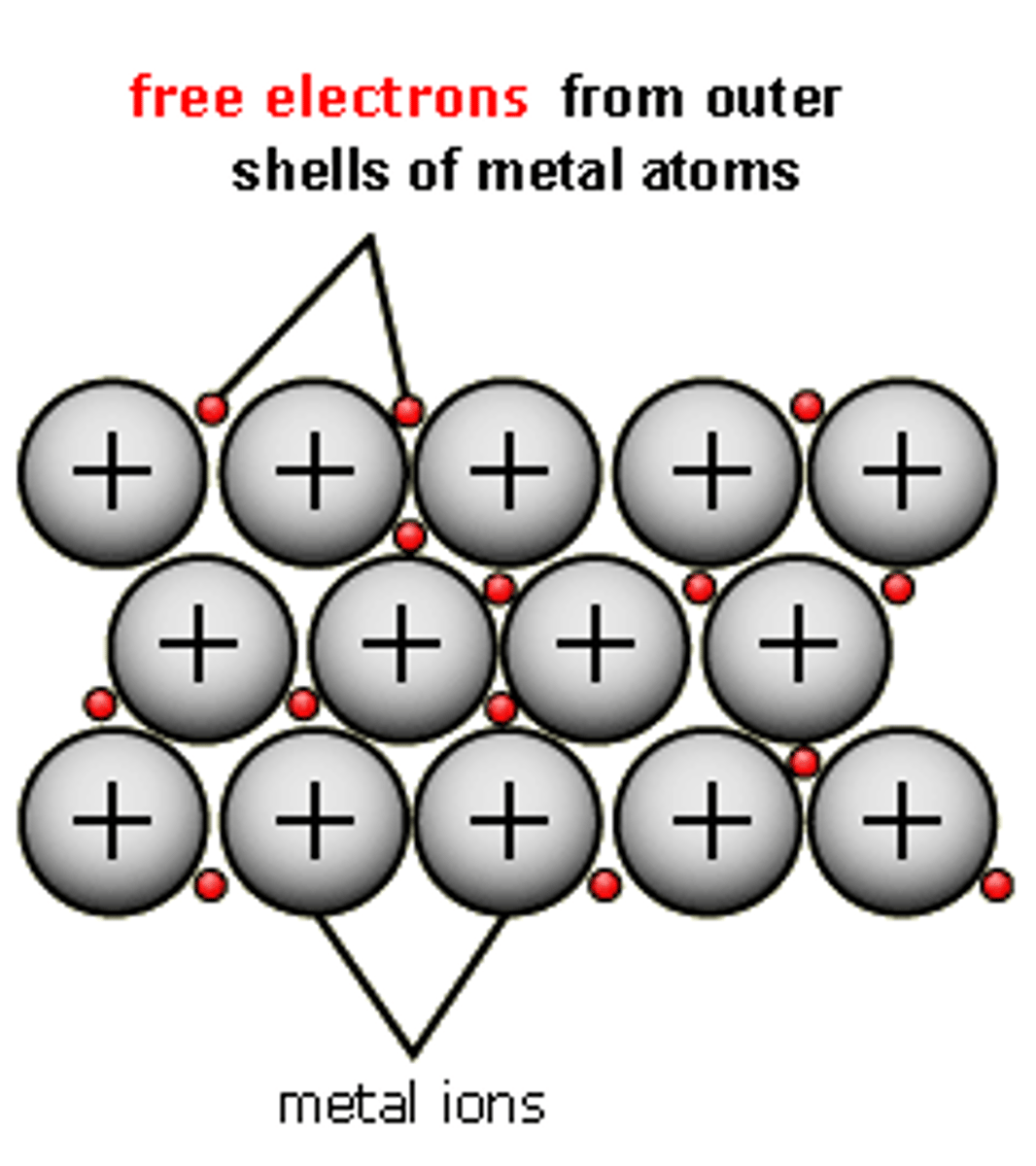
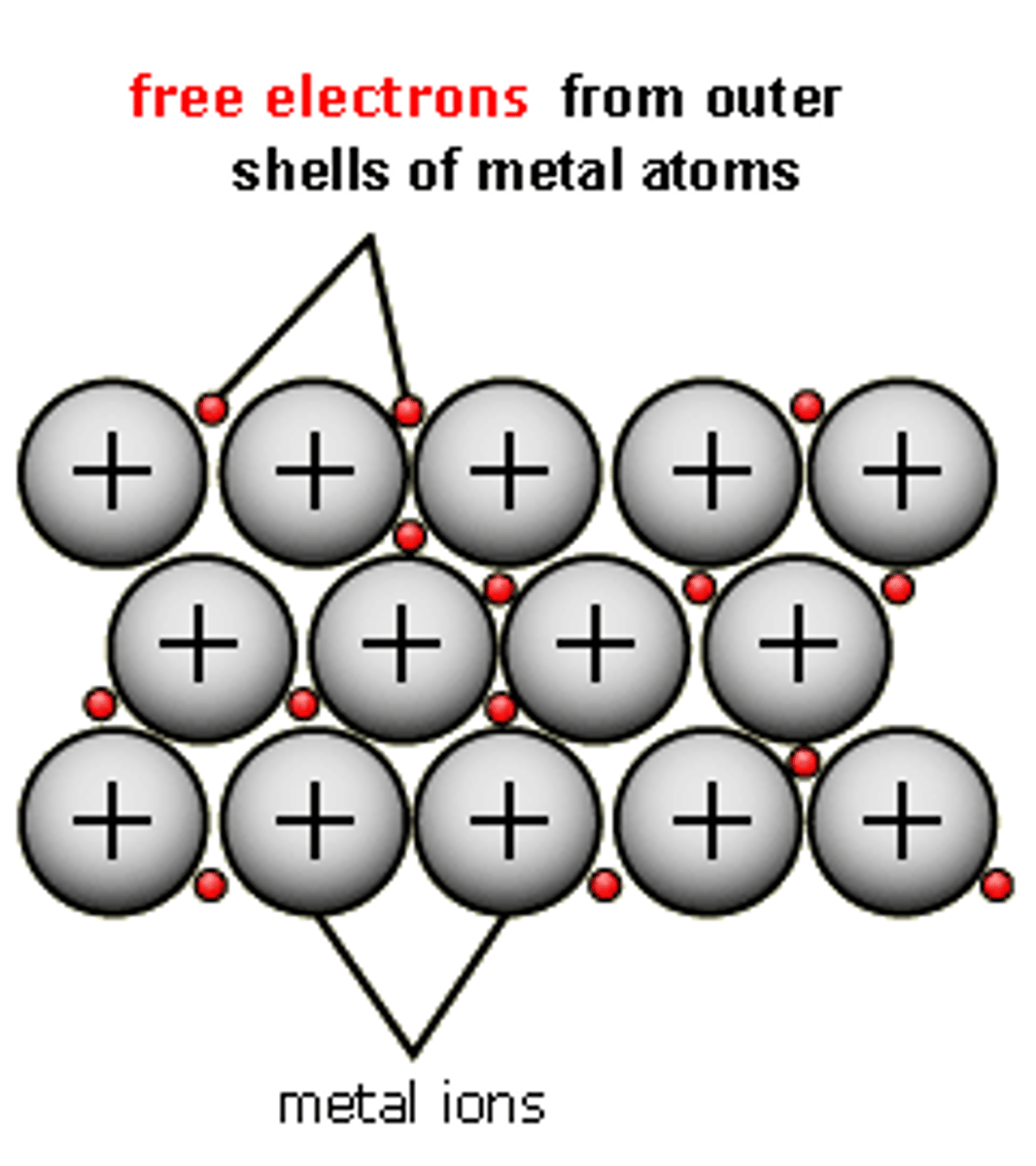
Metallic Bond
The electrostatic attraction between the lattice of positive metal ions and their delocalised electrons.
Ionic bond
The strong electrostatic attraction between a non-metal and a metal ion for each other. (Lost electrons of the metal are picked up by the non-metal.)
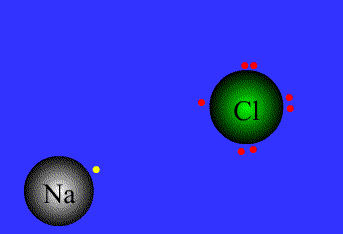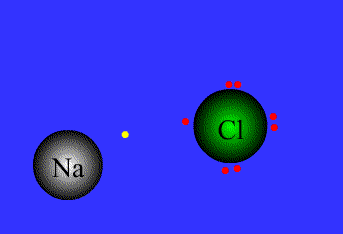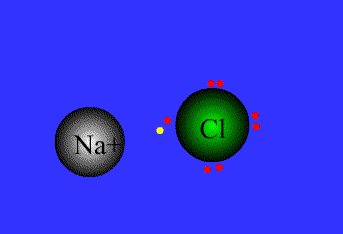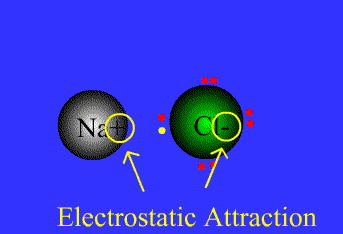
Covalent bond
The strong electrostatic attraction between two non-metal atoms and a shaired pair/shared pairs of electrons.
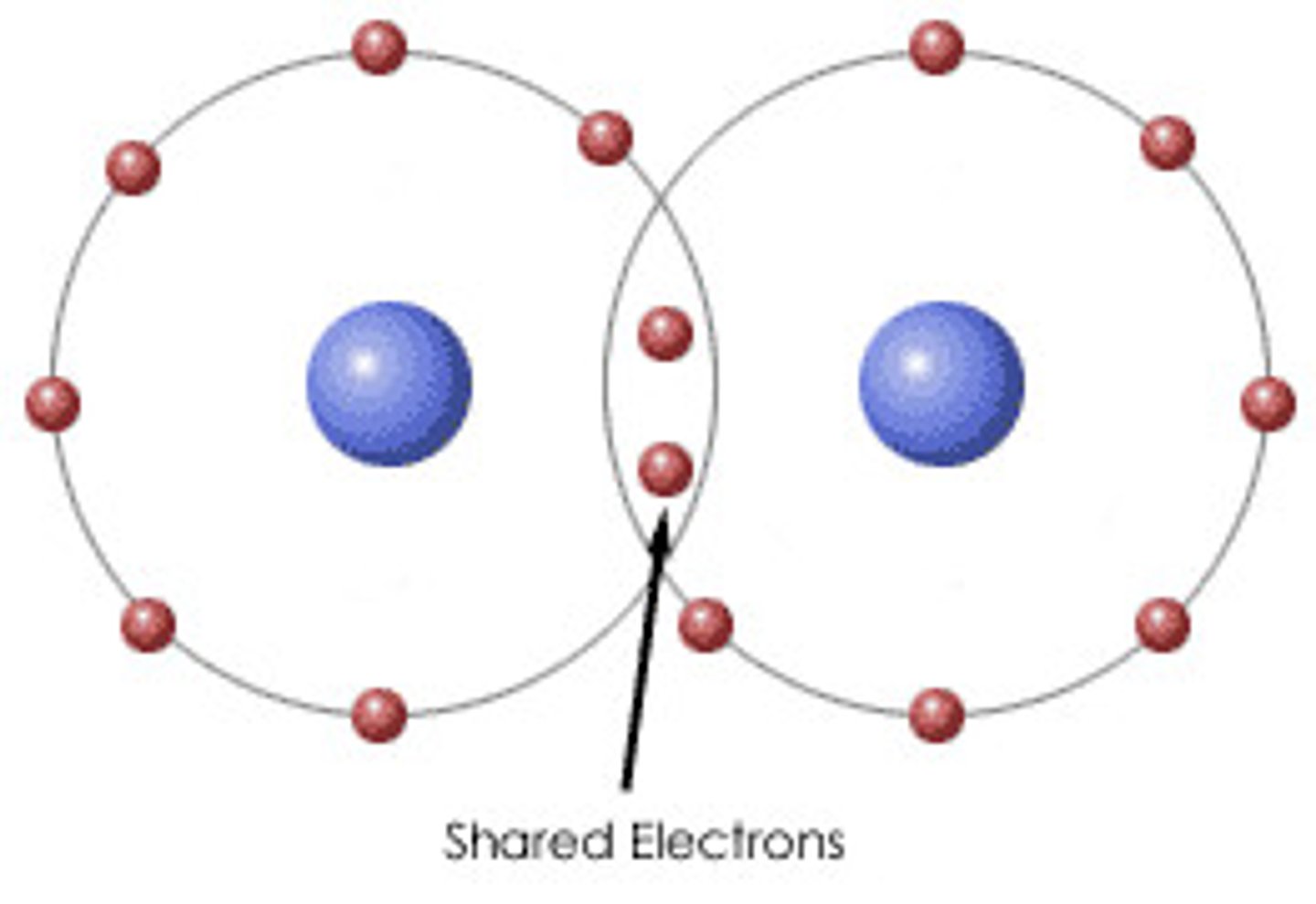
Valency
The number of bonds that is able to be made by atoms of an element.
Metallic lattice
Structure formed in metallic bonding. It is able to conduct electricity due to delocalised electrons and has high melting and boiling points.
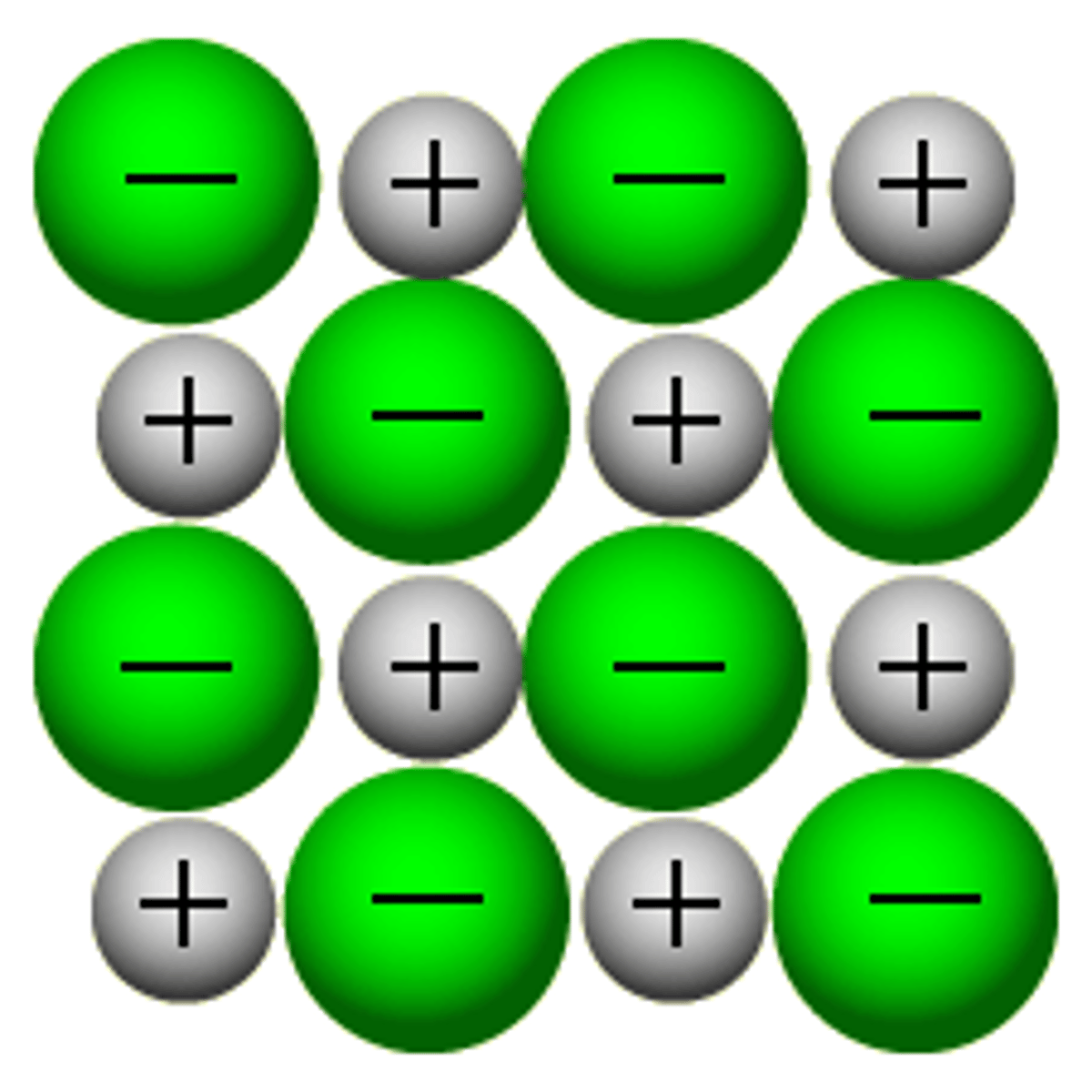
Ionic lattice
Structure formed by ionic bonding. It will not conduct electricity unless it is molten or dissolved, as then its ions are free to move. These structures tend to have high melting and boiling points.
Covalent molecular
One form of covalent compound. It has low melting and boiling points due to the fact that the molecules are held together by only weak forces of attraction, not bonds, that break easily. No covalent compound, excluding graphite, conducts electricity.

Covalent network
One form of covalent compound. It has high melting and boiling points due to the strong bonds between molecules and does not generally conduct electricity.

Relative atomic mass
The average mass of an element, taking into account the relative abundance of its isotopes.
Gram formula mass
Measurement of how many grams of an element would be present in one mole of that element.
Product
The name given to a substance formed in a chemical reaction.
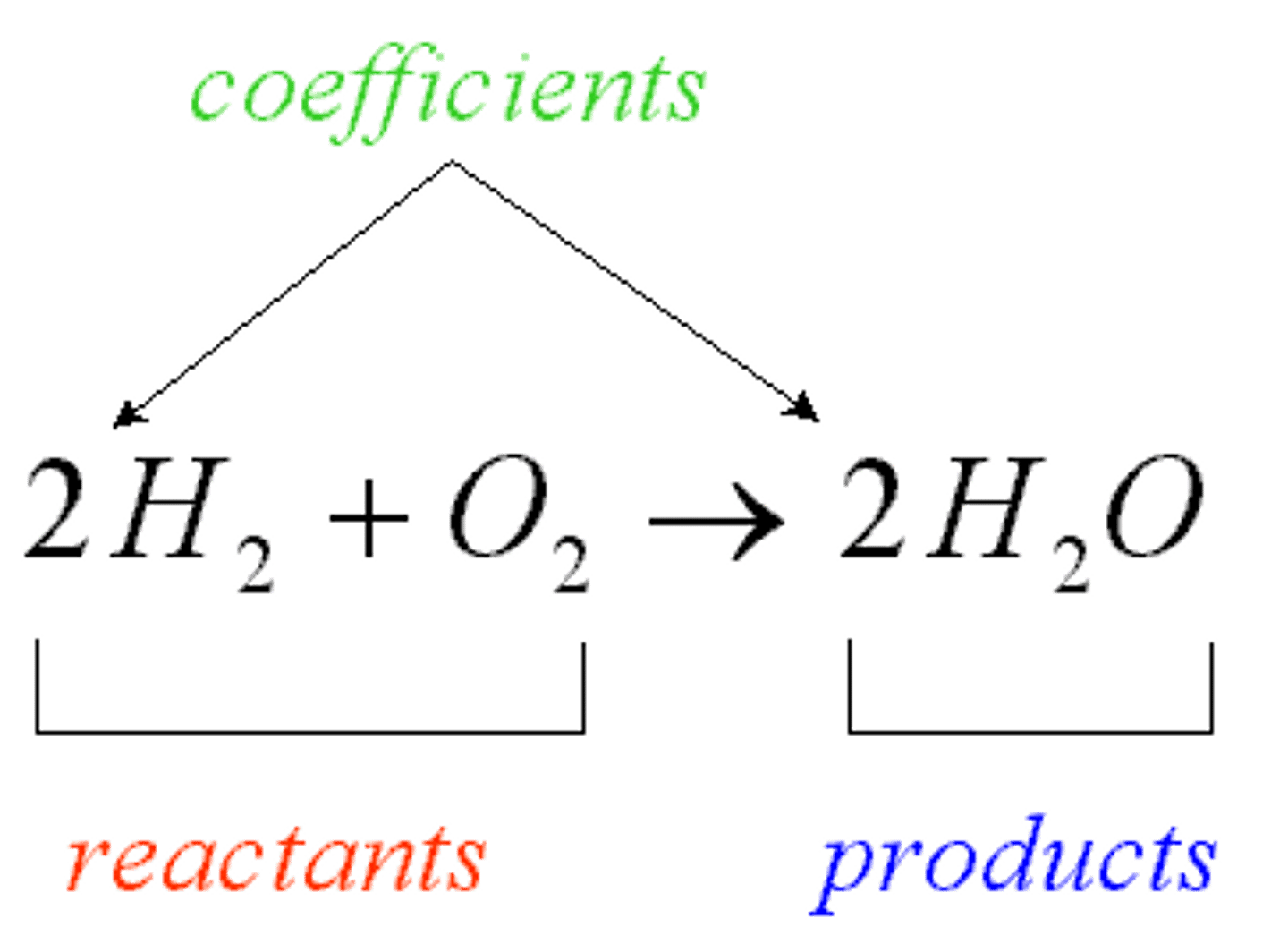
Reactant
Name given to a substance that reacts within a chemical reaction.
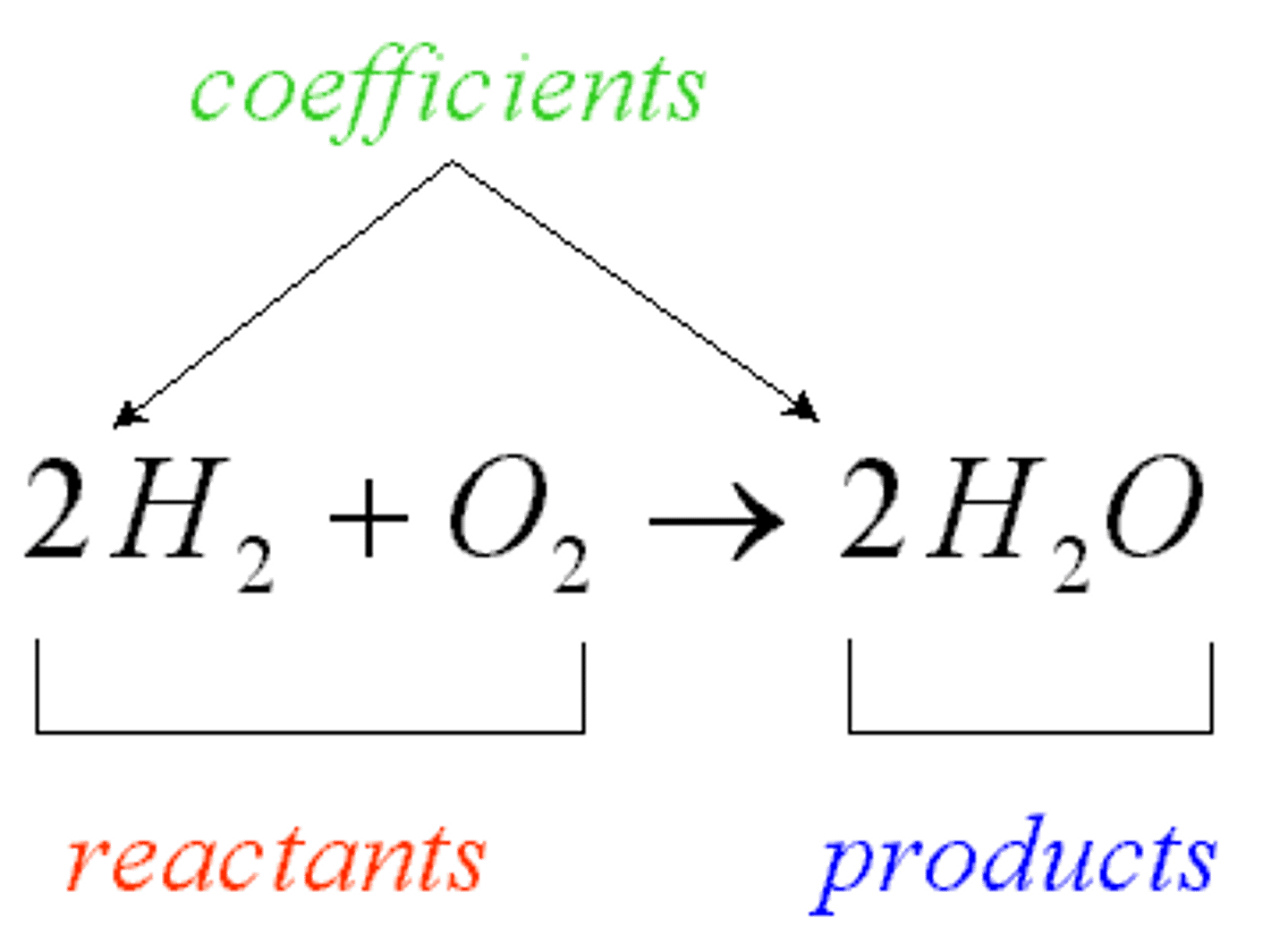
Exothermic
A reaction that gives off energy into its surroundings is _____. E.g, combustion.
Endothermic
A reaction that takes in energy from its surroundings is ______. E.g, a cool pack.

Alkali metals
Name given to group one of the periodic table. They are extremely unstable and reactive.

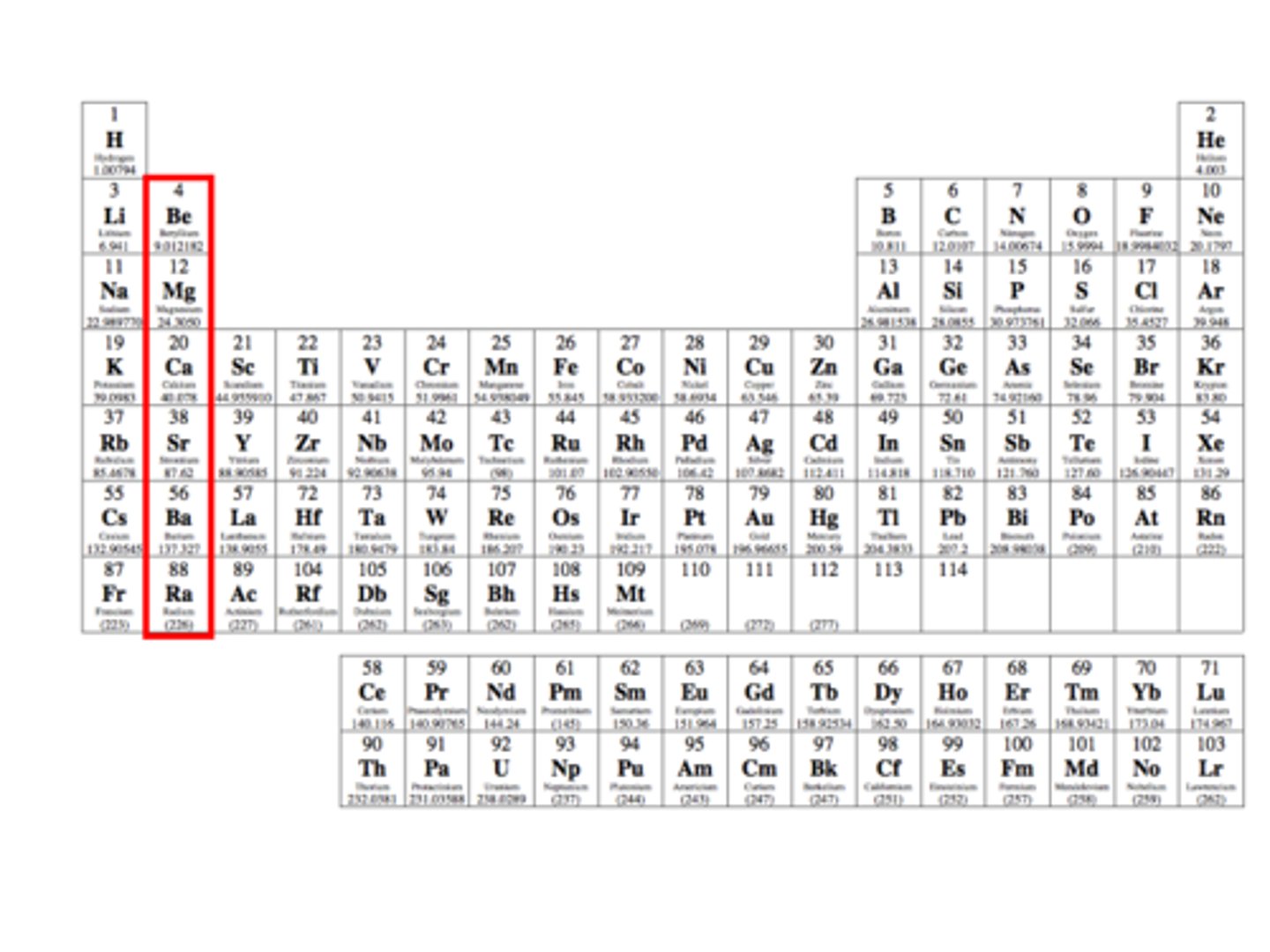
Alkali earth metals
Name given to group two of the periodic table.
Halogens
Name given to group seven of the periodic table.
Noble gases
Name given to group eight of the periodic table. These elements are all stable and so do not make bonds. They are unreactive.

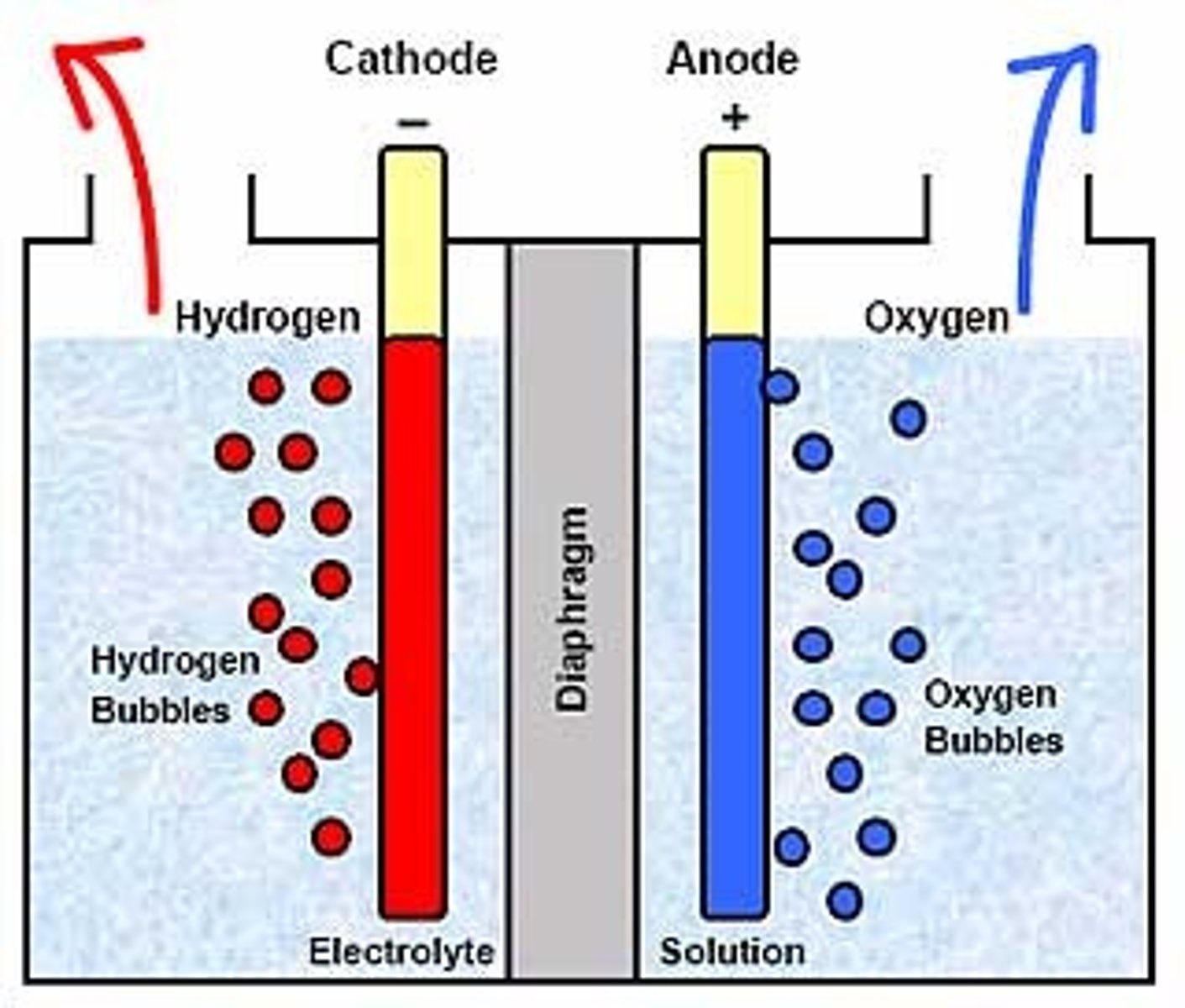
Electrolysis
The breaking down of an ionic compound using electricity.
Molecule
Group of atoms held together by covalent bonds.
Diatomic
Term that describes an element that exists on its own in pairs, such as Iodine, Bromine, Chlorine, Fluorine, Oxygen, Nitrogen and Hydrogen.

Linear
A shape of molecule as seen:
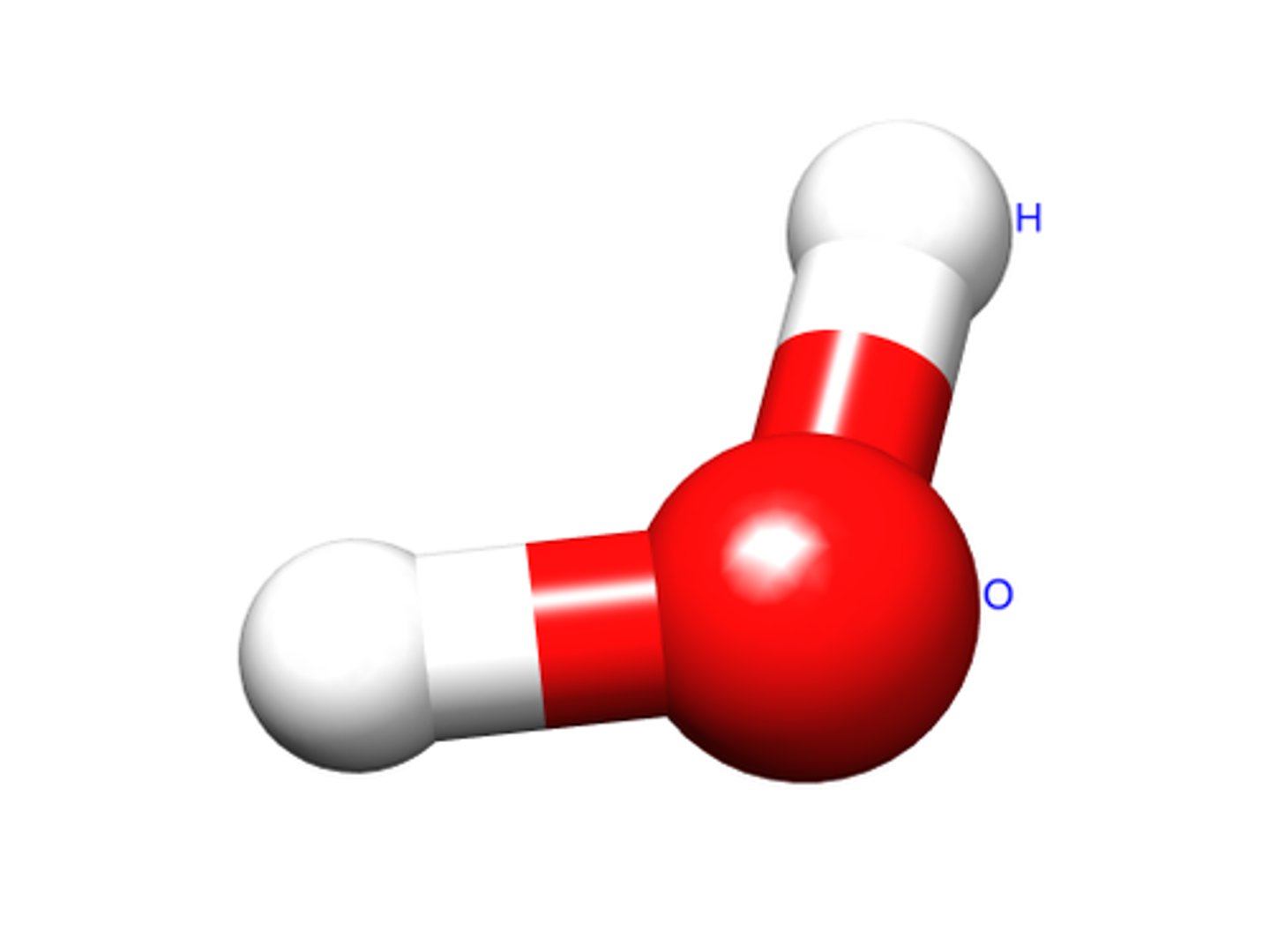
Angular
A shape of molecule as seen:

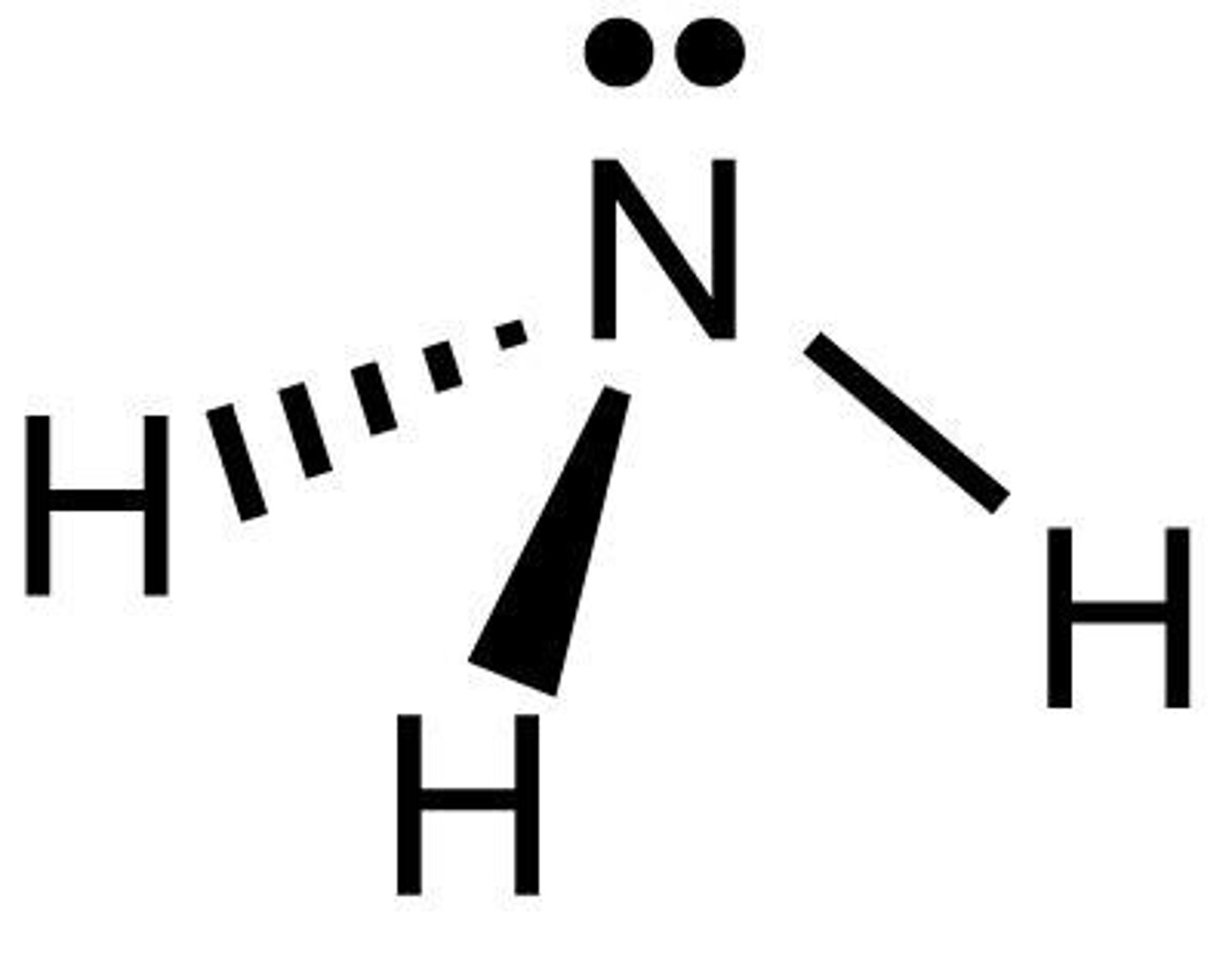
Trigonal pyramidal
A shape of molecule as seen:
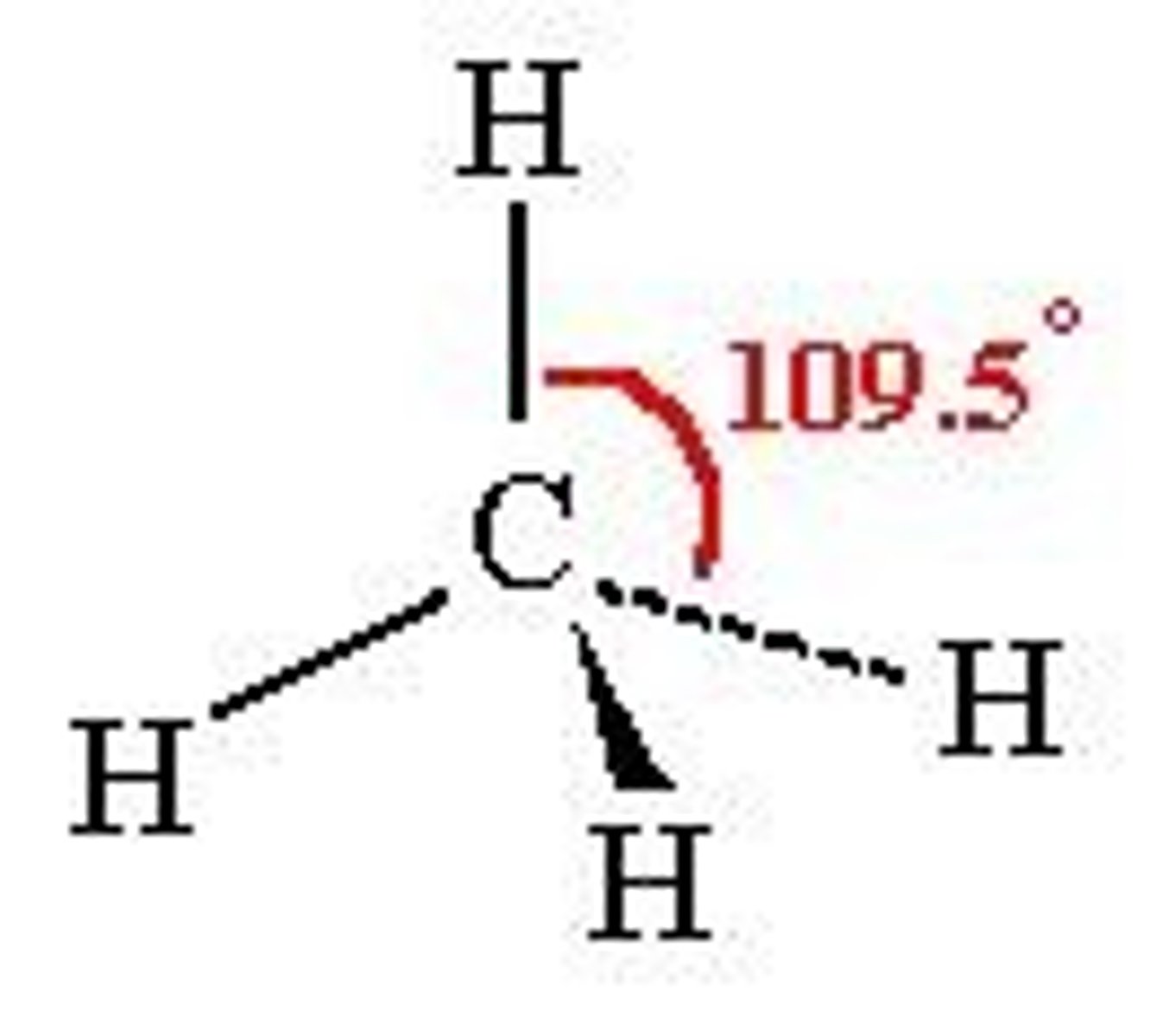
Tetrahedral
A shape of molecule as seen:
Group ion
An ion containing more than one element
Fuel
Name given to a substance that can be burned to release energy

Combustion
The reaction of a substance with oxygen, which gives out energy. Substances often need to be burned for this reaction to happen

Exothermic reaction
A reaction that gives out heat. The burning of a fuel is an example of this.

Endothermic reaction
A reaction that takes in heat from its surroundings

relight
The test for oxyen: when a glowing splint is put to the mouth of a test tube, it will ________.

Carbon dioxide
Product of the complete combustion of a hydrocarbon
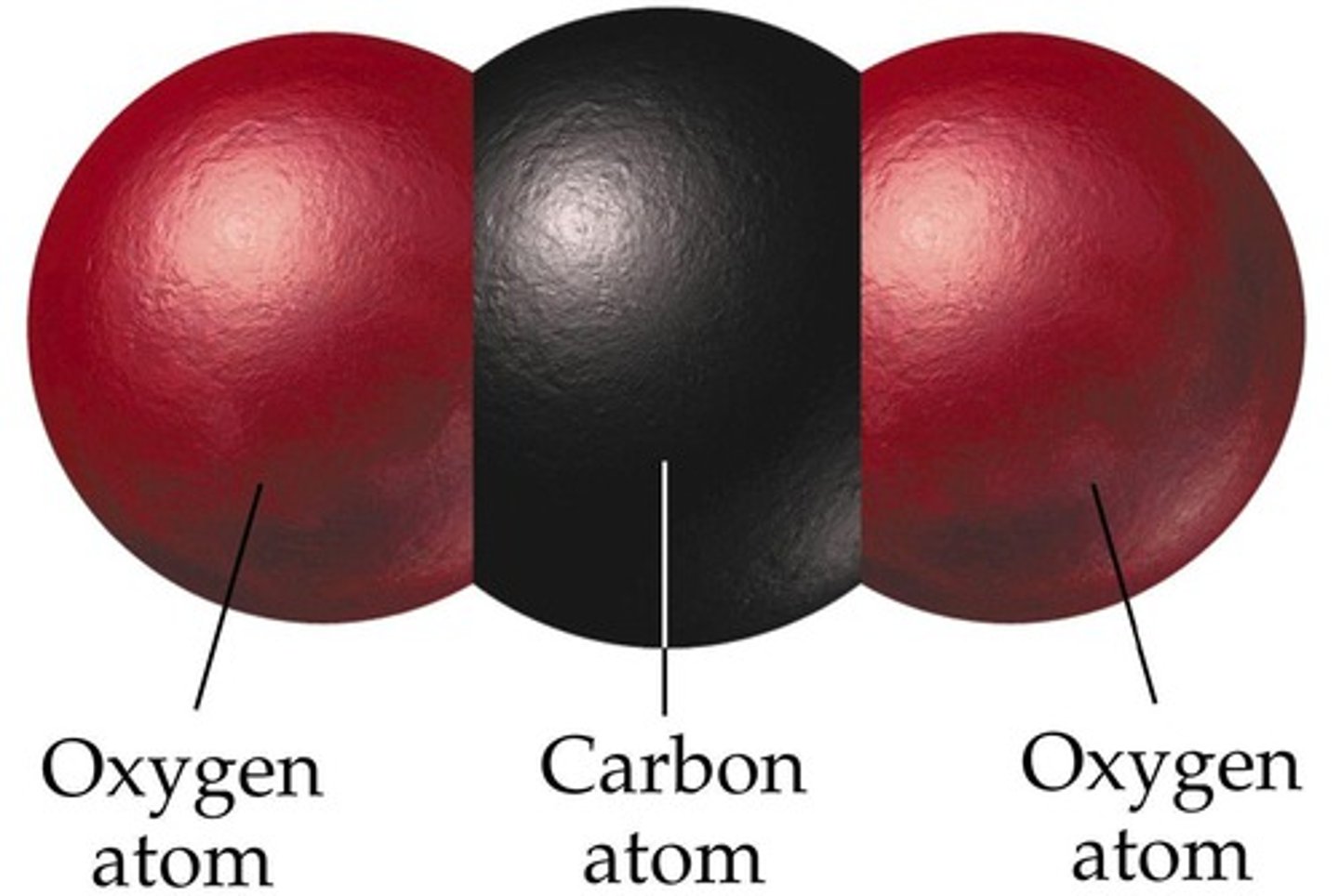
Hydrocarbon
Compound containing the elements carbon and hydrogen only. Crude oil is a mixture of these compounds and examples include alkanes and alkenes.
Products of combustion of a hydrocarbon
The products of this reaction are carbon dioxide and water.
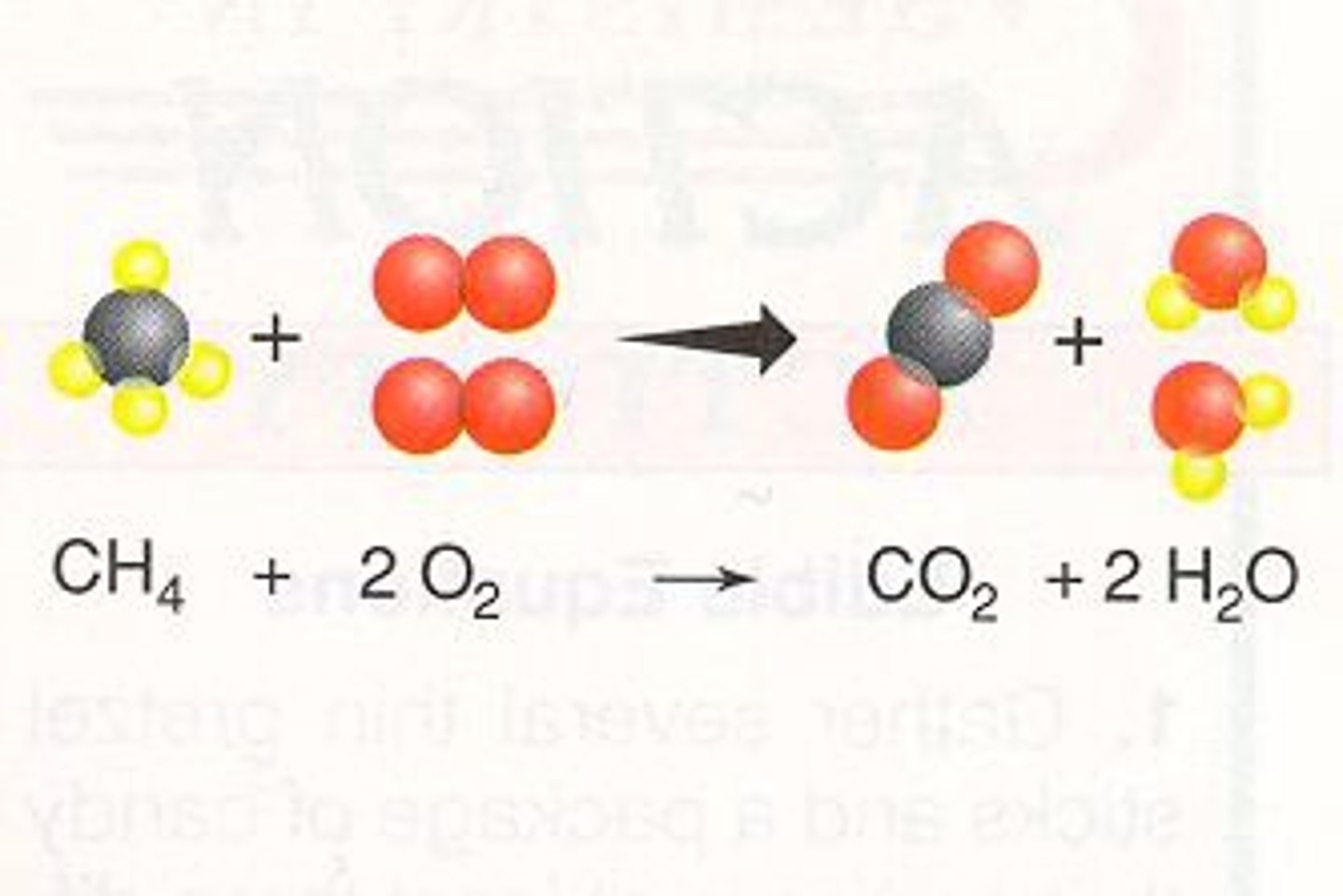
Homologous series
A group of compounds with similar chemical properties that can be represented by a general formula and have gradual change in physical properties such as melting and boiling points.
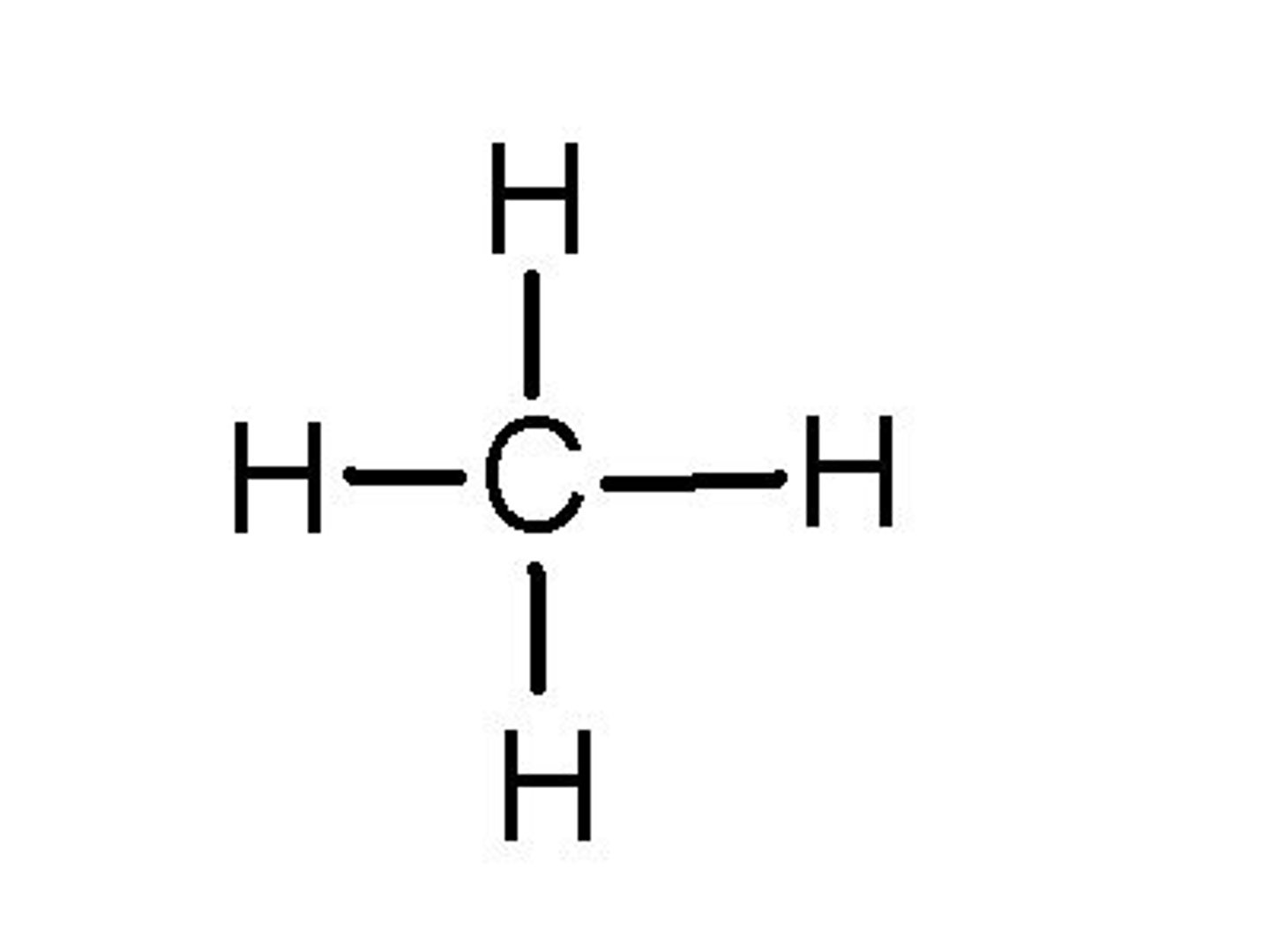
Alkanes
The simplest homologous series of hydrocarbons, named methane, ethane, propane, etc. They are saturated hydrocarbons due to the fact that they contain only single carbon-to-carbon bonds. Their general formula is Cn H 2n + 2.
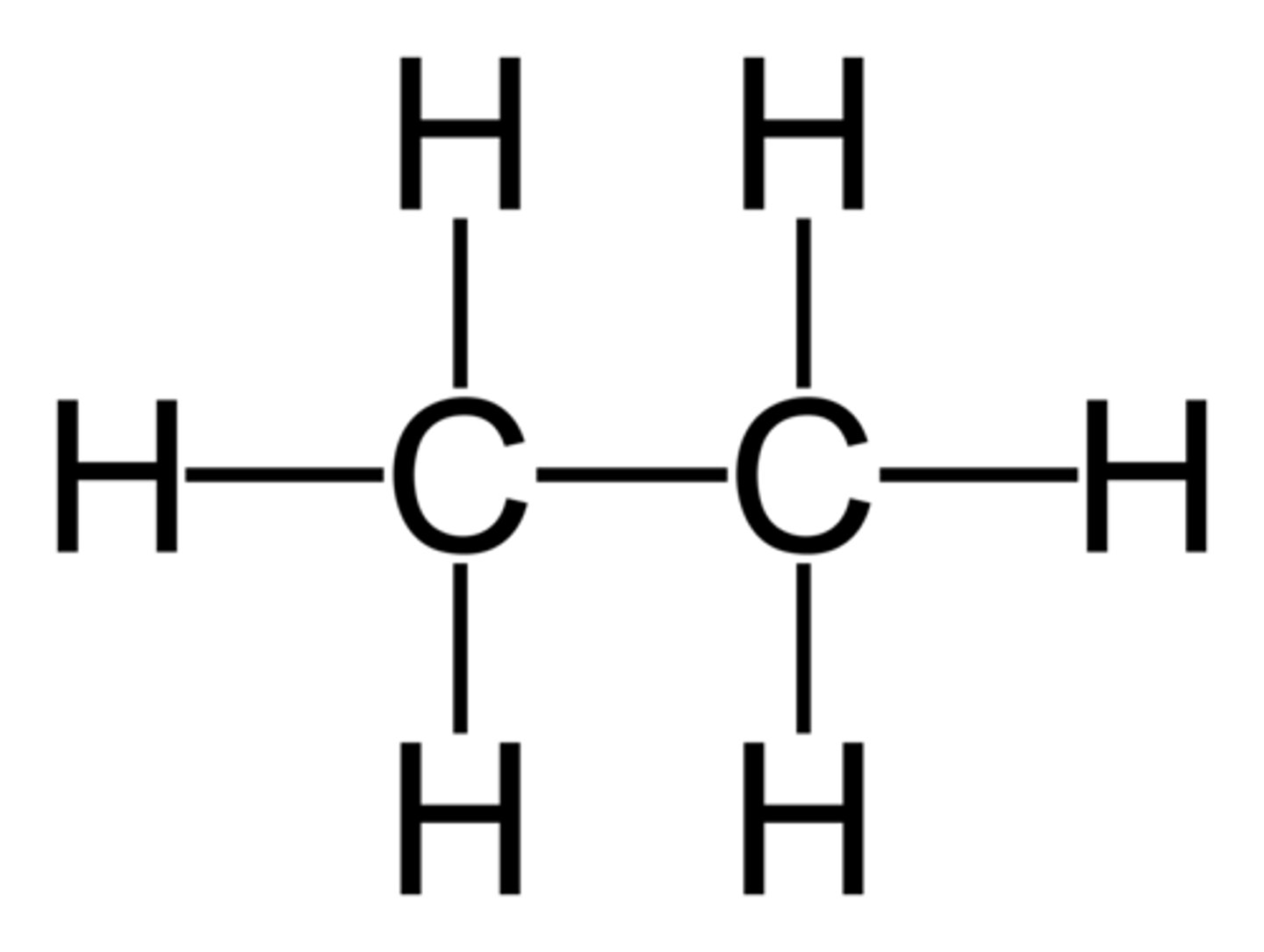
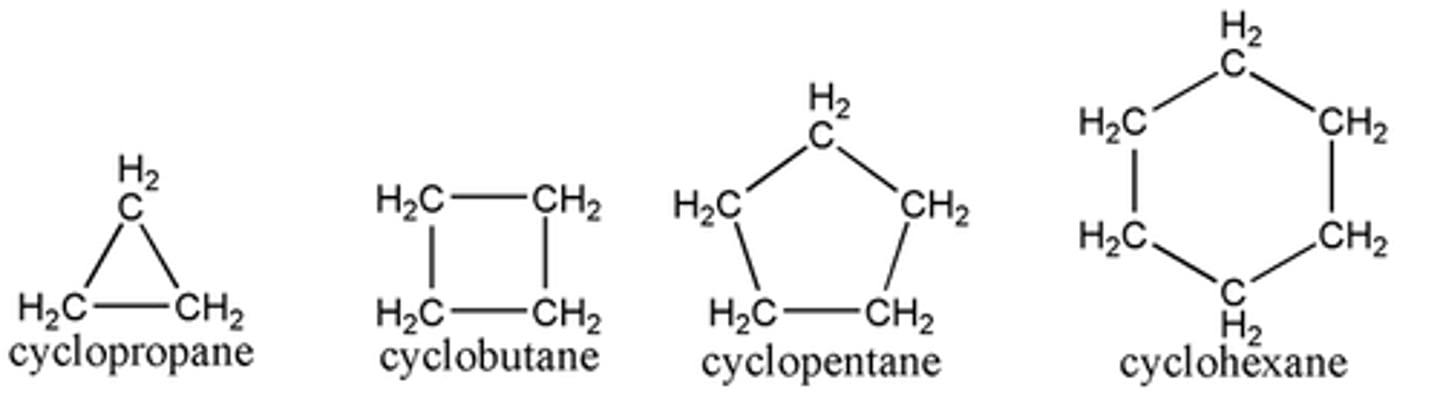
Cycloalkanes
A homologous series of hydrocarbons with cyclic shapes, named cyclopropane, cyclobutane, cyclopentane, etc. Their general formula is Cn H2n. They are saturated.
Alkenes
A homologous series of hydrocarbons with a carbon-to-carbon double bond. This is the functional group, and they are unsaturated because of this feature. Their general formula is Cn H 2n and their names are ethene, propene, butene, etc.
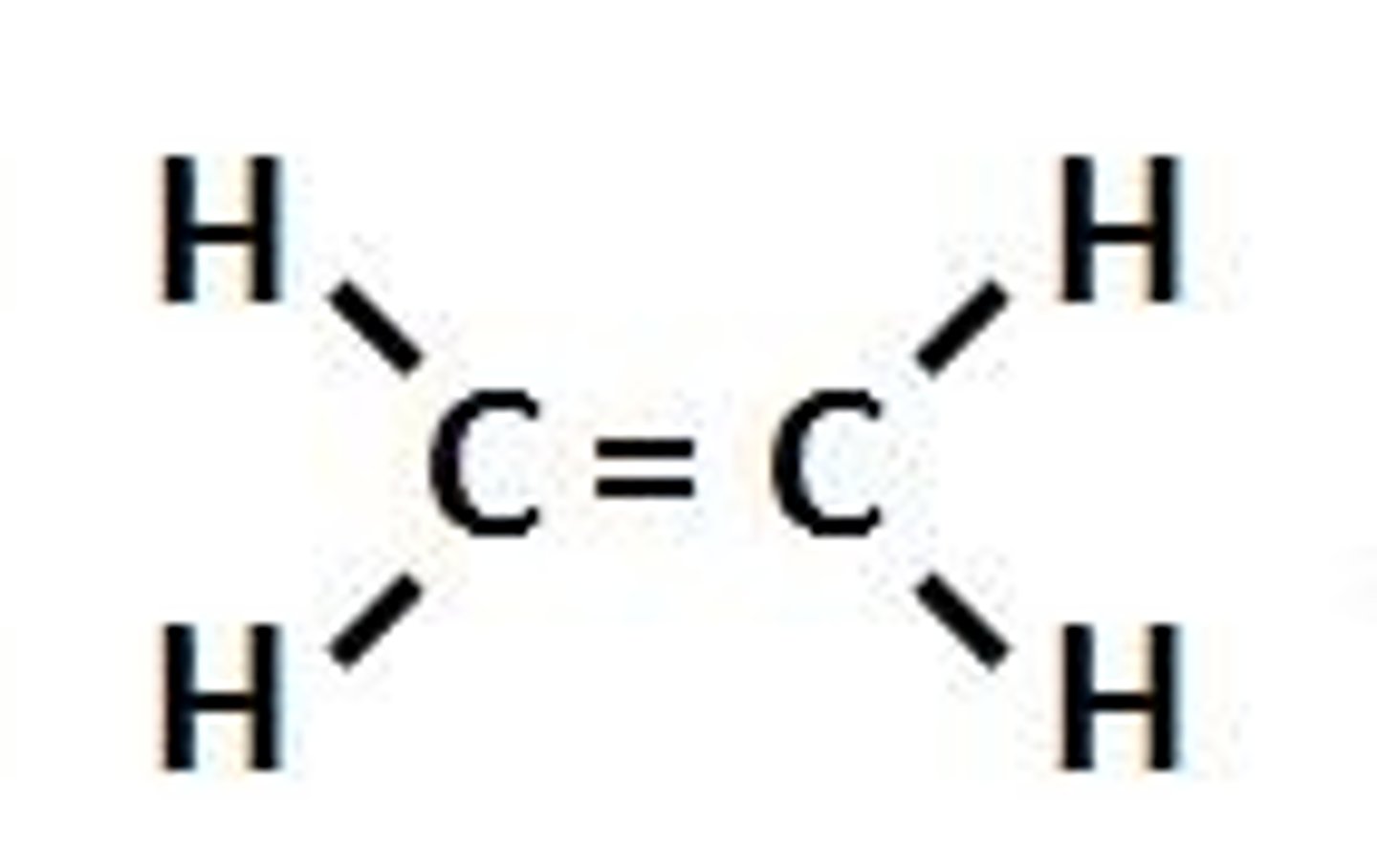
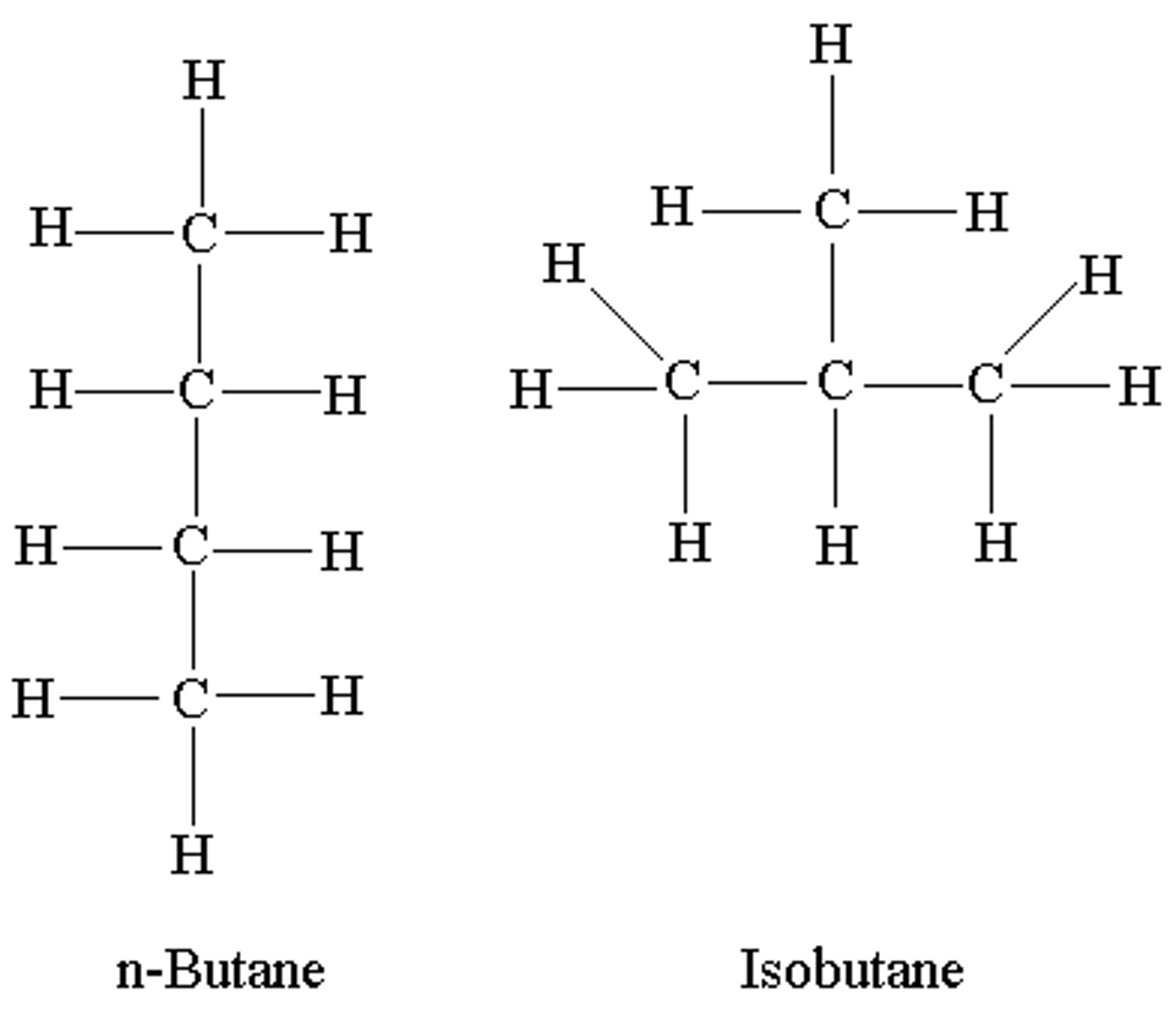
Isomers
Two compound that have the same molecular formula but different structural formulae are described as being ___________.
Saturated
A hydrocarbon is described as being __________ if it contains no carbon-to-carbon double bonds and so is not reactive. E.g, the alkanes.
Unsaturated
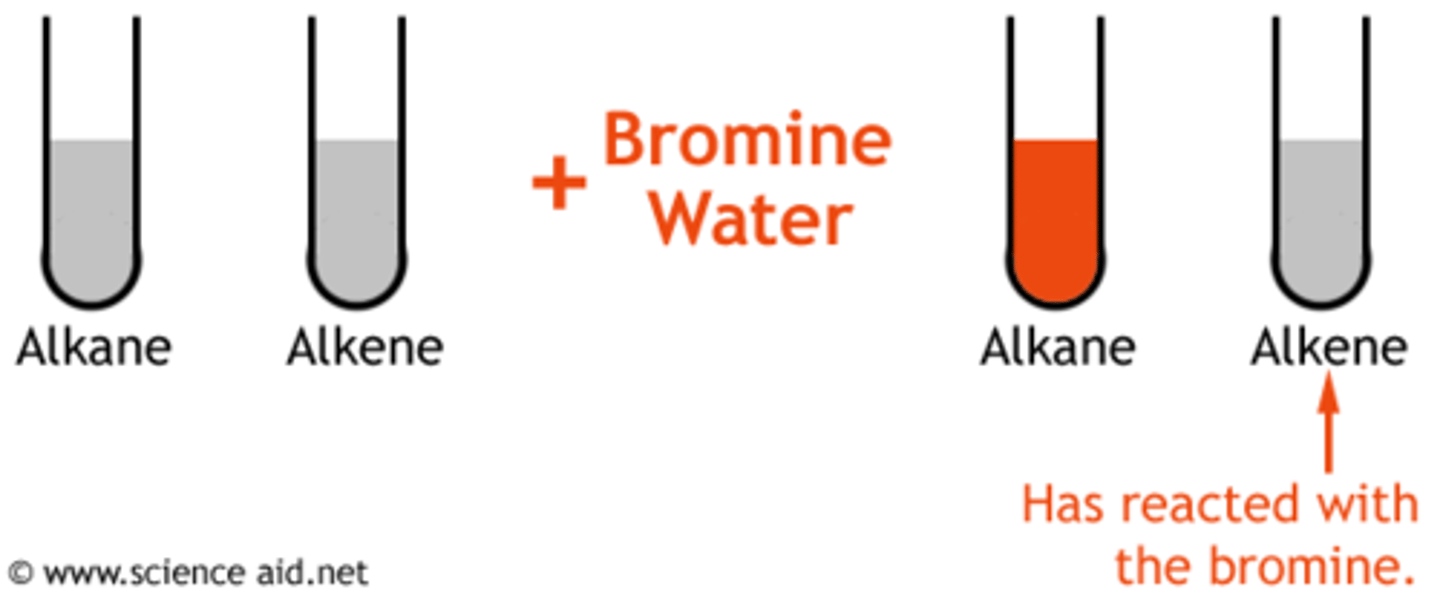
A hydrocarbon is described as being __________ if it contains carbon-to-carbon double bonds and so is reactive. E.g, the alkenes. Hydrocarbons of this variety will rapidly decolourise Bromine solution.
Decolourise
Unsaturated hydrocarbons will rapidly ___________ bromine solution. This is the chemical test for unsaturation.
Addition reaction
A type of chemical reaction where an unsaturated hydrocarbon reacts with another substance across the double bond. (The molecule effectively 'adds' in place of the double bond.)
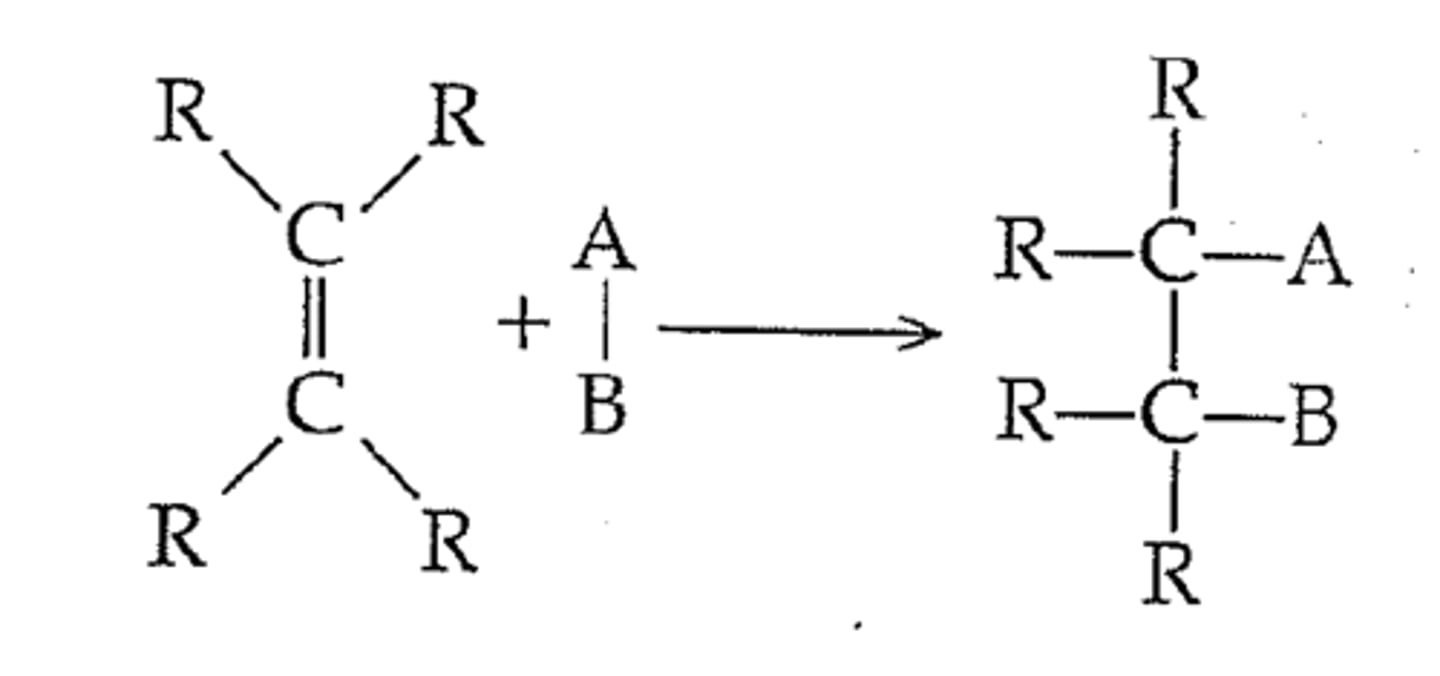
Hydrogenation
A reaction describing the addition of hydrogen to an alkene.
Hydration
A reaction describing the addition of water to an alkene.
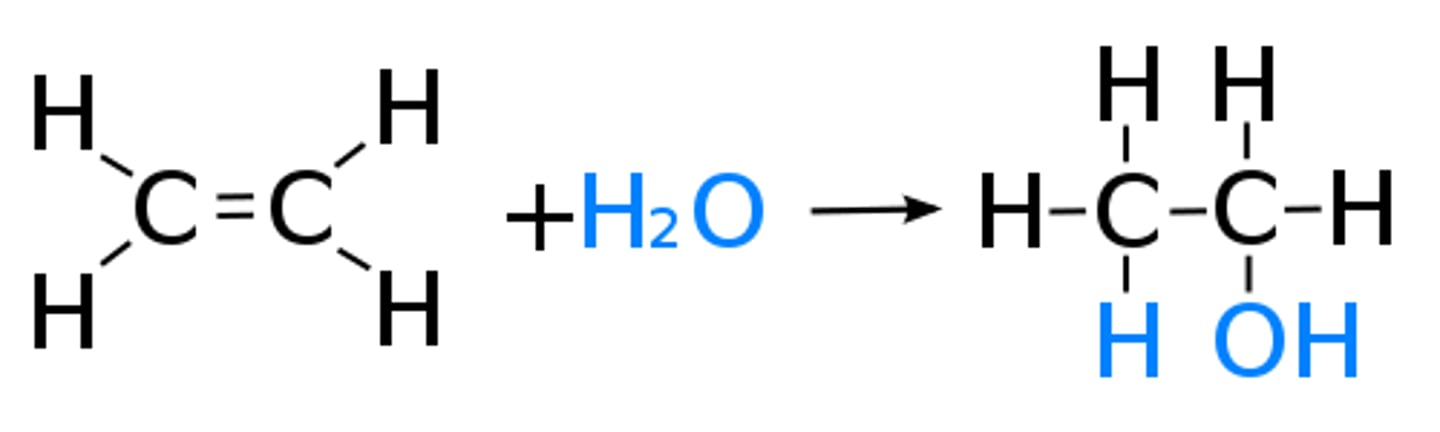
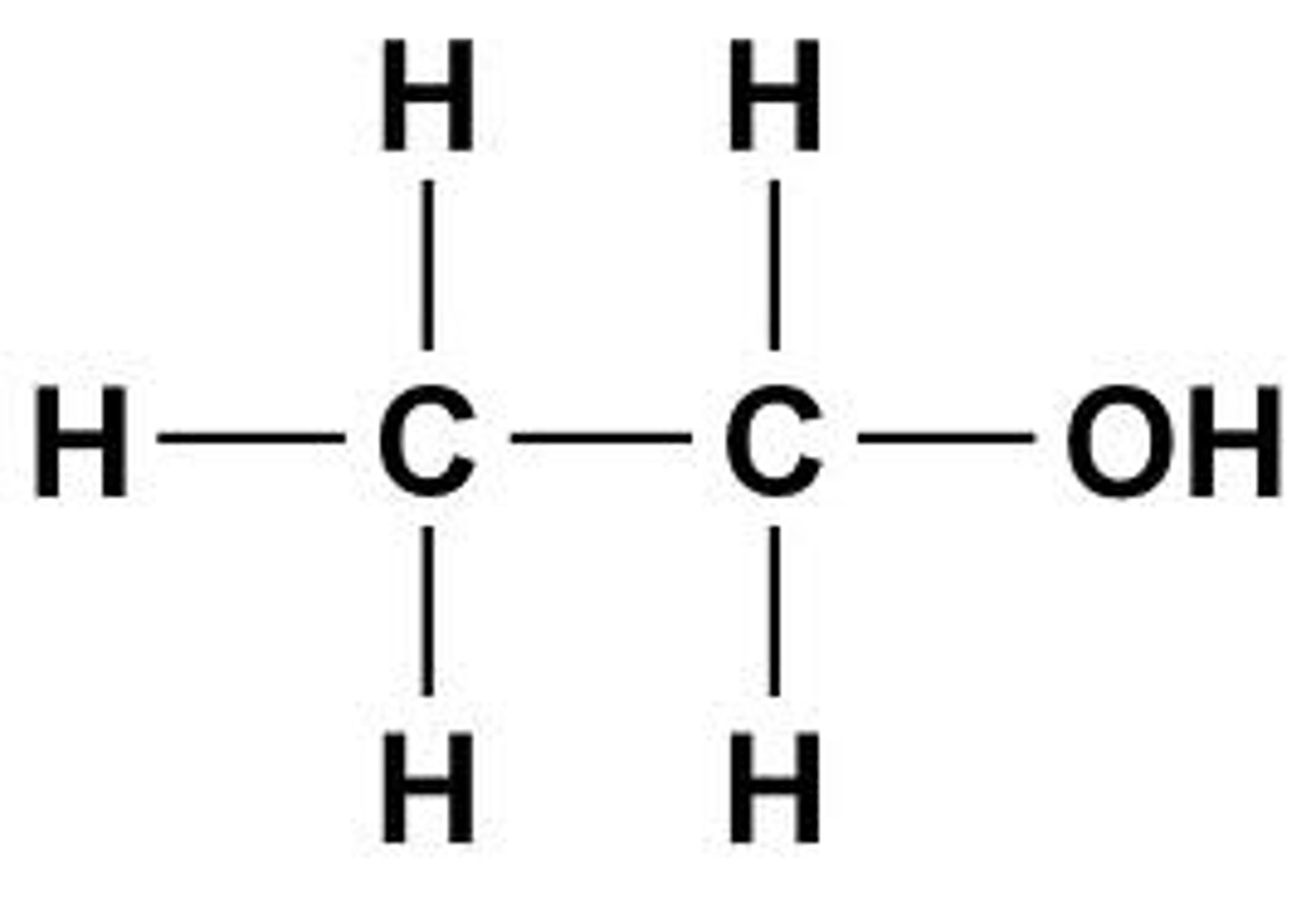
Ethanol
An alcohol with the formula C2H5OH.
Alcohols
Homologous series containing the hydroxyl functional group, -OH. Its general formula is CnH2n+1 OH.
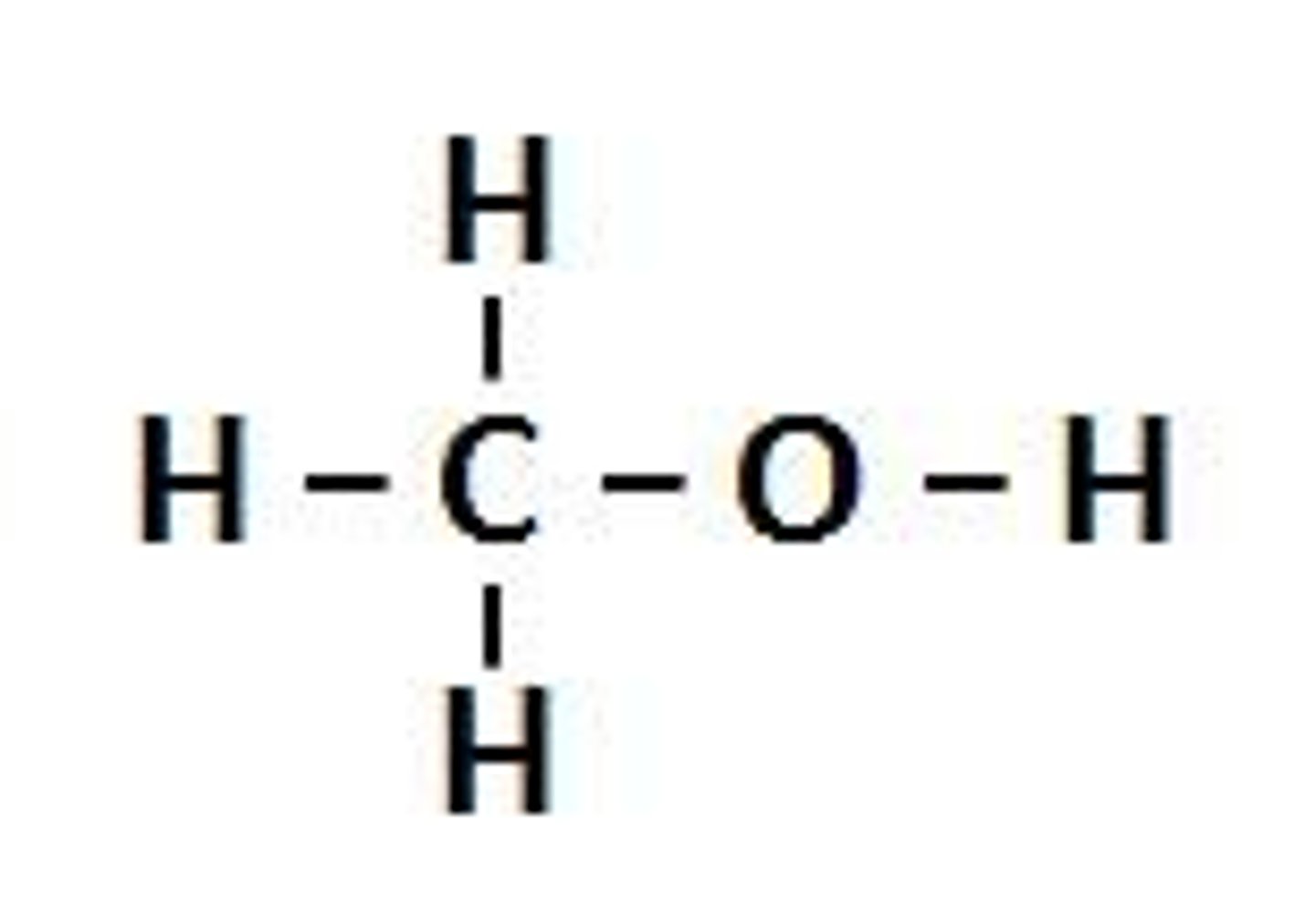
Carboxylic acid
Homologous series containing the carboxyl functional group, -COOH. General formula is CnH2n+1 COOH
Hydroxyl group
functional group of an alcohol -OH
Carboxyl group
functional group of a carboxylic acid -COOH
less than 7
pH of a carboxylic acid
Ethanoic acid
CH3COOH
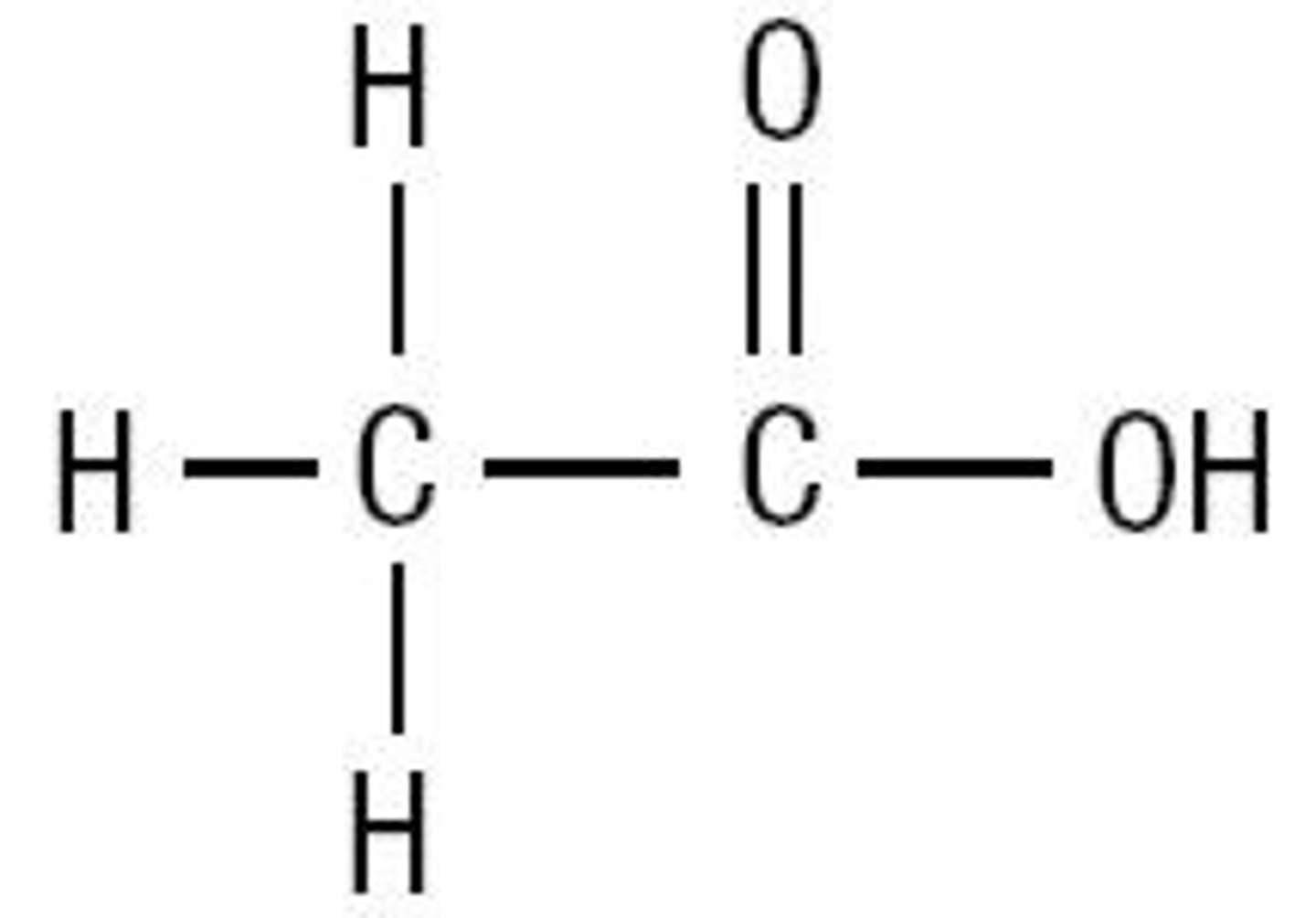
Properties of an alcohol
highly flammable and burns with a clean, blue flame. Methanol, ethanol and propanol are soluble in water, thereafter their solubility decreases as size increases.
Properties of a carboxylic acid
pH of less than 7. methanoic, ethanoic and propanoic acid are all soluble in water, thereafter their solubility decreases as size increases.
sodium hydroxide + ethanoic acid
Sodium ethanoate + water
magnesium + propanoic acid
magnesium propanoate + hydrogen