md4 - intercalators and telomerase inhibitors
1/101
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
What is the basic structure of DNA in the cell?
: In the cell, DNA is typically modeled as a double helix
What is supercoiling of DNA?
Supercoiling refers to the winding of circular DNA molecules multiple times, forming loops and twists.
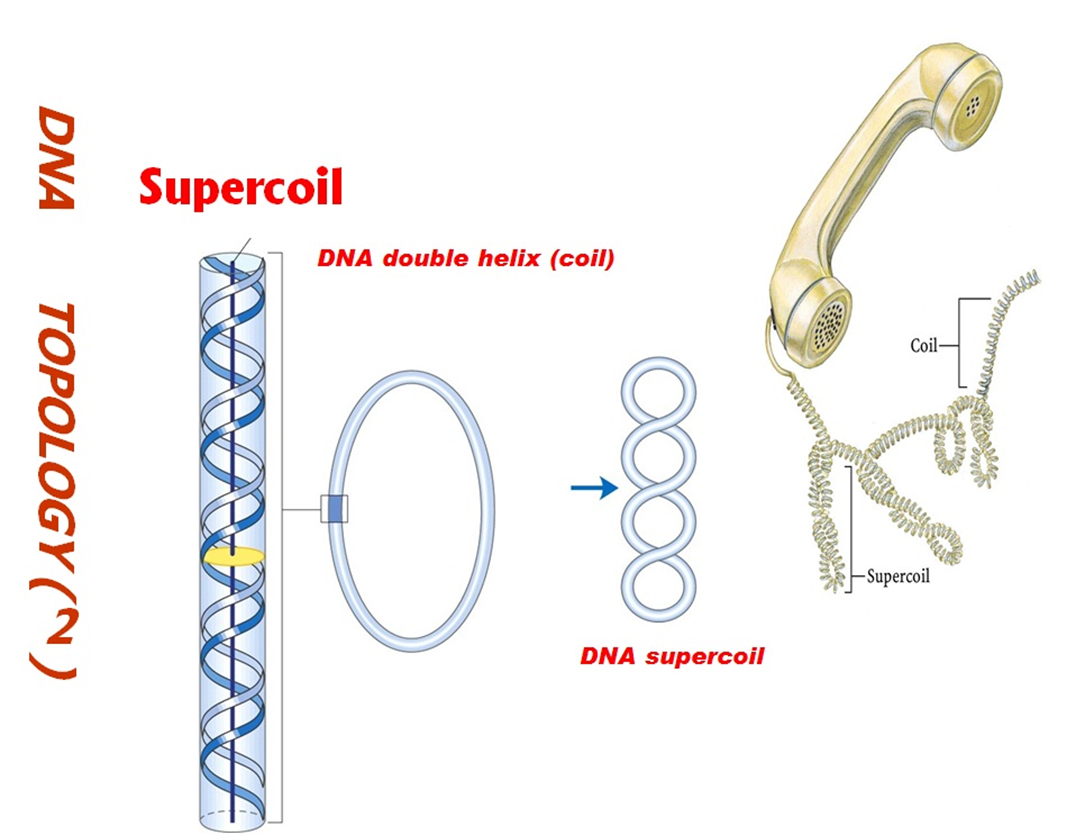
Why does supercoiling make DNA less accessible?
Because supercoiling compresses the DNA strands, making them tightly wound and less available for processes like transcription.
Which enzymes are responsible for managing DNA supercoiling?
Topoisomerases, specifically topoisomerase I and II, control DNA supercoiling.
How does topoisomerase I help manage supercoiling?
Topoisomerase I relaxes supercoiled DNA by cleaving one strand, allowing it to unwind, and then re-ligating the strand.
What is the function of topoisomerase II?
Topoisomerase II cleaves both DNA strands to manage tangles and supercoils that occur during DNA replication and transcription.
Why are topoisomerases important for DNA function?
They help maintain DNA in a relaxed and accessible form necessary for accurate replication, transcription, and overall cell function.
explain what topo 1 does:
topo 1 make transient single stranded breaks in the dna double helix and then researls the strand.
so there is a single strand nick, then it passes the other strand through the cut and researls the break .
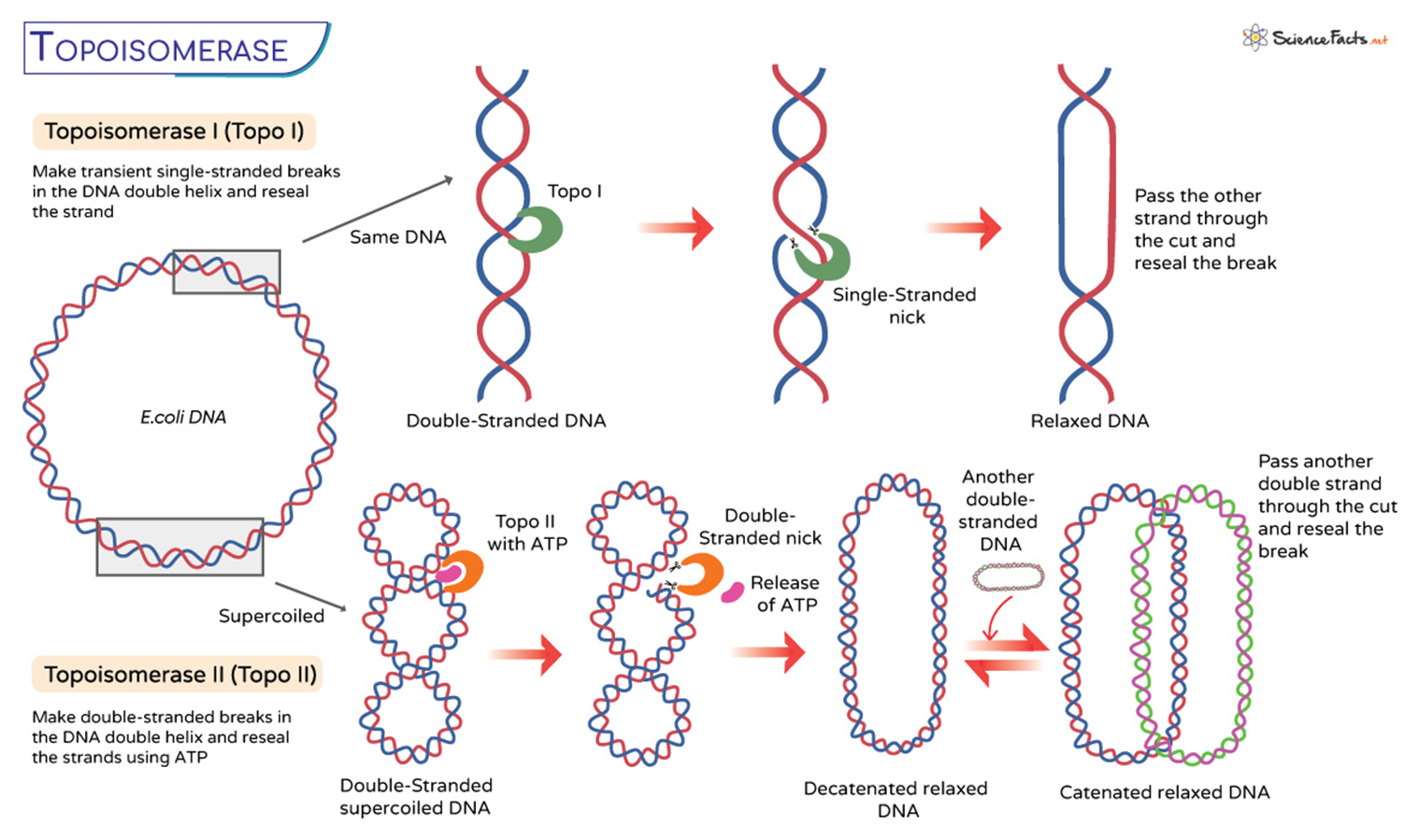
What type of DNA break does Topoisomerase I introduce to relax supercoiled DNA?
Topoisomerase I introduces a transient single-strand break.
How does Topoisomerase I restore DNA after relieving torsional stress?
It re-ligates the broken single strand, restoring the DNA’s integrity.
What kind of DNA break does Topoisomerase II create to manage DNA tangles and supercoiling?
Topoisomerase II creates a transient double-strand break.
During its action, how does Topoisomerase II resolve DNA tangles?
It passes another duplex of DNA through the double-strand break before re-ligating, helping to untangle DNA.
Why is the activity of topoisomerases crucial during DNA replication and cell division?
They manage supercoiling and tangles, ensuring proper separation of replicated DNA strands.
What would happen if topoisomerases did not function properly?
DNA would become overly supercoiled or tangled, impairing processes like transcription and replication.
explain what topoisomerase 2 does:
Topoisomerase 2 (Topo 2) is an enzyme that cuts both strands of DNA, allows another DNA segment to pass through the break, and then reseals the DNA using ATP.
It starts with supercoiled or tangled double-stranded DNA.
Topo 2 uses ATP to create a temporary double-stranded break (nick).
This allows another double-stranded DNA segment to pass through the break, helping to untangle (decatenate) or relax the DNA.
After this, Topo 2 reseals the DNA strands, resulting in relaxed or separated DNA molecules.
What does Topoisomerase II do to DNA?
It creates temporary double-stranded breaks, allows DNA strands to pass through, and then reseals the DNA.
What type of DNA does Topoisomerase II act on?
It acts on double-stranded DNA, especially supercoiled or tangled DNA.
What is the role of ATP in Topoisomerase II activity?
ATP provides the energy needed for cutting, passing DNA strands through, and resealing the DNA.
What happens after Topoisomerase II makes a double-stranded break?
Another double-stranded DNA segment passes through the break.
What is meant by “decatenation” of DNA?
It is the process of separating intertwined DNA molecules.
Q6: What is the final result after Topoisomerase II acts on DNA?
The DNA becomes relaxed or untangled and is properly separated.
what is the main difference between topo1 and topo2?
topo1 makes single stranded breaks and topo2 makes double stranded breaks in the dna double helix.
Q2: Does Topoisomerase I require ATP?
A: No, Topoisomerase I does not require ATP.
Q3: Does Topoisomerase II require ATP?
A: Yes, Topoisomerase II requires ATP to function.
Q4: How do Topo I and Topo II relieve DNA supercoiling differently?
Topo I relaxes DNA by cutting one strand, pass the other strand through the cut and reseal the break. NO ATP.
Topo II relaxes DNA by cutting both strands and passing another DNA segment through.
Q4: How do Topo I and Topo II relieve DNA supercoiling differently?
Topo I relaxes DNA by cutting one strand and allowing it to unwind.
Topo II relaxes DNA by cutting both strands and passing another DNA segment through.
Q5: Which enzyme is involved in decatenation of DNA?
A: Topoisomerase II, because it can separate intertwined DNA molecules.
Q6: Which enzyme is more complex in function?
A: Topoisomerase II, because it involves double-strand breaks and ATP use.
Q7: What happens after Topoisomerase I cuts DNA?
A: The DNA strand rotates to relieve tension, then the enzyme reseals the break.
Q8: What happens after Topoisomerase II cuts DNA?
A: Another double-stranded DNA segment passes through the break before it is resealed.
Q9: Why are topoisomerases important targets in cancer therapy?
Cancer cells divide rapidly and rely heavily on DNA replication, so inhibiting topoisomerases disrupts DNA processes and kills the cells.
Q10: What happens when Topoisomerase II is inhibited in cancer treatment?
DNA double-strand breaks cannot be properly resealed, leading to DNA damage and cell death.
Q11: What happens when Topoisomerase I is inhibited?
A: Single-strand breaks accumulate, which can lead to replication errors and cell death.
Q12: Why are Topoisomerase II inhibitors often more toxic?
A: Because they cause double-stranded DNA breaks, which are more damaging to cells.
Q13: Compare the mechanisms of Topoisomerase I and Topoisomerase II.
Topo I: single-strand break, no ATP, relieves supercoiling by rotation.
Topo II: double-strand break, requires ATP, passes DNA through break for decatenation.
Q14: Explain how Topoisomerase inhibitors can be used in cancer therapy.
A: They prevent DNA repair during replication, causing DNA damage accumulation and leading to cancer cell death.
Q15: Why are rapidly dividing cells more sensitive to topoisomerase inhibitors?
Because they rely heavily on DNA replication, making them more vulnerable to disruptions in DNA topology.
summary of topo1 and topo2:
Topoisomerase I (Topo I)
Cuts ONE DNA strand
DNA twists/rotates to relieve tension
No ATP needed
Topoisomerase II (Topo II)
Cuts BOTH DNA strands
Another DNA strand passes through the break
Uses ATP
Q1: What is camptothecin and where is it derived from?
Camptothecin is an anticancer compound originally isolated from the bark and stem of Camptotheca acuminata (the “Happy Tree”).
Q2: Which enzyme does camptothecin target?
Camptothecin targets Topoisomerase I.
Q3: How does camptothecin affect Topoisomerase I?
it prevents Topoisomerase I from resealing single-strand DNA breaks after cutting the DNA.
Q4: What is the consequence of inhibiting Topoisomerase I in cancer cells?
A: DNA damage accumulates, especially during replication, leading to cancer cell death.
Q5: Why are cancer cells particularly affected by camptothecin?
A: Because they divide rapidly and rely heavily on Topoisomerase I for DNA replication.
Q6: What are the limitations of camptothecin?
A: It has low solubility and can cause significant adverse drug interactions.
Q7: What are examples of camptothecin analogues used clinically?
A: Topotecan and Irinotecan.
Q8: Why were camptothecin analogues developed?
A: To improve stability, solubility, and reduce side effects for clinical use.
Q9: How does this relate to Topoisomerase II inhibitors?
camptothecin targets Topo I (single-strand breaks), whereas other drugs target Topo II (double-strand breaks).
camptothecin and why it is relevant in cancer:
Simple explanation
Camptothecin (from the “Happy Tree”) is a drug that targets Topoisomerase I.
It prevents Topo I from resealing the single-strand DNA break after it cuts it.
This causes DNA damage to build up, especially during replication → leads to cancer cell death.
Why this matters in cancer
Cancer cells divide rapidly → they rely heavily on Topo I
If Topo I is blocked, DNA can’t be properly unwound or repaired
This leads to cell death (especially in fast-dividing cancer cells)
how does topotecan and irinotecan work?
Topotecan and Irinotecan work during the S-phase (when DNA is being copied).
Normally, Topoisomerase I makes temporary single-strand cuts to relieve tension and then reseals them.
These drugs bind to the Topo I–DNA complex and stop the DNA from being resealed.
This causes the replication machinery to stall, leading to double-strand DNA breaks.
If the damage isn’t repaired, the cell dies.
Topotecan works by inserting itself into the DNA (intercalation) at the cut site.
This blocks the DNA from being rejoined.
Q1: During which phase of the cell cycle do Topotecan and Irinotecan act?
They act during the S-phase, when DNA is being replicated.
G0
G1 = growth
S = synthesis stage
G2 = growth and preparation for M (mitosis)
M = Mitosis = PMAT
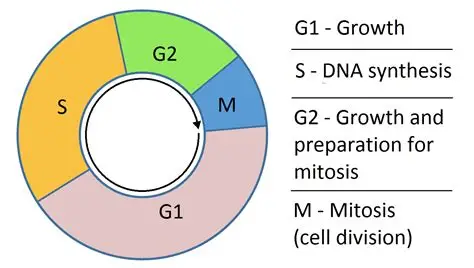
Q2: What is the normal function of Topoisomerase I?
It relieves DNA tension by creating temporary single-strand breaks and then resealing them.
Q3: How do Topotecan and Irinotecan affect Topoisomerase I?
They bind to the Topo I–DNA complex and prevent the resealing (re-ligation) of the single-strand breaks.
Q4: What happens when the DNA cannot be resealed?
A: The replication fork stalls, leading to double-strand DNA breaks.
Q5: Why are double-strand breaks dangerous for the cell?
A: If not repaired, they lead to cell death.
Q6: How does Topotecan specifically interact with DNA?
A: It intercalates (inserts) into the DNA at the cleavage site, where cut is, blocking re-ligation.
Q7: What is meant by intercalation in this context?
A: It means the drug inserts itself between DNA base pairs.
Q8: What is the overall effect of Topotecan and Irinotecan on cancer cells?
A: They cause accumulation of DNA damage, leading to death of rapidly dividing cancer cells.
Q1: What is a ternary complex in the context of Topoisomerase I inhibitors?
: A ternary complex is a three-part complex formed between Topoisomerase I, DNA, and a drug (e.g. Topotecan or Irinotecan).
enzyme + dna + drug
How is the ternary complex formed?
It forms when the drug binds to the Topo I–DNA complex after the DNA has been cut, preventing resealing.
Q3: What is the effect of forming a ternary complex?
A: It stabilises the DNA in a broken state, stopping repair of the single-strand break.
Q4: How does the ternary complex affect DNA replication?
A: It stalls the replication fork, preventing normal DNA synthesis.
Q5: What is the consequence of replication fork stalling?
A: It leads to double-strand DNA breaks, which are highly damaging.
Q6: Why is the ternary complex lethal to cancer cells?
Because the accumulated DNA damage cannot be repaired, leading to cell death, especially in rapidly dividing cells.
Q7: Why are rapidly dividing cells more affected by ternary complex formation?
because they are actively replicating DNA, so replication fork stalling occurs more frequently.
in simple terms, what is the significance of the ternary complex?
ternary complex = Topo I + DNA + drug → trapped broken DNA → cell death
Q1: What is the main function of Type II topoisomerases (TOP2)?
A: They cut both strands of the DNA helix simultaneously to manage DNA tangles and supercoiling.
Q2: How do Type II topoisomerases differ from Type I topoisomerases?
A: Type II cut both DNA strands, while Type I cut only one strand.
Q3: Why are TOP2 enzymes important during DNA replication?
A: They are essential for processes like separating daughter DNA strands and resolving tangles.
Q4: What types of small molecules target TOP2?
A: They are either TOP2 inhibitors or TOP2 poisons.
Q5: What do TOP2 poisons do?
A: They increase levels of TOP2-DNA covalent complexes, leading to DNA strand breaks.
Q6: What is the effect of TOP2 poisons on cells?
A: They cause DNA damage (including strand breaks), leading to cell death.
Q7: What do catalytic TOP2 inhibitors do?
A: They block the catalytic activity of TOP2 without increasing covalent DNA complexes.
Q8: How do catalytic inhibitors kill cells?
A: By preventing TOP2 function, disrupting essential DNA processes.
Q9: What structural feature do many TOP2 poisons have?
A: They are planar, aromatic, multi-ring molecules.
Q10: How do TOP2 poisons interact with DNA?
A: They act as intercalators, inserting between DNA base pairs.
what do topo2 poisons do?
These trap TOP2 on DNA after it has cut both strands, forming a covalent TOP2–DNA complex.
The DNA is broken, and because the enzyme is stuck, the break cannot be repaired, leading to DNA damage accumulation → cell death.
Example drugs: Doxorubicin
what do Catalytic TOP2 Inhibitors do?
These prevent TOP2 from cutting DNA in the first place.
No covalent complex forms, so there aren’t trapped DNA breaks, but DNA supercoiling and tangles cannot be resolved during replication.
This disrupts essential DNA processes, eventually leading to cancer cell death.
Q1: What type of drugs are most Topoisomerase II drugs used in the clinic?
A: Most are anthracycline-type drugs (doxorubicin)
Q2: What is the structure of anthracycline drugs?
A: They have a large, planar aromatic region and a flexible part that binds in the grooves of DNA.
Q3: What type of DNA damage is caused by TOP2-targeting drugs?
A: They cause DNA strand breaks and formation of protein–DNA covalent complexes.
Q4: How is TOP2-mediated DNA damage repaired in cells?
A: It is repaired by double-strand break repair pathways and pathways that remove protein–DNA adducts.
Q5: What is a major limitation of anthracycline use?
A: Their use is limited by cardiotoxicity.
Q6: Is the mechanism of anthracycline cardiotoxicity well understood?
A: No, the mechanism is poorly understood.
Q7: What is a serious long-term side effect of TOP2 poisons?
A: They can cause secondary cancers due to drug-induced chromosomal translocations.
summary so far:
Anthracyclines (e.g. doxorubicin) → TOP2 poisons → DNA breaks
Side effects = cardiotoxicity + peripheral neuropathy
Q1: What is telomerase?
A: Telomerase is an enzyme that adds DNA sequence repeats to the ends of chromosomes.
Q2: What DNA sequence does telomerase add in vertebrates?
A: It adds the repeat sequence TTAGGG to the 3’ end of DNA strands.
Q3: Where does telomerase act in the chromosome?
A: It acts at the telomeres, which are located at the ends of eukaryotic chromosomes.
Q4: What are telomeres?
A: Telomeres are regions of repeated, non-coding DNA at the ends of chromosomes.
Q5: What is the function of telomeres?
A: They protect chromosomes by preventing loss of important DNA during replication.
Q6: Why is telomerase important for cells?
A: It maintains telomere length, preventing excessive shortening that could damage genetic information.
Q1: What is hTERT?
A: hTERT (human Telomerase Reverse Transcriptase) is the catalytic subunit of telomerase.
Q2: What is the function of hTERT?
A: It is responsible for adding DNA repeats to telomeres, enabling telomerase activity.
Q3: In which cells is hTERT upregulated?
A: It is upregulated in rapidly dividing cells, including cancer cells.
Q4: How does hTERT contribute to cancer?
A: It allows cells to maintain telomere length, leading to cell immortalisation.
Q5: Why is hTERT an important therapeutic target?
A: Because inhibiting it can limit telomere maintenance and reduce cancer cell survival.
Q6: What is ALT?
A: ALT is a telomerase-independent mechanism for maintaining telomeres.
Q7: How does ALT maintain telomere length?
A: It uses homology-directed repair, copying sequences from other telomeres.
Q8: How does ALT differ from normal homologous recombination?
A: Instead of copying from a sister chromatid, it copies from matching telomere sequences.
Q8: What is the function of telomestatin?
A: Telomestatin binds to and stabilises G-quadruplexes, preventing them from being unwound.