Chem 1A Midterm 3- Acids and Bases
1/126
Earn XP
Description and Tags
Aaaauuughhhh
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
127 Terms
T/F: Whether you start with all reactants or products (or a mix of both), the composition of the final equilibrium mix is always the same
True
What does K represent?
Equilibrium constant - numerical value defining the ratio of product concentrations to reactant concentrations for a reversible reaction at equilibrium
What is a reversible reaction?
Process where reactants form products that can also turn back into reactants. This helps reach equilibrium. Symbolized by double arrow
When a given equation is aA + bB ⇌ cC + dD, what will the equilibrium equation look like?
[C]c [D]d / [A]a [B]b
What is the equation for K?
products/reactants
What forms of matter are NOT used to calculate equilibrium?
Solid and liquid - you ONLY use aqueous and gas
What do brackets [ ] represent?
Units of molarity
What is the equation for molarity (M)?
mol/L
What does the K equation look like when using partial pressures?
PCc PDd / PAa PBb
When do you use the partial pressure K equation?
ONLY with gases
When is a reversible equation reactant favored? (What size of K?)
When the denominator is bigger
or
K < 1
When is a reversible equation product favored? (What size of K?)
When the numerator is bigger
or
K > 1
What is a forward reaction rate in a reversible reaction?
Signals how “easy” the forward reaction is (reactants → products)
What is the equation for forward reaction rate?
Kf [X]
Where Kf = rate constant and [X] depends on the quantity of the reactant
What is a backward reaction rate in a reversible reaction? What does it depend on?
Signals how “easy” the backward reaction is (products → reactants)
Depends on how many products you have
What is the equation for backward reaction rate?
Kb [Y]
Where Kb = rate constant and [Y] depends on the quantity of the product
What is chemical equilibrium?
A state all reversible reactions reach where amounts are no longer changing because the forward and backward reates are equal
What is the reaction quotient (Q)?
Amounts of reactant and product in equation at any time (not at equilibrium)
How are K and Q different? How are they the same?
Same equation, but it is only K if the concentrations are at equilibrium (if it’s not equilibrium, it’s just Q)
What does a system under “stress” refer to?
System at a non-equilibrium condition
Le Chatelier’s Principle
An equilibrium system subjected to stress will move in the direction that relieves stress and returns system to equilibrium
According to Le Chatelier’s principle, if a system is Q > K, in which direction will the system shift?
There are too many products, so shift to reactants
According to Le Chatelier’s principle, if a system is Q < K, in which direction will the system shift?
There are too many reactants, so shift to products
If there are more collisions, what happens to the reaction speed?
Reaction becomes faster and opposite reaction speeds up to match
What kind of impact will a temperature change have on a system?
Stresses a system to change the K
If ΔH > 0, will heat be a product or reactant?
Reactant
If ΔH < 0, will heat be a product or reactant?
Product
What does a large Kb represent?
Easy backward reaction
What does a large Kf represent?
Easy forward reaction
What does Kc > 1 represent?
Mostly products @ equilibrium
What does Kp < 1 represent?
Mostly reactants @ equilibrium
What does Q > K represent?
Shift to reactants, too many products
What does Q < K represent?
Shift to products, too many reactants
If K > > 1, does that signify more dissolved products or more undissolved reactants?
More dissolved products
Will turn back into reactants to reach equilibrium (form a precipitate)
If K < < 1, does that signify more dissolved products or more undissolved reactants?
More undissolved reactants
Will create more products to reach equilibrium
What kind of solubility does a big k value signal?
Very soluble
What kind of solubility does a small k value signal?
Not very soluble
What does KS represent?
Whe a liquid dissolves in H2O
What does KH represent?
When a gas dissolves in H2O
What does KSP represent?
When a solid dissolves in H2O
Regardless of subscript, what do all K have in common?
PRODUCTS / REACTANTS !!!!!! !!!!
If a solution has more than the maximum amount of reactant, it’s a type of stress. What happens to the solution in this situation?
The solution is at its maximum saturation, so it forms a precipitate
How does K relate to solubility?
K is the maximum solubility
What will happen to a solution if there are molarities above K? What about below K?
If molarities above K, precipitate will form
If molarities below K, that’s okay — the solution just won’t be at max saturation
With an ionic solid solubilizing, what will the chemical equation look like?
Splits into either element
ex) PbCl2(s) ⇌ Pb2+ (aq) + 2Cl- (aq)
What is a RICE table?
Reaction, initial, change, equilibrium
What stages of matter are left out of a RICE table?
Solid and liquid
What is the common ion effect?
The presence/addition of common ions reduces solubility
When diluting, how do you account for total volume and M change?
M decreases and L water increases
Remember to add L of both reactants together!
What is dissociation?
Dissolution of charged ions in water
What is an acid?
Releases H+ in water (aka a proton or hydronium)
What is the generic form of an acid equation?
HA (aq) + H2O (l) ⇌ A- (aq) + H3O+ (aq)
What is partial ionization?
Some acids dissociate completely and make a lot of product, but others only dissolve a bit → this is partial ionization
Strong acids are (good/bad) at being acids
Good!
What is a characteristic of a strong acid?
Loses ALL protons to form lots of H3O+
What are the strong acids we need to memorize?
HBr, HCl, HI
Strong acids have a (large/small) Ka
Large
Weak acids are (good/bad) at being acids
Bad
What is characteristic of weak acids?
Lose some of their protons to make SOME H3O+
How can you identify a weak acid?
COOH group
Weak acids have a (large/small) Ka
Small!
What is a base?
Releases OH- in water
How can a base make OH?
1) Forming it from reactants
2) “Finding” it from the background H2O
What is the general form of a base equation?
B (aq) + H2O (l) ⇌ BH+ (aq) + OH- (aq)
Strong bases are (good/bad) at being bases
Good!
What is characteristic of a strong base?
Completely dissolves to create lots of OH-
How can you identify a strong base?
Metal + OH-
Strong bases have a (large/small) Kb
Large
Weak bases are (good/bad) at being bases
Bad
How do weak bases often form?
Stealing protons from water to make SOME OH-
How can you identify a weak base?
N with lone pair
Weak bases have a (large/small) Kb
Small
How can you tell if something is neutral?
If an OH- is not attached to metal or CO then it may be neutral
What does “p” mean?
-log( )
What is pH?
-log(H+)
What is pOH?
-log(OH-)
What does a low pH indicate? A high one?
Low - acid
High - base
What is x in -log(x) when finding pH?
mol/L aka M
What is “act one”? When do you use it?
Writing balanced equation → finding excess reactant
When you only have strong substances
What is “act two”? When do you use it?
RICE table, when you have only weak substances
What is neutralization?
When there are equal moles of strong acid and strong base
When combining unequal moles of strong acid and base, what determines pH?
Excess reactant
What is the equation for moles?
M x vol
How can you find pH from pOH?
pH = 14 - pOH
How are pH and pOH related?
Inversely
What is a conjugate base?
Product formed when acid loses proton
Acts as a base in reverse reaction
When should you “worry about” a conjugate base?
When acid is weak
What is a conjugate acid?
Product formed when base gains proton
When should you “worry about” a conjugate acid?
When base is weak
When do you know if you made pOH?
If the product of a reaction is OH
A strong acid is a (strong/weak) base
Weak
A strong base is a (strong/weak) acid
Weak
What is the equation for Kw?
Ka x Kb = Kw = 1.0 × 10-14
What is the equation for pKa and pKb?
pKa + pKb = 14
What does x represent when filling out a RICE table?
Reactant lost and product gained
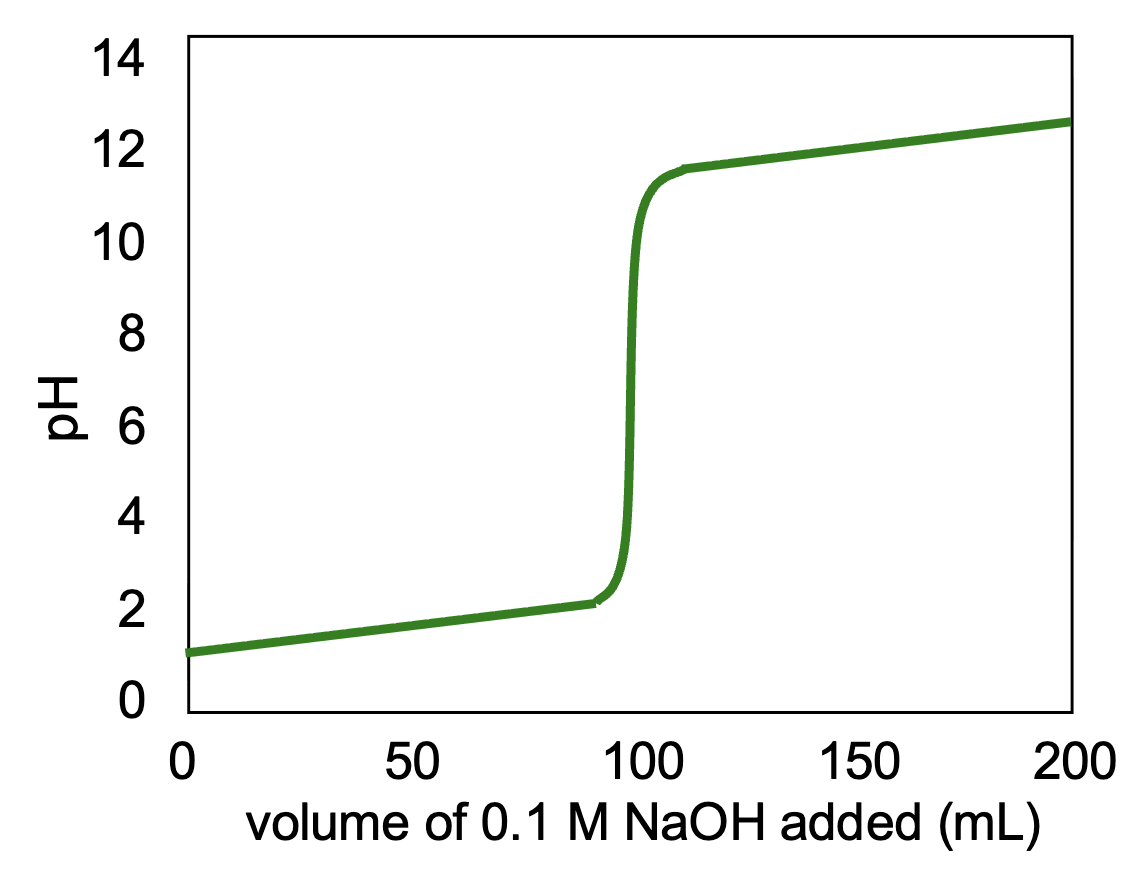
What does each segment of this graph represent?
First segment (lower horizontal stretch) = excess acid
Vertical segment = equal moles of titrant and analyte
Second segment (higher horizontal stretch) = excess base
What is titration?
Gradually adding a known acid or base to a sample to gain info about the system
What is known in a titration?
Volume
What is unknown in a titration?
Concentration or identity
What is titrant?
The thing being added