4.9. Biochemistry: Immune and Lymphatic system
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
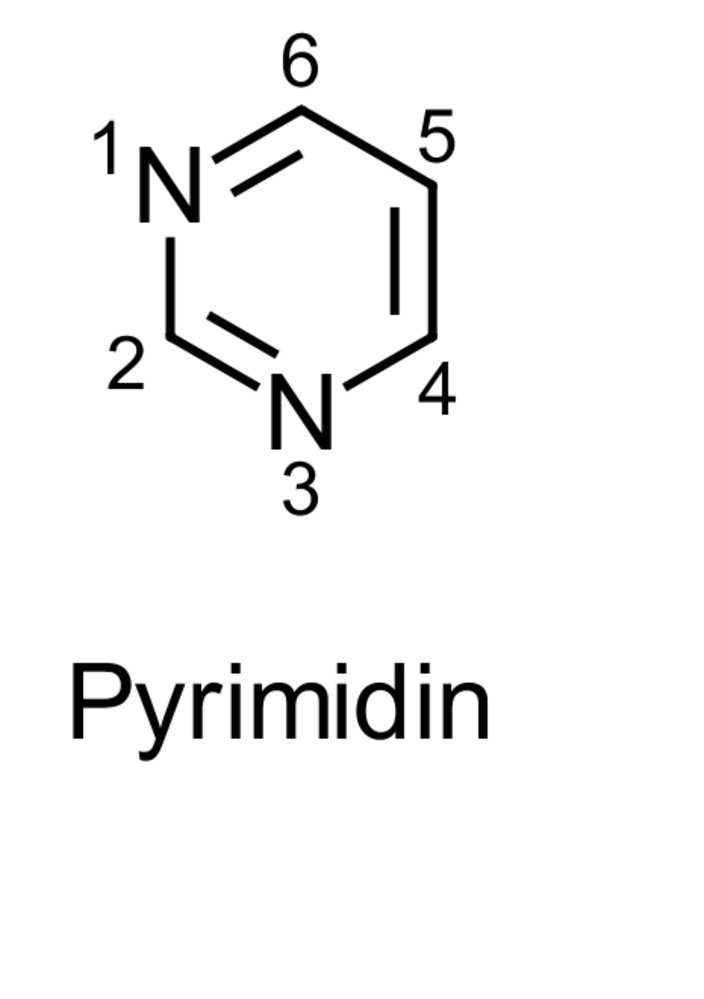
1. The basic structure of pyridoxal phosphate (PALP) consists of a pyridine ring, which differs from pyrimidine derivatives.
How does a pyridine ring differ from a pyrimidine ring?
A. 0 vs. 1 oxygen atom in the ring
B. 1 vs. 2 nitrogen atom(s) in the ring
C. Aliphatic vs. aromatic ring
D. Homocyclic vs. heterocyclic ring
E. Ring of 5 vs. 6 atoms
B. 1 vs. 2 nitrogen atom(s) in the ring
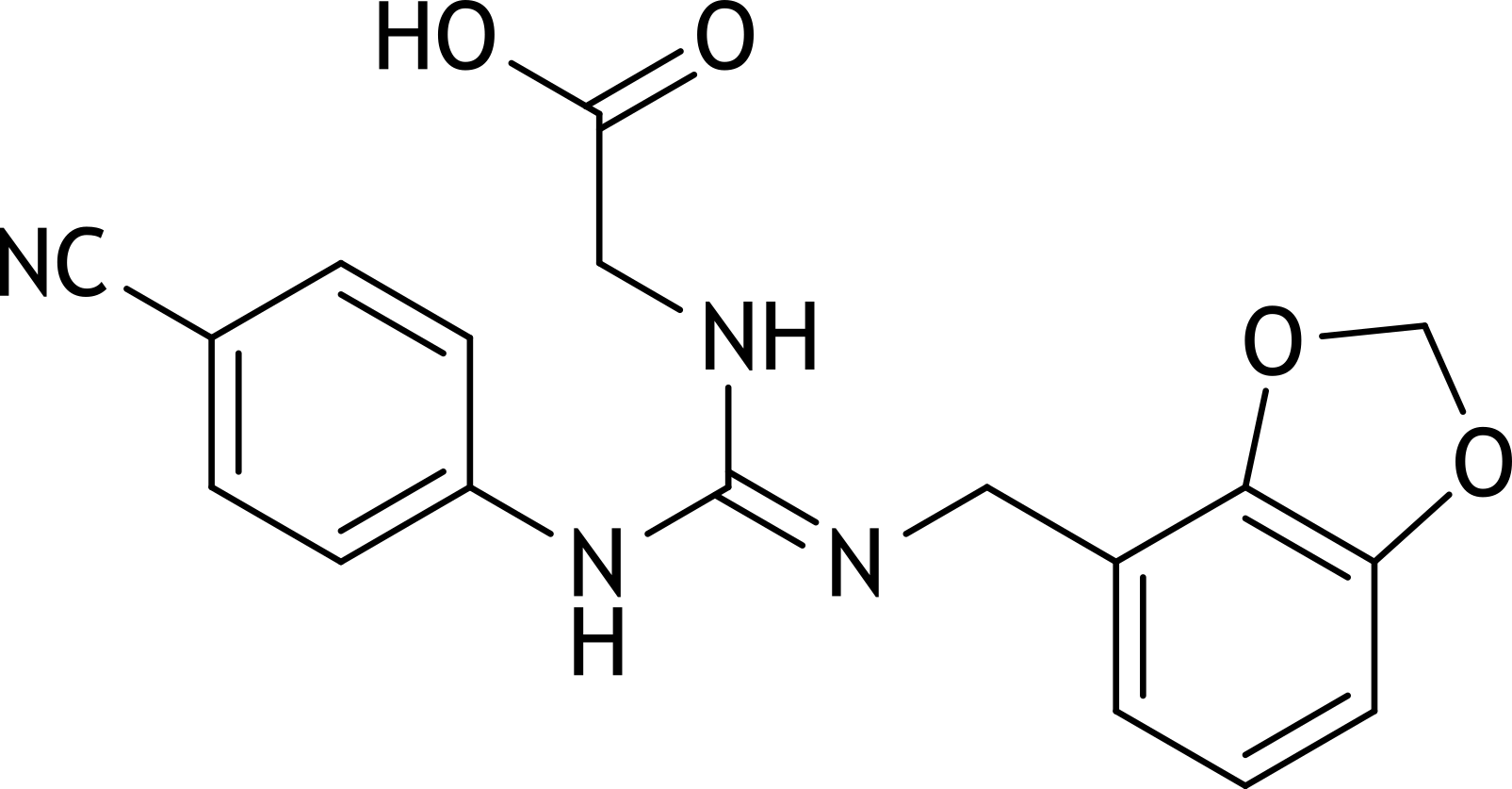
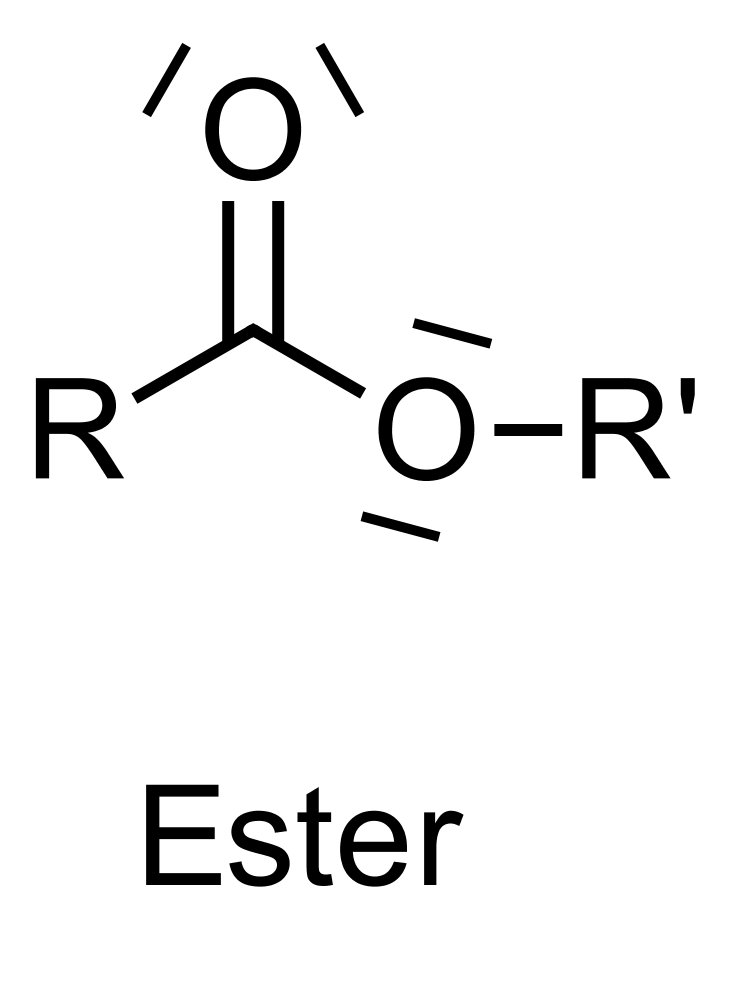
2. Lugdunam is a highly potent synthetic sweetener with approximately 200,000 times the sweetness of sucrose. It has the following chemical structural formula:
To which of the following classes of compounds does it belong?
A. Carboxylic acid amide
B. Carboxylic acid anhydride
C. Carboxylic acid ester
D. Dipeptide
E. Guanidine
E. Guanidine
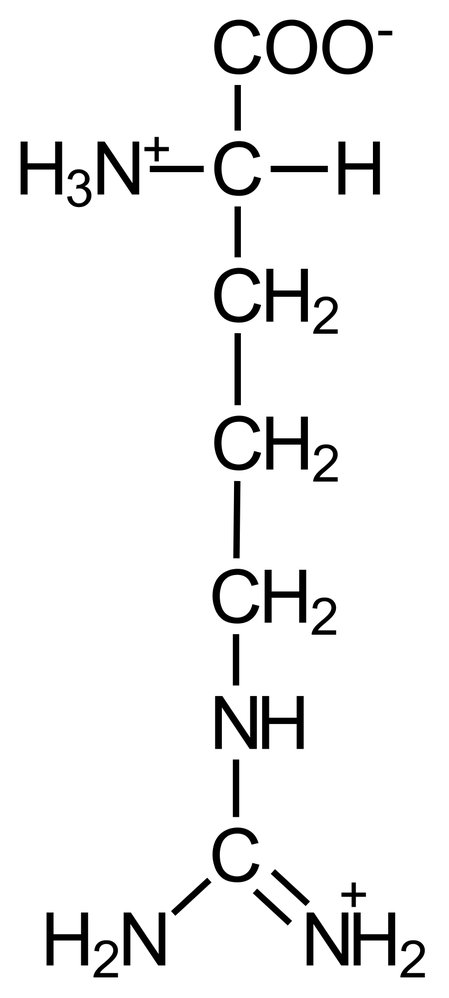
3. During translation, amino acids are covalently linked together to form a polypeptide chain.
In which derivative form does an amino acid exist immediately before being incorporated into the growing polypeptide chain at the ribosome?
A. Carboxylic acid amide
B. Carboxylic acid anhydride
C. Carboxylic acid ester
D. Carboxylic acid thioester
E. Schiff base
C. Carboxylic acid ester
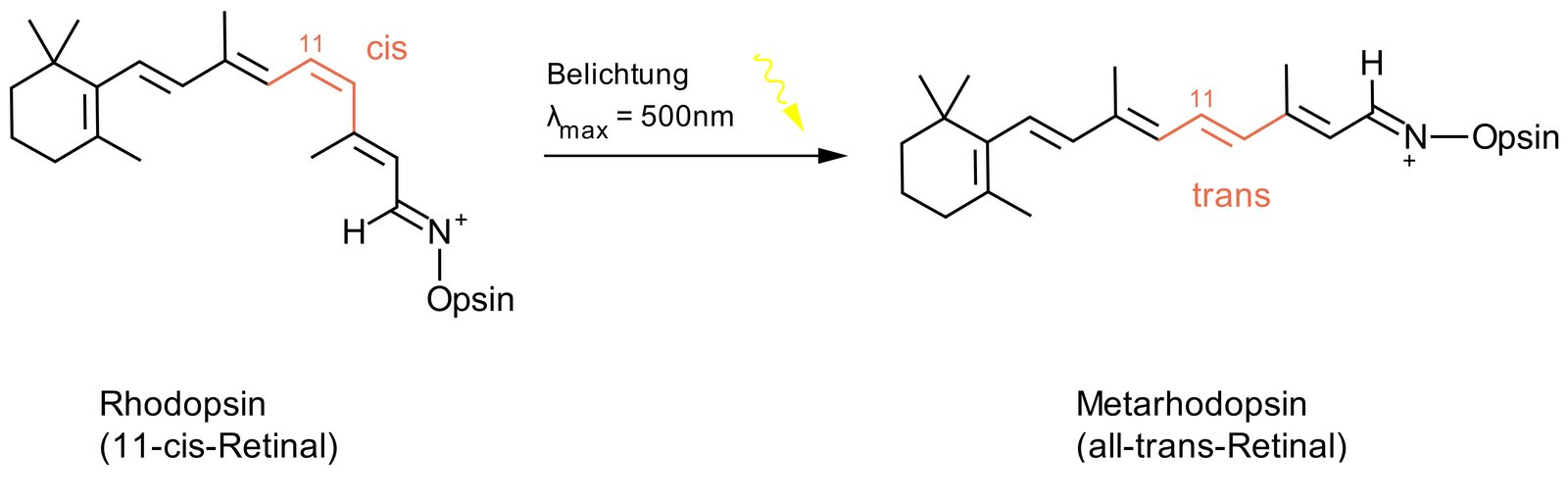
4. Molecules with the same molecular formula can adopt different structures, which can be interconverted under certain biochemical conditions.
What is the relationship between the two molecular forms that underlie light sensitivity in the primary photochemical reaction of vision?
They are:
A. Cis/trans isomers (geometric isomers)
B. Enantiomers
C. Conformational isomers
D. Constitutional isomers
E. Tautomers
A. Cis/trans isomers (geometric isomers)
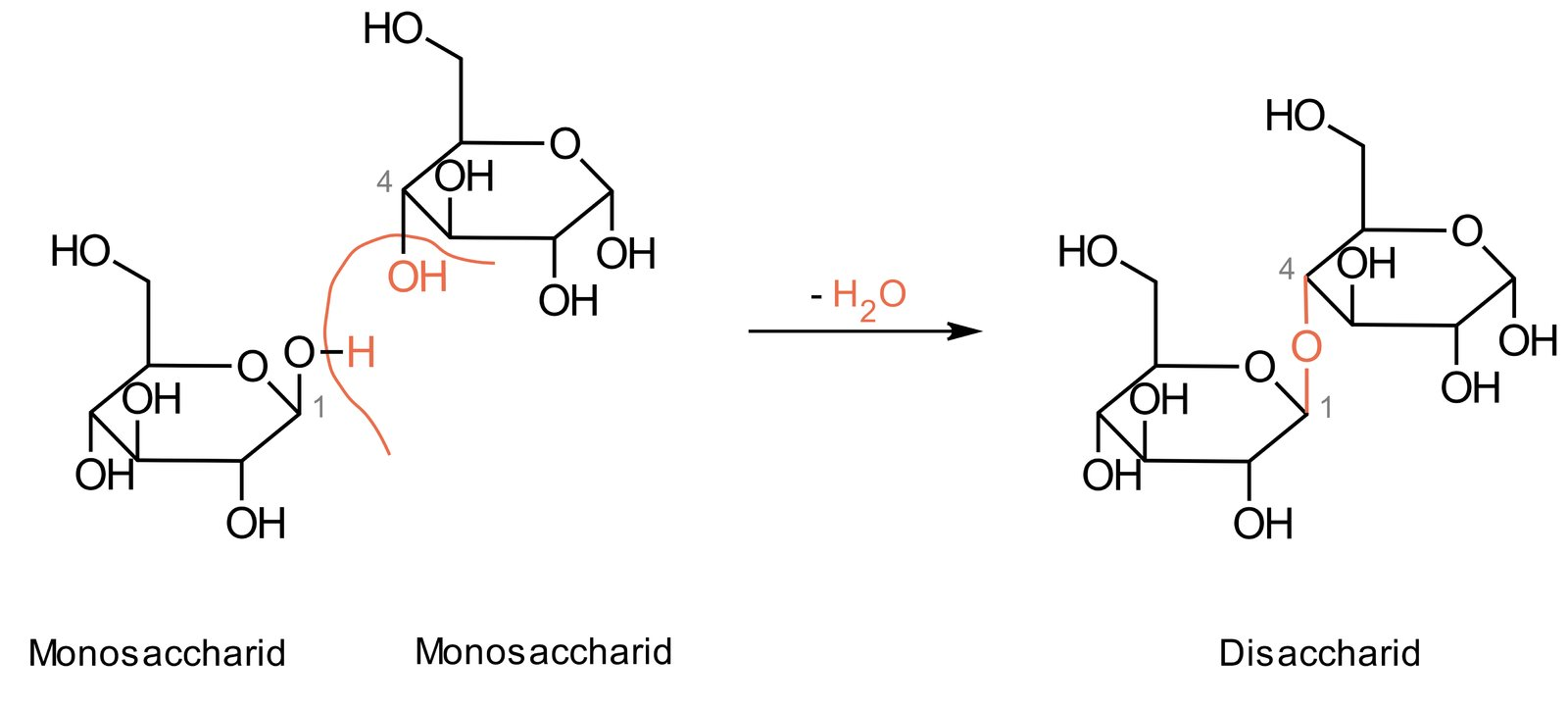
5. Compounds containing a glycosidic bond are called glycosides.
Which molecule is released net during the acid-catalyzed synthesis of a glycoside?
A. CO₂
B. H₂O
C. NH₃
D. NO
E. O₂
B. H₂O
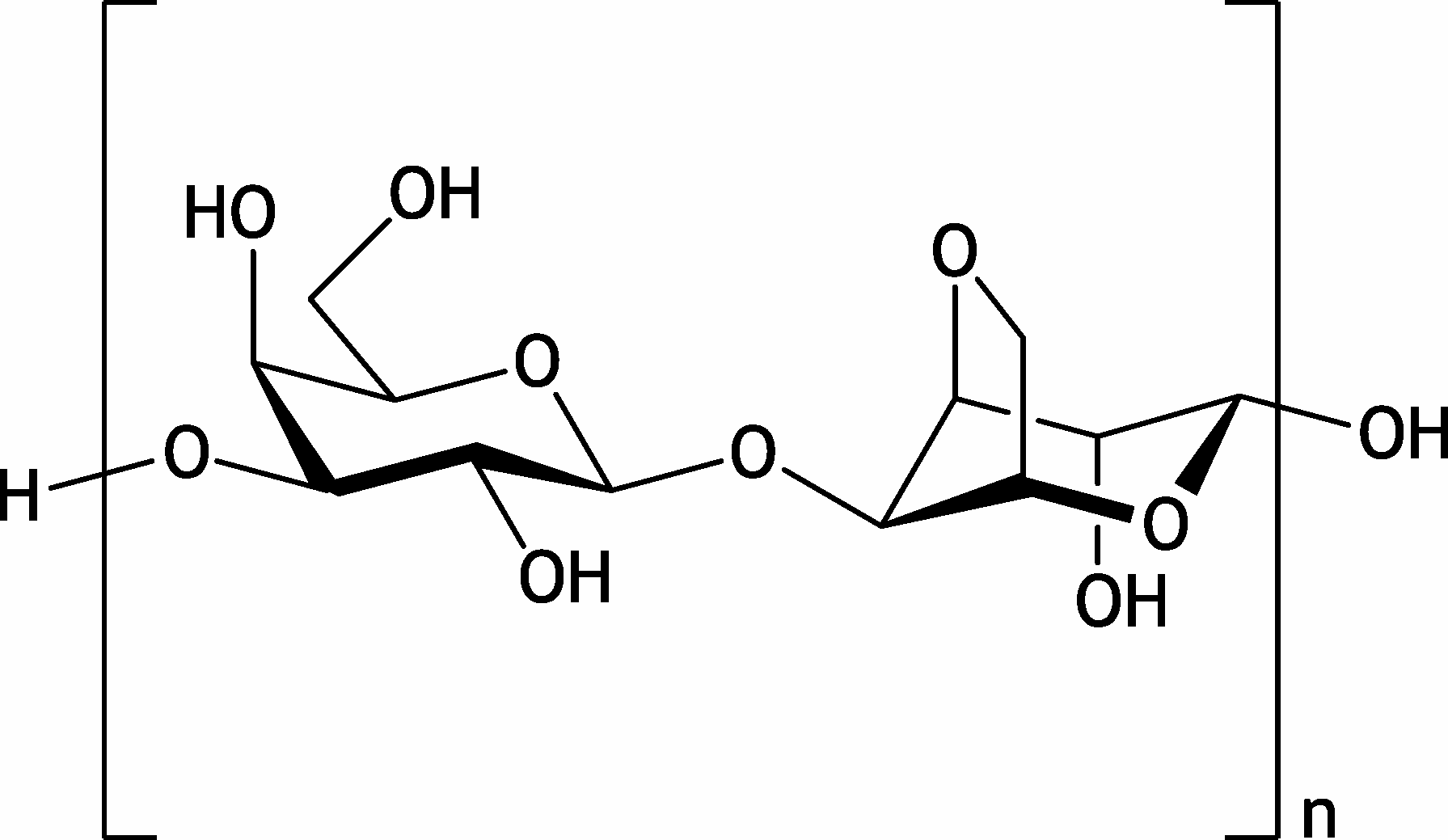
6. Agarose, used in agarose gel electrophoresis, is a polymer isolated from certain algae and composed of the shown disaccharide unit.
Which of the following monosaccharides is a component of this disaccharide unit?
A. β-D-galactose
B. α-D-glucose
C. β-D-glucose
D. β-L-glucose
E. β-D-mannose
A. β-D-galactose
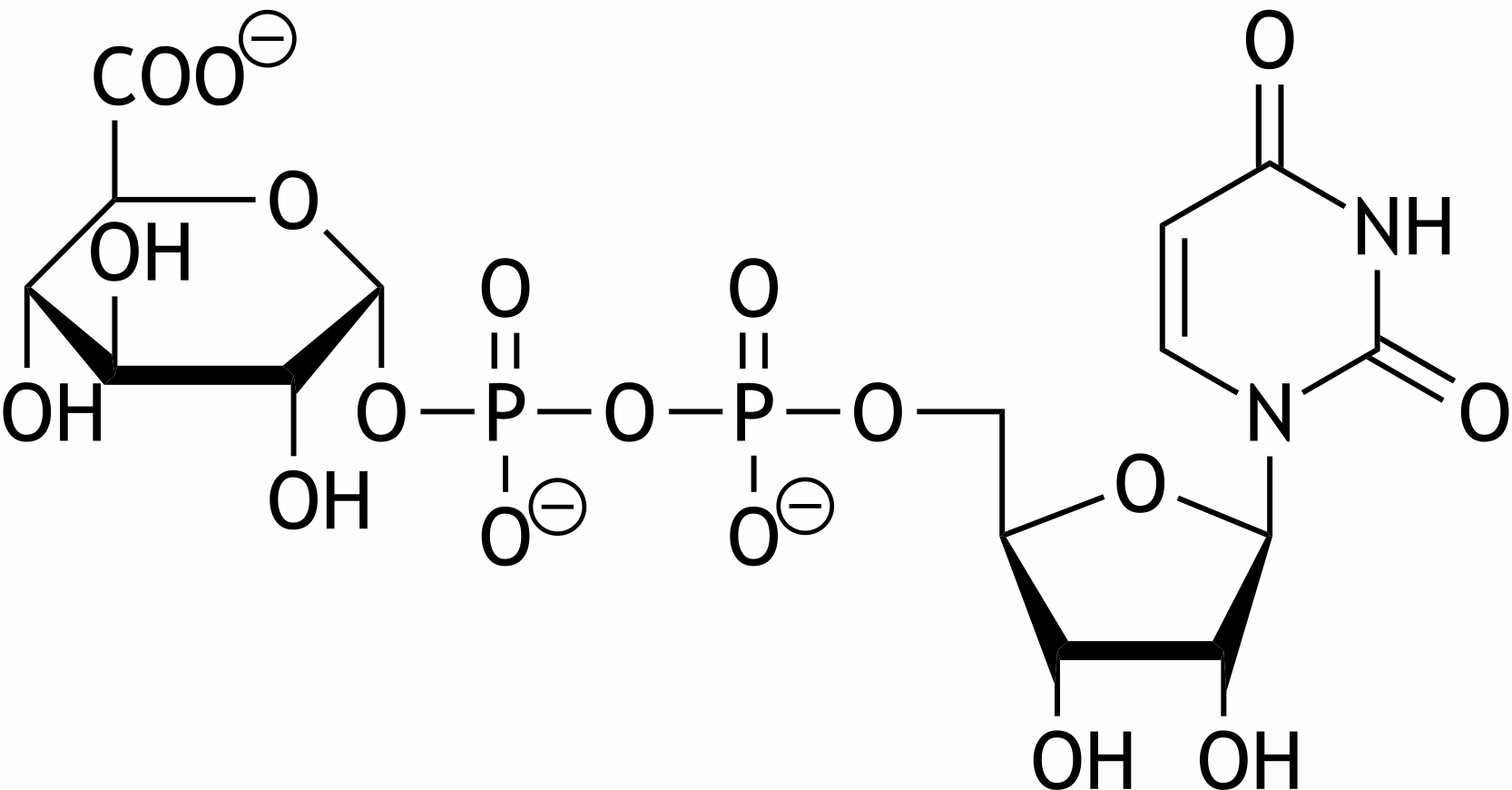
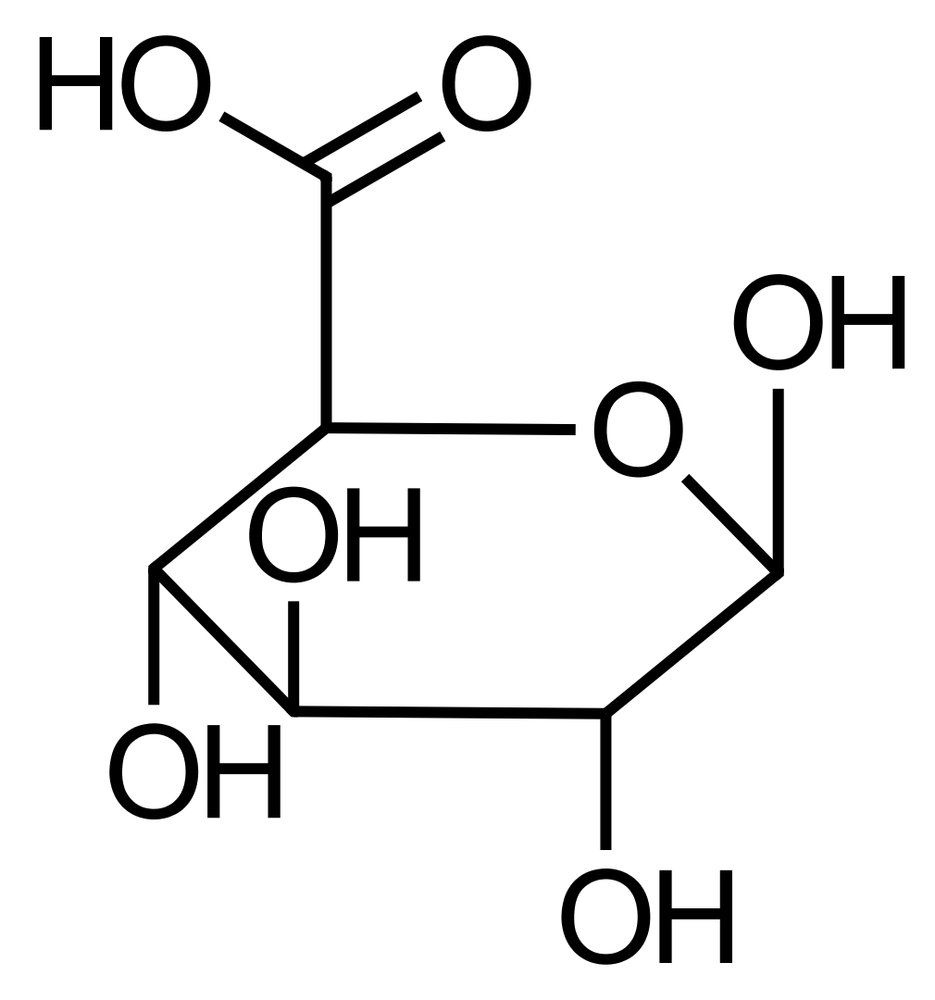
7. Carbohydrates and their derivatives must exist in a nucleoside diphosphate–activated form in metabolism in order to be transferred to another molecule. Below is a uridine diphosphate (UDP)–activated carbohydrate derivative.
Which of the following carbohydrate derivatives is it?
A. Galacturonic acid
B. Gluconic acid
C. Glucuronic acid
D. Hyaluronic acid
E. Sialic acid
C. Glucuronic acid
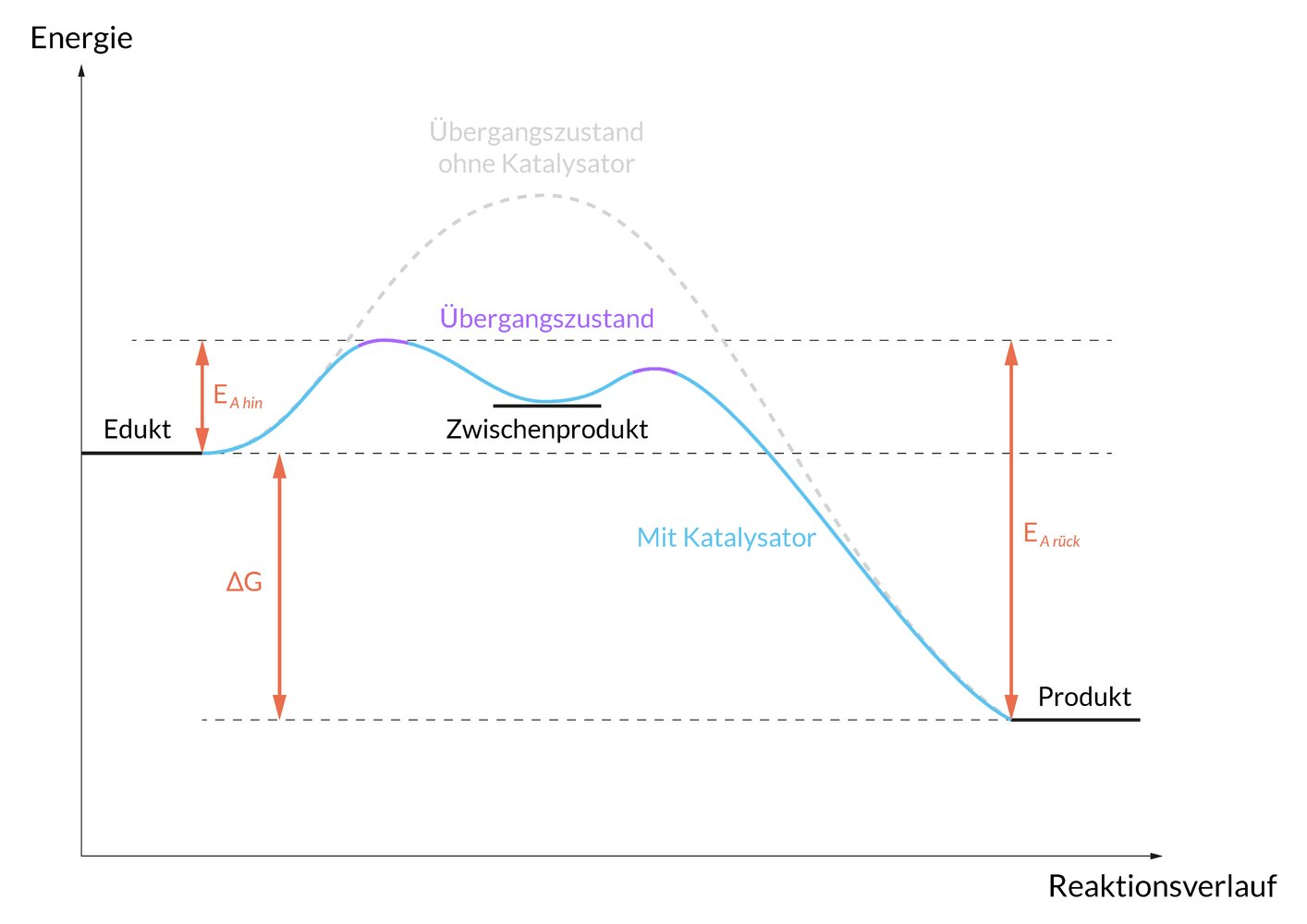
8. Enzymes accelerate chemical reactions as biocatalysts.
Which of the following mechanisms is most likely the basis of this catalytic effect?
A. Enzymes accelerate the establishment of equilibrium
B. Enzymes always form a covalent bond with the substrate
C. Enzymes increase the activation energy of a reaction
D. Enzymes shift the equilibrium toward the reaction educts
E. Enzymes shift the equilibrium toward the reaction products
A. Enzymes accelerate the establishment of equilibrium
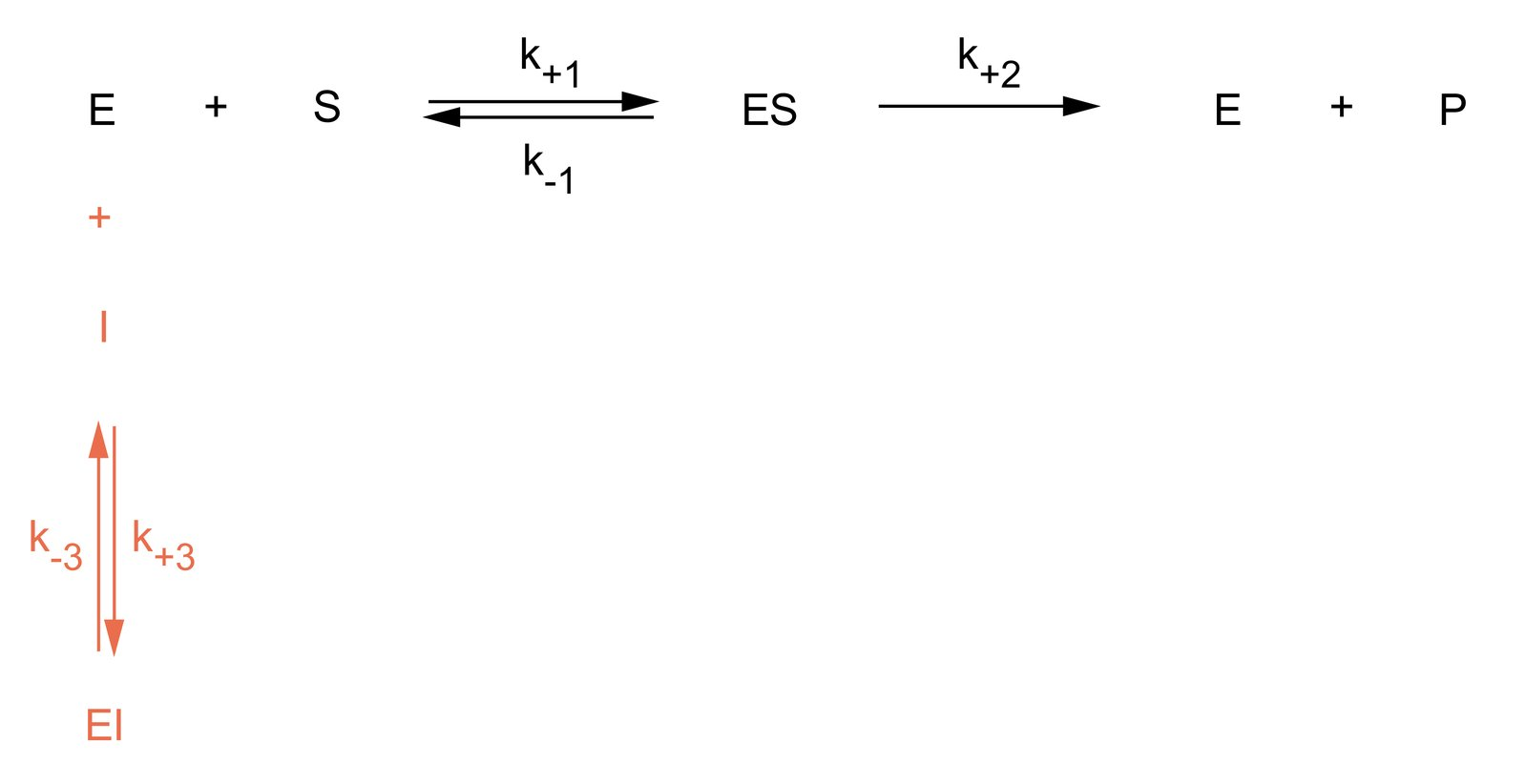
9. Enzymes can be influenced in their activity by interaction with low-molecular-weight substances (effectors). A competitive inhibitor has structural similarity to the substrate.
What is characteristic of competitive inhibition?
A. The binding of the inhibitor blocks the binding of allosteric activators
B. A strong increase in substrate concentration largely abolishes the effect of the inhibitor
C. The inhibition requires simultaneous binding of the inhibitor and the substrate
D. The inhibition results from the binding of the inhibitor outside the active site
E. The Michaelis-Menten constant (Michaelis constant, KM, Km) is decreased by the inhibitor
B. A strong increase in substrate concentration largely abolishes the effect of the inhibitor
10. For an enzymatically catalyzed reaction that follows Michaelis-Menten kinetics, the kinetic parameters KM (Michaelis-Menten constant, Michaelis constant, Km) and vmax of an enzyme were determined in a first measurement at a defined enzyme concentration. This determination is now repeated in a second measurement, but with double the enzyme concentration.
Which changes are most likely to occur regarding KM and vmax compared to the first measurement?
A. KM remains unchanged, vmax is doubled
B. KM and vmax both remain unchanged
C. KM and vmax are both doubled
D. KM is halved, vmax remains unchanged
E. KM is doubled, vmax remains unchanged
A. KM remains unchanged, vmax is doubled
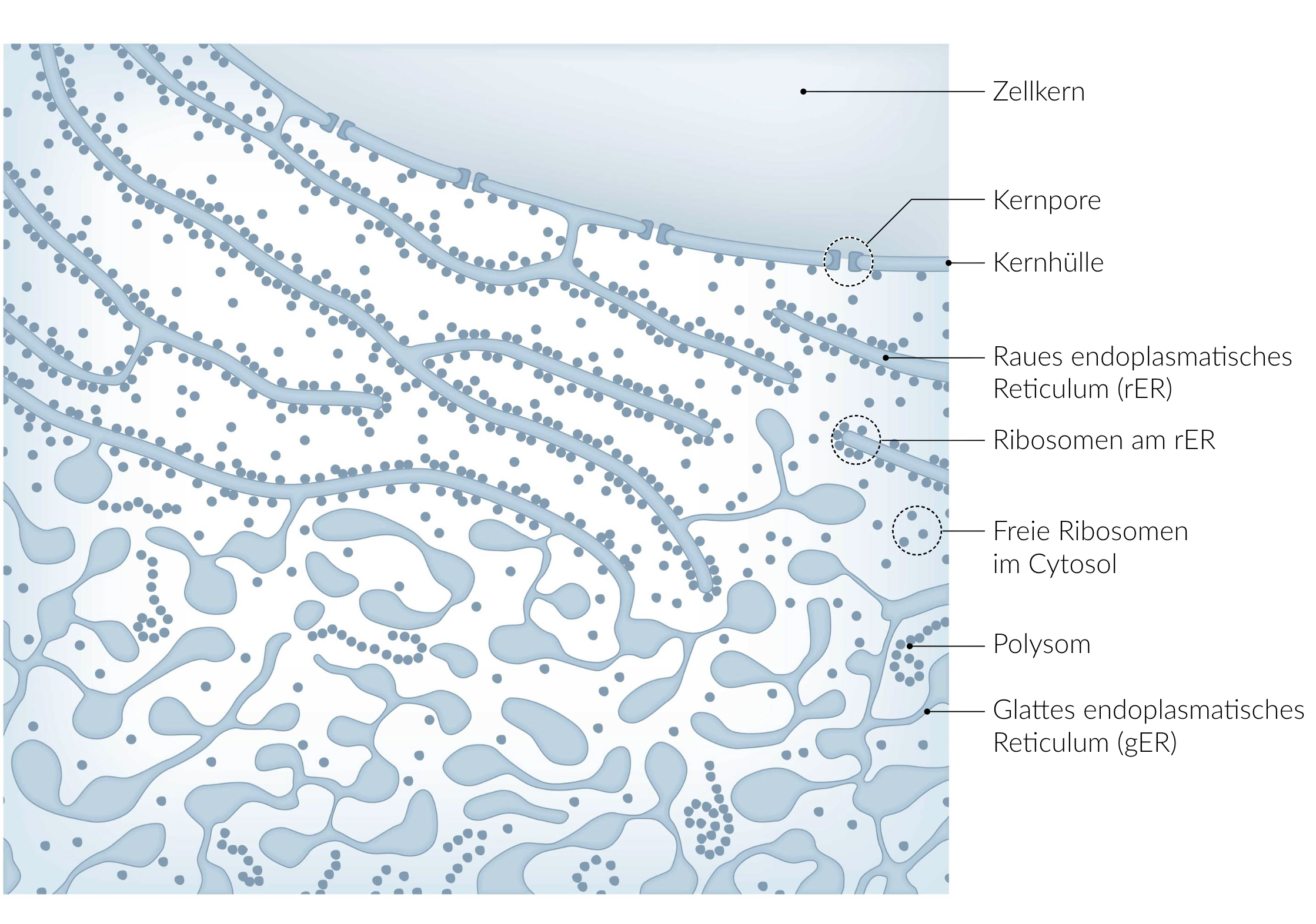
11. In lysosomes, phagocytosed material is degraded and thus made available to the cell. If lysosomal enzyme activities are reduced or absent due to decreased or missing synthesis of the corresponding enzymes, or due to the production of defective enzymes, this can cause so-called lysosomal storage diseases.
Where does the main part of the biosynthesis of most lysosomal enzymes take place in healthy individuals?
A. At the rough endoplasmic reticulum
B. In the Golgi apparatus
C. In the lysosomes
D. In the mitochondria
E. In the peroxisomes
A. At the rough endoplasmic reticulum
12. Hereditary defects of the enzyme sphingomyelinase cause a lysosomal storage disease known as Niemann-Pick disease. Sphingomyelinase catalyzes the cleavage of sphingomyelin.
Which of the following compounds is formed in this cleavage in addition to ceramide?
A. Arachidonic acid
B. Glucose
C. Inositol-1,4,5-trisphosphate
D. Phosphorylcholine
E. Sialic acid
D. Phosphorylcholine
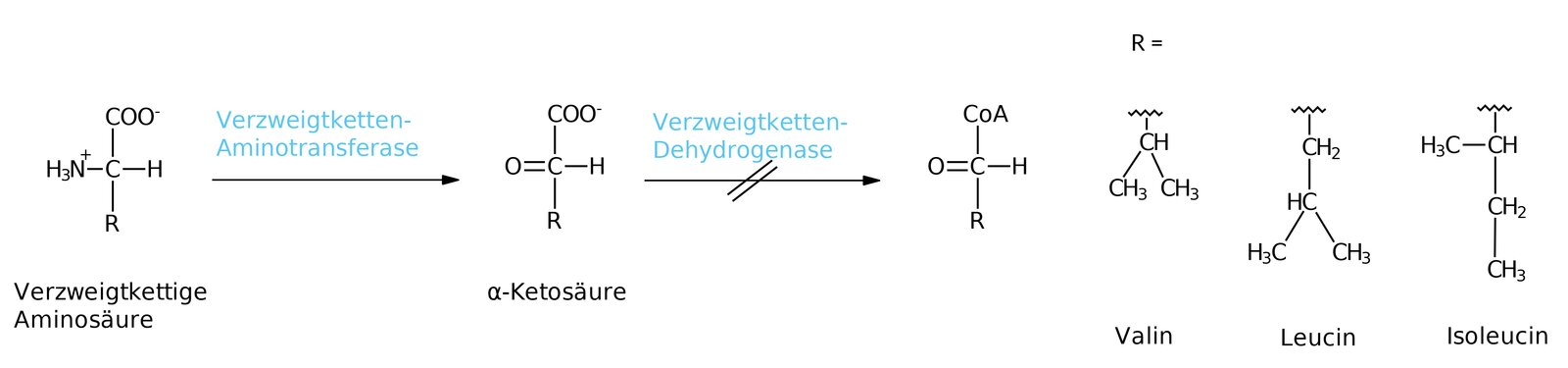
13. Maple syrup urine disease is based on a genetic disorder of amino acid metabolism. In affected patients, branched-chain α-keto acids are only decarboxylated to a reduced extent.
The degradation of which of the following amino acids is directly impaired in this context?
A. Leucine
B. Lysine
C. Methionine
D. Phenylalanine
E. Threonine
A. Leucine
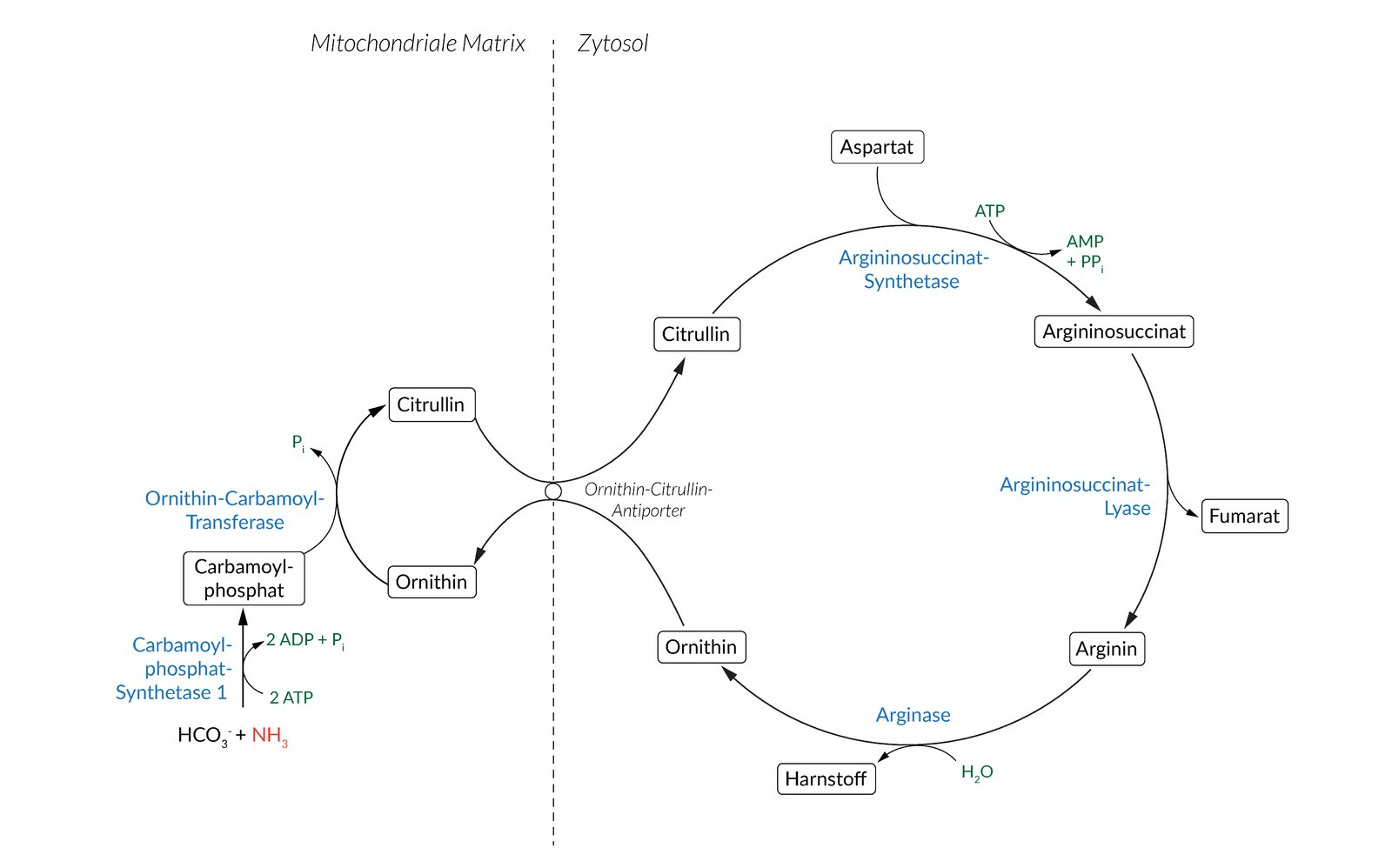
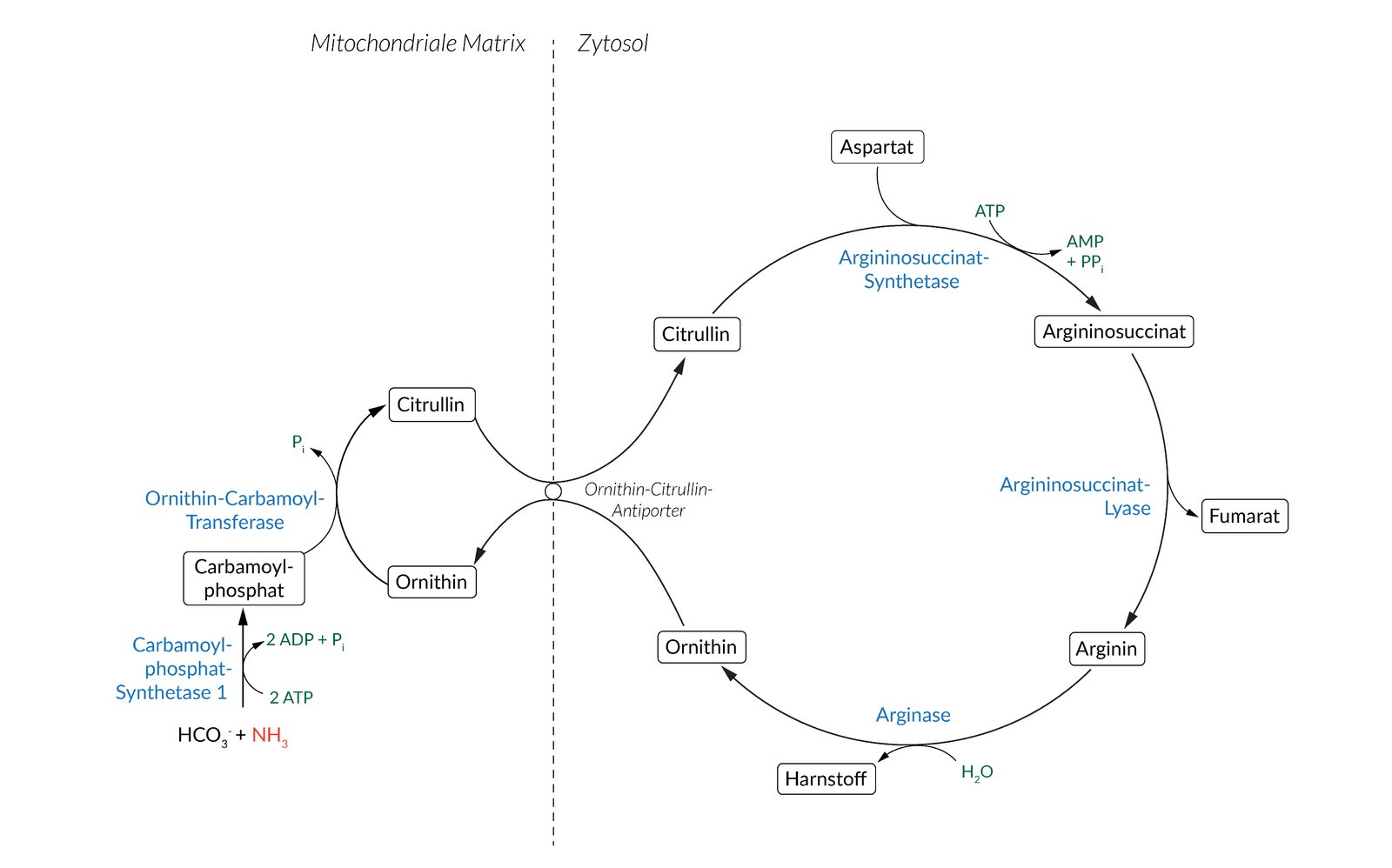
14. Defects of the enzyme N-acetylglutamate synthase can lead to disorders of the urea cycle.
Which of the following enzymes is allosterically activated by N-acetylglutamate?
A. Adenylosuccinate synthetase
B. Arginase
C. Argininosuccinate lyase
D. Carbamoyl phosphate synthetase I
E. Ornithine transcarbamylase
D. Carbamoyl phosphate synthetase I
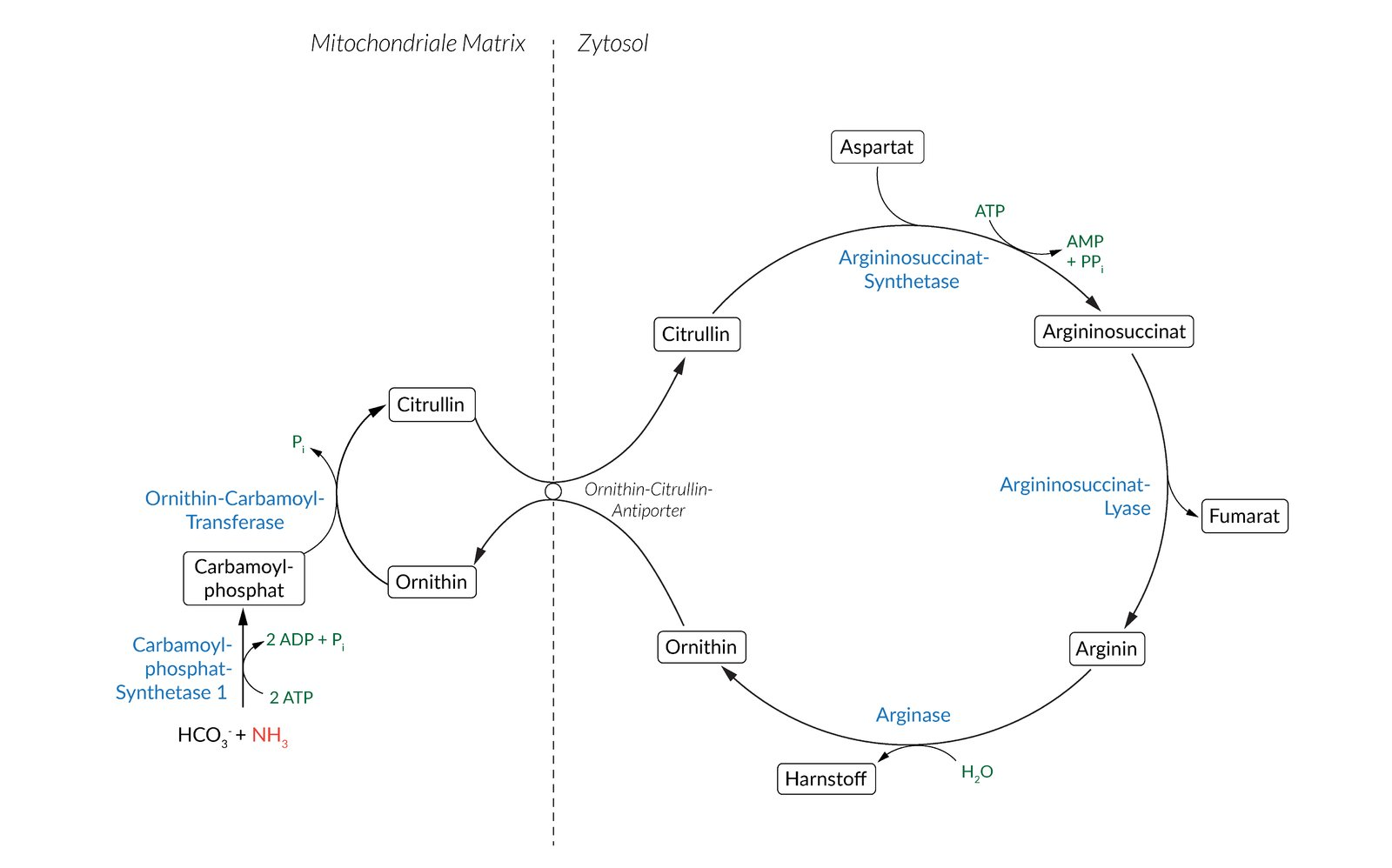
15. The reactions of the urea cycle are distributed across two cellular compartments — the mitochondria and the cytosol. A metabolite carrier in the inner mitochondrial membrane is therefore essential for the overall functionality of the urea cycle.
Which two compounds does this carrier exchange as an antiporter?
A. Arginine and guanidinoacetate
B. Aspartate and fumarate
C. Citrate and malate
D. Citrulline and ornithine
E. Glutamate and α-ketoglutarate
D. Citrulline and ornithine
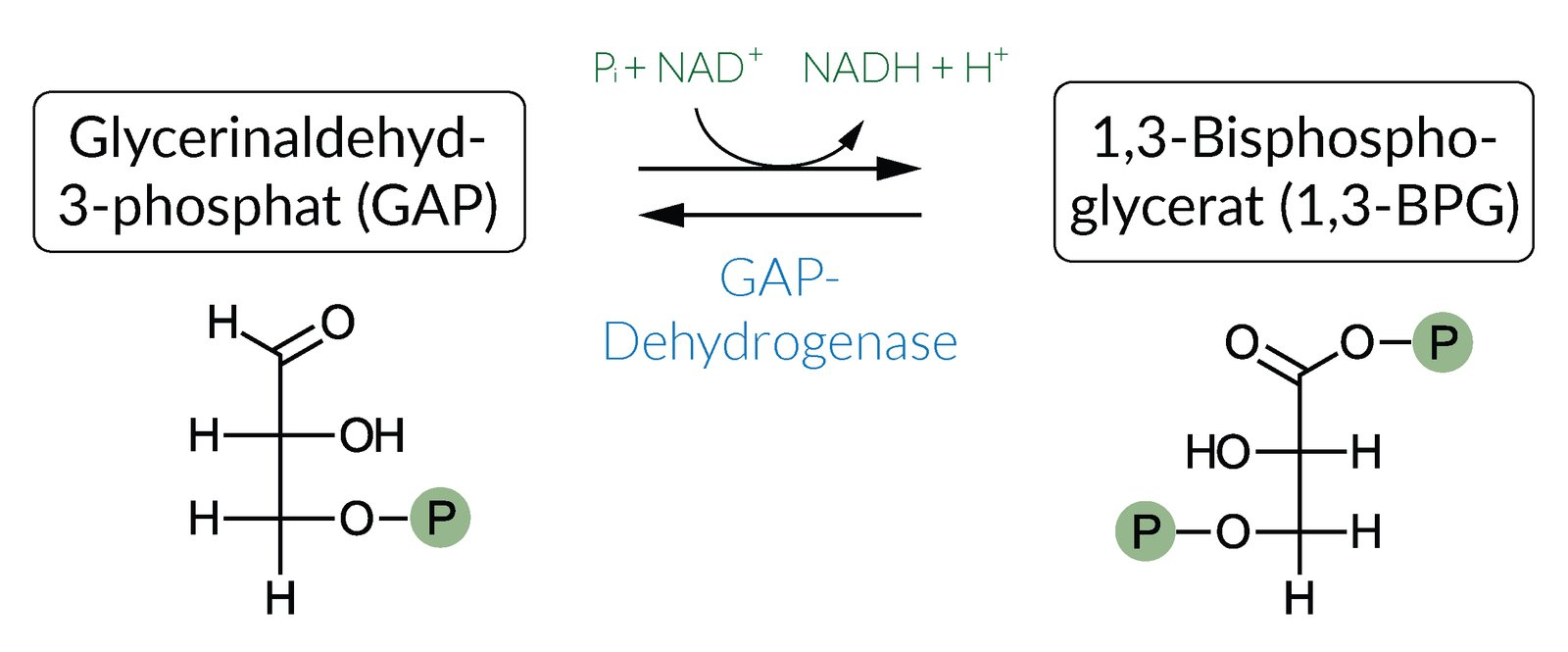
16. In anaerobic glycolysis, ATP is generated from ADP by substrate-level phosphorylation. In the reaction that occurs directly before this substrate-level phosphorylation, inorganic phosphate is linked to a carbon skeleton via a high-energy bond.
Which enzyme catalyzes this reaction?
A. Aldolase A
B. Glyceraldehyde-3-phosphate dehydrogenase
C. Hexokinase
D. Phosphofructokinase-1
E. Pyruvate kinase
B. Glyceraldehyde-3-phosphate dehydrogenase
17. The pyruvate dehydrogenase complex consists of different subunits and requires various cofactors for its enzymatic activities.
Which of the following molecules is a cofactor of the pyruvate dehydrogenase complex?
A. Ascorbic acid
B. Glutathione
C. Lipoamide
D. NADPH
E. Pyridoxal phosphate
C. Lipoamide
18. Metabolites of the citric acid cycle serve as precursors for the biosynthesis of numerous important compounds in the body. To prevent the citric acid cycle from coming to a halt due to these processes, they must be balanced by so-called replenishing reactions (anaplerotic reactions).
Which of the following enzymes catalyzes such an anaplerotic reaction?
A. Acetyl-CoA carboxylase
B. δ-Aminolevulinate synthase
C. α-Ketoglutarate dehydrogenase
D. Pyruvate carboxylase
E. Pyruvate dehydrogenase
D. Pyruvate carboxylase
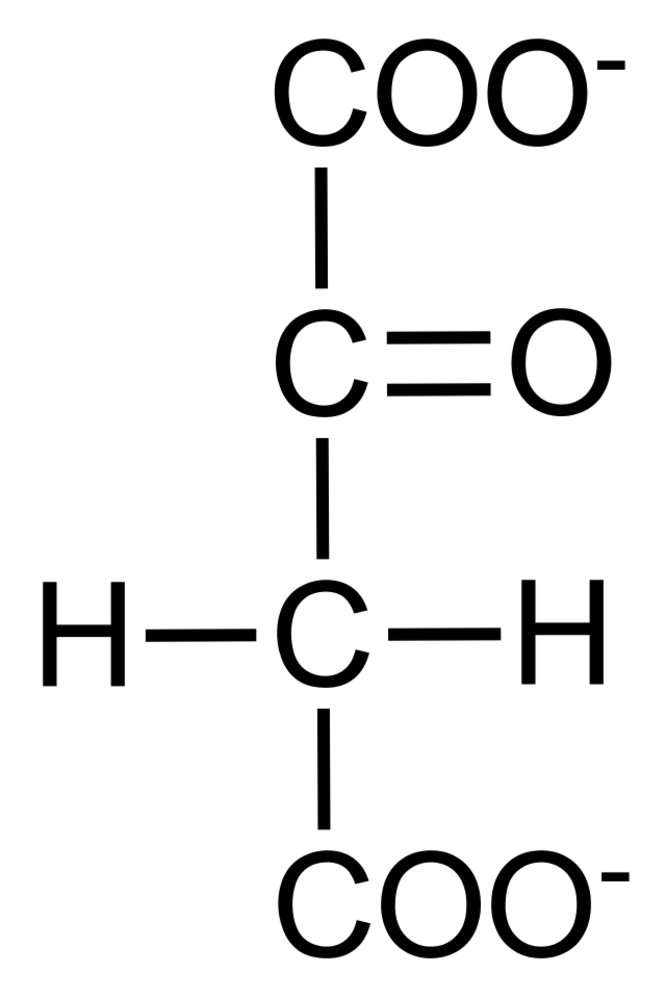
19. In the respiratory chain, oxygen is ultimately reduced with the formation of water. The reducing equivalents NADH and FADH₂ are oxidized in the process.
How many moles of water are formed at most in the respiratory chain per mole of oxidized NADH?
A. 0.5
B. 1
C. 1.5
D. 2
E. 4
B. 1
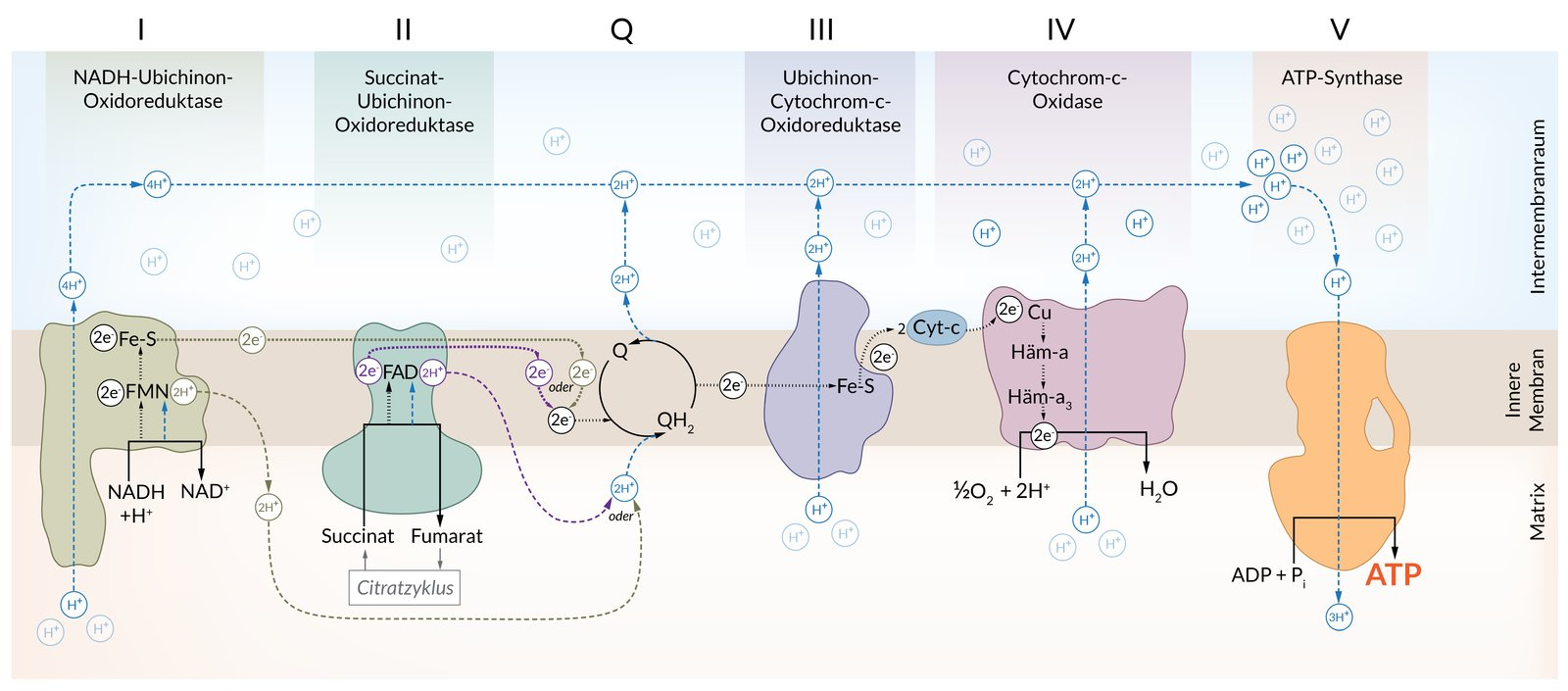
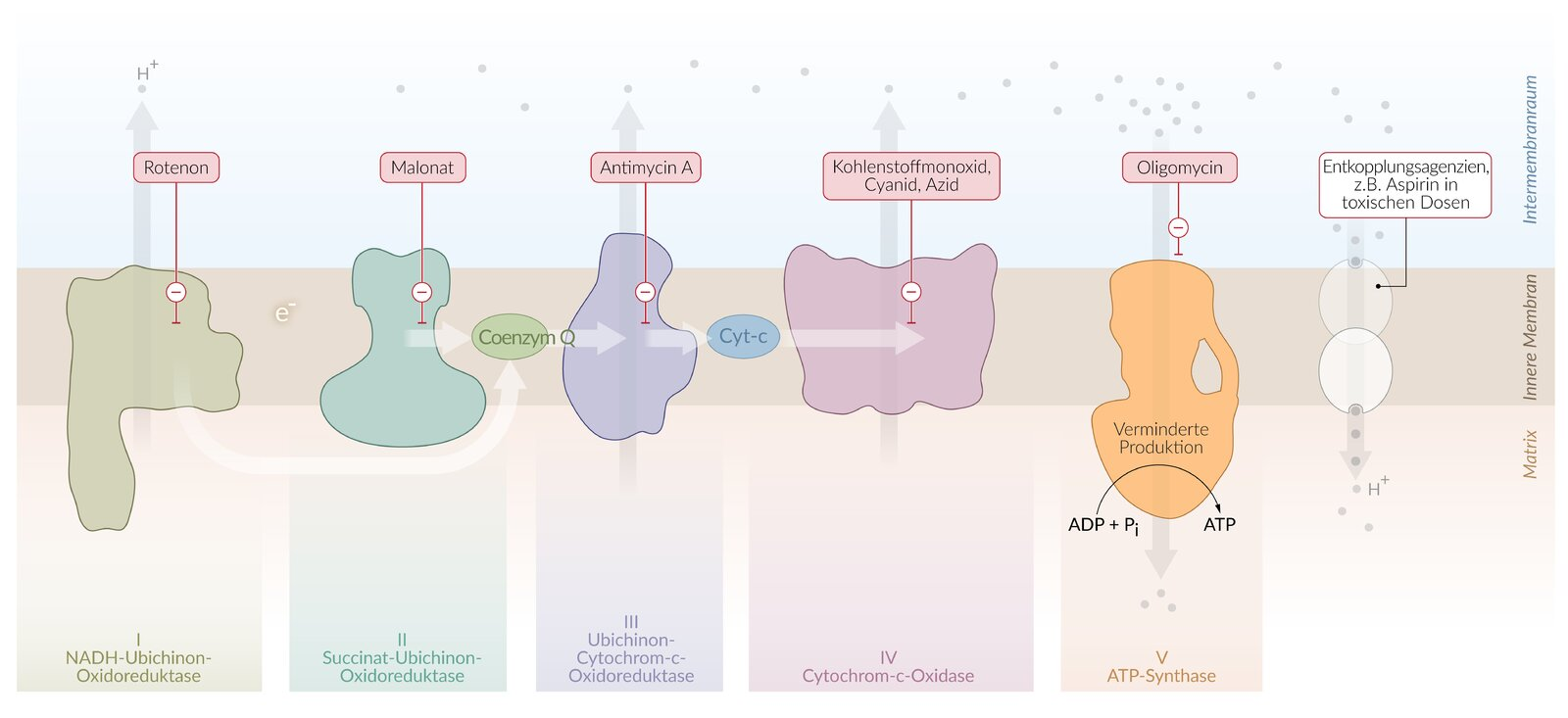
20. Uncouplers are small organic molecules or proteins that influence the process of ATP synthesis through oxidative phosphorylation in mitochondria.
Which of the following effects on mitochondrial energy metabolism best describes the immediate consequence of the presence of an uncoupler?
A. Release of cytochrome c from the intermembrane space into the cytosol
B. Ubiquitinylation of subunits of NADH–ubiquinone oxidoreductase (complex I)
C. Increased enzymatic degradation of the electron carrier ubiquinone
D. Decreased activity of succinate dehydrogenase (complex II)
E. Decrease of the H⁺ gradient across the inner mitochondrial membrane
E. Decrease of the H⁺ gradient across the inner mitochondrial membrane
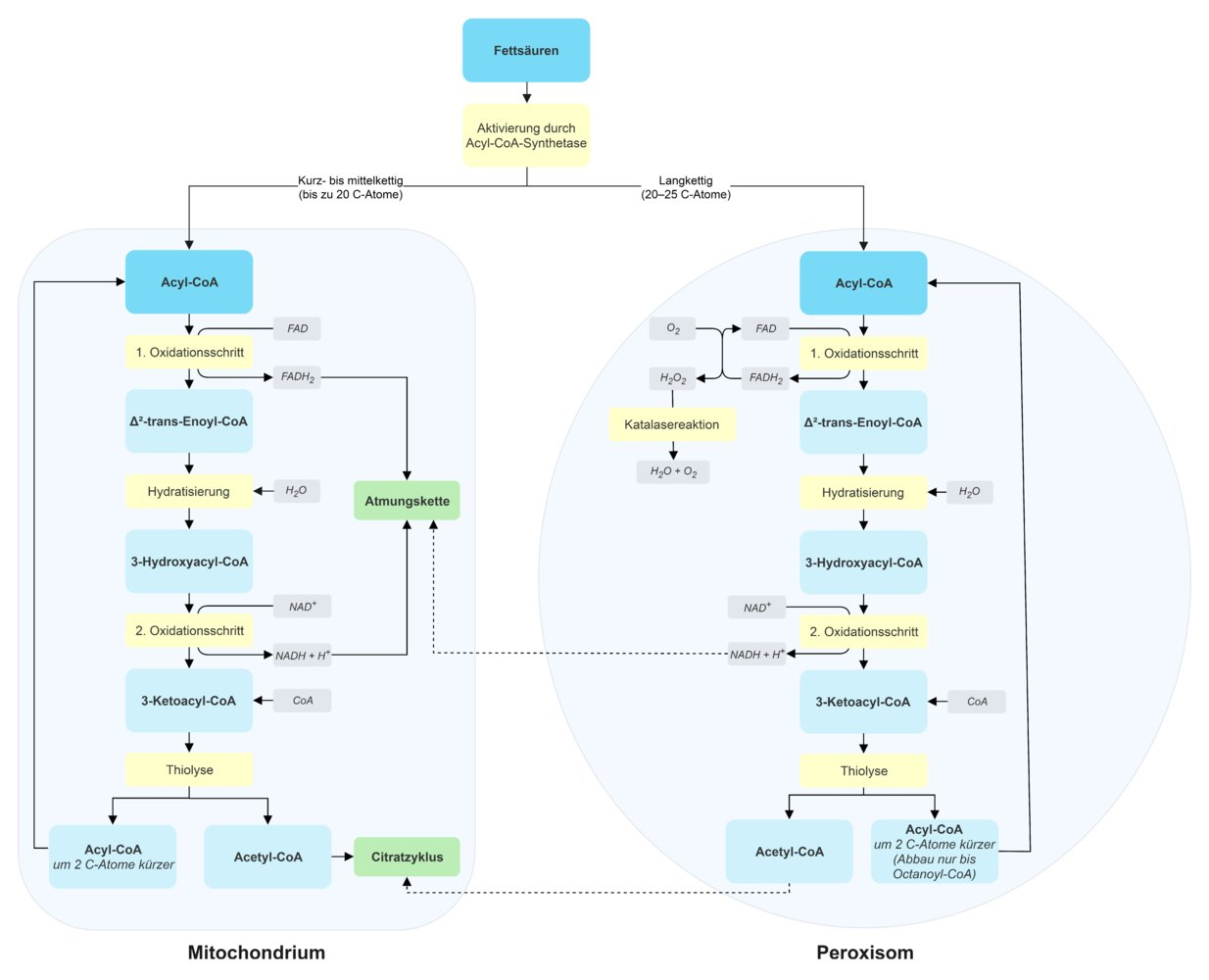
21. As part of newborn screening, a defect in mitochondrial β-oxidation of fatty acids is detected in a girl. This condition is typically associated with hypoglycemic crises.
The absence of which allosteric activator of pyruvate carboxylase is most likely to lead to such a hypoglycemic crisis in this patient?
A. Acetyl-CoA
B. Malonyl-CoA
C. Methylmalonyl-CoA
D. Propionyl-CoA
E. Succinyl-CoA
A. Acetyl-CoA
22. For the synthesis of fatty acids, large amounts of reducing equivalents in the form of NADPH are required.
Which of the following biochemical reactions in the cytosol is mainly responsible for providing NADPH when the pentose phosphate pathway is insufficient in this regard?
A. The conversion of dihydrofolate to folate
B. The conversion of glutamate to α-ketoglutarate
C. The conversion of glutathione to glutathione disulfide
D. The conversion of malate to pyruvate
E. The conversion of sorbitol to fructose
D. The conversion of malate to pyruvate
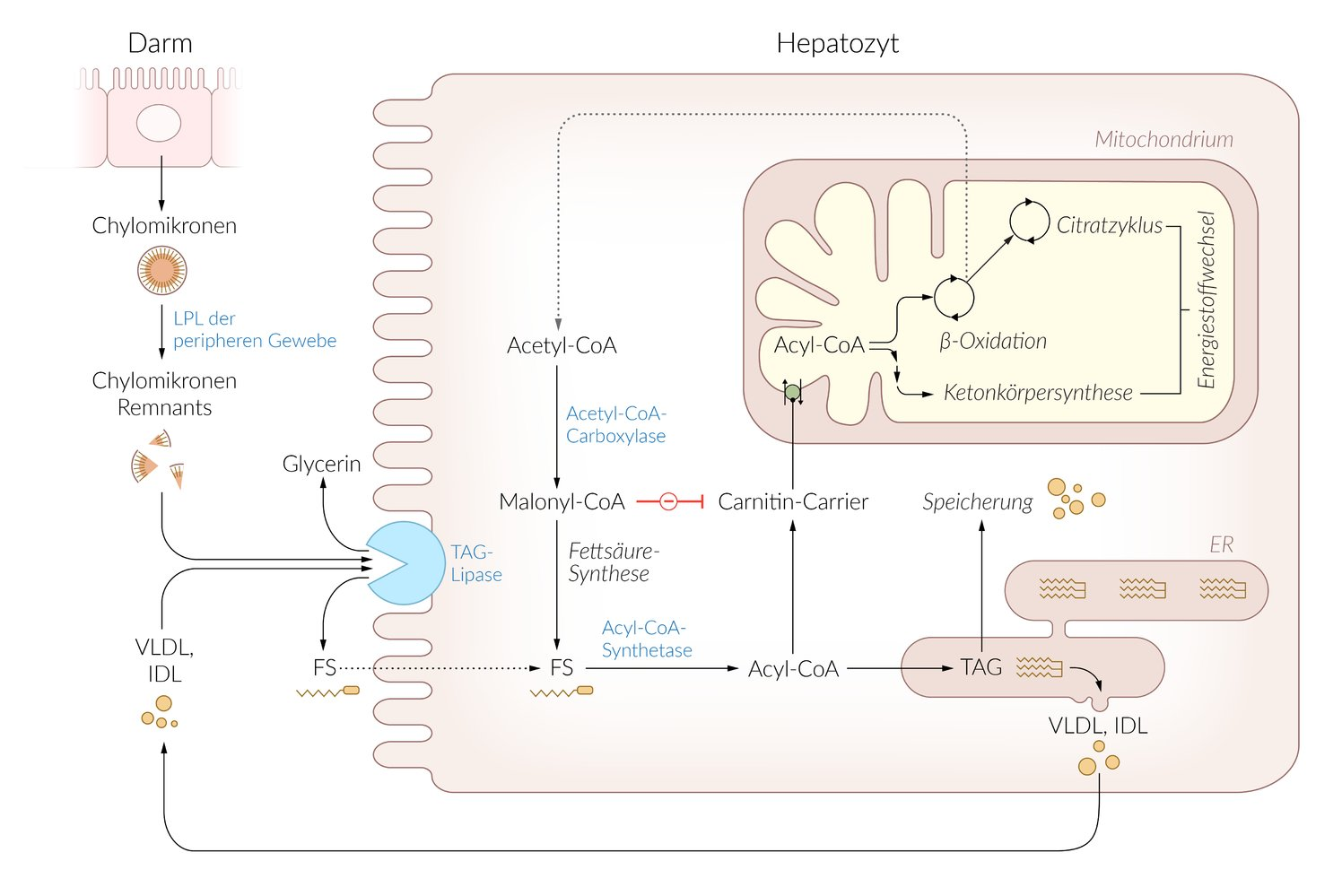
23. In an animal model, muscle metabolism is studied under conditions of hyperglycemia and hyperinsulinemia. After several hours of rest, muscle biopsies are taken, and the concentration of malonyl-CoA is measured. Compared to untreated controls, the concentration of malonyl-CoA in the muscle tissue of the test animals is markedly increased.
Which of the following metabolic processes is most likely inhibited in the test animals due to the strongly increased concentration of malonyl-CoA?
A. Citric acid cycle
B. Fatty acid oxidation
C. Fatty acid synthesis
D. Glycolysis
E. Glycogen breakdown
B. Fatty acid oxidation
24. In the course of the apoptosis process, certain enzymes called caspases play a central role.
To which enzyme class do caspases belong?
A. Hydrolases
B. Isomerases
C. Lyases
D. Oxidoreductases
E. Transferases
A. Hydrolases
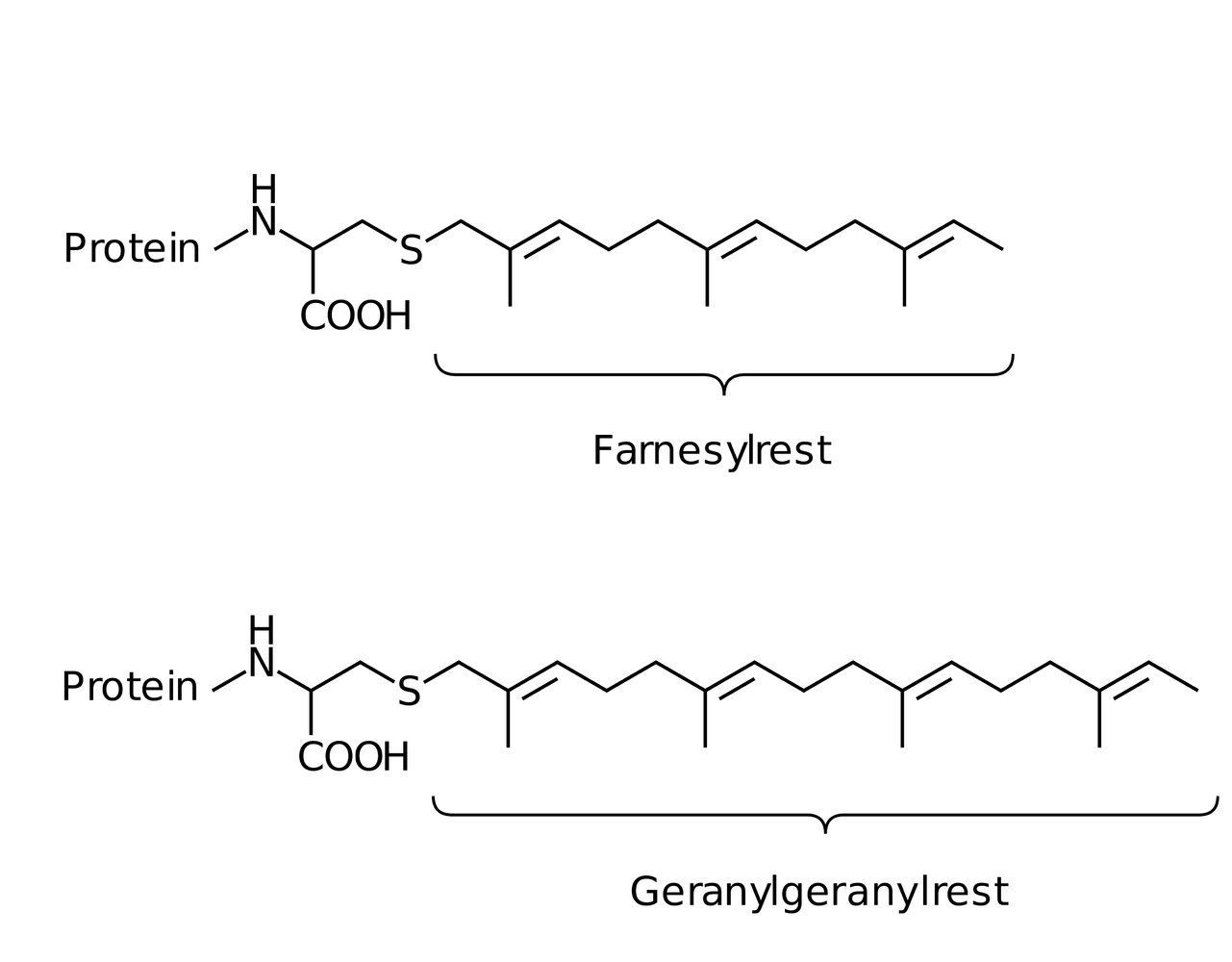
25. In pancreatic carcinomas, the activity of the G protein Ras is usually impaired. This signaling protein is anchored to the plasma membrane intracellularly through a specific modification.
Which of the following modifications most likely causes this stable membrane attachment?
A. Acetylation
B. Farnesylation
C. Glycosylation
D. Phosphorylation
E. Ubiquitinylation
B. Farnesylation
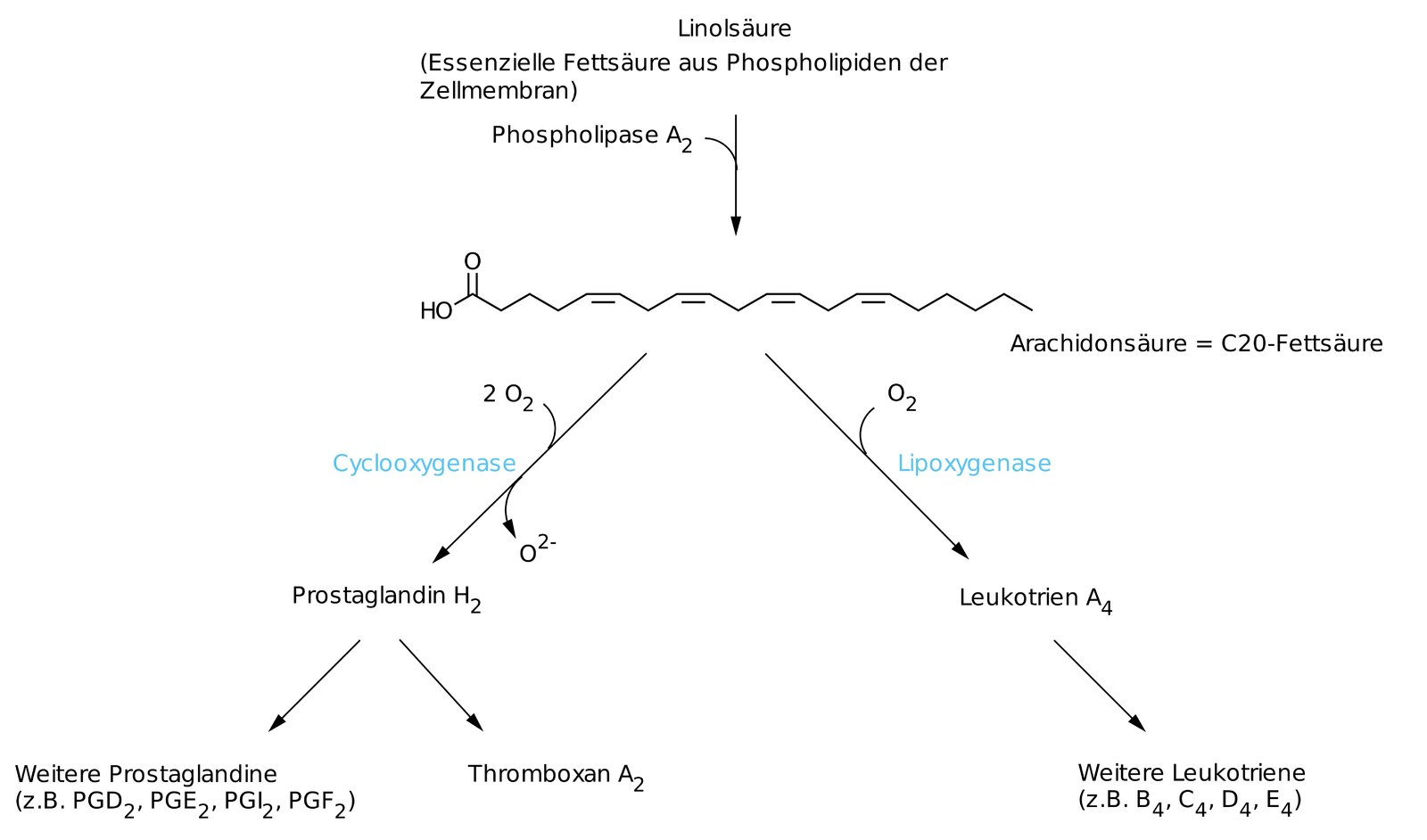
26. Polyunsaturated fatty acids are essential dietary components for humans.
For which of the following are they therefore absolutely required?
A. Formation of a membrane potential
B. Cardiolipin synthesis
C. Eicosanoid synthesis
D. Detoxification of reactive oxygen species
E. Decrease in the fluidity of the plasma membrane
C. Eicosanoid synthesis
27. Starting from cholesterol, various compounds can be synthesized enzymatically—partly through several intermediate steps.
Which of the following compounds is a product of a biosynthetic pathway involving cholesterol 7α-hydroxylase (CYP7A1)?
A. Aldosterone
B. Cholecalciferol
C. Cholic acid
D. Pregnenolone
E. Testosterone
C. Cholic acid
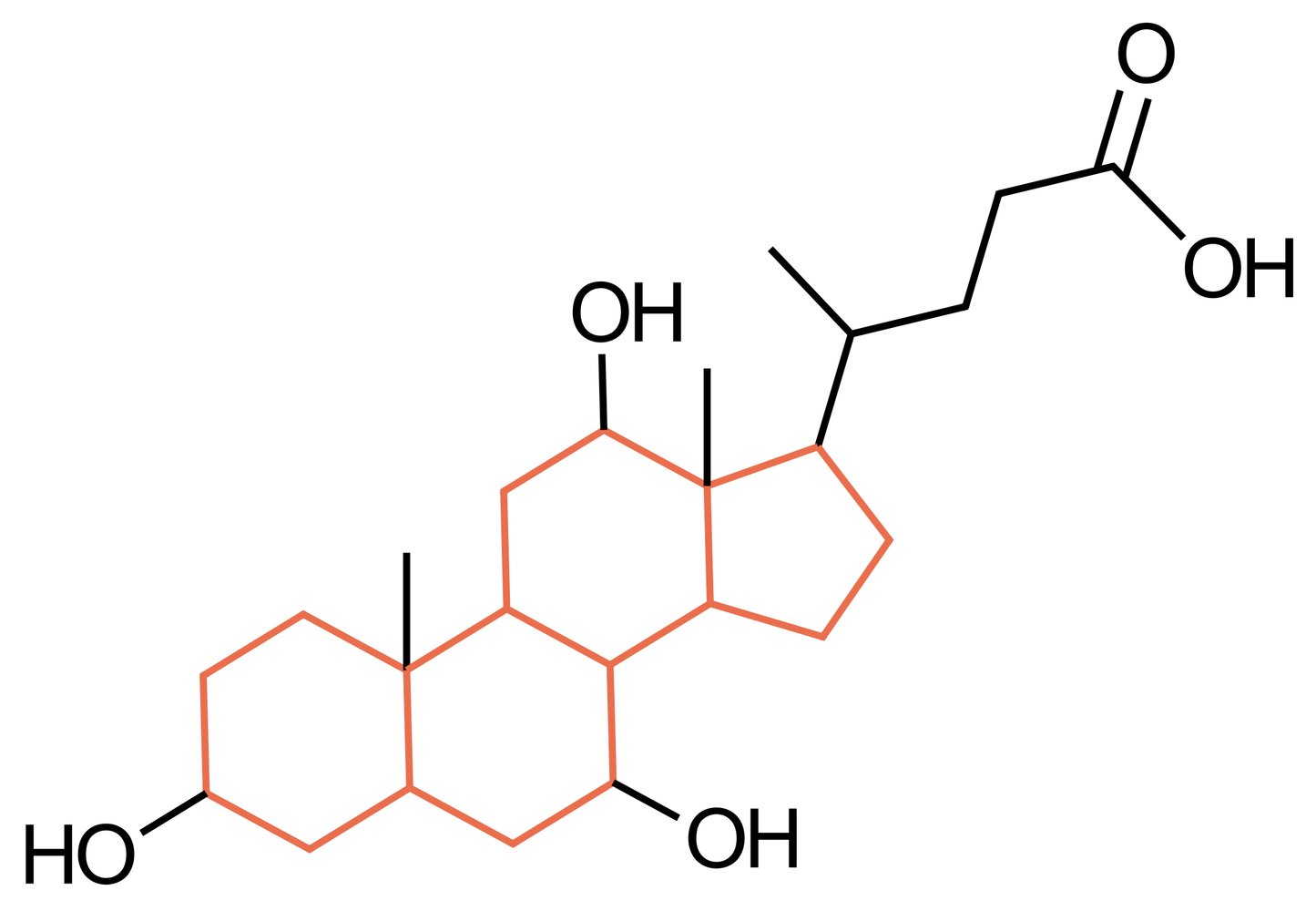
28. An 80-year-old patient is admitted to the hospital with muscle cramps and difficulty swallowing. A few days earlier, she sustained a cut while gardening. A microbiological examination of the wound confirms the suspected diagnosis of an infection with Clostridium tetani, which can release tetanus toxin.
The release of which of the following neurotransmitters is most likely inhibited in this patient by tetanus toxin?
A. Acetylcholine
B. Dopamine
C. Glutamate
D. Glycine
E. Norepinephrine
D. Glycine
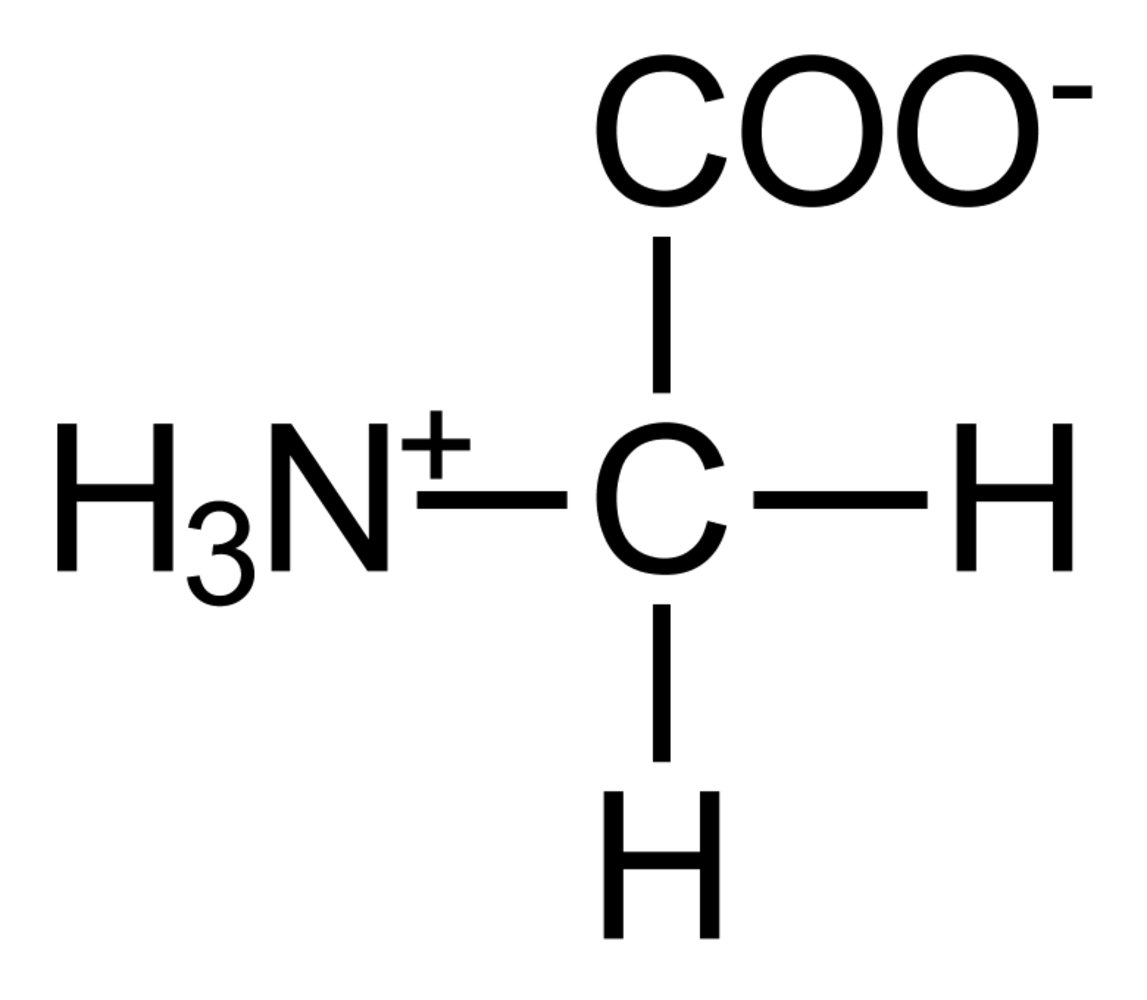
29. Levodopa (L-Dopa), a drug used in the treatment of Parkinson’s disease, as well as catecholamines and other biogenic amines, can be methylated and thereby inactivated by catechol-O-methyltransferase (COMT).
Which metabolite serves directly as the methyl group donor for COMT in this process?
A. Methylcobalamin
B. N⁵,N¹⁰-Methylene-tetrahydrofolate
C. N⁵-Methyl-tetrahydrofolate
D. Phosphatidylcholine
E. S-adenosylmethionine
E. S-adenosylmethionine
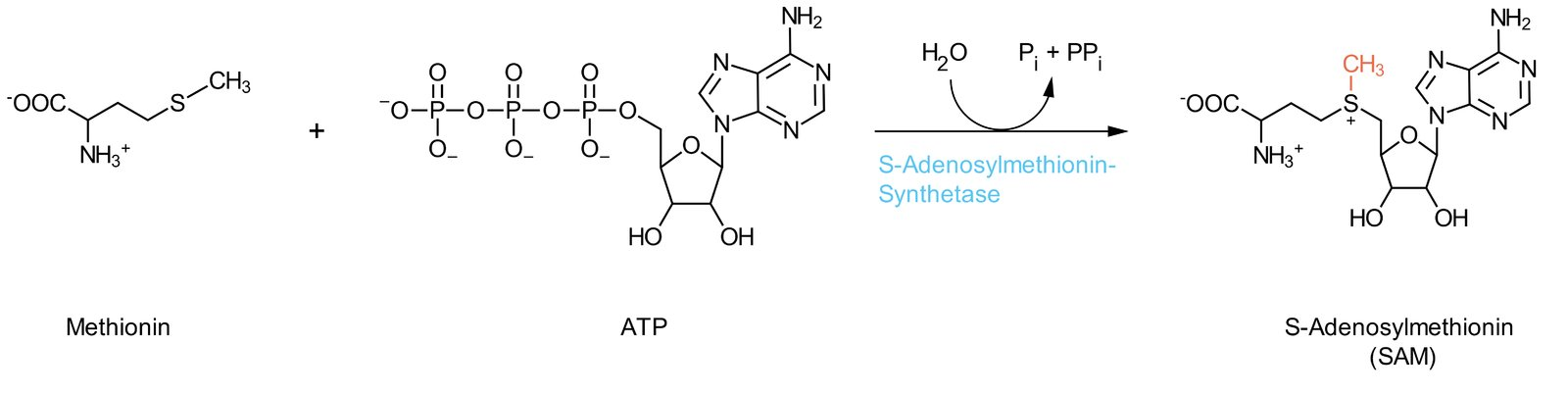
30. A 44-year-old patient with the autoimmune disease psoriatic arthritis is being treated with the immunosuppressive drug leflunomide. In the body, the active metabolite teriflunomide is formed from leflunomide. This metabolite inhibits dihydroorotate dehydrogenase (DHODH).
The synthesis of which of the following biomolecules is directly blocked by this enzyme inhibition?
A. Glycolipids
B. Prostaglandins
C. Purines
D. Pyrimidines
E. Steroid hormones
D. Pyrimidines
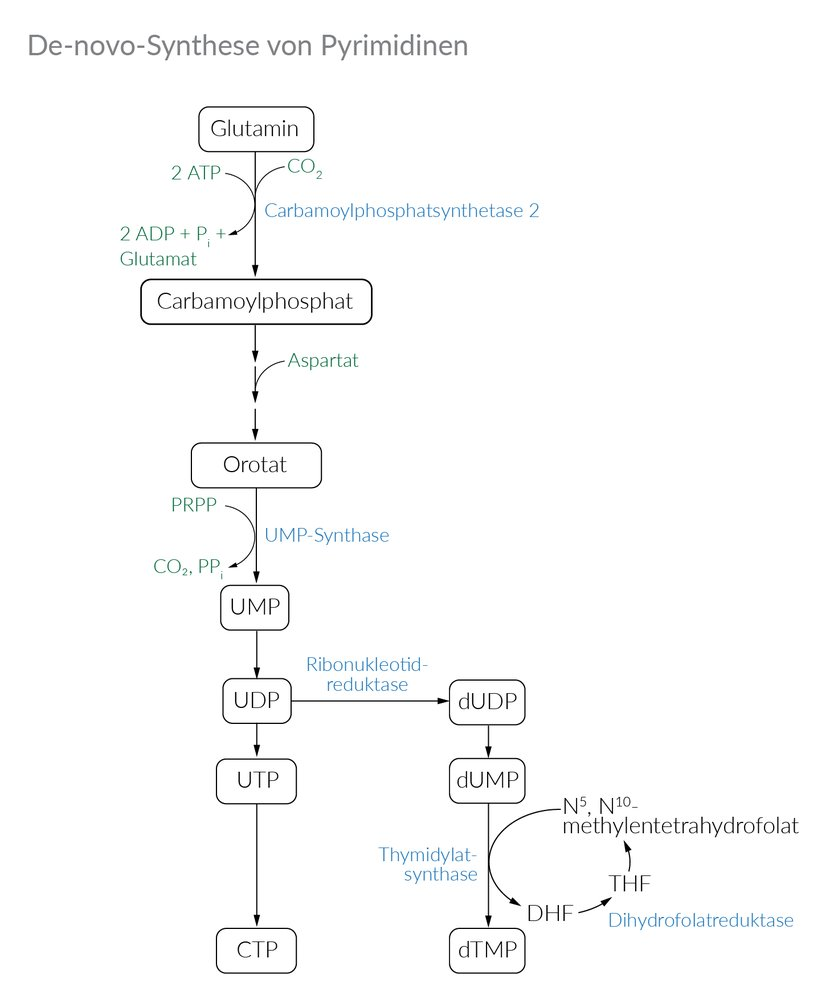
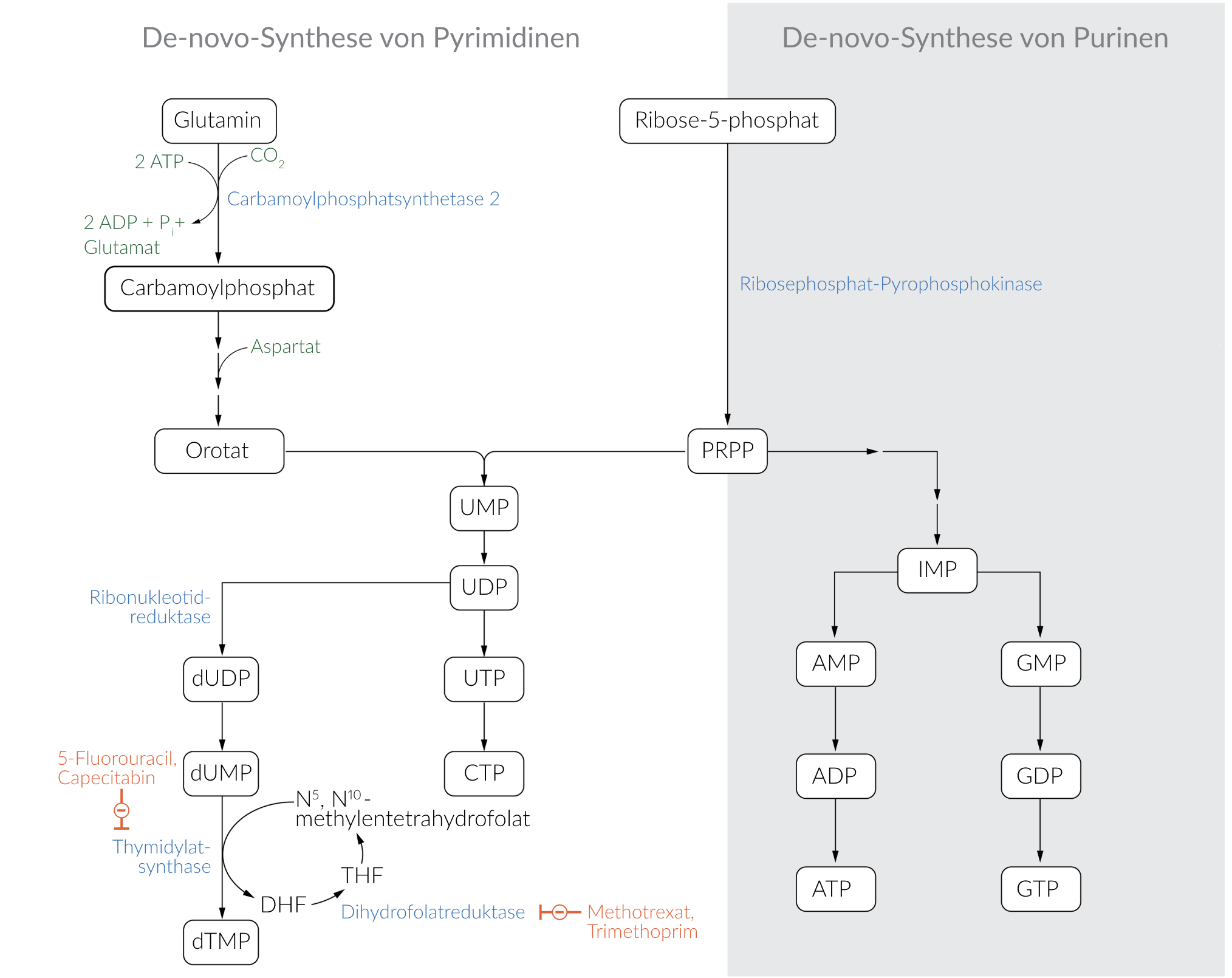
31. A 55-year-old patient is undergoing chemotherapy for colorectal carcinoma, which includes the drug 5-fluorouracil (5-FU). In target cells, 5-FU is converted into the active intermediate 5-fluoro-2’-deoxyuridine monophosphate (FdUMP). This compound specifically inhibits the de novo synthesis of nucleotides.
Which of the following enzymes or enzymatic activities is most likely inhibited by FdUMP?
A. Adenosine deaminase
B. Aspartate carbamoyltransferase
C. Dihydrofolate reductase
D. Hypoxanthine-guanine phosphoribosyltransferase
E. Thymidylate synthase
E. Thymidylate synthase
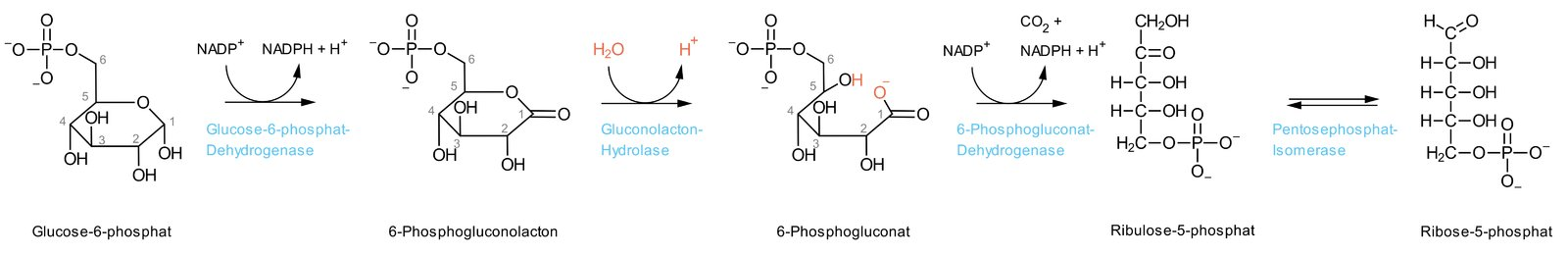
32. In the pentose phosphate pathway, ribulose-5-phosphate is first synthesized; however, ribose-5-phosphate is required for nucleotide synthesis.
How is ribose-5-phosphate produced from ribulose-5-phosphate?
A. Isomerization of a ketose to an aldose
B. Oxidation by an NAD⁺-dependent dehydrogenase
C. Oxidation by an NADP⁺-dependent dehydrogenase
D. Spontaneous decarboxylation
E. Thiamine pyrophosphate–dependent rearrangement
A. Isomerization of a ketose to an aldose
33. In a variety of tumors, the protein “Proliferating Cell Nuclear Antigen” (PCNA, sliding clamp) is overexpressed.
What is the function of PCNA?
A. PCNA activates the origins of replication to open the replication bubble
B. PCNA serves as a primase
C. PCNA enables DNA polymerase δ to perform processive DNA synthesis
D. PCNA holds the sister chromatids together during the G2 phase
E. PCNA separates the two single strands of double-stranded DNA
C. PCNA enables DNA polymerase δ to perform processive DNA synthesis
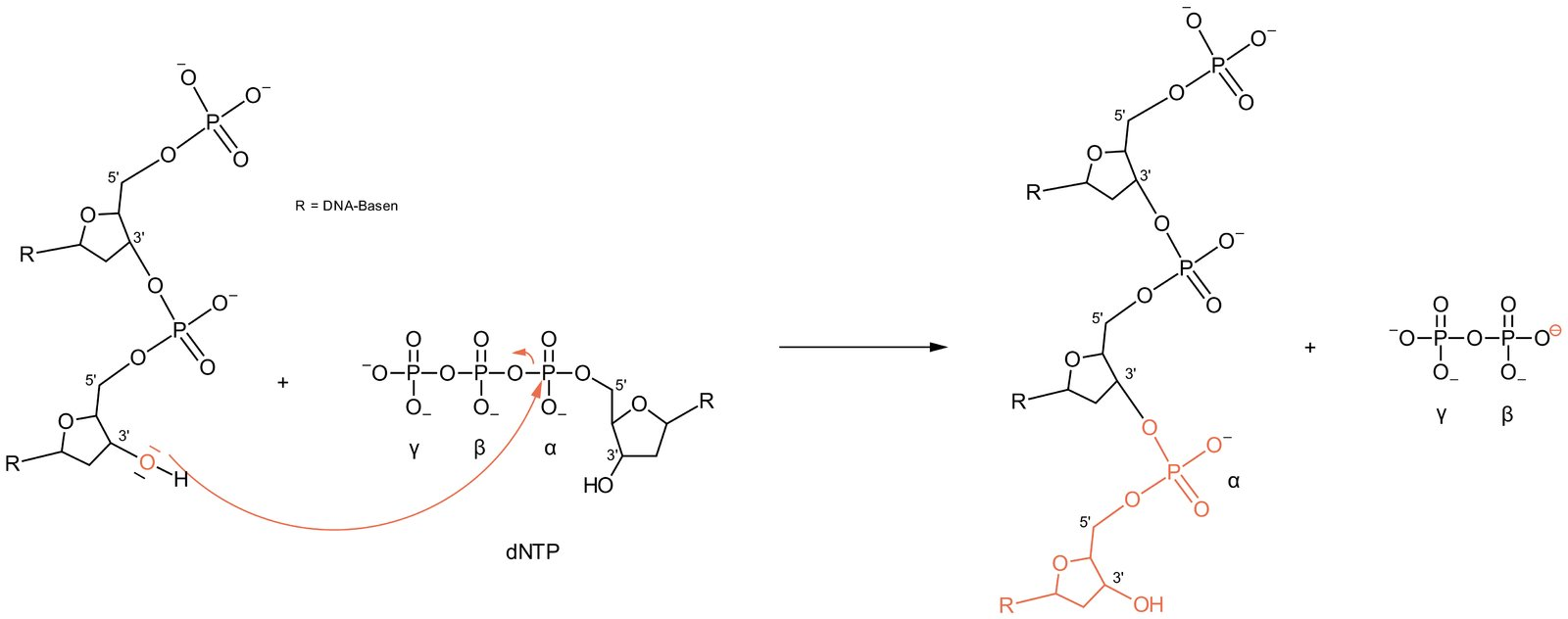
34. During DNA and RNA synthesis, an existing nucleic acid chain is gradually extended by the addition of (deoxy)ribonucleotides.
With which functional group of the existing nucleic acid chain does each subsequent nucleotide react during this elongation process?
A. Amino group
B. Carboxyl group
C. Hydroxyl group
D. Methyl group
E. Phosphate group
C. Hydroxyl group
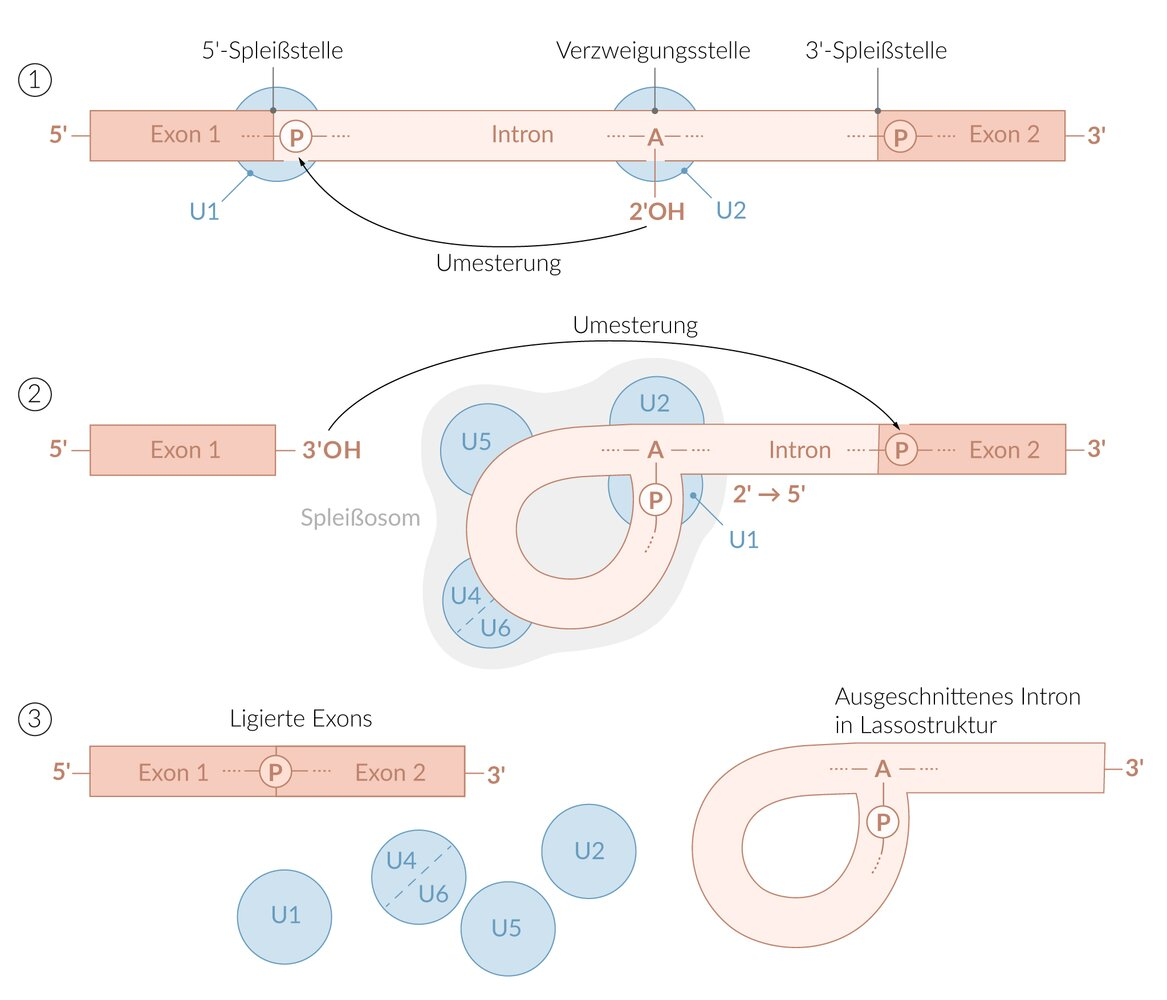
35. The pentose of a nucleotide can carry one or two free OH groups.
In which process is the reaction of a phosphate group with the 2'-OH group of a nucleotide involved?
A. 3'-5' exonuclease activity of DNA polymerases
B. Purine nucleotide synthesis
C. Pyrimidine nucleotide synthesis
D. Splicing of mRNA
E. Synthesis of RNA primers during DNA replication
D. Splicing of mRNA
36. Ribosomes consist of proteins and ribosomal RNA (rRNA).
Which functional structure is formed by the 28S rRNA of the ribosomes?
A. The catalytic center of the peptidyl transferase
B. The binding site for the initiation factor eIF2
C. The decoding site where the anticodon of the tRNA associates with the mRNA
D. The small subunit of the ribosomes
E. A specific binding site for GTP
A. The catalytic center of the peptidyl transferase
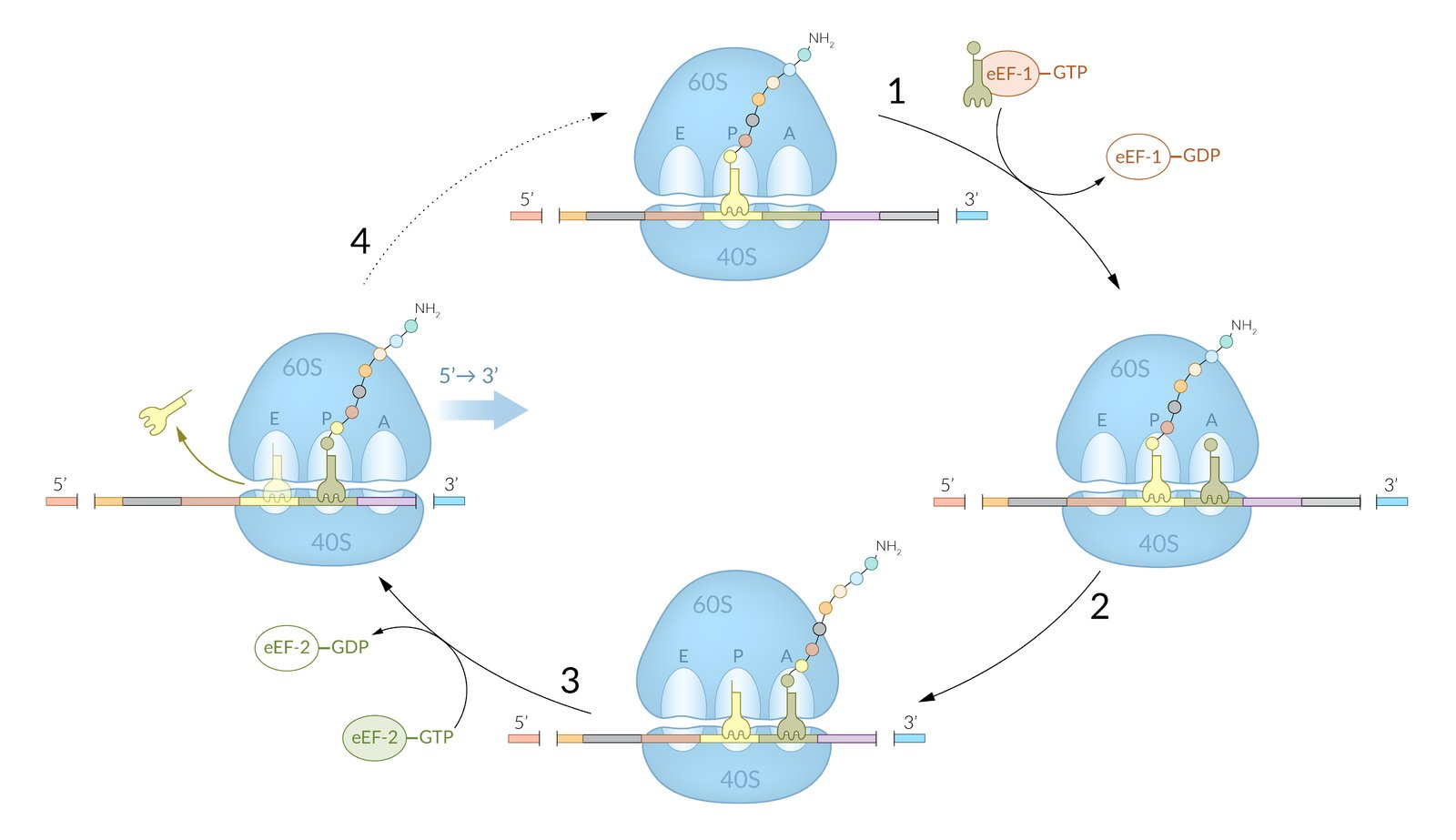
37. Diphtheria toxin ADP-ribosylates the eukaryotic elongation factor 2 (eEF2). This completely inhibits protein biosynthesis. A single toxin molecule is sufficient to kill the affected cell.
Which step of translation is specifically blocked by diphtheria toxin?
A. Insertion of the correct aminoacyl-tRNA
B. Recognition of the stop codons
C. Search for the start codon
D. Movement along the mRNA by 3 nucleotides
E. Joining of the cap and poly-A structures of the mRNA
D. Movement along the mRNA by 3 nucleotides
38. To study protein function, mammalian cells in culture are often transfected with siRNAs (“small interfering RNAs”), which induce a targeted reduction in the amount of a specific protein and the resulting cellular phenotype.
On which biochemical principle is this specific depletion of individual proteins by siRNA primarily based?
A. Direct inactivation by binding to the active site of the protein
B. Inhibition of tRNA binding to ribosomes
C. Methylation of the promoter of the corresponding gene
D. Stimulation of protein degradation
E. Enhanced degradation of the corresponding mRNA
E. Enhanced degradation of the corresponding mRNA
39. Under aerobic conditions, pyruvate from glycolysis is converted to acetyl-CoA by the pyruvate dehydrogenase complex. However, certain cells form lactate from pyruvate under aerobic conditions instead of acetyl-CoA.
Which of the following cells exhibit this behavior?
A. Erythrocytes
B. Granulocytes
C. Lymphocytes
D. Monocytes
E. Thrombocytes
A. Erythrocytes
40. Gel filtration is a method used to determine the size of a native protein. Using this method, the molecular weight of an antibody is found to be approximately 1,000 kDa. Subsequent gel electrophoresis with a reducing SDS–polyacrylamide gel and staining with Coomassie Brilliant Blue detects two bands with molecular weights of about 25 kDa and 75 kDa, corresponding to the light and heavy chains of the antibody, respectively.
Which antibody class most likely corresponds to this antibody?
A. IgA
B. IgD
C. IgE
D. IgG
E. IgM
E. IgM
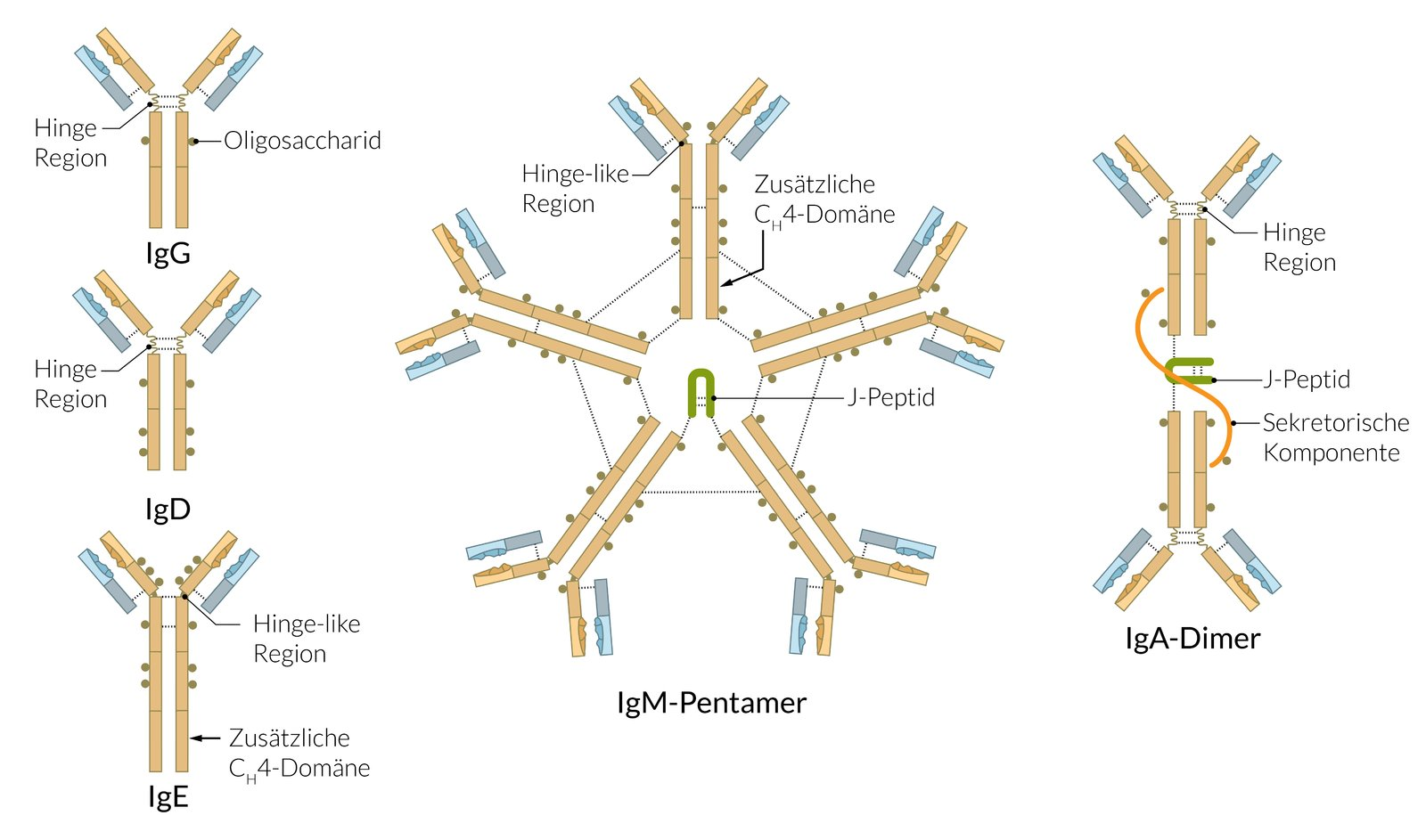
41. Infection with the coronavirus SARS-CoV-2 induces a humoral immune response, resulting in the presence of SARS-CoV-2–reactive antibodies in the serum.
Which two immunoglobulin classes make up the majority of SARS-CoV-2–reactive antibodies that can be detected in the serum about 10–14 days after initial exposure?
A. IgA and IgD
B. IgA and IgE
C. IgD and IgE
D. IgE and IgG
E. IgG and IgM
E. IgG and IgM
42. In the detection of SARS-CoV-2 antigens using rapid antigen tests, monoclonal antibodies are employed.
Which property of monoclonal antibodies is most likely utilized by these tests?
A. A monoclonal antibody forms precipitates in aqueous environments due to its very low solubility
B. A monoclonal antibody binds an antigen covalently
C. A monoclonal antibody binds identical antigens in the same environment with the same affinity
D. A monoclonal antibody binds different epitopes of an antigen
E. A monoclonal antibody has only one specific antigen-binding site (monovalent)
C. A monoclonal antibody binds identical antigens in the same environment with the same affinity
43. The four polypeptides of an immunoglobulin of class G are held together by intermolecular covalent bonds between the side chains of identical amino acids.
Which amino acid is involved in these bonds?
A. Arginine
B. Asparagine
C. Cysteine
D. Methionine
E. Serine
C. Cysteine
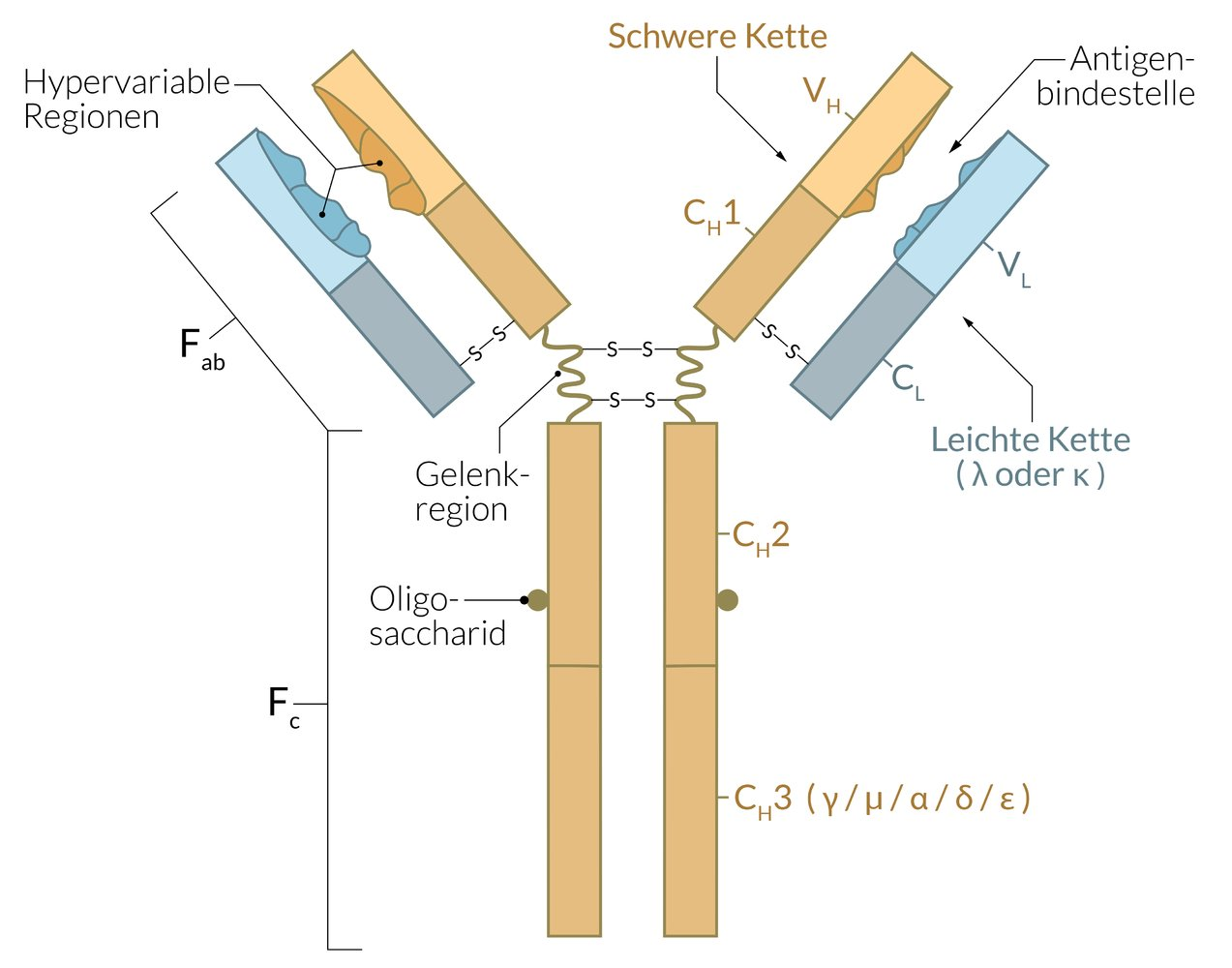
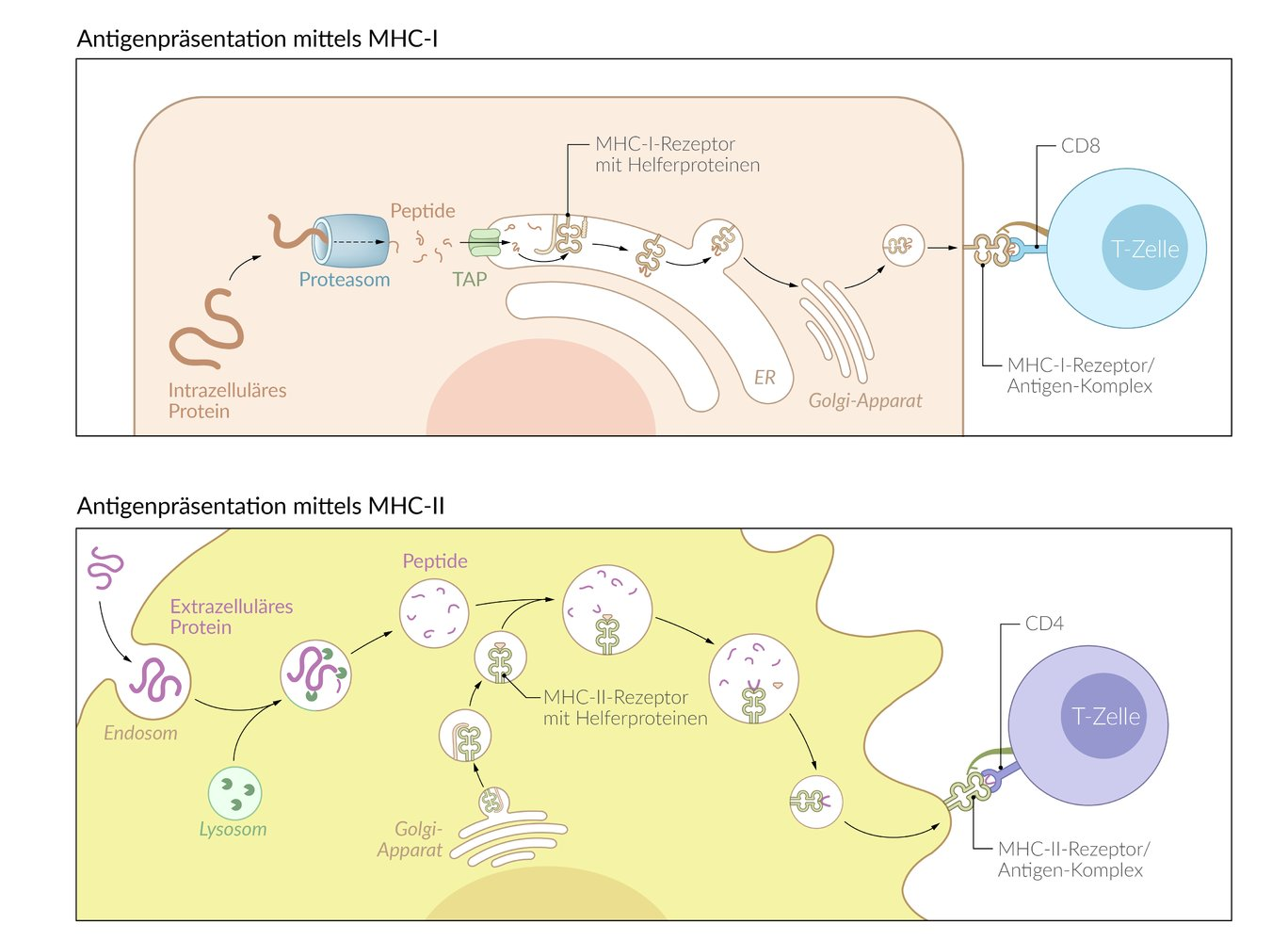
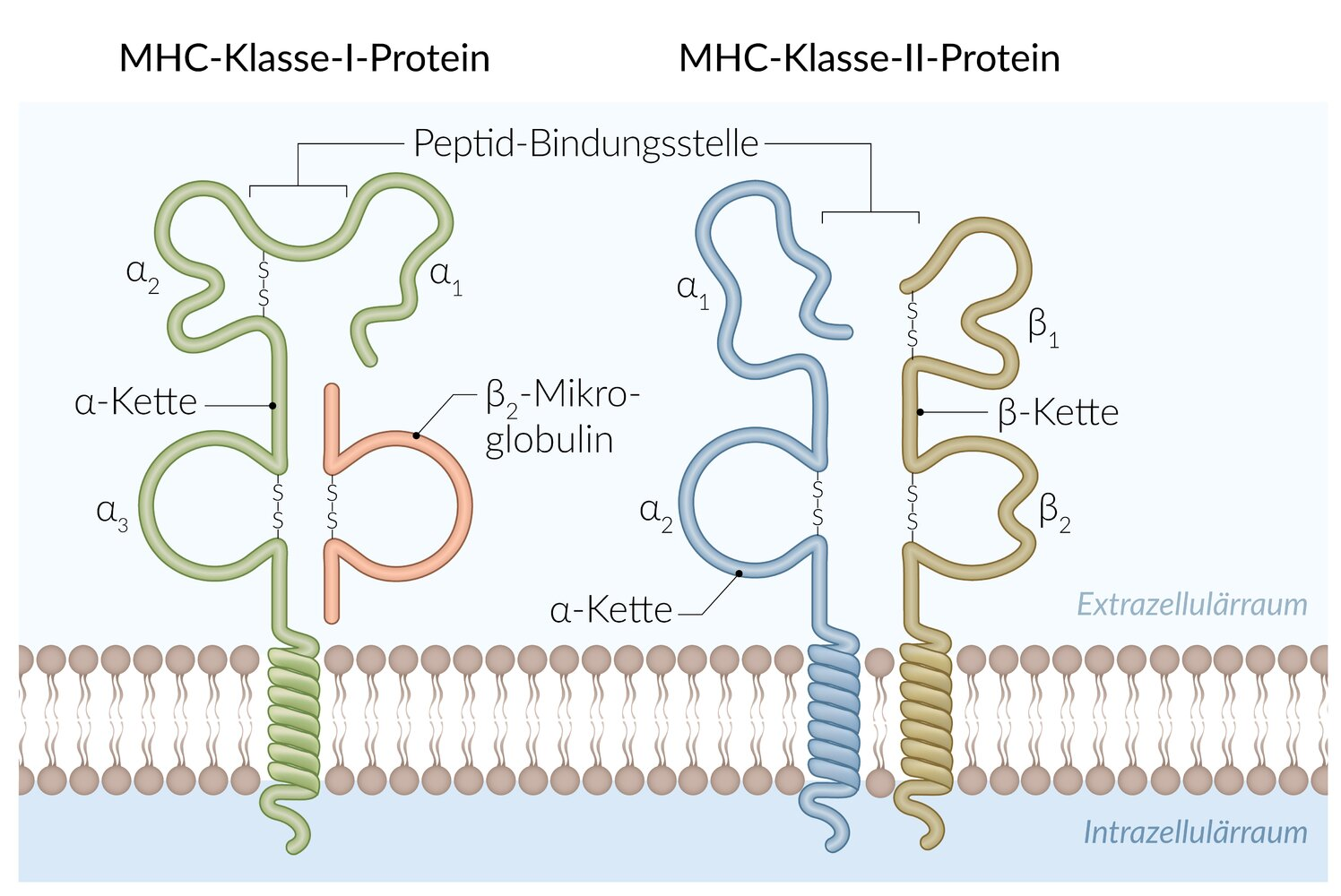
44. Peptides bound to MHC class II proteins on the surface of antigen-presenting cells serve as an important signal for immune system activation.
By which biochemical process are such peptides generated for presentation?
A. Premature termination of translation of cellular mRNA
B. Expression of specific nuclear-encoded antigens
C. Proteolytic cleavage of external antigenic proteins after endocytosis
D. Cleavage of intracellularly synthesized antigenic proteins by the proteasome
E. Trypsin-like cleavage of viral proteins
C. Proteolytic cleavage of external antigenic proteins after endocytosis
45. The majority of plasma proteins are synthesized primarily by the liver. However, there are exceptions.
Which of the following plasma proteins is synthesized predominantly by extrahepatic tissues?
A. α1-Antichymotrypsin
B. Ceruloplasmin
C. Haptoglobin
D. α2-Macroglobulin
E. β2-Microglobulin
E. β2-Microglobulin
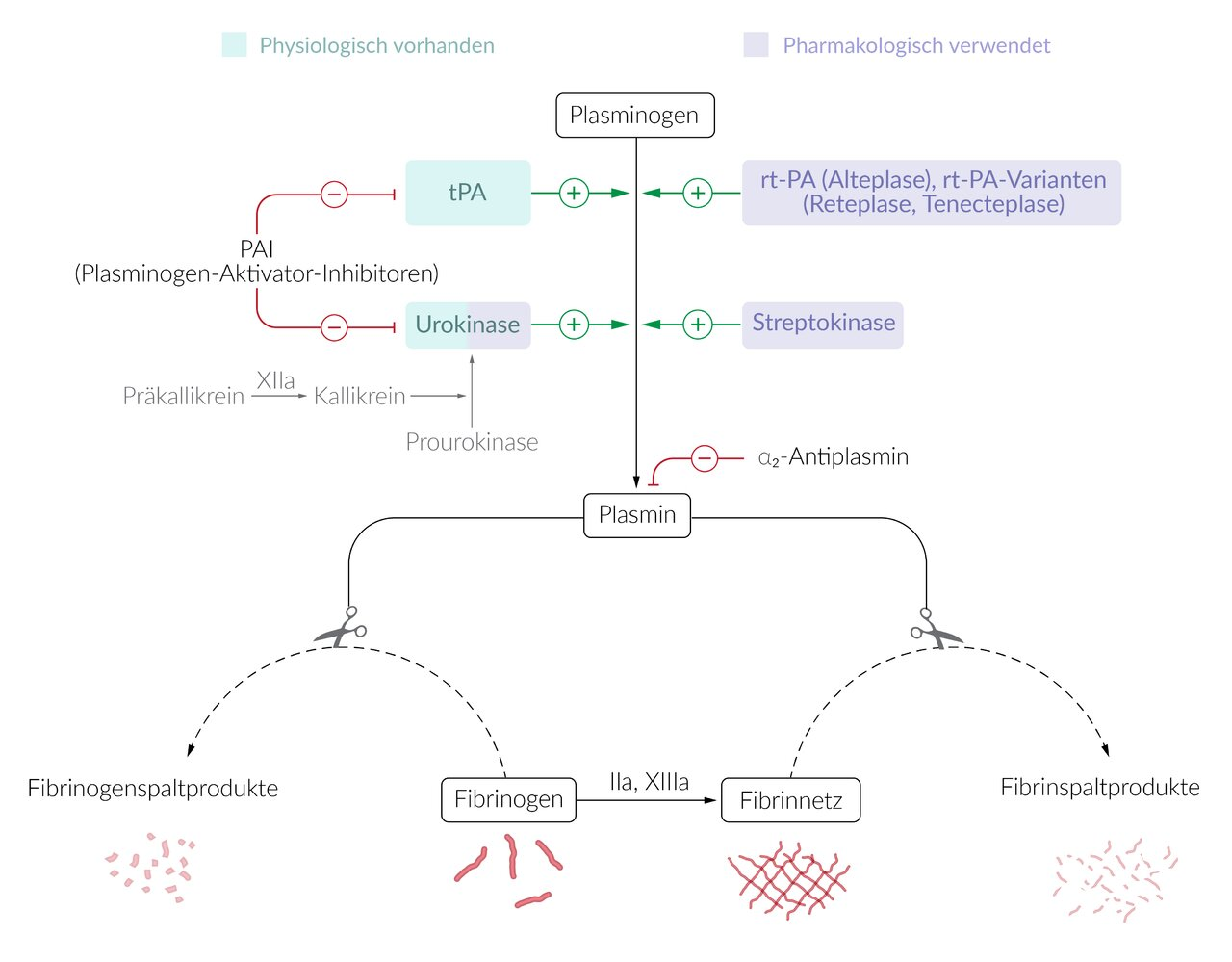
46. After blood coagulation has occurred, clot formation must be limited and intravascular blood flow restored.
Which of the following enzymes directly dissolves fibrin clots in the body?
A. Antihemophilic factor A
B. Plasmin
C. Prothrombin
D. Tenase complex
E. Thrombin
B. Plasmin
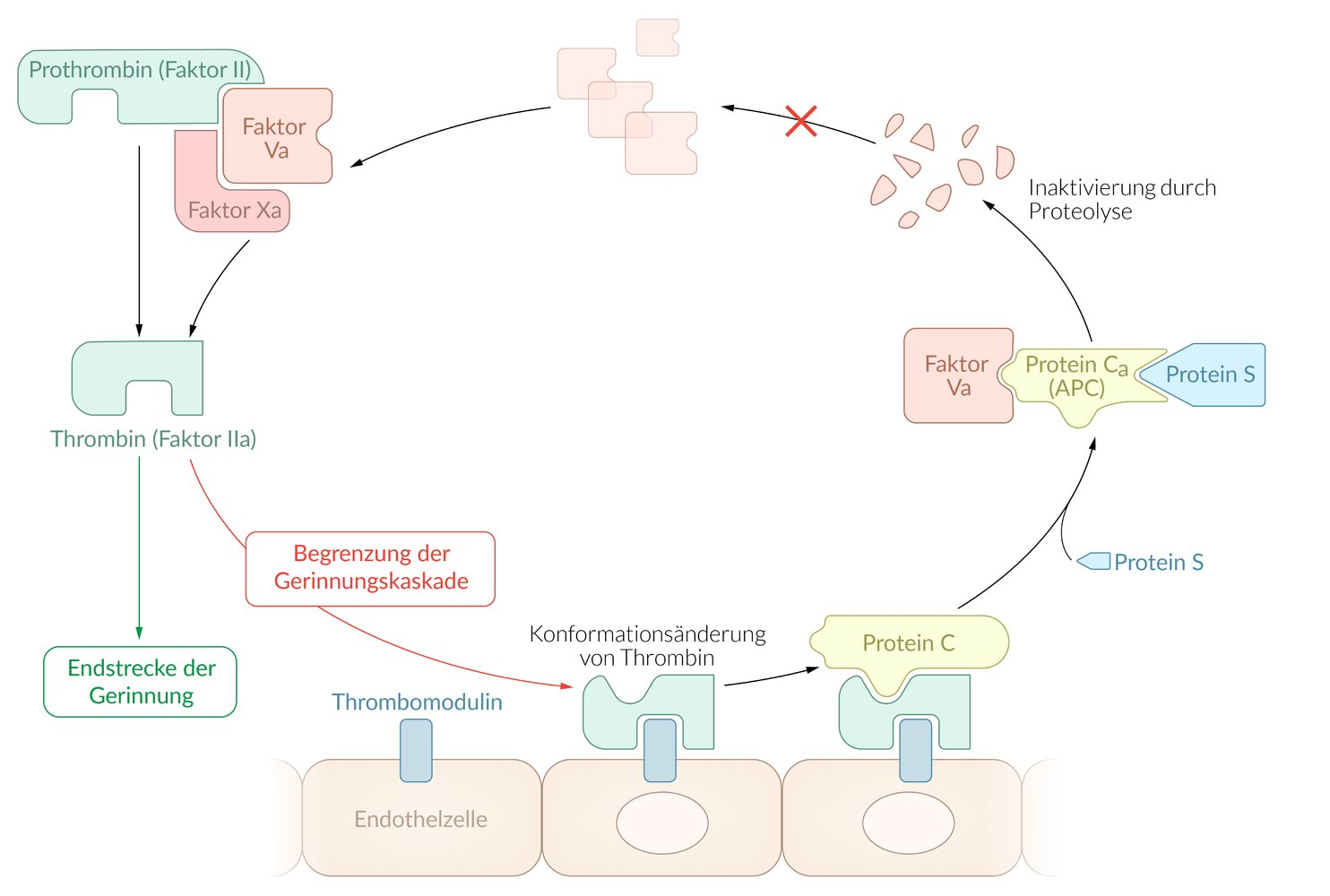
47. A tendency toward thrombosis (thrombophilia) is, in many cases, genetically determined. Missense mutations in the gene for factor V can cause familial thrombophilia. Due to such mutations, activated factor V (Va) is inactivated less efficiently by a specific protein and is therefore particularly stable.
Which of the following proteins is most likely responsible for this effect?
A. Activated vitamin K–dependent γ-glutamyl carboxylase
B. Activated factor X
C. Activated protein C
D. Activated protein S
E. Activated thrombomodulin
C. Activated protein C
48. Hypoxia-inducible factors (HIF) are transcription factors that play a key role in regulating the expression of genes involved in erythropoiesis and aerobic metabolism.
Which oxygen-dependent posttranslational modification of HIF ultimately leads to their inactivation through proteasomal degradation of the modified subunit?
A. Formation of an N-terminal cyclic glutamate
B. Dephosphorylation of tyrosine side chains
C. Hydroxylation of proline side chains
D. Phosphorylation of serine and/or threonine side chains
E. Proteolytic cleavage of an N-terminal peptide
C. Hydroxylation of proline side chains
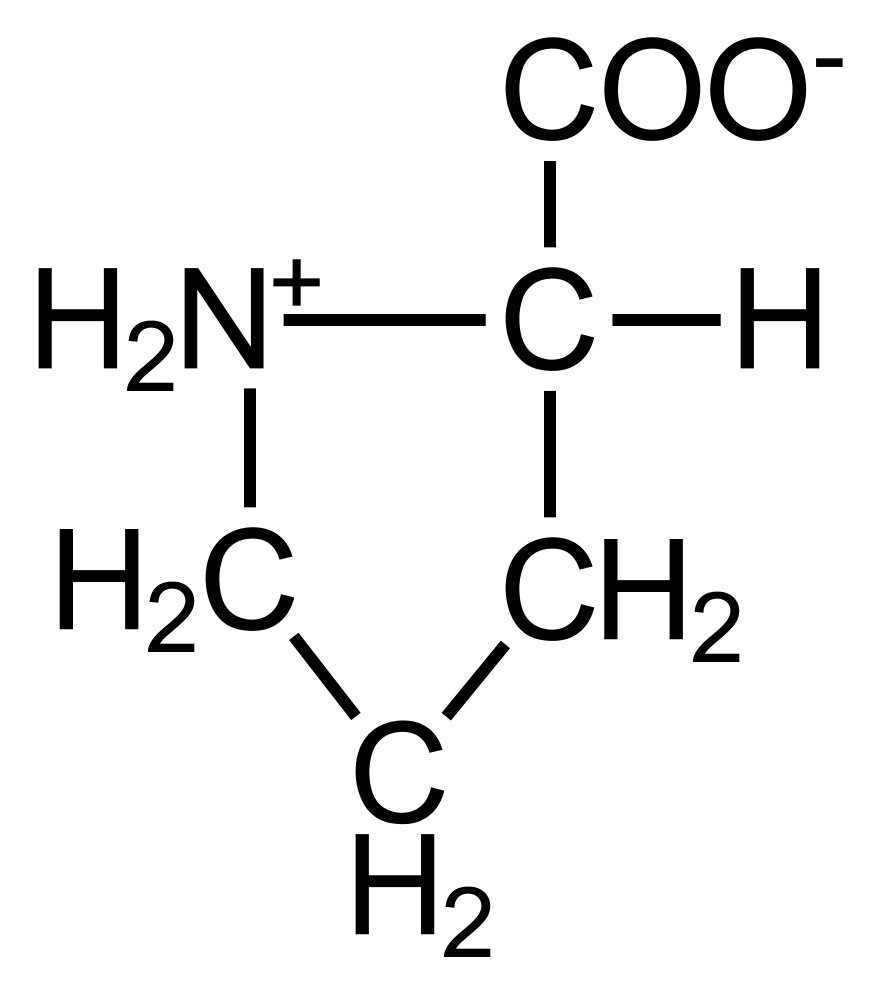
49. In sickle cell anemia, the β-globin gene is mutated so that, in the β-globin protein, another amino acid — amino acid X — is incorporated at position 6 instead of glutamate. The binding (hydrophobic interaction) of amino acid X of one β-globin protein in a hydrophobic pocket of another β-globin protein causes the fibril formation of deoxygenated sickle cell hemoglobin (HbS).
Which of the following amino acids is most likely X?
A. Aspartate
B. Histidine
C. Lysine
D. Serine
E. Valine
E. Valine
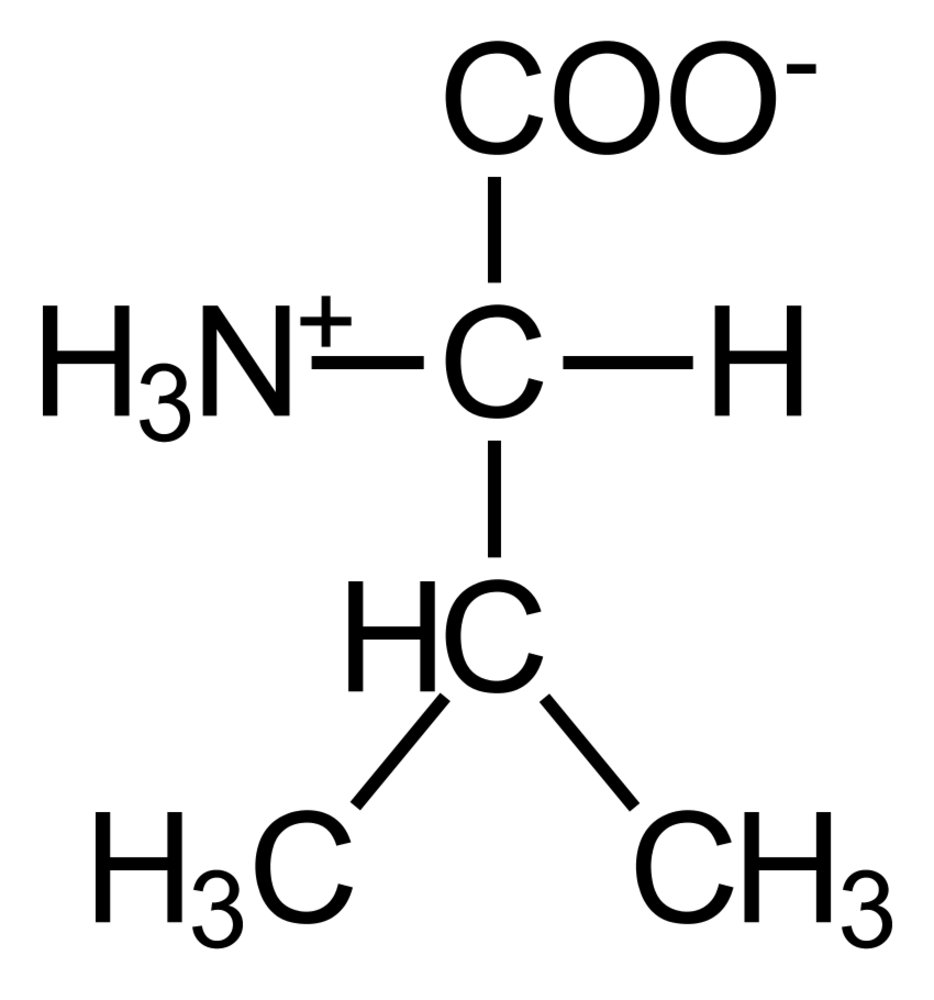
50. Glycogen serves as an important energy reserve in muscle cells, which can be mobilized by hormonal and neuronal stimuli.
Which of the following events most likely leads to enhanced breakdown of muscle glycogen?
A. Activation of phosphorylase kinase by Ca²⁺ ions
B. Dephosphorylation of glycogen phosphorylase
C. Increase in the intracellular ATP/AMP ratio
D. Decrease in cAMP levels by phosphodiesterases
E. Inhibition of protein kinase A by phosphorylation
A. Activation of phosphorylase kinase by Ca²⁺ ions
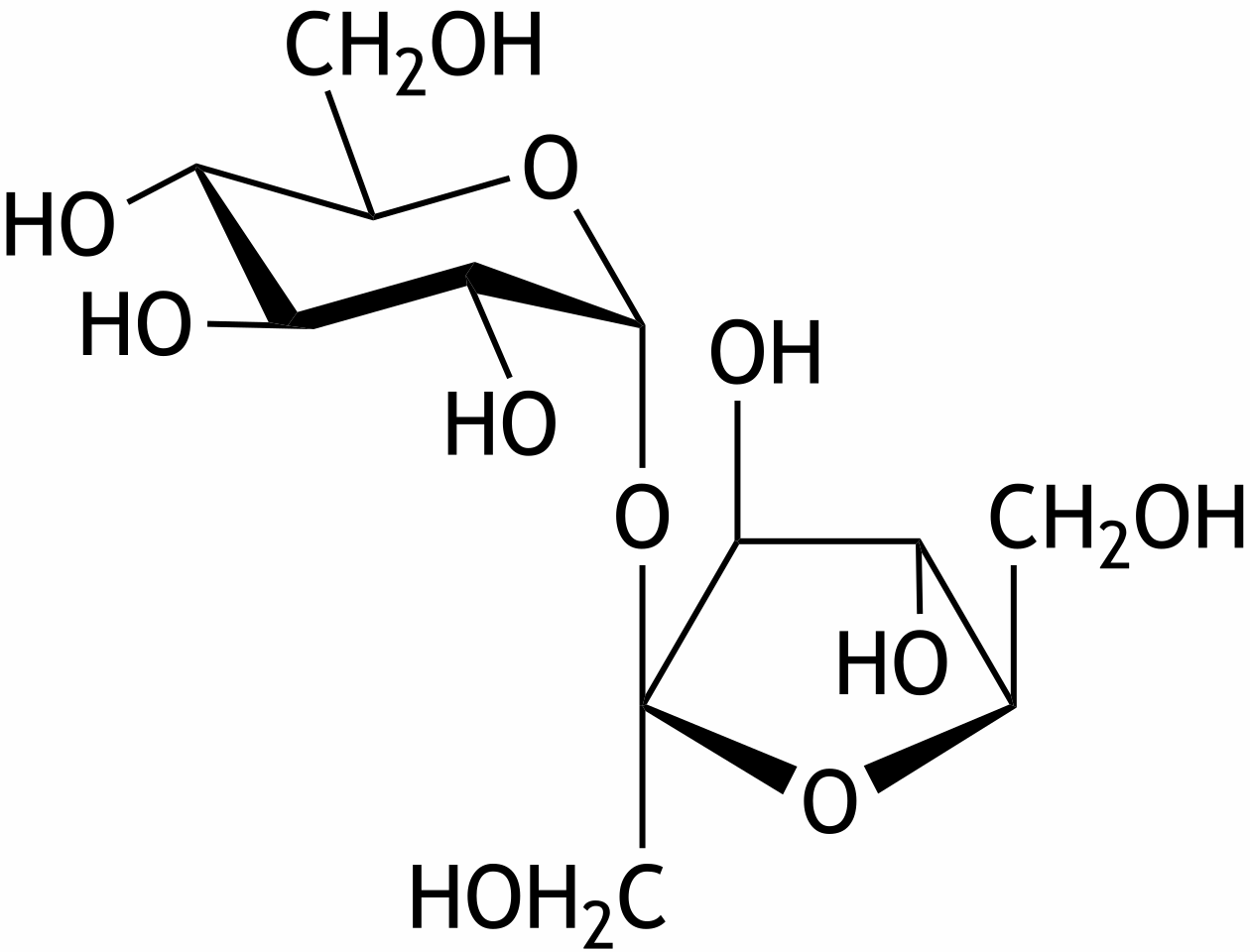
51. Which disaccharide is shown here?
A. Cellobiose
B. Isomaltose
C. Lactose
D. Maltose
E. Sucrose
E. Sucrose
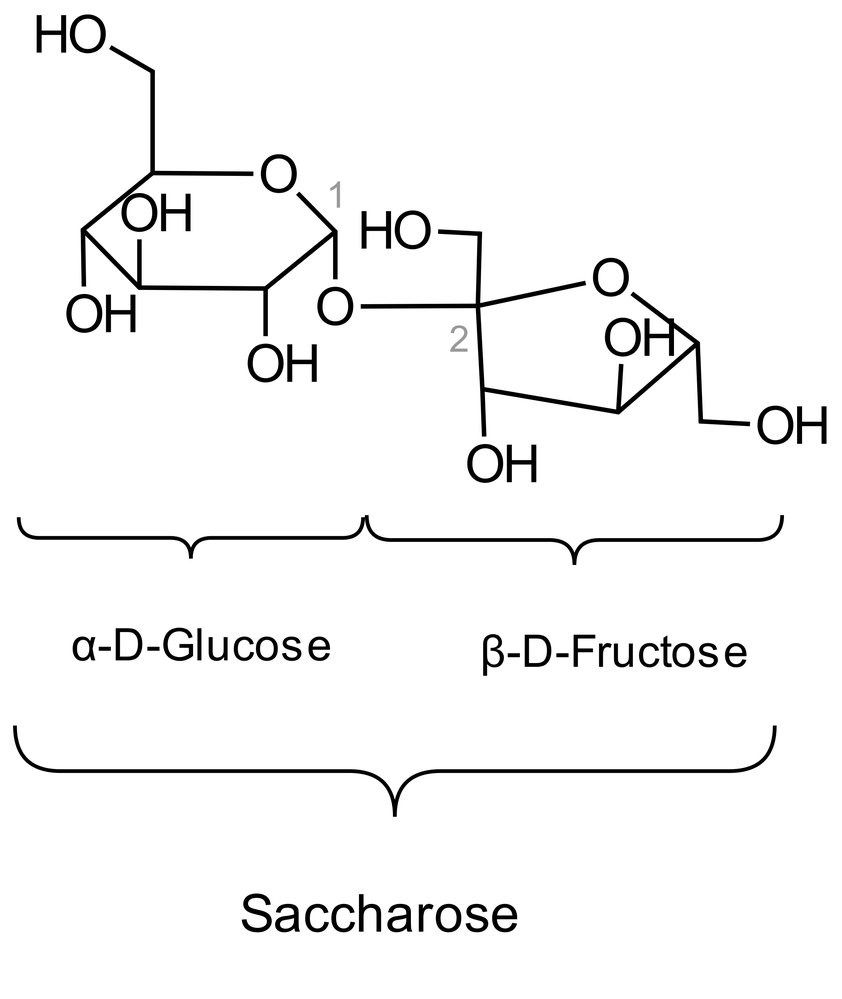
52. Adult hypolactasia, a form of lactose intolerance, is caused by reduced expression of the lactase gene. Lactase cleaves lactose into two monosaccharides.
Into which of the following monosaccharide combinations is lactose split by lactase?
A. Fructose and galactose
B. Fructose and glucose
C. Fructose and mannose
D. Galactose and glucose
E. Galactose and mannose
D. Galactose and glucose
53. Hereditary fructose intolerance is a rare inherited disorder caused by an enzyme defect in fructose metabolism. Affected individuals, typically very young children such as infants after the introduction of fructose-containing foods, suffer from hypoglycemia and acidosis. In this disease, an intermediate of fructose metabolism cannot be degraded.
Which of the following metabolites is most likely involved?
A. Dihydroxyacetone phosphate
B. Fructose-1-phosphate
C. Fructose-6-phosphate
D. Fructose-1,6-bisphosphate
E. Glyceraldehyde-1-phosphate
B. Fructose-1-phosphate
54. As part of newborn screening, a breastfed infant is diagnosed with galactosemia.
Which of the following metabolites is most likely elevated in the erythrocytes and other body cells of this infant?
A. Fructose-1,6-bisphosphate
B. Fructose-2,6-bisphosphate
C. Galactose-1-phosphate
D. Glucose-1-phosphate
E. Glucose-6-phosphate
C. Galactose-1-phosphate
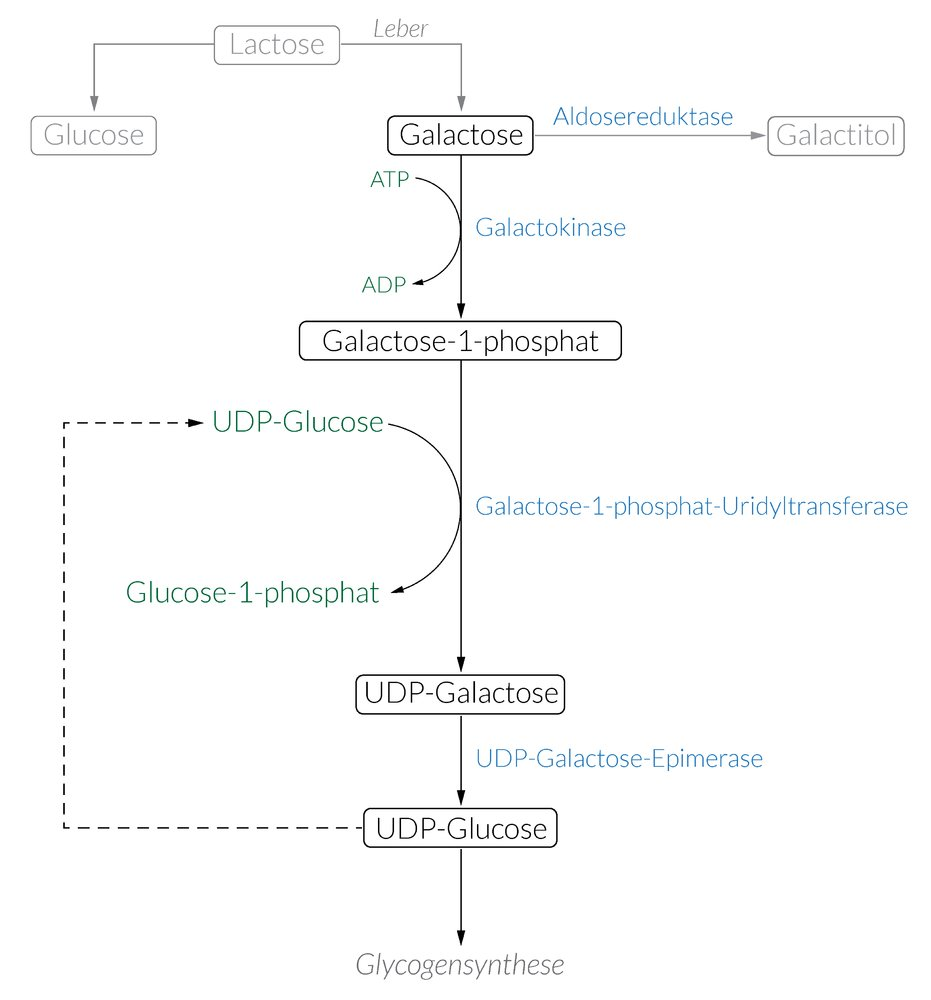
55. HMG-CoA lyase catalyzes the cleavage of β-hydroxy-β-methylglutaryl-CoA (HMG-CoA) with the release of acetyl-CoA.
Which is the second cleavage product formed in this reaction?
A. Acetoacetate
B. β-Hydroxybutyrate
C. Carbon dioxide
D. Mevalonate
E. Succinate
A. Acetoacetate
56. Transaminases (aminotransferases) act as key enzymes in hepatic amino acid metabolism by catalyzing the exchange of functional groups.
Which of the following amino acids can be formed directly from the α-keto acid pyruvate by transamination?
A. Alanine
B. Aspartate
C. Glutamate
D. Serine
E. Valine
A. Alanine
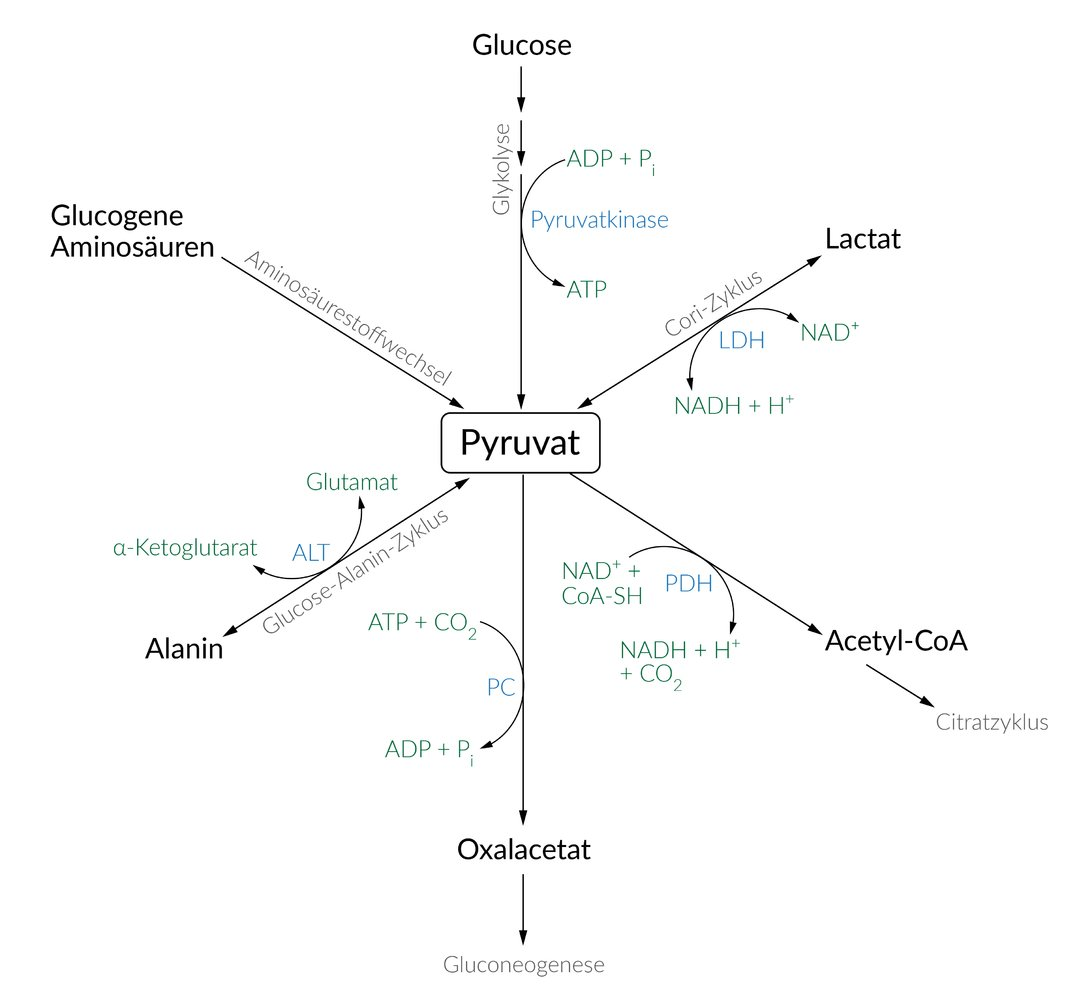
57. Hepatic encephalopathy is a functional disorder of the central nervous system caused by advanced liver dysfunction (e.g., in the context of liver cirrhosis). A typical feature is hyperammonemia (a pathological increase in blood ammonia concentration).
Impairment of which of the following hepatic enzymatic activities is the direct cause of hyperammonemia?
A. Carbamoyl phosphate synthetase I
B. Cytochrome P450–dependent monooxygenase
C. β-Hydroxy-β-methylglutaryl-CoA lyase
D. Phosphoenolpyruvate carboxykinase
E. UDP-glucuronyltransferase
A. Carbamoyl phosphate synthetase I
58. Night blindness can be caused by a specific hypovitaminosis.
A deficiency of which of the following vitamins is most likely indicated by night blindness?
A. Vitamin A
B. Vitamin B12
C. Vitamin D
D. Vitamin E
E. Vitamin K
A. Vitamin A
59. Vitamins perform various vital functions in human metabolism.
Which pairing of vitamin and function is correct?
A. Biotin (vitamin B7) — transamination of amino acids
B. Pantothenic acid (vitamin B5) — fatty acid biosynthesis
C. Phylloquinone (vitamin K1) — oxidative decarboxylation of malonyl-CoA
D. Thiamine (vitamin B1) — γ-carboxylation of glutamyl residues
E. α-Tocopherol (vitamin E) — electron transfer in the respiratory chain
B. Pantothenic acid (vitamin B5) — fatty acid biosynthesis
60. To monitor the course of a viral hepatitis treated with an antiviral drug, alanine aminotransferase (ALT) activity is measured. For this purpose, a coupled enzymatic-optical assay is performed, in which the consumption of NADH is monitored at 340 nm. The substrates of ALT as well as NADH are present in excess.
Which of the following enzymes must most likely be added in excess to the assay mixture to enable determination of ALT activity?
A. Alcohol dehydrogenase
B. Aspartate aminotransferase
C. Lactate dehydrogenase
D. Malate dehydrogenase
E. Branched-chain aminotransferase
C. Lactate dehydrogenase
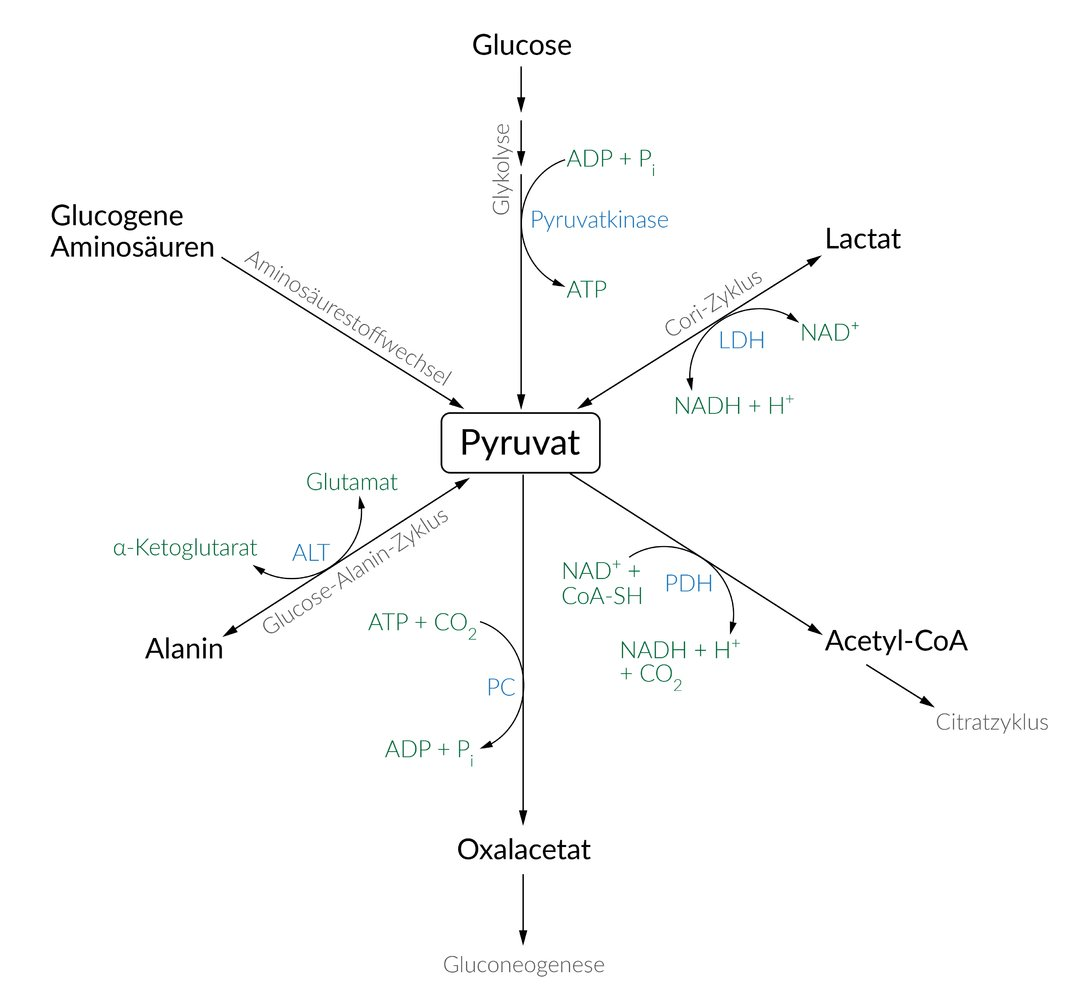
61. G proteins act as molecular switches regulating a variety of cellular processes.
Which of the following statements about GTP is most likely correct in this context?
A. GTP serves as a substrate for ADP-ribosylation
B. GTP stabilizes the active conformation of the G protein
C. GTP transfers a phosphate group to a substrate of the G protein
D. GTP is converted into cyclic GMP
E. GTP is covalently linked to a substrate protein
B. GTP stabilizes the active conformation of the G protein
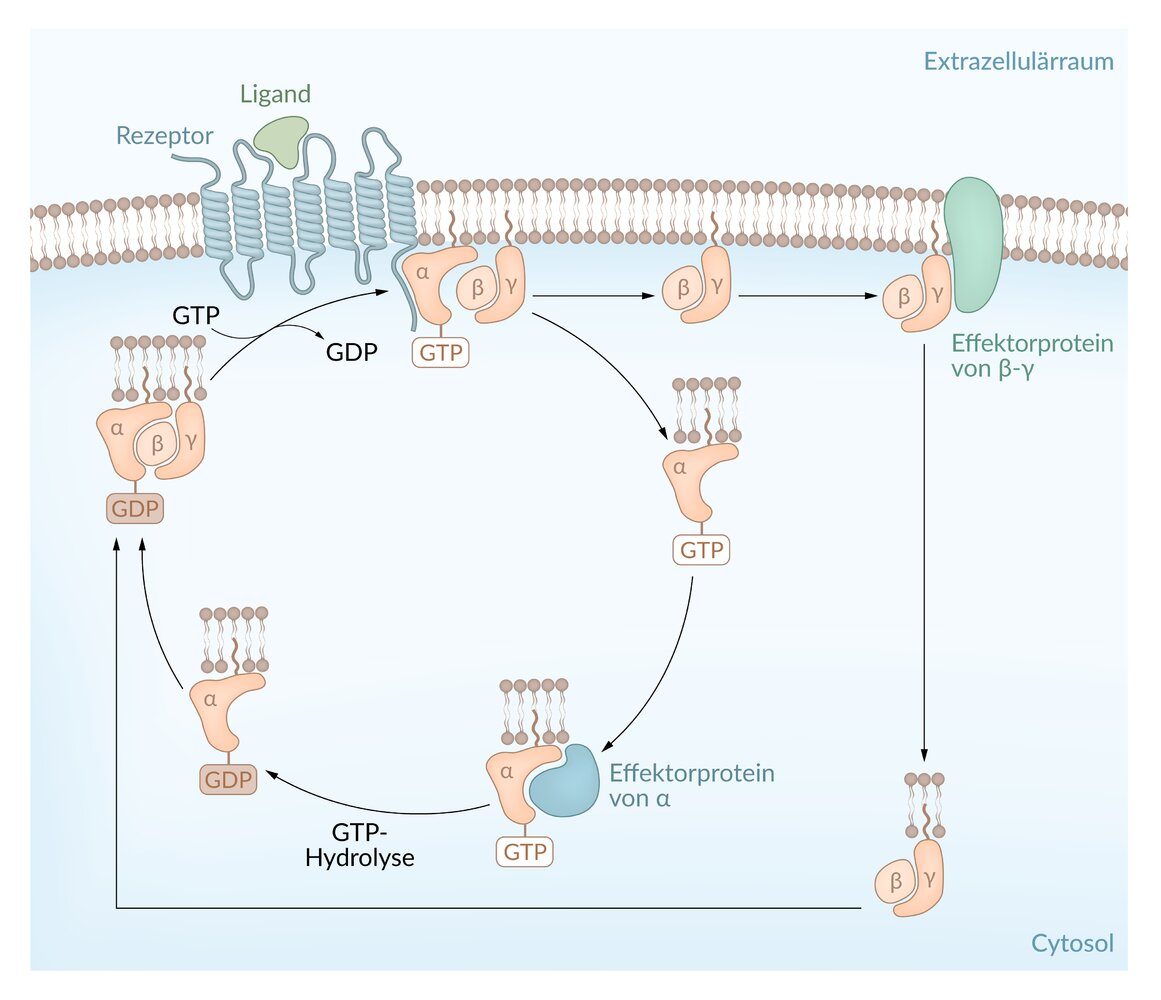
62. Contraction of a vascular smooth muscle cell leads to vasoconstriction. This process is mediated, among other factors, by the binding of norepinephrine to the α₁-adrenoceptor, which thereby becomes activated. This receptor activation subsequently causes an increase in intracellular Ca²⁺ concentration.
Which of the following enzymes is critically involved in this increase in intracellular Ca²⁺ concentration?
A. Adenylate cyclase
B. Phospholipase Cβ
C. PI3-kinase
D. Protein kinase A
E. Protein kinase B
B. Phospholipase Cβ
63. Hormones are either synthesized in advance and stored in intracellular vesicles until secretion or synthesized by the secreting cells immediately before release in response to a stimulus.
Which of the following hormones is typically synthesized only immediately before secretion?
A. Adiuretin (ADH, vasopressin)
B. Adrenaline
C. Aldosterone
D. Histamine
E. Growth hormone (GH)
C. Aldosterone
64. Serotonin is a tissue hormone and neurotransmitter that influences, among other things, the function of the central nervous system, the cardiovascular system, and the gastrointestinal tract.
Which of the following chemical names is one of the formally correct designations for serotonin?
A. γ-Aminobutyric acid
B. 3,4-Dihydroxyphenylalanine
C. β-Hydroxy-β-methylglutaric acid
D. 5-Hydroxytryptamine
E. α-Ketoadipic acid
D. 5-Hydroxytryptamine
65. A woman injects herself intravenously with a large amount of insulin in a suicide attempt and is later found unconscious. Despite being rescued, she suffers permanent damage to the central nervous system (CNS).
Which mechanism most likely explains the CNS damage caused by the insulin overdose?
A. Insulin accelerates gluconeogenesis in the liver, and the resulting glucose release causes fluid shifts (osmotic shock) out of CNS neurons.
B. Insulin blocks the glucose transporters that supply the CNS.
C. Insulin inhibits the transport of ketone bodies into the CNS.
D. Insulin stimulates the insertion of glucose transporters into muscle cell membranes, thereby lowering the glucose concentration in the blood and consequently in the CNS.
E. Insulin reduces glucose uptake in muscle cells, weakening the muscle pump of the leg veins, venous return, and thus CNS perfusion.
D. Insulin stimulates the insertion of glucose transporters into muscle cell membranes, thereby lowering the glucose concentration in the blood and consequently in the CNS.