BI1014 - acid base reactions
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Bronsted-lowry theory
Acid: Proton donor (H⁺)
Base: Proton acceptor
equilibrium
Governed by:
Rate constants (Kₒᵣ, K₆ₐ꜀ₖ)
Le Chatelier’s Principle: System shifts to oppose change (concentration, pressure, temperature)
Ka
Ka (acid constant):
Large Ka → strong acid (complete dissociation)
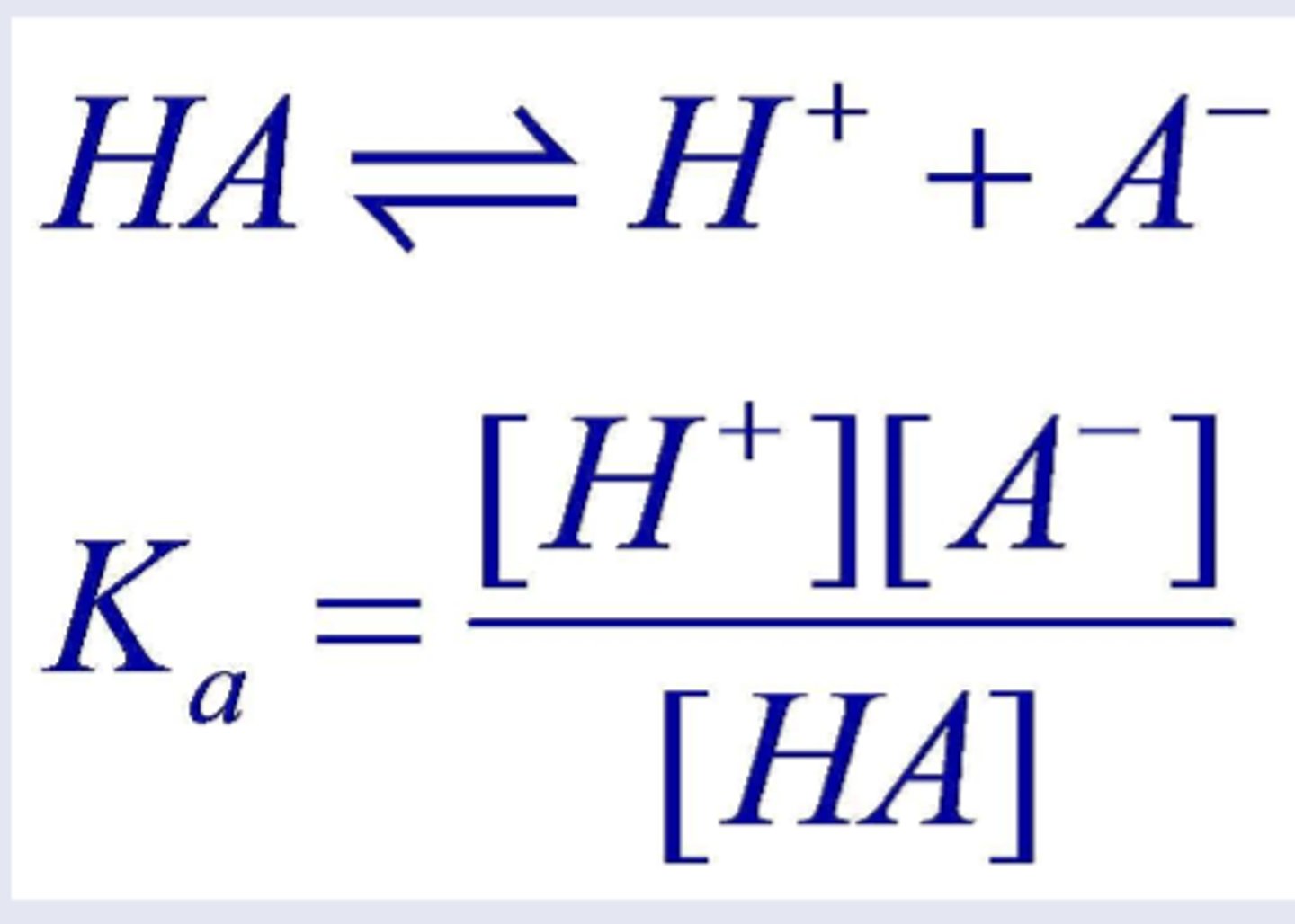
Kb
(base constant)
Large Kb → strong base

Kw
ionic product of water
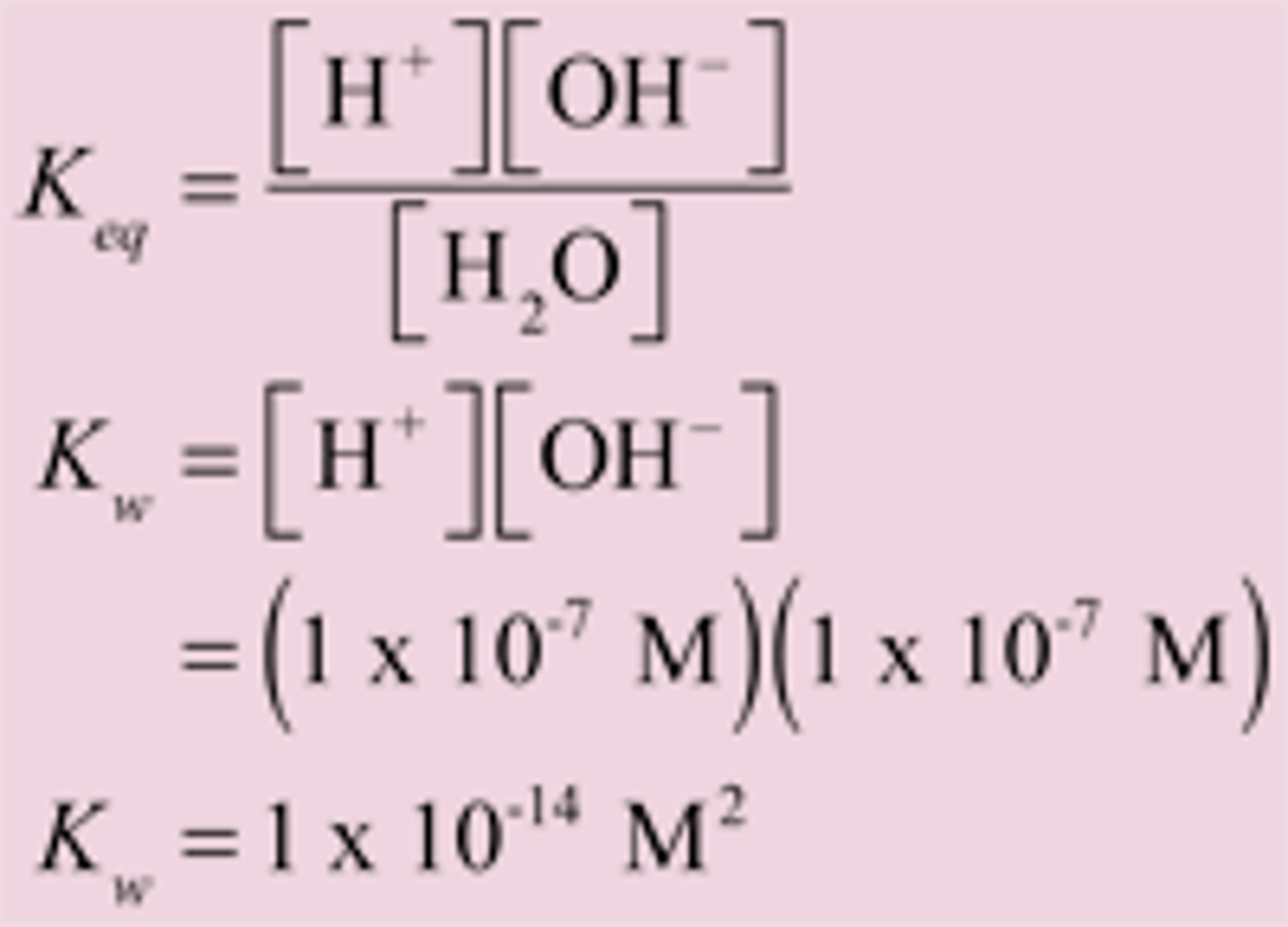
strong acids and bases
Ka or Kb -> >1
Dissociation -> Full
Examples (Acid) HCl, HNO₃, H₂SO₄
Examples (Base) NaOH, KOH
weak acids and bases
Ka or Kb -> <1
Dissociation -> Partial
Examples (Acid) CH₃COOH, citric acid
Examples (Base) NH₃, amines
conjugate base pairs
Every acid has a conjugate base
Every base has a conjugate acid
example of a conjugate pair
CH₃COOH ⇌ CH₃COO⁻ + H⁺(Acid ⇌ Conjugate Base + H⁺)
neutral salts
Strong acid + strong base → pH ~7
acidic salts
Strong acid + weak base → pH <7
basic salts
Weak acid + strong base → pH >7
ampholytes
Can act as both acid & base
Examples: Amino acids, hydrogen phosphate
buffers
Solution that resists pH change
components of a buffer
Weak acid + conjugate base (e.g. CH₃COOH + CH₃COONa)
Weak base + conjugate acid (e.g. NH₃ + NH₄Cl)
buffer action
Add acid (H⁺) → base component neutralizes it
Add base (OH⁻) → acid component donates H⁺
Henderson-Hasselbalch equation
Change of 1 pH = 10× concentration ratio shift
Not valid if [HA] or [A⁻] = 0
![<p>Change of 1 pH = 10× concentration ratio shift</p><p>Not valid if [HA] or [A⁻] = 0</p>](https://knowt-user-attachments.s3.amazonaws.com/bc428632-dbf4-4d22-9c7f-4411c8ab3fd9.jpg)
titration curve
four stages
1. Initial pH: pH of analyte before titration begins.
2. Buffer Region: Gradual change in pH as titrant is added (especially with weak acids/bases)
3. Equivalence Point:
4. After Equivalence: Excess titrant determines the pH
equivalence point
Chemically, when moles of acid = moles of base
Acid = Base (stoichiometrically)
All acid/base is neutralised
Sharp pH change
end point
When the indicator changes colour (should be close to equivalence point).
indicators
An indicator is a weak acid or base that changes colour depending on the pH of the solution.
how do indicators work
Indicator exists in two forms: HInd (acid) and Ind⁻ (base)
Colour changes depending on pH and which form dominates
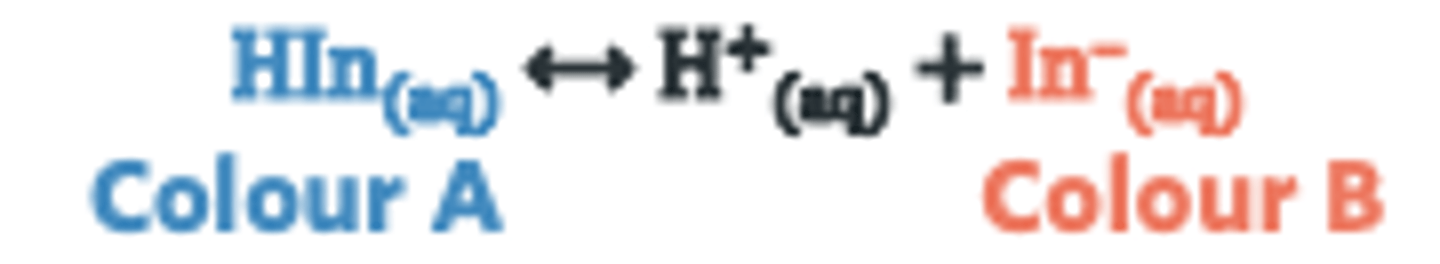
types of indicators
Strong acid + strong base
Phenolphthalein or Methyl orange
Colourless → pink or red → yellow
Weak acid + strong base
Phenolphthalein
Colourless → pink
Strong acid + weak base
Methyl orange
Red → yellow
Weak acid + weak base
No good sharp change; use pH meter
pH equation
pH = -log[H+]
![<p>pH = -log[H+]</p>](https://knowt-user-attachments.s3.amazonaws.com/2d4419ab-98e3-49d4-bfaa-6004592c1e39.jpg)