Lecture 2: Non-covalent interactions; acids, bases , and pH
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
describe water as a solvent
polarity of H2O breaks electrostatic interactions (increasing entropy → randomness)
allows high concentration of solutes
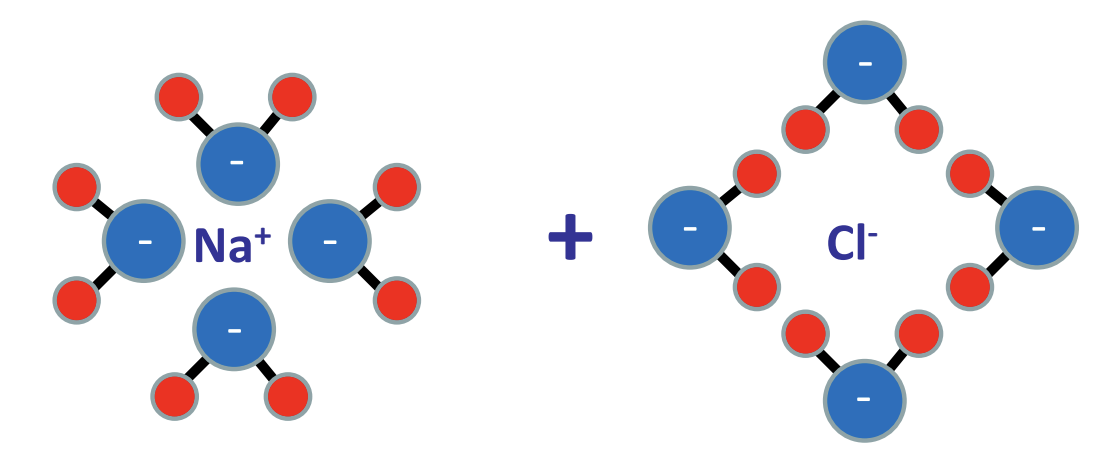
describe the hydrophobic effect
Fatty acids have polar heads and nonpolar tails (amphipathic)
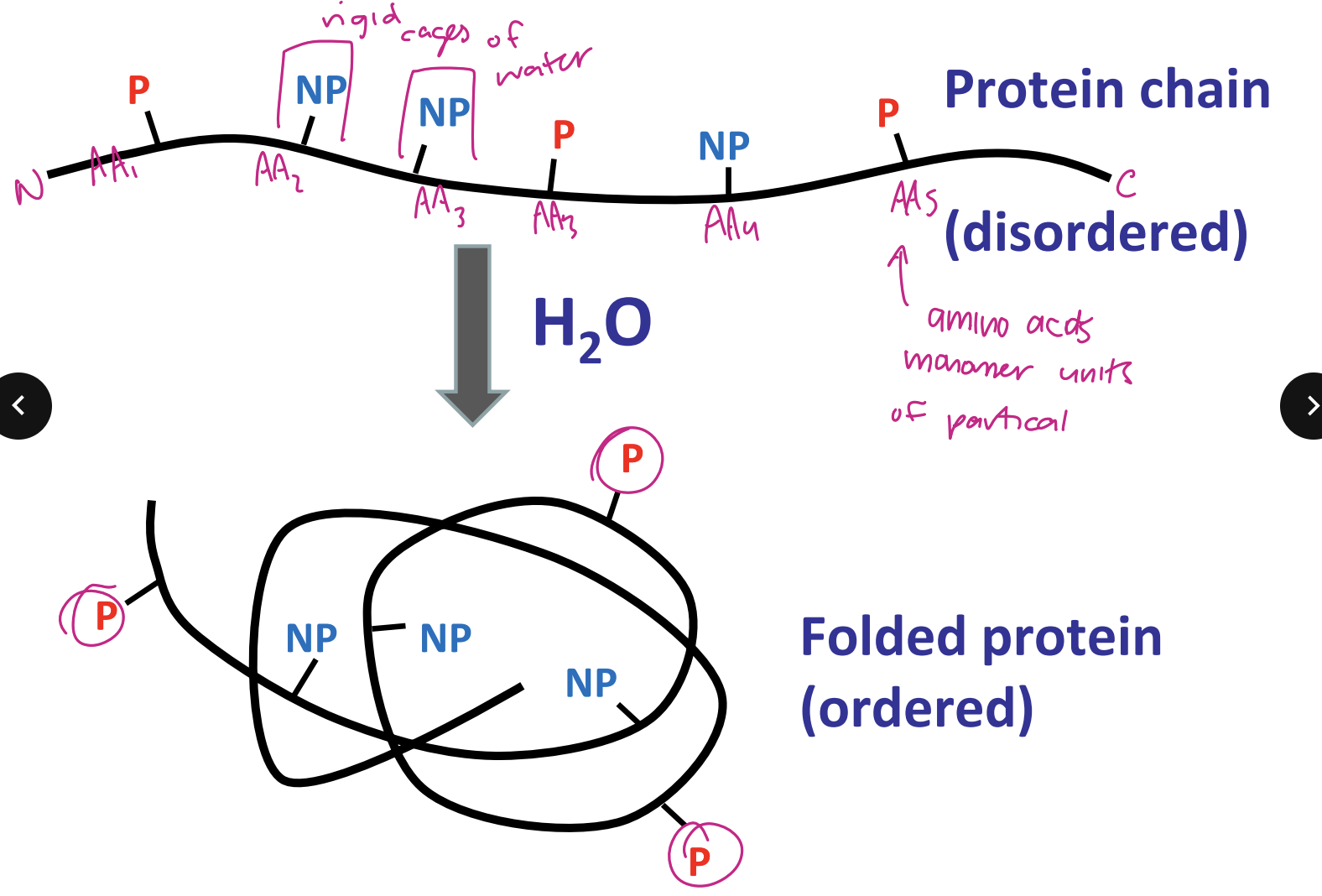
entropy of H2O cages drive. interactions
→ fewer H2O’s in cage
highly structured and ordered
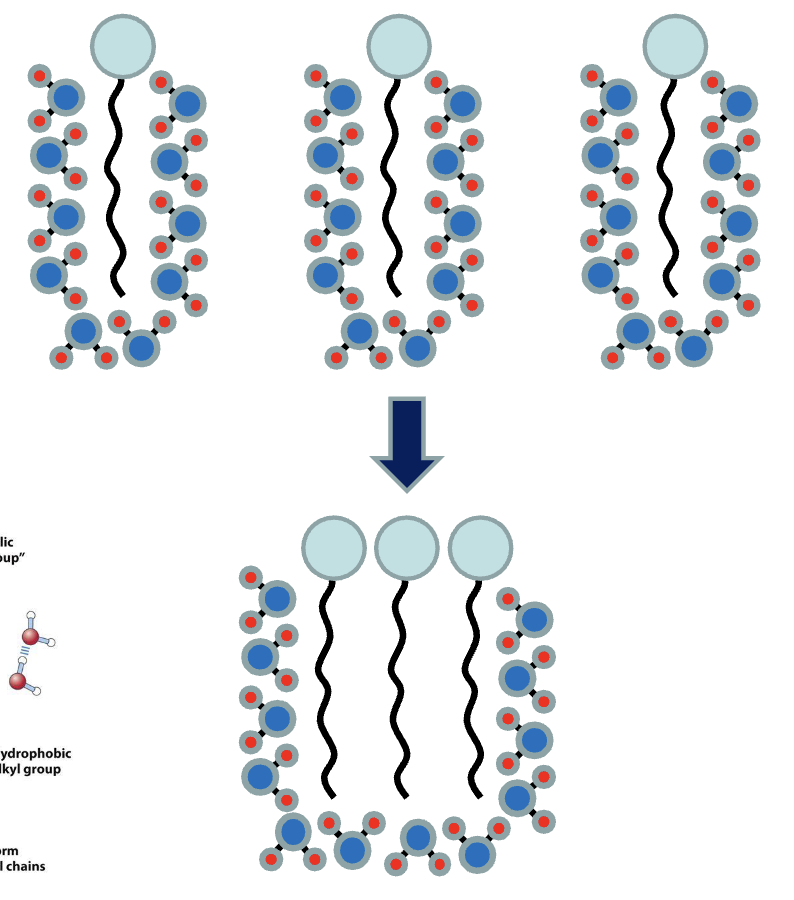
hydrophobic effect drives formation of structures ex. lipid bilayer
hydrophobic effect drives protein folding ex. protein chain (disordered) → folded protein (ordered)
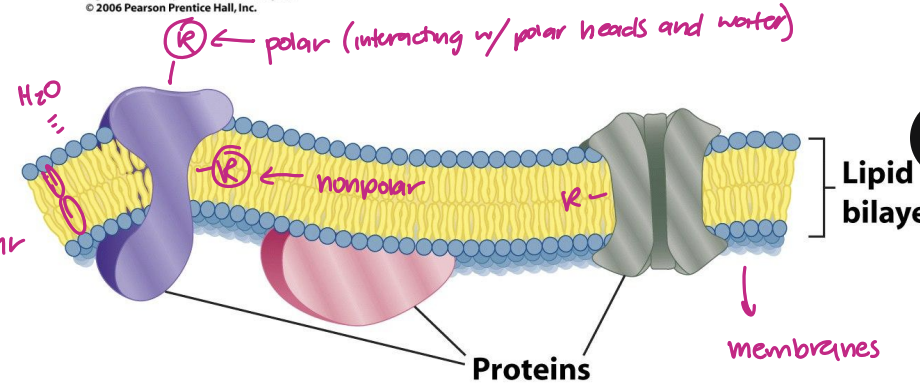
NP inside
Polar outside
describe water as a reactant: hydrolysis and condensation
hydrolysis: water breaking
condensation: production of water
ex. hydrolysis of a peptide
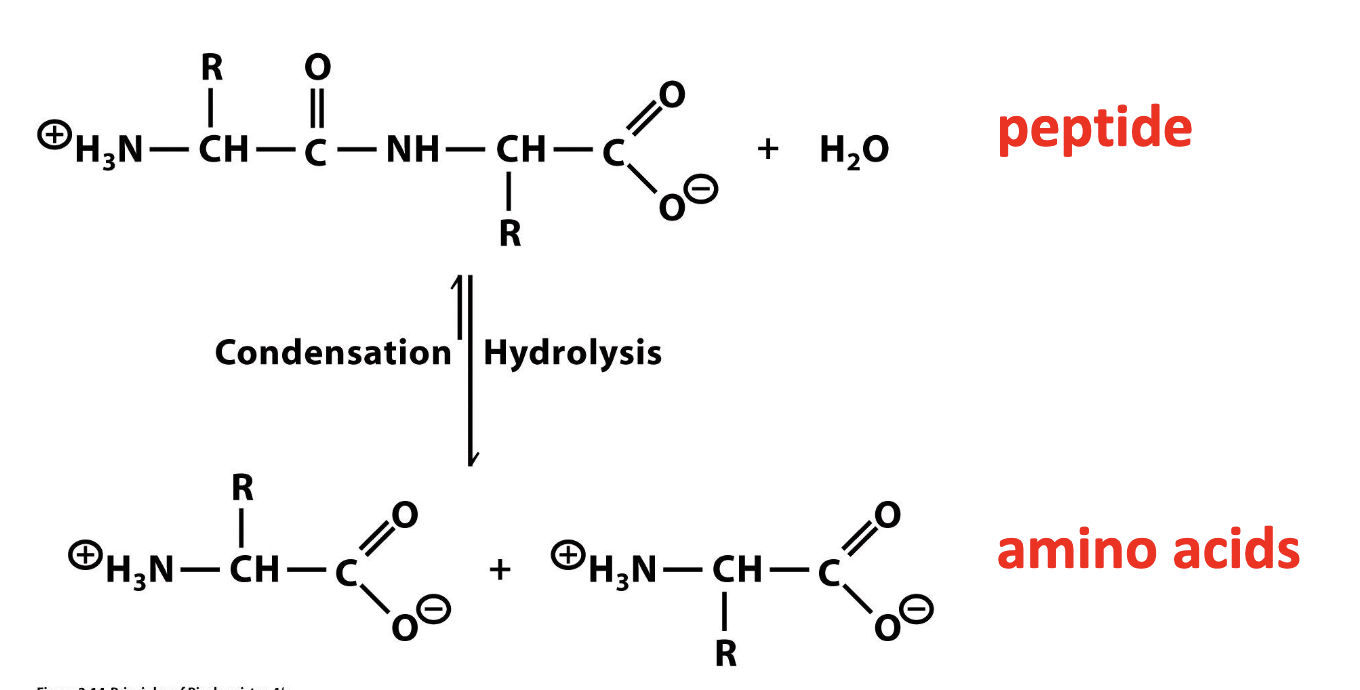
O=C-NH group = peptide bond
R = special diverse groups
water as a reactant: hydration/dehydration
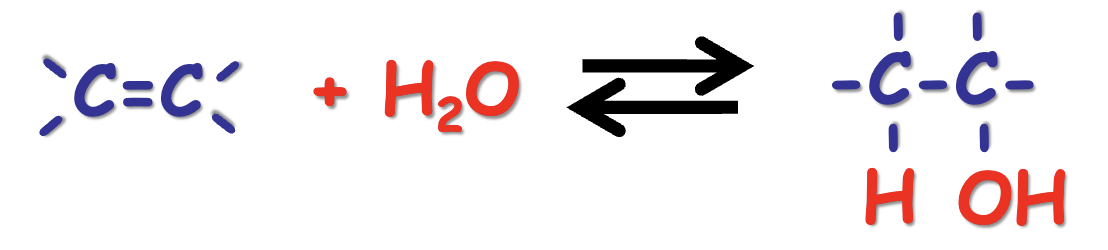
hydration forward
dehydration reverse
water as a heat buffer
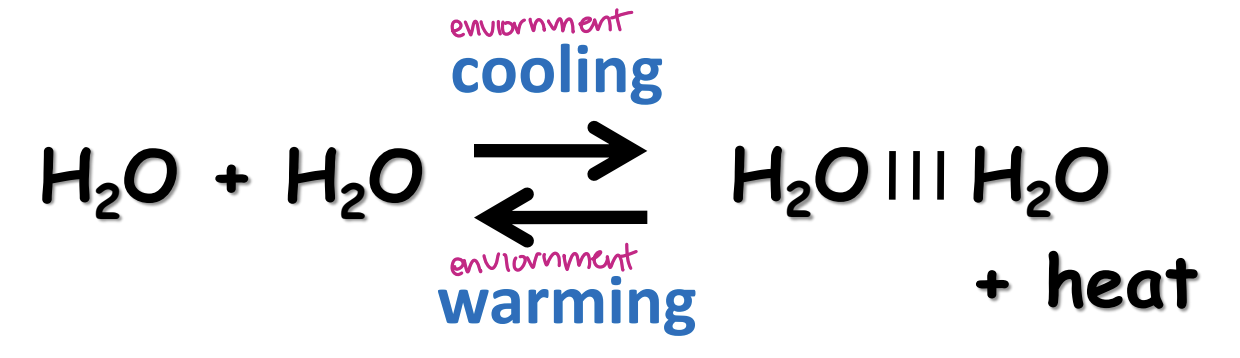
high specific heat of water
cooling counteracted by heat release
warming counteracted by heat uptake
perspiration requires heat input to vaporize
water moderates enviornmental temperature
what are the properties of water
electrical asymmetry
hydrogen bonding
polar solvent
hydrate ions
H-bonds with solutes
hydrophobic interactions
reactant - hydrolysis and hydration
ionization (pH)
formula fo equillibrium constant Keq
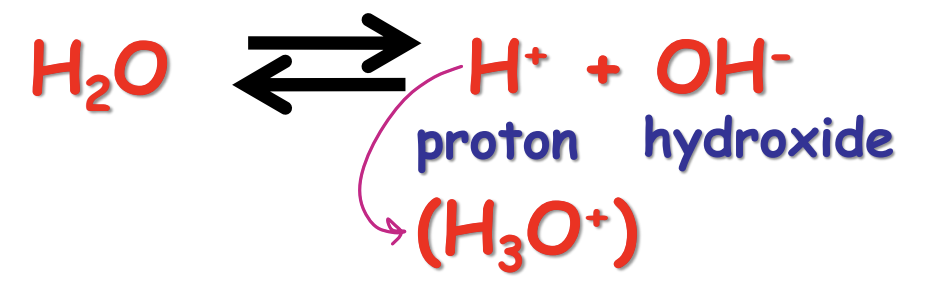
[H+][OH-]/[H2O]
Describe Kw
Kw = [H+][OH-] = Keq [H2O] = 10-14 M2 @ 1 atm, 25 c)
for pure water (neutral): [H+] = [OH-] = 10-7 M
ex.
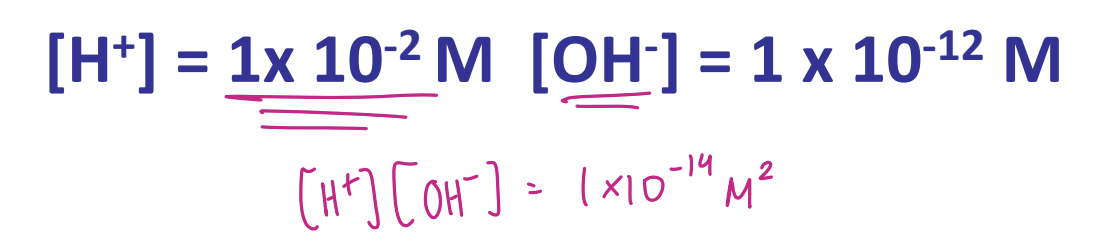
describe the equations for pH and pOH
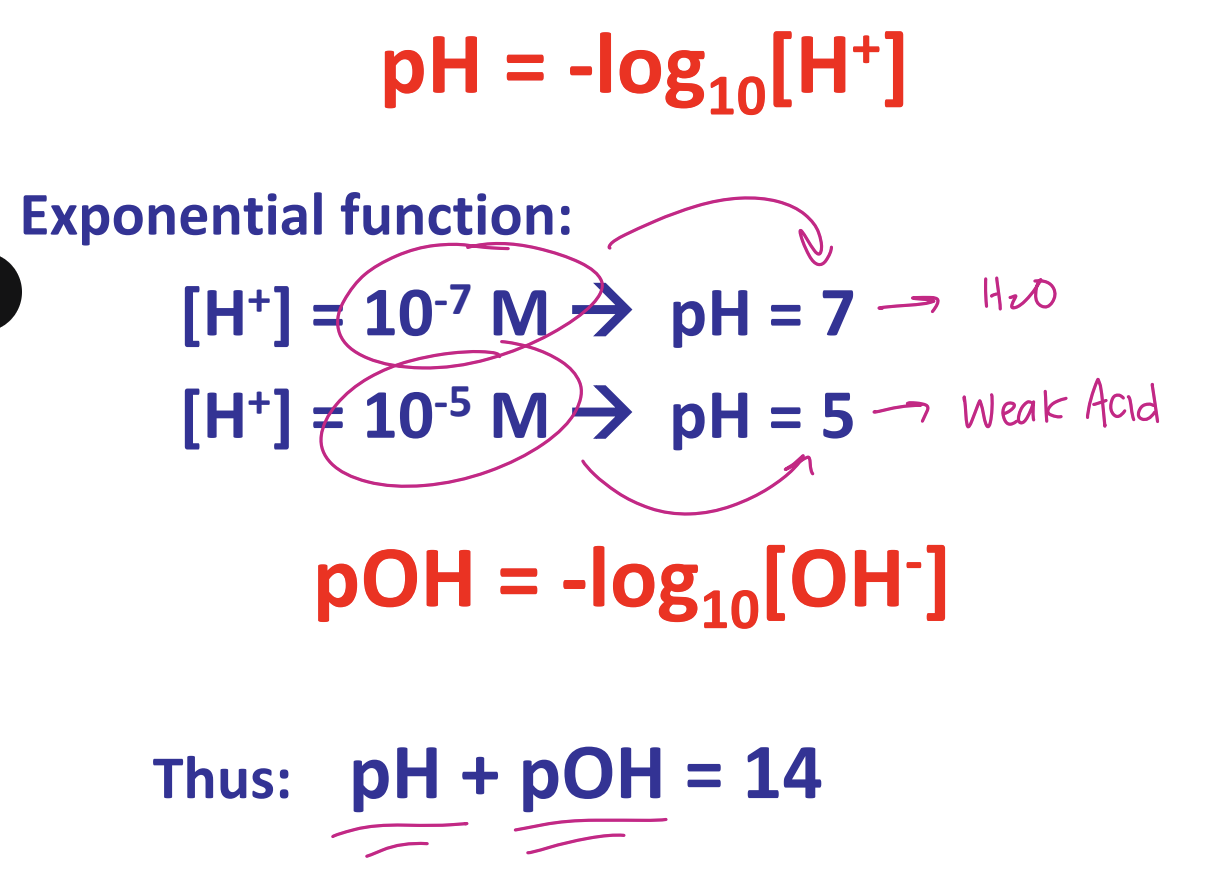
discuss the pH of common solutions
excess OH basic (alkaline): → strong bases NAOH or Ammoia
[H+] = [OH-] → 7.0 → human blood plasma/cows milk
excess [H+] acidic → strong acids human gastric contents HCl
waht are teh definitions of Acid-Base
acid
proton donor (bronsted)
electron pair acceptor (lewis)
base
proton acceptor
electron pair
describe teh relationship between acid strength and pka
stronger the acid = lower the pKa
what is the henderson hasselbach equation
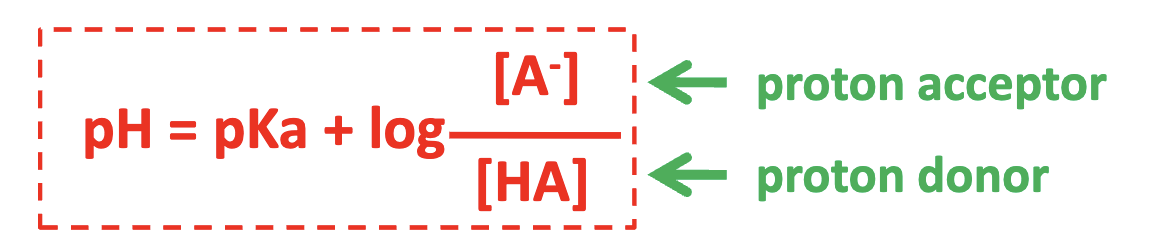
what is a buffer solution
[H+] and pH change very little
micture of a weak acid and its salt buffers pH
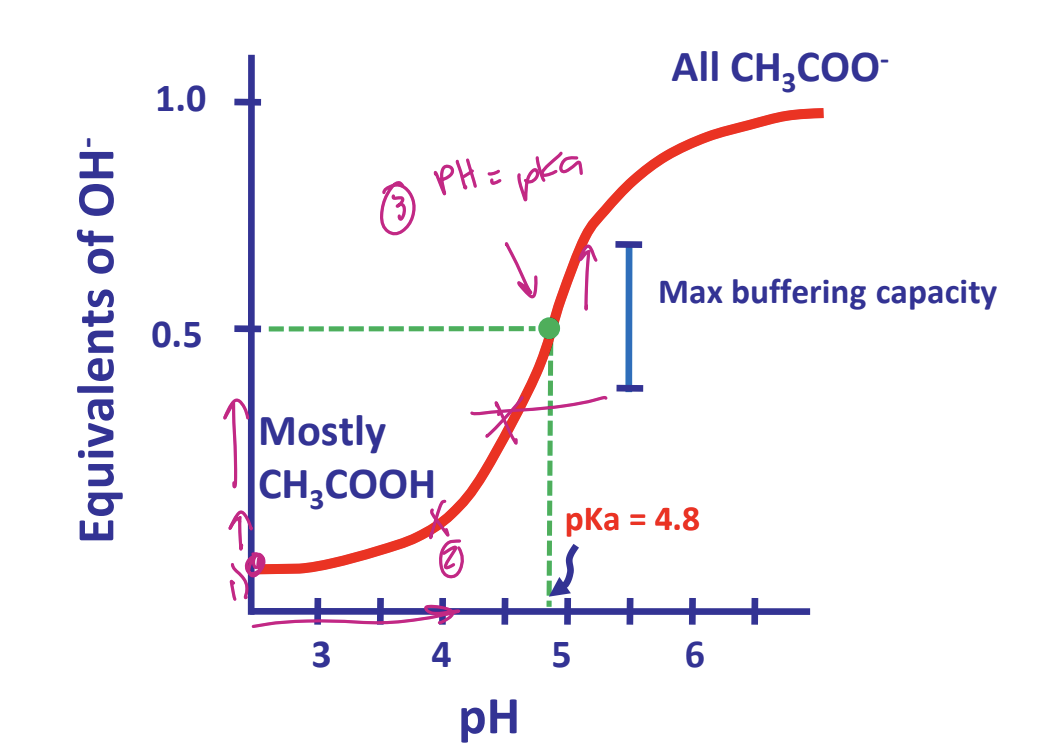
titration curves represent henderson-hasselbach equations
@ buffer region pH = pKa
buffering capacity of a buffer is greatest near its pKa
when [A-] = [HA]
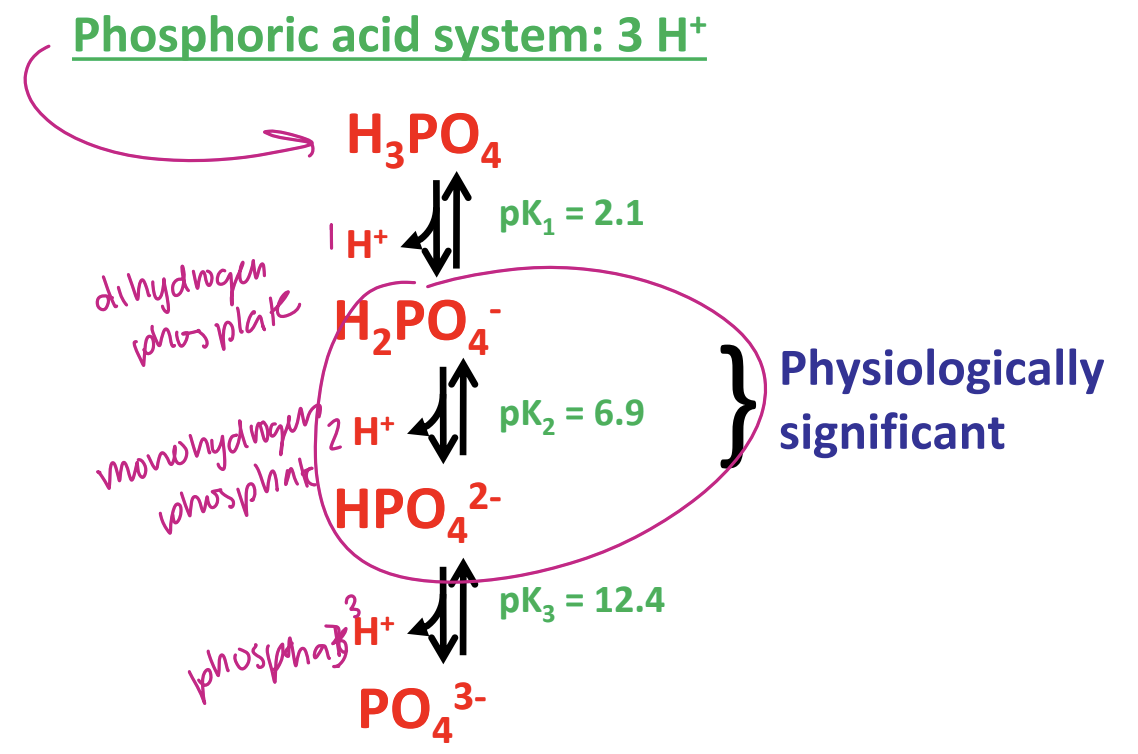
what is the biological buffering of pH
intracellular and extracellular fluids kept at a constant (6.9-7.4) by buffering systems
critical for enzymes and other activities
phosphoric acid system
describe the carbonic acid system (principal blood buffer)
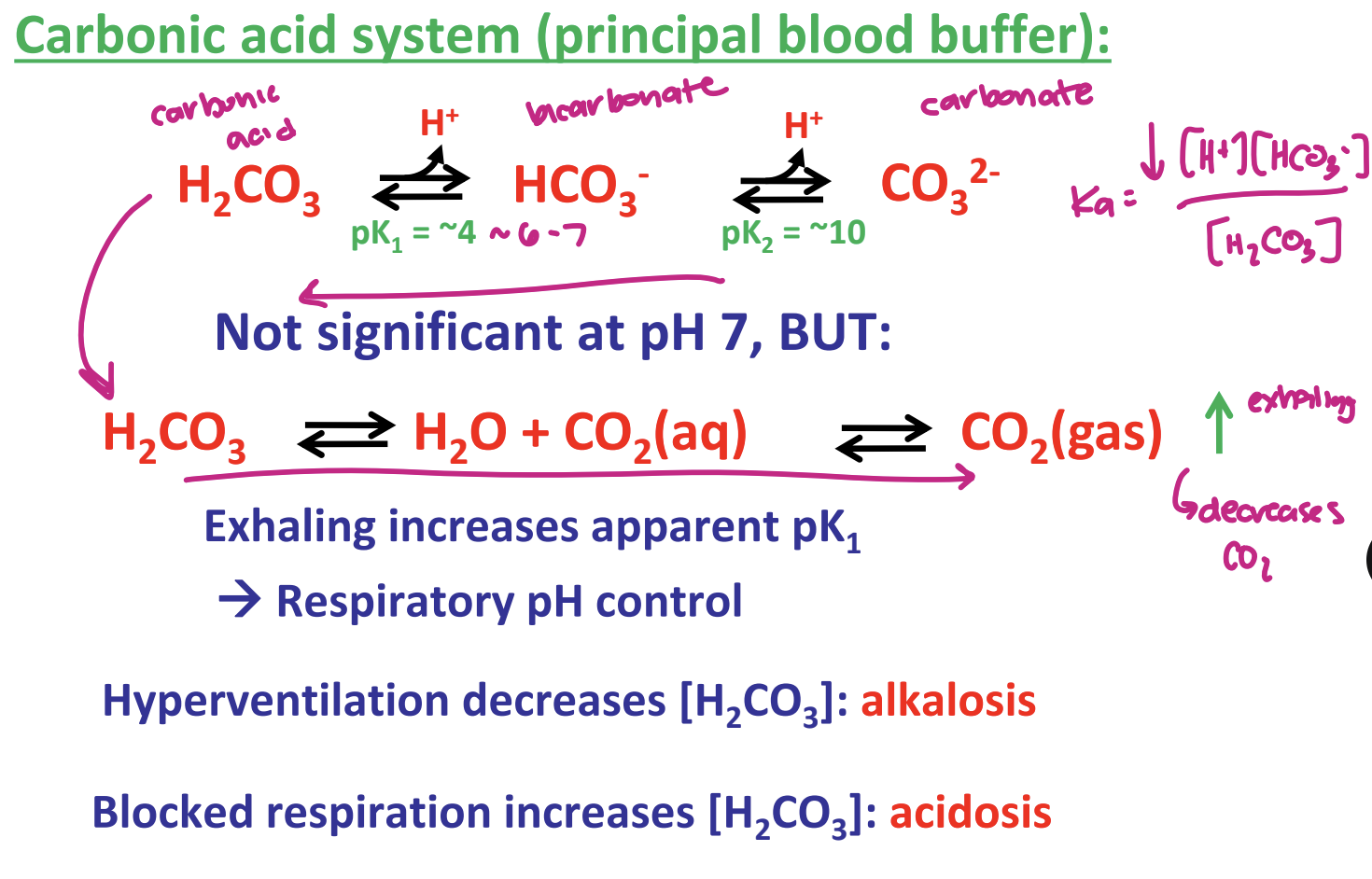
exhaling increases pK1 → respiratory pH control