Chem 241L Final UNC
1/116
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
117 Terms
Buffer capacity is at a maximum when:
pH = pKa
moles HA = moles A-
moles HA = moles OH-
A, B, and C
Both A and C
Both A and B
Both A and B
Which of the following is NOT a reason for which the calibration of laboratory glassware important?
It increases the accuracy of our measurements so that our measurement lies closer to the true value of the quantity of interest.
The calibration of glassware and equipment ensures that the answers we arrive at after the experiment is complete is a better reflection of the true answer.
It provides us with information about how our specific glassware measures the quantity in question as opposed to what the manufacturer claims it can do.
It increases the precision of our measurements and ensures that the results are repeatable.
It increases the precision of our measurements and ensures that the results are repeatable.
What is the required PPE inside the lab?
Goggles, Closed toed shoes, Lab coat, Shirt with sleeves (at least capped sleeves), and long pants that cover ankles or socks that cover ankles; gloves (must be removed before exiting the lab)
What are the key components of the SDS
Hazards of each chemical and proper disposal
Always add ___ to ____
Water and Acids
Acid to Water
AAA always add acid to water
Disposal rules
Any unused liquid or liquid waste goes into liquid waste container
Do NOT return unused reagents to stock bottles
any used solids go in solid waste container (kim wipes, gloves, etc go here)
Glass must be discarded in specially designed containers
Ensure waste containers are fully closed
Make sure not to put chemicals down the sink because the toxicity may cause explosions later down the drain
How would you transfer 250 microliters to another beaker? (explain all steps)
1. Remember to first add a tip to the pipette by pressing the tip cone into the larger hole of the pipette tip
2. Turn the dial until the volume display reads 0250 (if using a 1mL pipette)
3. Push push button to first stop
4. Submerge tip in liquid
5. Release the push button to draw up the liquid
6. Remove pipette from liquid and place over second beaker
7. Push the push button to the second stop to dispense the liquid
8. press the tip ejector to eject the pipette tip
9. Dispose of the pipette tip in the solid waste disposal
10. Put micropipette back in chemical fume hood.
How do you use the separatory funnel?
1. Make sure stopcock is closed before adding liquid
2. Hold stopper in place and vent often when inverting.
How do you read a graduated cylinder?
Bottom of the meniscus
Key objectives of Experiment 4
4.1:
1. Learn to calibrate laboratory equipment (pH electrode and micropipette)
2. Create a calibration curve
3. Prepare strong acid and strong base solutions using volumetric glassware
4. Find the buffer capacity of water
4.2:
1. Validate theoretical buffer calculations using Henderson-Hasselbach equations
2. Prepare a theoretical buffer
3. Prepare a Practical buffer
4. Determine the buffer capacity of ONE solution (theoretical or practical)
How did we accomplish the key objectives of experiment 4
4.1: Used gravimetric analysis to calibrate a micropipette. Weighed the water distributed by the micropipette, converted the mass into a volume using the known density of water at a given temp, then compared the volume of the reading to determine the correction factor. Then we created a calibration curve for the scale of the micropipette allowing us to correct the measurements we make
Buffer solutions were made using calibrated micropipettes. In lab we calculated the amount of stock solution needed to make each solution (one HCL other NaOH). Then diluted each with water. Then we set up a pH probe and titrated each solution with either a strong acid or strong base
4.2. Developed a procedure to make a theoretical buffer based on calculations; used calibrated micropipette to make the buffer; used practical method to make practical buffer; Titrated each buffer with Strong acid and then strong base; calculated buffer capacity for each buffer
what is Gravimetric Analysis
lab technique used to determine the mass or concentration of a substance by measuring a change in mass;
in this lab done by measuring mass of water put into a beaker and changing it to volume
How do you pick an acid/base pair?
Want a pKa closest to the target pH of the buffer
How do you calculate the appropriate amount of solution to make a buffer?
Use the pH and pKa given
HH first; then set up concentrations so F-[HA] / [HA] (or vice versa
Solve for [HA]
Use Formal concentration (F) to solve for [A-]
Use M1V1=M2V2 to find volumes needed for each
What is a practical buffer?
The practical approach involves choosing one of the reagents you selected above (either the weak acid or weak base) and using either a strong acid or a strong base to form the conjugate. There are two ways to do this.
How do you make a practical buffer?
selecting either the weak acid or weak base and adding either strong base or strong acid to produce the conjugate (You will need to partially neutralize your strong base or acid to form the conjugate)
1. Weigh out the desired number of moles of your weak acid OR weak base and dilute in a beaker containing 80 mL of deionized water.
2. Place a calibrated pH electrode in the solution to monitor the pH.
3. Add either strong acid or strong base into the beaker until your desired pH (4.50) is met.
4. Transfer the solution to a 100.00-mL volumetric flask; wash the beaker with deionized water and add the washings to the flask as well. Dilute to the line with water and mix.
Pros/cons of a practical buffer
Do not need to calculate amount of strong acid/base you are adding; you just monitor the pH as you add the strong acid or base
limited buffering capacity; can be susceptible to contamination and more complex to make
What is a theoretical buffer?
The theoretical approach involves selecting two reagents, based on your assigned pH, and using the Henderson-Hasselbalch equation to calculate the amount of each reagent needed.
You then add those reagents together into a volumetric flask of the desired volume followed by dilute to the line.
For this experiment you will need to select a weak acid —weak base conjugate pair.
How do you make a theoretical buffer?
prepared by selecting two reagents -- a weak acid and its conjugate base, where the pKa is close to the desired pH and preparing the solution is based on calculated values
1. Select the appropriate acid/base pair
2. Perform calculations to determine appropriate masses/volumes of weak acid and conjugate base
Pros/cons of a theoretical buffer?
Ideally has precise pH control and a wide buffering capacity; hypothetical so it has practical limitations so doesn't exist in reality
Which technique (theoretical or practical) is best in a laboratory setting? why?
Practical is best in the laboratory setting because they can be physically prepared and are the most realistic
What is the henderson hasselbach equation?
pH = pKa + log ([A-]/[HA])
What is buffer capacity?
The number of moles of a strong acid or base needed to change the pH of 1L of buffered solution by 1pH unit;
Describes the resistance of a solution to a change in pH
Will always be positive
What are the calculations associated with buffer capacity?
B = d(Cbase)/d(pH) = -d(Cacid)/d(pH)
Cacid and Cbase are formal concentrations (prior to any reactions)
In this class we did not take first derivative so we estimated it using the slope between consecutive points within the data (ex: Abs value of C1 - C2 over pH 1 - pH 2)
First derivative of the titration curve shows how resistant the solution is to change in pH at any point
Buffer
a solution that is resistant to a change in pH;
consists of a weak acid and its conjugate base
the pH of a buffered solution is generally unaffected by dilution
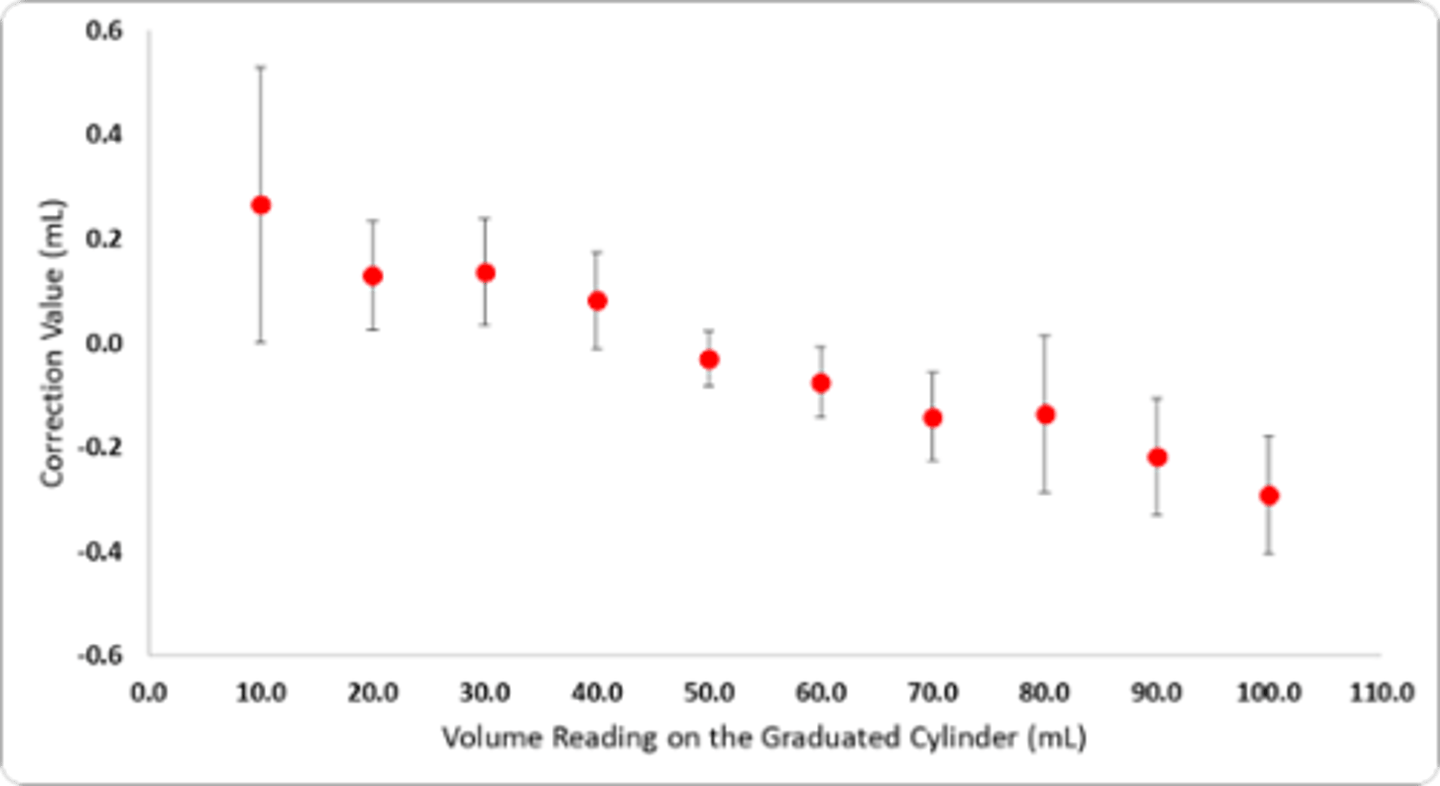
Making and using a calibration curve
Plot correction value for each volume
•Each of the red dots represent the correction value at a specific volume. Notice how it fluctuates with each volume
•To find the true volume at one of the measured values simply add (or subtract, if the correction value is negative) to the volume reading
•In order to find a value in between two points (say, 17). Take the slope of the line between 10mL and 20mL and then determine the correction value based on the line. (as seen in the purple line/green dot)
Calibration and Tolerance
calibration = process of relating the actual physical quantity (such as volume) to the quantity indicated on the scale of an instrument
Tolerance = accuracy to some degree (ex: +- 0.6mL) so acceptable variation in precision
In Experiment 4 you calibrated a micropipette and determined the correction factor. How
would you apply the correction factor if the volume delivered was between two points on your calibration curve?
•In order to find a value in between two points (say, 17). Take the slope of the line between 10mL and 20mL and then determine the correction value based on the line. (as seen in the purple line/green dot)
Correction factor
adjustment that is necessary for physical quantity to equal quantity indicated on the scale of an instrument
You are tasked with making a pH 6.0 buffer. In your experiment you titrate sodium hydroxide (pH 10) into a sample of your buffer and hydrochloric acid (pH 2) into another sample of your buffer. You then determine the buffer capacity. These experiments are completed independently. The buffer is able to resist a pH change upon the addition of which solution (HCl or NaOH)?
Both
Experiment 3 Key objectives and how we achieved them
1. Determine the pKa of bromothymol blue
2. Construct absorbance spectra using UV-Vis Spectrophotometer
3. Use Beers Law to draw conclusions about concentrations of H+
4. Perform a solvent-solvent extraction to qualitatively analyze solubility
We prepared 2 standards, 6 bromothymol blue solutions, and 2 extraction samples; then analyzed all samples with the spectrophotometer
pH and pKa equations from lab manual
pKa = -logKa
pH = -log[H3O+]
Ka = [A-]*[H3O+] / [HA]
Indicator used in lab; range of pH it is able to be used for; color in basic form and acidic form
Bromothymol blue
(pH 6.0-7.6)
Yellow in acidic form (pH 5; lambda max = 433.33nm)
Blue in basic form (pH 10; lambda max = 617.9nm)
How does a UV Vis Spectrophotometer work?
Used to measure absorbance as a function of wavelength or at a specific wavelength
Draw an instrument diagram of a UV Vis Spectrophotometer
Light source --> wavelength selector --> cuvette --> detector
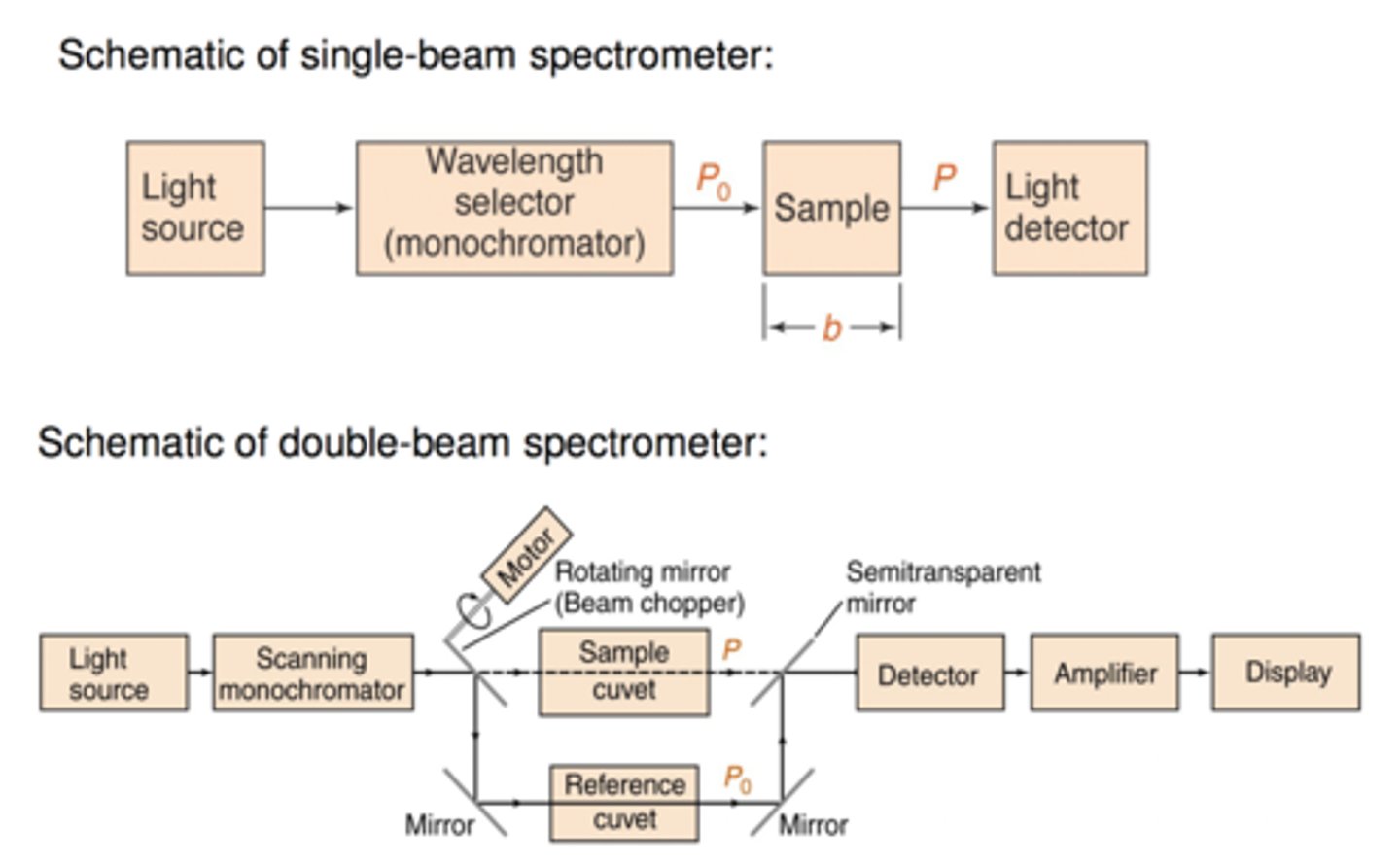
Why is a UV Vis spectrophotometer used for experiment 3?
We need to measure absorbance so we can plot absorbance vs pH and find inflection point of first derivative graph
Solutions are colored in the experiment
Concentration remains constant and cuvette size remains constant
How is concentration related to absorbance?
Concentration is directly proportional to absorbance (according to Beers Law) when measured at a single wavelength
Beers Law
A = Ebc(where E is molar absorptivity)
What is the purpose of a solvent-solvent extraction
to illustrate the varying solubilities in aqueous and organic solvents of BB due to the difference in molecular charge
overall: technique exploits differences in analyte polarity or charge to separate them into different liquids
What are the structural changes in the BB indicator at varying pHs
The bonds in the ring reorganize to delocalize the negative charge due to deprotonation
The 5 membered ring that contains the sulfur atom is broken in the rearrangement accounting for the dramatic color change to blue
Increase in bond conjugation (forms a double bond to an C-O)
forms a new O-S bond
What is absorbance?
the ability of a substance to absorb a certain wavelength of light
What is transmission?
wavelengths of light that pass through the sample
How are absorbance and transmission related?
Absorbance = -log(Transmission)
Absorbance equation (with log)
A = log (Io/I)
Io = Irradiance of light measured in absence of absorbing solution; I = irradiance of light measured after it passes through a solution of interest
In experiment 3 which was the organic layer and which was the aqueous layer? what was in each layer? what color was each layer?
Which layer was necessary for the experiment? and why do we care?
top layer = organic
bottom layer = aqueous
Most Acidic solution:
top layer = yellow
bottom layer = clear
Most Basic Solution
top layer = clear
bottom layer = blue
measuring the concentration of bromothymol blue → use the blue, aqueous, deprotonated, bottom layer
use top layer of the Most Acidic solution because the protonated (acidic) form of BB is present in that layer
In experiment 3, you completed a solvent-solvent extraction. Which form of BB was more soluble in the aqueous layer? Which form was more soluble in the organic layer?
Why?
The basic form was more soluble in the aqueous layer because it was deprotonated and negatively charged.
The acidic form of BB is fully protonated and therefore more soluble in the organic layer
When looking at the full spectra of the BB ranging from pH 5 to pH 10, the absorbance values converge. What is this point called and what is its significance?
Isosbestic point
Significant because it is a specific wavelength at which the total absorbance does not change so the concentration across all solutions measured is the same
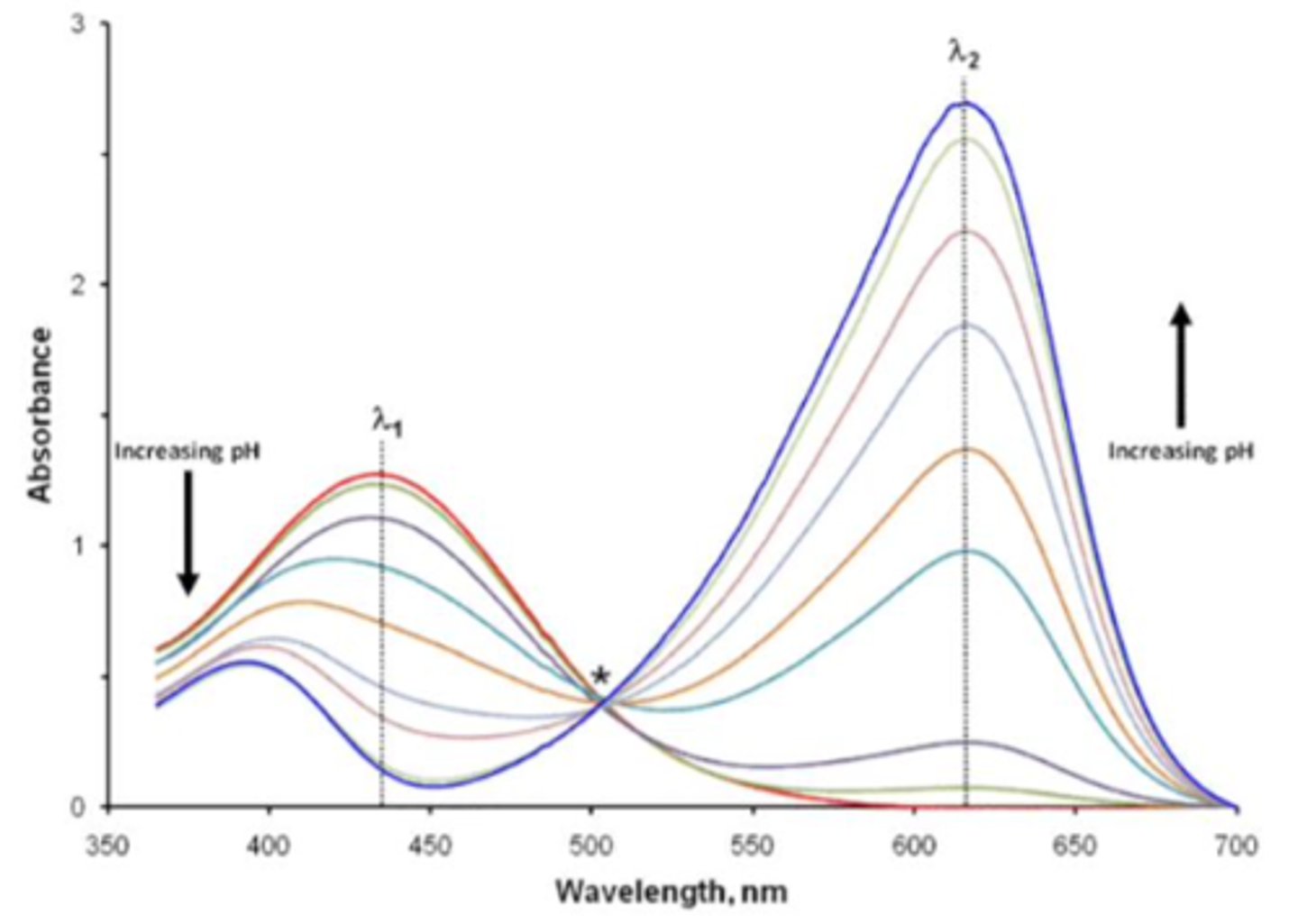
In this lab you were tasked to find the pKa of the indicator, bromothymol blue. How was this done?
Analyze the acidic and basic forms of the compound
We measure absorbance so we can plot absorbance as a function of pH for each max wavelength; the inflection point is where pH = pKa;
Use excel to calculate change in absorbance / change in pH and plot this as a function of average pH for both wavelengths on the same graph
this is a first derivative plot. the peak is where pH = pKa (the inflection point)
What does pKa tell us about a reagent?
It tells us the strength of the acid (lower pKa stronger acid and higher pKa weaker acid)
Tells us the pH value at which a chemical species will accept or donate a proton
For the analysis of the solvent-solvent extraction, you were required to use a glass cuvette instead of a plastic one. Why was this necessary?
ethyl acetate will dissolve the plastic
Kpartition equation
Kpart = [solute]organic / [solute]aqueous
(not multiplied organic and aqueous are just labels)
Key objectives of Experiment 1 and how we achieved them?
1. prepare a set of standard EtOH solutions in 1-pentanol 2%, 4%, and 6%
2. Analyze the solutions using gas-liquid chromatography
3. Construct a calibration plot using data (ethanol peak area vs % ethanol)
4. Determine partition coefficient between water and pentanol using pre-prepared extraction solution (plug in absorbance to calibration plot to get percent ethanol and then solve)
1. prepare 4 samples with % ethanol
2. analyze solutions via gas-liquid chromatography
3. construct a standard addition plot (ethanol peak area vs % ethanol added to beverage A)
4. Determine the alcohol content of beers
Partition Coefficient (GC)
K = C stationary phase/ C mobile phase
Standard calbiration curve
peak area vs %ethanol
x intercept is percent ethanol (from experiment 1) for peak area of zero
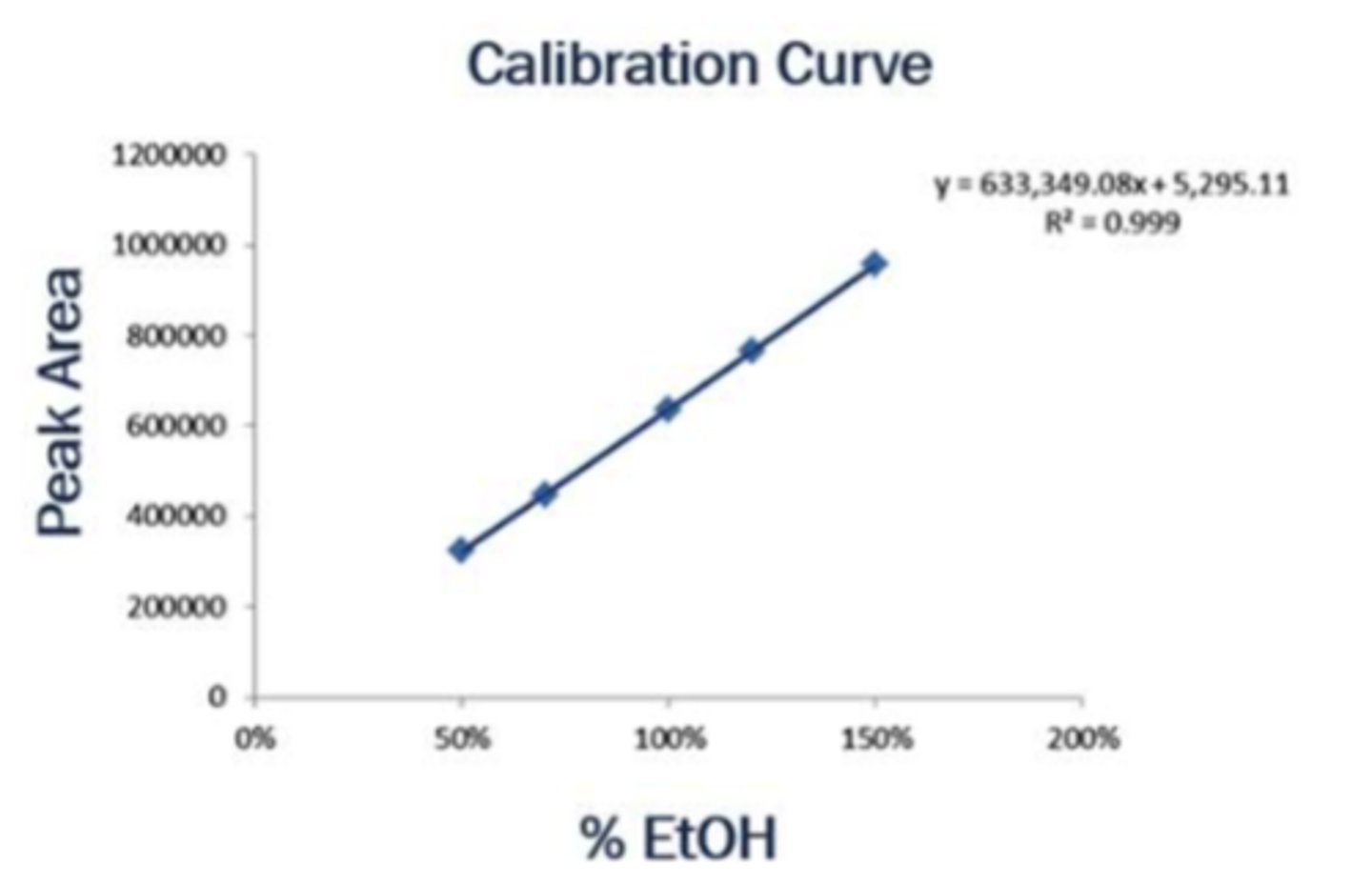
Standard addition curve
peak area vs % ethanol added to beverage A
X-intercept is the ethanol percentage in just beverage A
line of best fit is used to estimate for all other beers
How to perform a Solvent-solvent extraction?
1. solvents must be immiscible
what is the purpose of the salt and the pentanol in the solvent-solvent extraction in Experiment 1?
extraction is better when what you are trying to extract is less soluble in the layer you dont want it in; therefore adding salt decreases solubility of a molecule in aqueous layer so it can enter the organic layer easier
Why is it necessary to do a solvent-solvent extraction for all the samples for GC?
The sample is a complex mixture with a lot of chemicals so we need to isolate the compound of interest -- in this case Ethanol
How does the GC instrument work?
consists of liquid stationary phase (carbowax) and gaseous mobile phase (Helium)
used to separate volatile organic compounds
elution is based on boiling point for hydrocarbons
elution is based on interactions with the stationary phase for polar and non-polar compounds (non-polar elutes first due to less interactions)
. It is commonly used to determine the purity of substances, separate the components of mixtures, quantify the relative amounts of the components in a mixture, and qualitatively identify compounds in a mixture.
Draw and label a GC
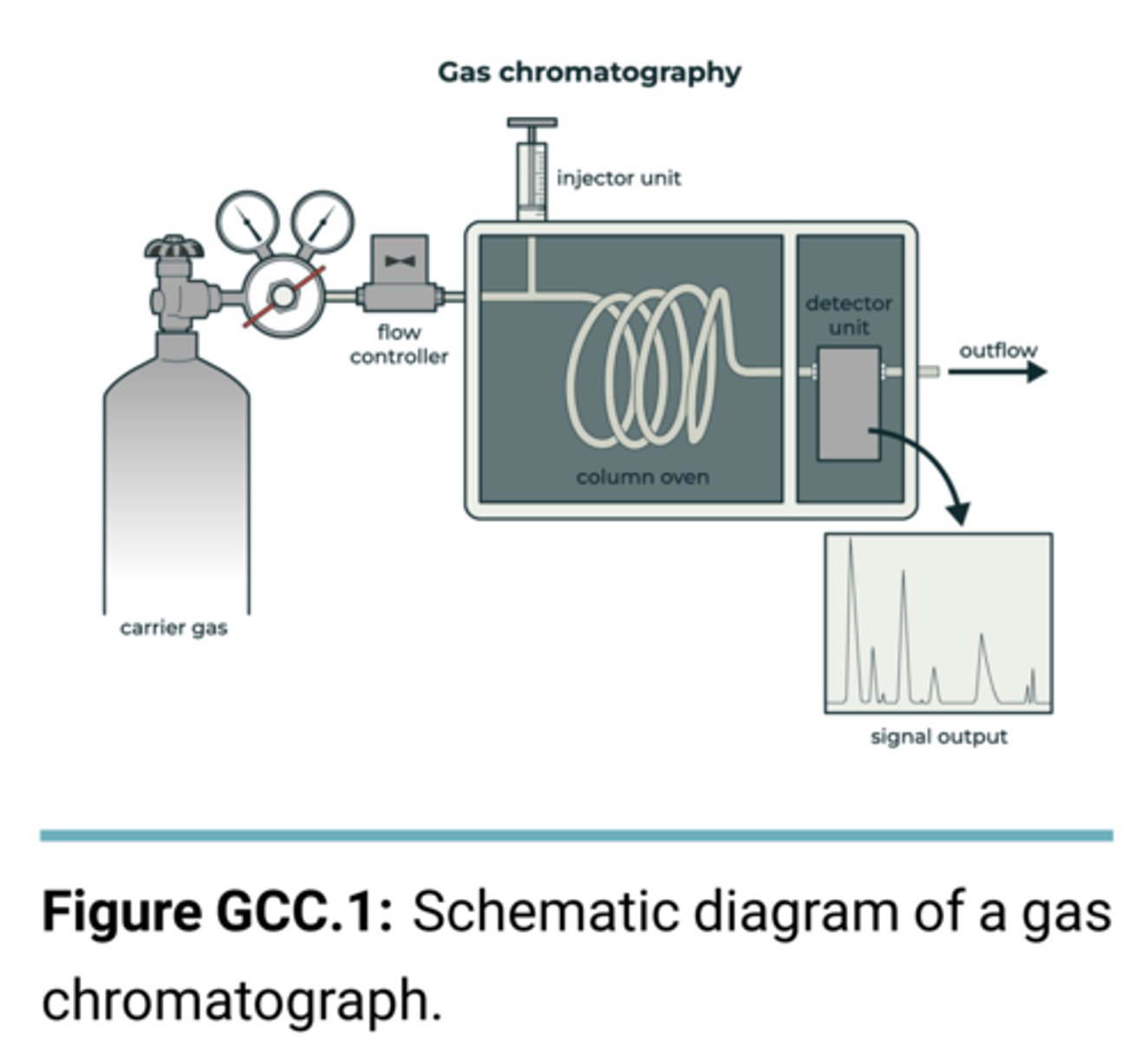
Why is GC used for the samples of experiment 1?
because our samples are volatile
What is retention time?
the time it takes a component in a mixture to pass through the column during gas chromatography
Tr = Tm + Ts (time in mobile plus time in stationary)
Equation for amount of analyte left in aqueous layer (q)
q = Vaq / (Vaq + (K*Vorg))
to the n power (n = number of extractions)
What is peak area?
the area underneath the peak on the gas chromatogram; relates to concentration of the compound in the sample mixture
How were retention time and peak area used qualitatively and quantitatively in this experiemnt?
qual - retention time show the elution order
quant - peak area is used and the standard addition plot to help calculate the percent ethanol in each solution
What are assumptions that were made about the matrix of the three beer samples? what were limitations of the experiment?
We assumed they are complex but all have similar matrices; therefore we needed to use standard addition for the experiment;
the limitations included assuming the molar absorptivity were the same for each sample
What is a baseline correction?
moving the plot up to zero to make it easier to measure the peak area; (formal def: to remove interference or background effects)
why is a baseline correction necessary for quantitative data?
Necessary to ensure accuracy in the quantitative data since we are looking at the area (which relates to concentration) and not just qualitative (there or not) data
What is the difference between a standard calibration curve and a standard addition curve?
Standard calibration
Analyte and standard are same compound
Standard solutions of known
increasing concentration are analyzed
Assume no matrix effect
Standard Addition
Analyte and standard are same compound
Add increasing amounts of standard solution by constant volume of unknown solution, basically making standards out of the unknown solution
Used when sample matrix is unknown or complex and hard to duplicate
unknown matrix effects anticipated
Why must we use a standard addition curve when determining the amount of ethanol in beer samples?
We do not know the sample matrix so we cannot duplicate it
After a solvent-solvent extraction, ethanol has a calculated partition coefficient of 1.33,
does more of the ethanol reside in the aqueous or organic layer? How do you know?
Organic layer; know because the partition coefficient is equal to concentration in organic over concentration in aqueous so a coefficient of over 1 means more is in the organic layer
What chemical property can be used to determine elution order? (GC)
elution is based on
boiling point for hydrocarbons
interactions with the stationary phase for polar and non-polar compounds
(non-polar elutes first due to less interactions)
Compare and contrast the stationary and mobile phases of Gas Chromatography and
Liquid Chromatography.
GC = liquid stationary; gas mobile
LC = solid stationary; liquid mobile
What chemical characteristics do samples need to be good candidates for GC?
volatile
Why must the sample be rapidly injected into the GC?
the solutions instantly vaporized as soon as they are put through the injection port
Key objectives of lab 5 and how we were able to achieve them
1. perform solvent-solvent extraction to isolate plant pigments from spinach
2. separate pigments using liquid chromatography
3. analyze pigments using spectroscopic detection
4. evaluate relative retention times and resolution values for the pigments
Relative retention
A = (tr2 - tm) / (tr1 - tm)
retention time 2 - mobile phase time over retention time 1 - mobile phase time
resolution
R = 2deltaTr / (Wa + Wb)
2 times change in retention time over width peak 1 plus width peak 2
gradient vs isocratic elutions
gradient elution = composition of mobile phases changes with time
Isocratic = mobile phase is constant
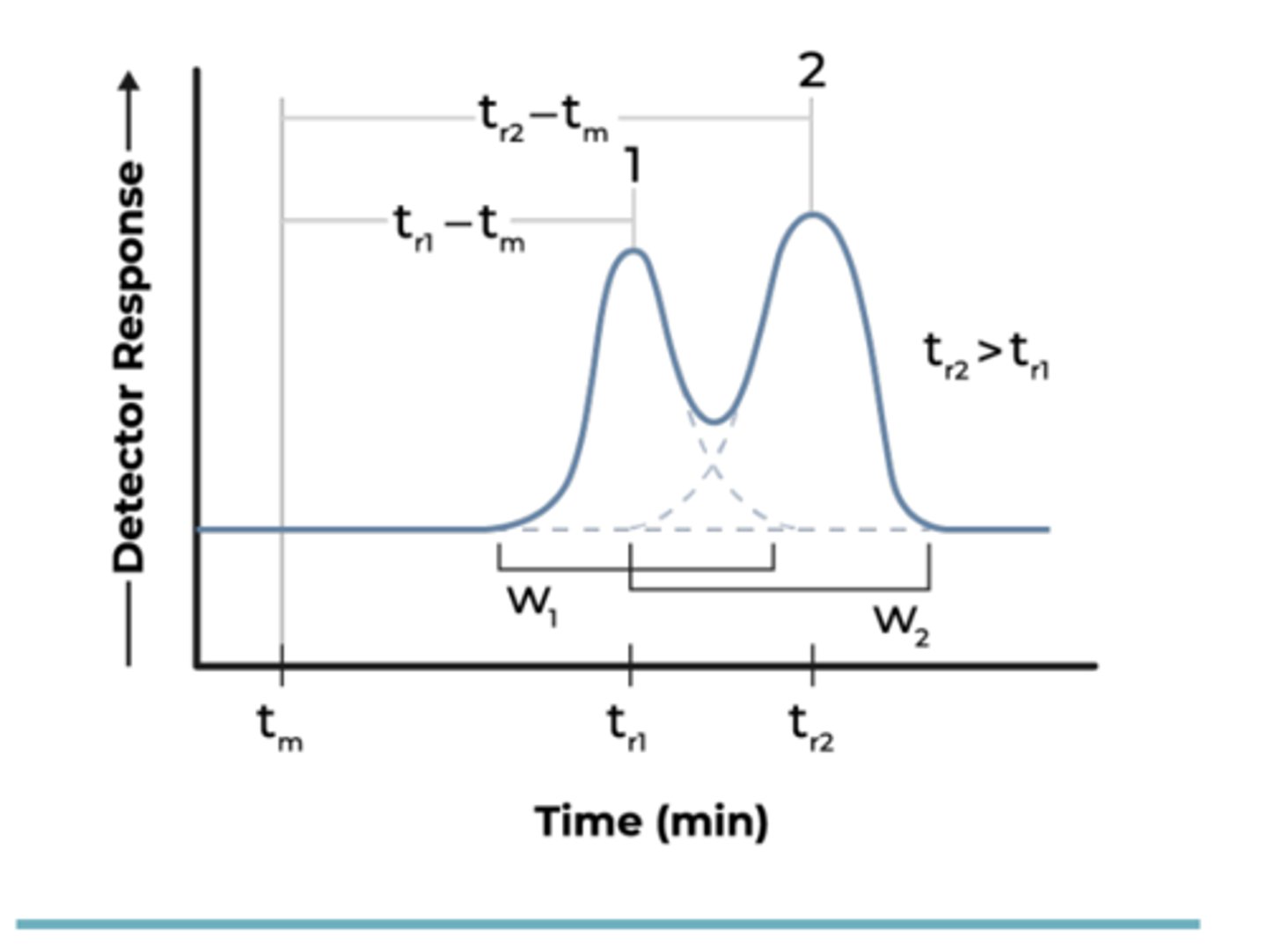
How do you find peak width
draw line along the peak and then subtract large from small
Relative retention time and resolution describe what?
the efficiency of the separation
How do you find retention time
Add time in mobile phase and time in stationary phase... or look at time it took for elution to occur
Which layer of the extraction in lab 5 do we care about (organic or aqueous)?
organic because that is where the pigment molecules reside after the extraction
Outline/diagram how LC works
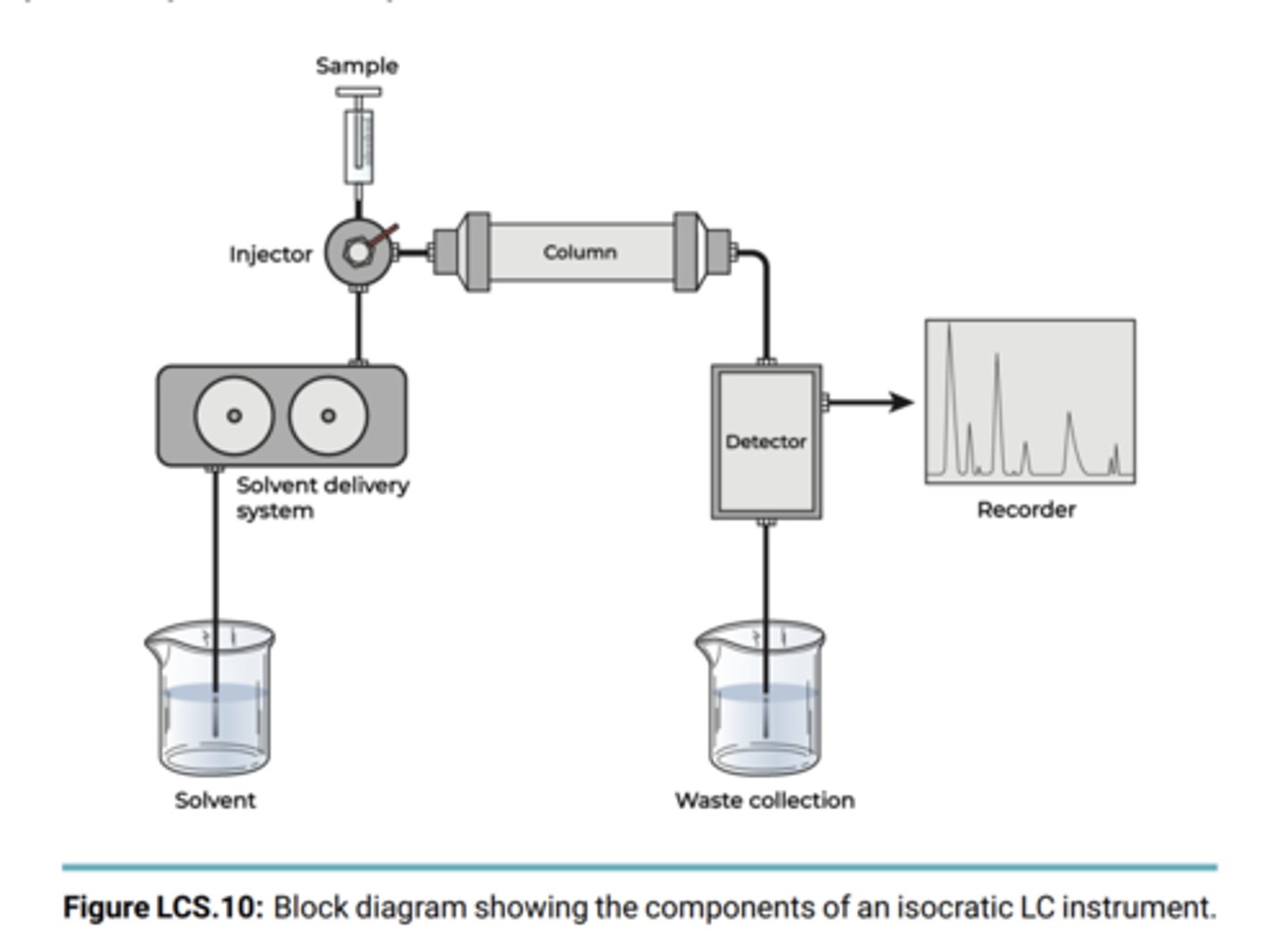
What was the elution order of the compounds and why?
xanthophylls -> chlorophylls -> carotenes
most polar to most nonpolar because we are running reverse phase which involves a polar mobile phase and a nonpolar stationary phase
Stationary and mobile phase of experiment 5
stationary phase = column packing material
Mobile phases 1 2 and 3 which get progressively more nonpolar
Normal phase LC vs Reverse phase LC
what did we run in exp 5?
Reverse = np stationary , polar mobile
Normal = polar stationary, np mobile
Reverse phase
What is the salting-out effect and why is it necessary for experiment 5?
If a solvent-solvent extraction is performed using water (no salt) and an organic solvent, the water molecules would hydrate many of the intermediate to high polarity pigments, thus making them more soluble in the polar aqueous phase.
If instead an aqueous electrolyte solution (saturated NaCl) is used, the solubility of the plant pigments in the aqueous phase decreases because the water molecules hydrate the sodium cations. This is termed the salting-out effect.
makes the pigments easier to move to organic layer which we are measuring
How to use UV Vis spectrophotometer?
1. follow lab manual to calibrate the instrument
2. Rinse the cuvette with about 1mL of the sample you are about to measure
3. Make sure the cuvette is facing the correct direction when measuring
4. Don't dispose of samples until the end of lab
**NO organic solvents with plastic cuvettes (use Glass cuvettes)
Poor resolution vs good resolution
poor resolution = combined peaks
good resolution due to high efficiency = separate peaks more pointy/thin
good resolution due to high selectivity = separate peaks, broader peak
What were the different mobile phases of experiment 5? why were they used
(most polar) water >> acetonitrile > ethylacetate (least polar)
gradient elution used because the polarities of the compounds in the mixture vary widely
In the LC experiment, you were tasked with determining the flow rate of the liquid chromatograph. Why is this important to the experiment? How was it completed?
put a grad cylinder under the tube that used to go to the waste container; set a timer for 5 mins; divide mL by 5 mins to get mL per min
important because it tells us if the solution is running through the column at an appropriate rate
In the LC experiment, you were to separate plant pigments on what type of column?
What pigments would you expect to elute first based on this? (Hint: will nonpolar or polar elute first?)
Reversed phase LC; Nonpolar stationary and Polar mobile; Polar elutes first then nonpolar
(xanthophylls then chlorophylls then carotenes)
If you were to switch from a reverse phase column (stationary phase) to a normal phase column but used the same mobile phase, what would you expect to happen?
Reverse elution so nonpolar —> polar
carotenes → chlorophylls → xanthophylls
What would happen if you switched both the stationary phase and made the mobile phase non-polar. Would you expect the pigments to elute in the same order? All together? Not at all?
stationary is polar —> polar molecules would not elute off the column
nonpolar pigments would elute off the column
In your experiment, you discover that your peaks have poor resolution. What are two ways to combat this experimentally?
Increase column length; decreasing flow rate
Describe how you would determine the peak width of a particular pigment in the
chromatogram. (Hint: draw a sketch).
What forces govern interaction of plant pigment with stationary and mobile phases?
-Electrostatic
-dipole-dipole
-dispersive interactions
Key objectives of lab 6 and how they were achieved?
1. construct a calibration curve for absorbance vs [glucose]
2. use a spectroscopic enzyme assay to detect sugars in: Gatorade sample; Acid digested sugar solution; Nondigested sugar solution
3. Determine how enzyme reaction rates are affected by temperature, reaction time, and amount of enzyme
Monosaccharide vs Disaccharide
Monosaccharide: one sugar unit; Disaccharide: two sugar units