Thermal Properties of Engineering Materials
1/62
Earn XP
Description and Tags
Brief summary of topic but will expand over time
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
What governs macroscopic thermal properties?
heat capacity, thermal expansions and thermal conductivity
Thermal stresses lead to …
catastrophic failure
Heat capacity is …
the ability to absorb heat from external surroundings.
MORE SPECIFICALLY heat capacity is…
amount of energy required to produce a unit temperature rise
Heat capacity in mathematical terms…
C = dQ/dT
What are the two ways of measuring heat capacity?
At constant volume or at constant pressure
What is usually slightly bigger at room temp and above, Cv or Cp?
Cp is slightly greater - BUT difference usually negligible
How is heat primarily stored in solids?
Vibrational energy
Atoms in solid crystal lattice constantly vibrating.
When thermal energy added to material…
there’s an increase in amplitude and frequency of vibrations
Effect of adjacent atoms being coupled due to atomic bonding when thermal energy applied…
vibrations travel through the crystal lattice in the form of elastic waves
How is energy is energy in the form of elastic waves quantised?
Single quantum of vibrational energy is called a phonon
What does thermal energy absorption generate more of ?
Phonons
Total vibrational energy of a solid is …
sum of energy of all its phonons
Heat capacity is not constant.
It is dependent on …
Temperature
At absolute zero, vibrational energy at minimum and Cv is 0.
As T rises, what happens to Cv
it increases rapidly, then plateaus at higher temperatures
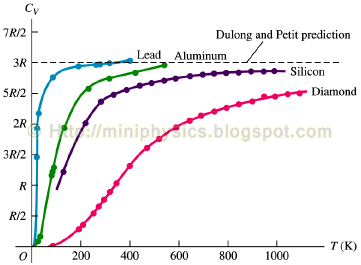
What is Cv like at very low temperatures, mathematically?
Cv proportional to T³
Change in dimensions with temperature quantified by …
coefficients of thermal expansion, al
What is al
material property which indicates the extent to which a length of material expands upon heating
Define al mathematically
change in length/original length = al * change in T
volume coefficient of thermal expansion av describes what?
fractional change in volume with temperature
For isotropic materials, approximate av
av = 3*al
What is shown by the potential energy versus interatomic distance curve?
the relationship between the potential energy of a system of atoms and their relative positions
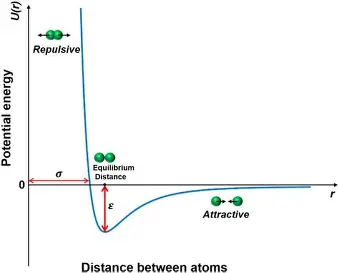
What happens, in terms of potential energy, when atoms move closer together?
their potential energy decreases due to attractive forces between them What happens, in terms of potential energy, when atoms move closer together
What happens, in terms of potential energy, when atoms reach equilibrium bond length?
potential energy reaches a minimum and the atoms are most stable
Effect of increase in energy on a perfectly symmetric energy trough …
most energy troughs for solids are not symmetric
would not change the average interatomic distance - SO no thermal expansion would occur
Describe the typical energy trough for solid materials
asymmetric, repulsive force curve rises much more steeply than the attractive force curve
What do very deep, narrow energy troughs indicate about a material?
strong interatomic bonding and likely a lower coefficient of thermal expansion
Describe expansion in metals
Coefficient of thermal expansion: 5×10-6 to 25×10-6
intermediate al, mid expansion
strong metallic bonding keeps expansion moderate
metals// higher melting point: stronger bonds, deeper potential energy troughs, lower al values
al dependent on melting point
Describe expansion in ceramics
Coefficient of thermal expansion: 0.5×10-6 to 15×10-6
very low al, least expansion
very strong interatomic bonds: ionic or covalent
highly resistant to thermal shock
fused silica has al close to 0
Describe expansion in polymers
Coefficient of thermal expansion: 50×10-6 to 400 ×10-6
high al, highest rates of thermal expansion
weaker van der waals bonds holding polymer chains together
leads to significant macroscopic expansion - T increases and weak bonds allow chains to easily move further apart
Define thermal conductivity
the phenomenon by which heat is transported from high temperature regions to low temperature regions within a solid
Define thermal expansion macroscopically, mathematically
Fourier’s Law: q = -k(dT/dx)
q is the heat flux
dT/dx is the temperature gradient within the medium
k is the thermal conductivity of the medium
What is the significance of the negative sign in Fourier’s law
Heat flows down the temperature gradient - hot to cold
The two primary mechanisms by which heat is transported through a solid are …
phonons: lattice vibration waves
translational motion of free electrons
Mathematically, the total thermal conductivity is …
the sum of the lattice/phonon conductivity and the electronic conductivity
k = kl + ke (respectively)
Atomically, what two factors increase when a region of a solid is heated?
the amplitude and frequency of atom vibrations
How do enhanced vibrations, in a solid when heated, travel through the lattice?
as phonons, carrying thermal energy to cooler regions
What additional feature of metals carries heat to cooler regions?
(not phonons)
free electrons in the hot region gain kinetic energy and migrate to cooler regions
here energy is transferred to the lattice via collisions
What is the Wiedemann-Franz law?
The Wiedemann-Franz law states that the ratio of the electronic contribution of the thermal conductivity (κ) to the electrical conductivity (σ) of a metal is proportional to the temperature (T).
What is the formula of the Wiedemann-Franz law?
σκ=LT
thermal conductivity (κ)
electrical conductivity (σ)
proportionality constant, Lorenz number (L)
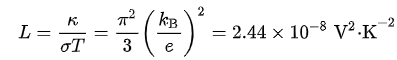
In what materials do the electron mechanism heavily dominate the phonon mechanism
high-purity metals
Effect of alloying metals on the thermal conductivity and why does this occur?
decreases thermal conductivity
impurity atoms introduce severe scattering centers
impede on both electron and phonon motion
What materials have practically no electron contribution?
non metallic materials such as glass and ceramics
lack a significant concentration of free electrons
(also polymers)
What mechanism are ceramics dependent on for thermal conductivity, atomically?
almost entirely dependent on phonons (lattice vibrations)
What are phonons and how are they scattered in ceramics?
elastic waves
scattered by imperfections within the crystal lattice
in ceramics: scattered by lattice defects, grain boundaries, and other phonons
as T increases, more atomic vibrations and more frequent phonon-phonon collisions (Umklapp scattering)
What happens when heat energy can no longer travel through a ceramic due to Umklapp scattering?
Thermal conductivity drops dramatically
Heat conduction in ceramics primarily relies on phonon transport.
If Umklapp scattering becomes so strong that phonons lose their ability to carry heat efficiently, the material’s thermal conductivity plummets.
could lead to structural or functional consequences

How does porosity effect thermal conductivity of ceramics
pores are gas filled voids
they have exceptionally low thermal conductivity
engineers exploit this by designing highly porous ceramic structures for use as a thermal insulator
What mechanism are polymers dependent on for thermal conductivity, atomically?
scattering of vibrational energy
caused by complex, intertwined and largely amorphous structure of polymer chains
What atomic movements does heat transfer within polymers rely heavily on?
translation, rotation and vibration of molecular chain segments
BUT movements highly restricted so heat transfer is very slow
How can thermal conductivity be increased (slightly)?
increase the crystallinity of the polymer
more ordered lattice allows more efficient vibrational energy transfer compared to an amorphous structure
Define thermal stresses
stresses induced on a body as a result of temperature change
What can thermal stresses cause in materials?
plastic deformation
catastrophic fracture - especially in brittle materials
What two sources usually lead to thermal stresses?
mechanical constraint of uniform thermal expansion/contraction
presence of temperature gradients across a material
Effect of constrained expansion is …
internal compressive stresses
Effect of cooling a constrained body so that it cannot contract is …
internal tensile stresses
What is the magnitude of internal stresses caused by constraining thermal expansion/contraction, mathematically?
σ = E*al*change in T
magnitude of stress (σ)
modulus of elasticity (E)
coefficient of thermal expansion (al)
At what point will permanent damage or failure occur due to thermal expansion/contraction?
when stress exceeds yield strength or fracture strength
When can thermal stresses occur in material when body is completely unconstrained?
when temperature varies from one part of the body to another
Effect of rapid heating or cooling is …
surface changes T quicker than core
one wants to expand/contract whilst the other is restricting it
Effect: massive tensile stresses on surface whilst core experiences compressive forces to balance the load
Effect of rapid cooling on materials such as ceramics:
ceramics: no capacity for plastic deformation to relieve stress
rapid cooling induces surface tensile stresses that exceed fracture strength
leads to rapid crack propagation and explosive failure AKA thermal shock
What is the capacity of a material to withstand failure caused by thermal stress?
thermal shock resistance
High thermal shock resistance favored by …
high fracture strength
high thermal conductivity
low modulus of elasticity
low coefficient of thermal expansion
Example of material with good thermal shock resistance and why?
fused silica
near to zero coefficient of thermal expansion, al