L21 - Amino Acid Metabolism
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
what are amino acids used for
when glucose and fats are low, they can be catabolized as energy and carbon skeleton (can be made into an energy source) - most available AAs are used to make proteins, signaling molecules, and other products like hemes, purines, pyrimidines, coenzymes, and biogenic amines
when does amino acid catabolism occur
during starvation - too much nitrogen is toxic, so the amino group has to be removed and N excreted through uric acid (noraml) or urea (when in excess)
what are carbon chains of amino acids converted into
things we can work with such as pyruvate, OAA, acetyl CoA, or alpha-ketoglutarate
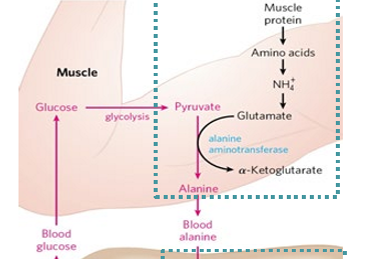
how does nitrogen get excreted in the muscles
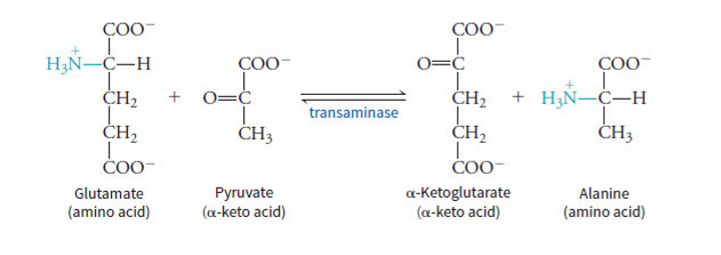
aminotransferase moves amino group to pyruvate from glutamate to produce alanine
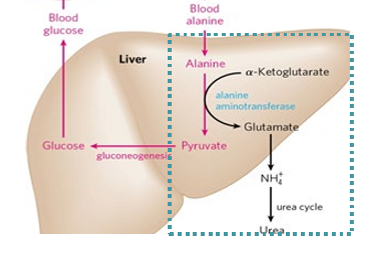
how does nitrogen get excreted in the liver
alanine is a source for pyruvate and aspartate/urea cycle - moving amino acids to the liver allows for gluconeogenesis to happen
what does it mean if nutritional stress in a negative energy balance mean
in a caloric deficit - amino acids can be sources of carbon for gluconeogenesis
what happens in the liver when there is excess nitrogen, and the carbon is harvested during gluconeogenesis
the amino groups are funneled into alpha-ketoglutarate to form glutamate which is oxidatively deaminated by glutamate dehydrogenase to give ammonia to alpha-ketoglutarate - the ammonia is made into urea for excretion and the carbon atoms are metabolized to major metabolic intermediates
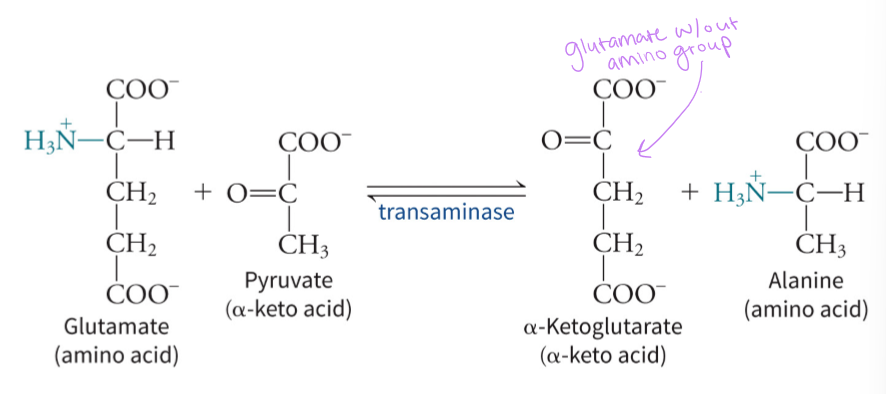
what are transaminases in amino group catabolism
can move the amino group around to make glutamate to be able to enter urea cycle - the alpha-amino groups are transferred onto alpha-keto acids to make the corresponding alpha-keto acids and alpha-amino acids - glutamine + alpha-ketoglutarate get converted to alpha-keto acid + glutamate

what enzymes perform deamidation in amino group catabolism
asparaginase and glutaminase
what does glutamate dehydrogenase do in amino group catabolism
glutamate will be metabolized to generate ammonia
what is the urea cycle in amino group catabolism
the cycle that is required to eliminate excess ammonium in the form of urea - a special cycle utilized to rid the body of nitrogen during times of nutrient deprivation when you are degrading muscle proteins to use the carbon for gluconeogenesis
what is the purpose of the tranamination reactions
to transfer amino group from amino acid to alpha-keto acid which uses vitamin B6 derivate pyridoxal phosphate (PLP) - set the stage for ammonia excretion
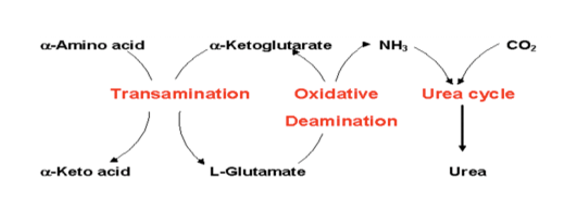
what is the purpose of oxidative deamination
removes ammonia to produce urea which uses glutamate dehydrogenase
what is pyridoxal-5’-phosphate (PLP) and
a derivate of pyridoxine, which requires flavin mononucleotide (FMN) as a cofactor for this reaction
how does transaminase bind to Pyridoxal-5’-phosphate (PLP)
must form an enzyme-PLP schiff base which stabilizes the intermediate allowing for transfer of the amino group from the first to the second molecules
how are amino groups moved
(enzyme with amino linked to carbon from PLP) pyridoxal phosphate is the coenzyme for amino transaminase that is required for the reaction that stabilizes the intermediate - aldehyde group of PLP forms a Schiff-base linkage (internal aldimine) with the E-amino group of a specific lysine group of the aminotransferase enzyme
after PLP stabilizes the intermediate, how does the amino group get transferred from the first molecule to the second
the enzyme-bound pyridoxal phosphate reacts with the amino acid, which forms a schiff base of C=N and after the shift to the double bond, alpha-keto acid is released through hydrolysis of the schiff base and PLP is produced - pyridoxamine phosphate then forms a schiff base with alpha-keto this time, and after the second double bond shift, amino acid is released through hydrolysis of the Schiff base and enzyme-bound pyridoxal phosphate is regenerated
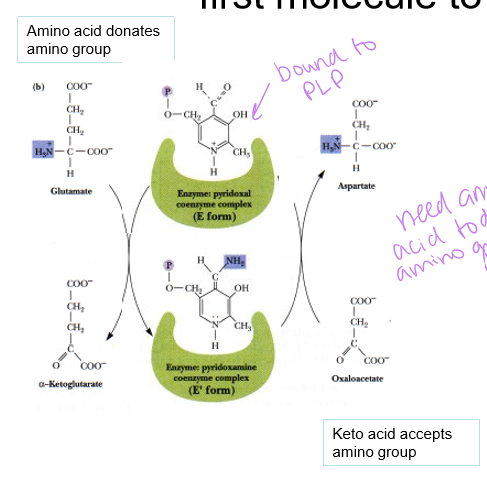
what donates and accepts the amino group when it is being transferred from molecules 1 (enzyme pyridoxal enzyme complex - E form) to molecule 2 (enzyme pyridoxyamine coenzyme complex - E’ form)
amino acid donates and keto acid accepts - results in turning glutamate into alpha-ketoglutarate, and aspartate into OAA
how is the nitrogen moved around between the amino acids
through three transamination reactions - to do this, transamination of alpha-ketoglutarate can be paired with the transfer of the amino group from alanine or aspartate and generates TCA intermediates that can enter the cycle
why is it important that the transfer of nitrogens between amino acids is reversible
it allows the body to seamlessly switch between amino acid degradation and amino acid synthesis based on immediate metabolic needs - vital for N homeostasis
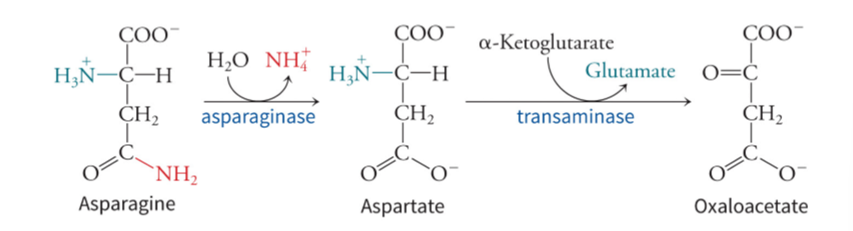
what does asparagine require in amino acid catabolism
requires deamidation because it has two amino acids to get to OAA - asparagine to (asparaginase) aspartate to (transaminase) OAA - produces ammonia from water and glutamate from alpha-ketoglutarate
how is glutamine deamidated
turns glutamine into glutamate using water and glutaminase, and releases ammonium
how is the amino group removed from glutamate
glutamate dehydrogenase reaction which removes the alpha amino group from glutamate to produce free ammonia and alpha-ketoglutarate
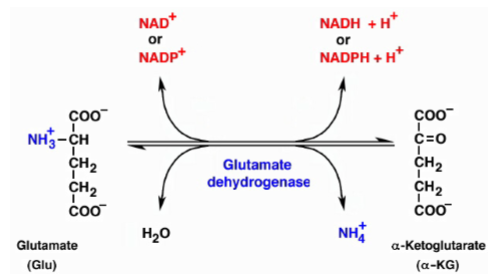
what does glutamate dehydrogenase reaction require
cofactor NAD+/NADP+ or NADH/NADPH depending on what direction it is running in - produces ammonia which moves on to get used in the carbamoyl synthase reaction, and alpha-ketoglutarate is recycled back to the transamination reaction or TCA cycle
why does the body need to generate new carbon molecules through amino acid catabolism
for the TCA cycle and ketogenesis - because of this, amino acids can alter flux of gluconeogenesis, although it doesn’t alter it directly, but instead through enzymatic reactions, regulation, and hormone signaling
what are the glucogenic fates of the catabolism of amino acids
alanine, arginine, asparagine, and glutamate
what are the glucogenic and ketogenic-catabolic fates of amino acids
threonine
how does serine get converted to pyruvate
through transamination where the amino group is not transferred to another compound
what happens in valine degradation
three step process where the alpha-amino group is removed via transamination using alpha-ketoglutarate and releases glutamate, and then the branched-chain alpha-keto acid dehydrogenase complex catalyzes an oxidative decarboxylation reaction where the carbon skeleton of valine becomes attached to CoA (releases NADH and CO2), and finally, the third step is catalyzed by acyl-CoA dehydrogenase which is the same enzyme that participates in fatty acid oxidation
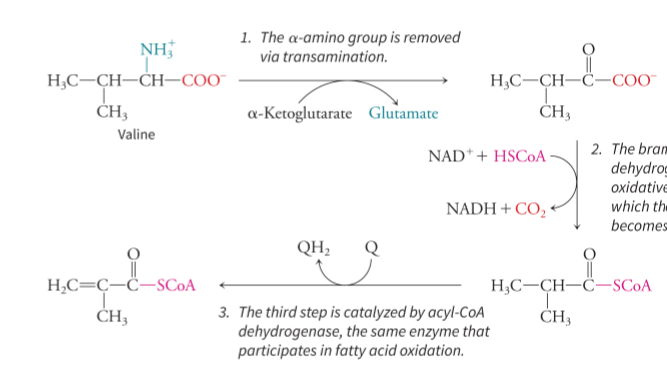
what are the enzymes and cofactors used in the three steps of valine degradation
transaminase with alpha-ketoglutarate, branched-chain alpha-keto-acid dehydrogenase complex with NAD+ and CoA, and acyl-CoA dehydrogenase with Q
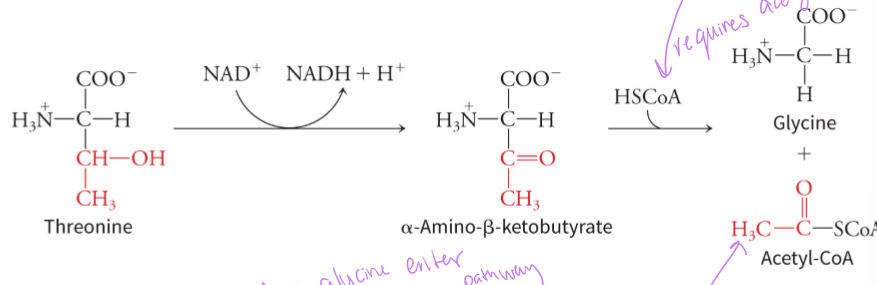
why is threonine both glucogenic and ketogenic
its metabolism breaks down into acetyl-CoA (ketones) and glycine (glucose) which can enter both glucose production pathways and fat/ketone body production pathways - glycine is first converted to serine which can be further converted into pyruvate which can enter gluconeogenesis to make glucose
how does the body get rid of excess ammonia that amino acid catabolism just produced
the urea cycle which runs during starvation when amino acid catabolism is high, so there are high levels of ammonia production
what does the urea cycle require
CO2, ammonium ion, and arginine as substrates, but then also needs 3 ATP total
how is urea the is produced in urea cycle excreted
into the blood stream, then cleared by the kidney and dumped into the urine - 2 reactions occur in the mitochondria with the remaining 3 in the cytosol
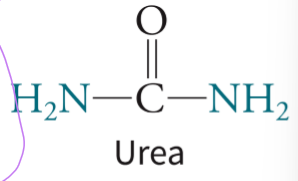
urea - has 2 nitrogen atoms
where is the first nitrogen in urea derived from
from glutamate that generates ammonia to enter the carbamoyl phosphate reaction, where ammonia, ATP, and bicarbonate are synthesized to carbamoyl phosphate, which combines with ornithine to make citrulline
where is the second nitrogen in urea derived from
from aspartate after a transamination reaction from OAA - aspartate combines with citrulline to form argininosuccinate (both nitrogen atoms are on this molecule)
what happens to argininosuccinate after it is generated containing the 2 nitrogens of urea
argininosuccinate is cleaved into arginine and fumarate, and then arginine is cleaved by arginase to form urea with the 2 Ns
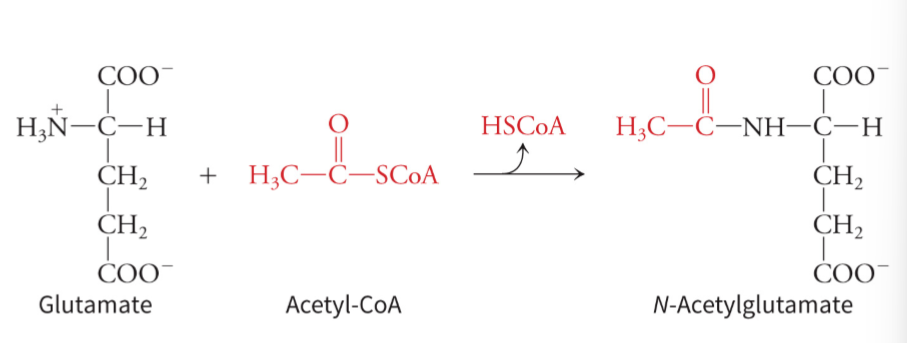
what activates carbamoyl phosphate synthetase
N-acetylglutamate, which is derived from glutamate combining with acetyl-CoA - allosterically activates
what is the first mitochondria reaction in ammonia detoxification
bicarbonate, ammonia, and 2 ATPs are required for carbamoyl phosphate synthetase I to make carbamoyl phosphate - this is the rate-limiting step
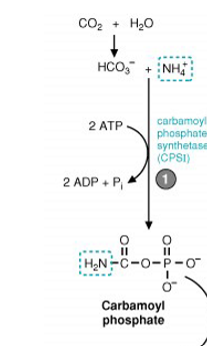
what is the second mitochondria reaction in ammonia detoxification
carbamoyl phosphate combines with ornithine to make citrulline, using ornithine transcarbamoylase - citruline then exits the mitochondria
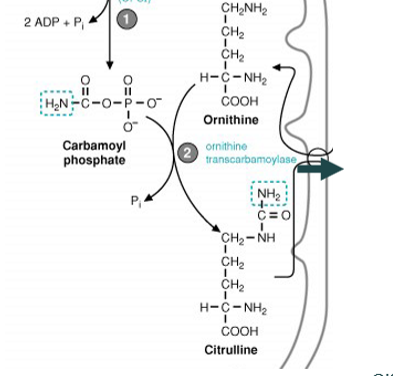
what happens once citrulline exits the mitochondria into the cytosol
it combines with aspartate (generated by the transamination reaction) to form argininosuccinate, which is catalyzed by argininosuccinate synthetase and requires ATP
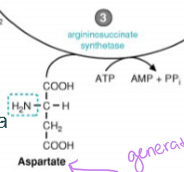
what happens to arginnosuccinate in ammonia detoxification in the cytosol
it gets cleaved by argininosuccinate lyase and makes arginine and fumarate - arginine is then cleaved by arginase and forms urea
where do the 2 nitrogens of urea come from
one comes from ammonia, while the other comes from aspartate
how does nitrogen get removed removed from the body during normal function/not when starvation is happening
normally during nutrient abundance or simply as a course of normal metabolism, we harvest carbon from dietary sources and get rid of nitrogen via uric acid synthesis and secretion - uric acid eventually moves to the urine for excretion
what is used to produce uric acid
purine metabolism (starting with the sugar backbone) which requires synthesis of purine ring, and glutamine is a key nitrogen donor
what is hypoxanthine
an intermediate product to uric acid
what does purine metabolism lead to
hypoxanthine production and the xanthine oxidase reaction - first is purine breakdown, then enzyme conversion, and then excretion
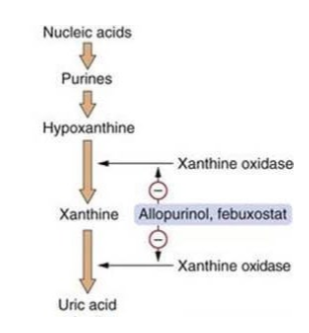
what are purines broken down into
xanthine and hypoxanthine
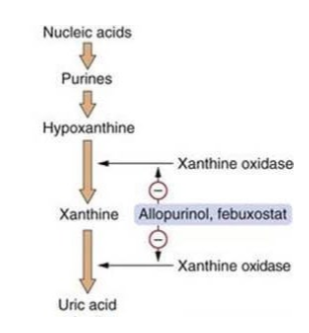
what does xanthine oxidase do
converts xanthine and hypoxanthine into uric acid
what are the intermediate that start synthesis
transamination reactions, arginine (urea cycle), asparagine/glutamine (synthetase reaction), and serine for one-carbon metabolism
what are the 5 starting molecules to build amino acids
alpha-ketoglutarate, OAA/aspartate, pyruvate, 3-phosphoglycerate, and phosphenolpyruvate/erythrose-4-phosphate
what are the amino acids in the alpha-ketoglutarate family
glutamate, glutamine, proline, arginine, and lysin
what are amino acids that are in the OAA/aspartate family
aspartate, asparagine, methionine, theronine, and lysine
what are the amino acids in the pyruvate family
alanine, valine, leucine
what are the amino acids in the 3-phosphoglycerate family
serine, glycine, cesteine
what are the amino acids in the phosphoenolpyruvate/erythrose-4-P family
phenylalanine, tyrosine, and tryptophan
how many amino acids does transamination directly synthesize
3 directly and another 2 amino acids require synthetase reactions - amino group provides the nitrogen
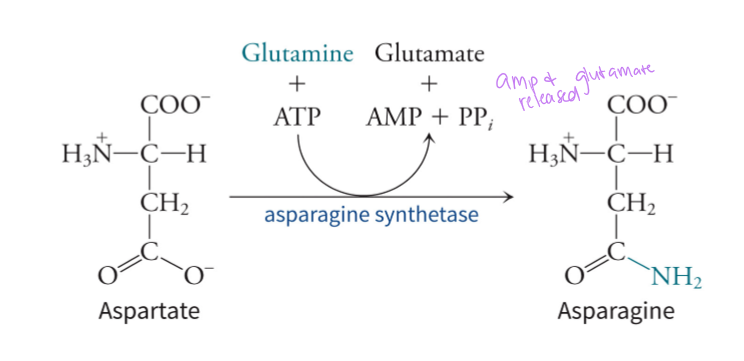
how is aspartate converted into asparagine
by using glutamine as an amino acid and ATP, asparagine synthetase can convert aspartate into asparagine - in the same vein, glutamine synthetase uses ammonia as a donor to synthesize glutamine from glutamate
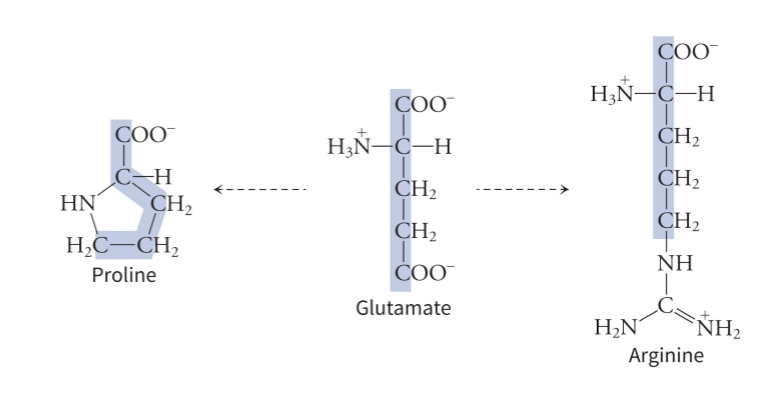
what is the importance of glutamate
is a starting molecule for arginine and proline - has the same 5 carbon chain
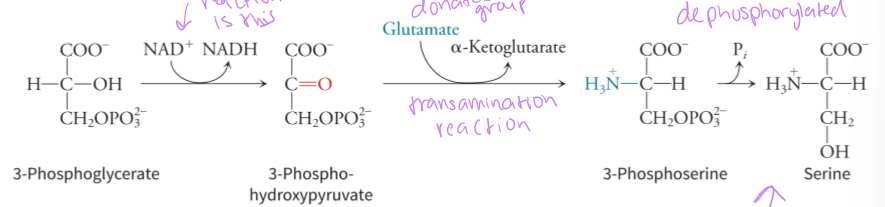
how is serine derived from glutamate
3-phosphglycerate is the starting molecule in the glycolysis pathway, and there are 3 reactions that first oxidation the substrate, then transamination where the amino group is received from glutamate and then a dephosphorylation
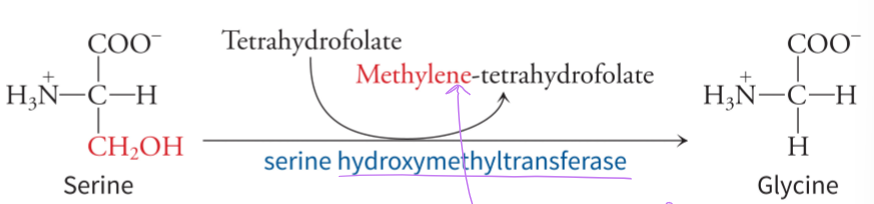
what does serine use to generate glycine
uses one-carbon metabolism and the enzyme serine hydroxymethyltransferase, and the tetrahydrofolate functions as a carrier of one-carbon units in several reactions of amino acid and nucleotide metabolism - serine can give up 1 methyl group to tetrahydrofolate
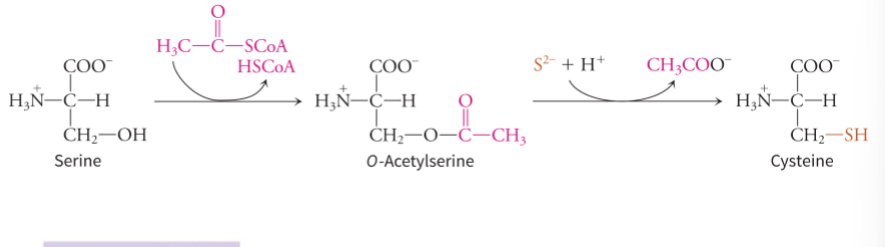
how does serine generate cysteine
cysteine contains a sulfur group, so does a two step enzymatic pathways containing sulfur incorporation
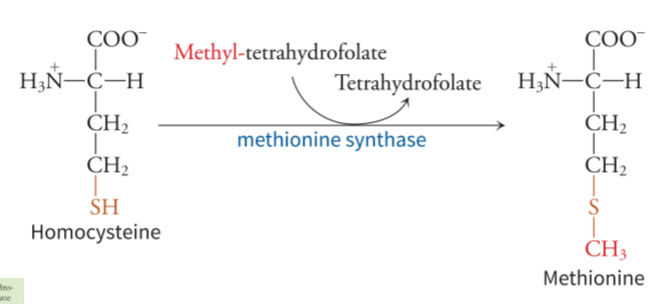
what is the methionine synthase reaction
leads to methionine, and requires cysteine and uses one carbon metabolism - converts homocysteine to methionine using methionine synthase
why are amino acids with aromatic group more difficult to synthesize
phosphoenolpyruvate and erythrose-4-P are the starting molecules, and this requires many steps for full synthesis of these amino acids - very high metabolic cost, and the complexity of the rings requires specialized, multi-step enzymatic pathways
what is one-carbon metabolism
sometimes metabolism needs to move one carbon units around in order to carry out a synthetic or degrading process - this system uses several different one-carbon carriers
what are some of the sources of one-carbon units
serine, glycine, histidine, formaldehyde, and formate