BMB 211 Exam 4
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Where is the oxygen produced during photosynthesis?
In the thylakoid
Where is the chlorophyll located in a plant?
thylakoid membrane
What is the function of Rubisco?
carbon fixation
Atrazine is a chemical that blocks the electron from leaving photosystem II. What would be the immediate effect on the plant?
prevents ATP synthesis
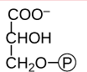
Name this molecule which is the 1st product of photosynthesis.
3-phosphoglycerate
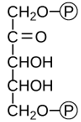
Name the substrate for the 1st step in photosynthesis.
Ribulose-1,5-bisphosphate
For every three molecules of CO2 that enter the cycle, how many molecules of Glyceraldehyde-3-phosphate (G3P) actually exit to be used for sugar synthesis?
1
During the reduction phase, what molecules provided by the light reactions are used to convert 3-PGA into G3P? _______ and _________
NADPH and ATP
Why is the membrane considered 'semi-permeable' rather than 'permeable'?
It regulates the passage based on size, charge and solubility
Which of the following best describes the movement of molecules during facilitated diffusion?
Molecules move down their gradient through a transport protein
If a cell is placed in a hypertonic solution, what will be the net movement of water?
water moves out of the cell causing it to shrivel
Which of the following is an example of exocytosis in a physiological context?
The release of neurotransmitters into a synapse
What happens to the surface area of the plasma membrane during heavy endocytosis?
It decreases as membrane sections are internalized via vesicle formation
How does the cell balance the change in surface area of plasma membrane due to endocytosis?
Cells often use exocytosis to rapidly replenish the membrane surface, maintaining a dynamic equilibrium
In the Na+/K+ pump cycle, what is the primary role of ATP phosphorylation?
To induce conformational change that releases sodium ions to the extracellular space
![<p><span>Name this molecule [1]</span></p>](https://assets.knowt.com/user-attachments/154aae71-1bbd-4765-9d10-14564bd16f51.png)
Name this molecule [1]
3-phosphoglycerate
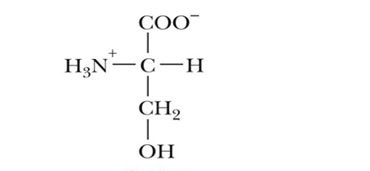
name this molecule
serine
Pyrimidines are [1]-membered rings
6
Purines are 6-membered rings fused to a 5-membered ring. true or false
true
Identify pyrimidines in the list provided:
adenine
guanine
thymine
cytosine
uracil
thymine
cytosine
uracil
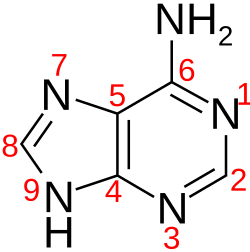
Identify the following nitrogenous base:
adenine
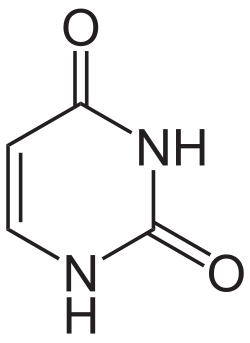
Identify the following nitrogenous base:
uracil
Which of the following is/are differences between DNA and RNA?
A. DNA is single stranded
B. RNA is double stranded
C. RNA contains uracil
D. RNA is single stranded
Both C and D are correct
Both C and D are correct
Which of the following describes the flow of genetic information as stated in the central dogma of molecular biology?
DNA → RNA → Protein
The DNA "backbone" is composed of
A phosphodiester bond between 3! and 5! hydroxyl groups of adjacent deoxyribose residues
There are approximately _________ human genes.
20,000
Which of the following statements describes the eukaryotic chromosome?
It consists of a single linear molecule of double-stranded DNA plus proteins.
Which of the following statements is true of histones?
Histone H1 is not present in the nucleosome bead; instead, it draws the nucleosomes together.
Why do histones bind tightly to DNA?
Histones are positively charged, and DNA is negatively charged.
What is the function of topoisomerase?
relieving strain in the DNA ahead of the replication fork
A genome is
All the DNA in an organism
In nucleosome structure the histone ________ aids in stabilizing the wrapping of DNA around the protein octomer.
H1
Where do the enzymatic reactions of the Calvin cycle take place?
stroma of the chloroplast
What is the primary function of the Calvin cycle?
synthesize simple sugars from carbon dioxide
The reactions that produce molecular oxygen (O2) take place in
the light reactions alone
Which of the events listed below occurs in the light reactions of photosynthesis?
Light is absorbed and funneled to reaction-center chlorophyll a.
Which of the following are directly associated with photosystem I?
receiving electrons from the thylakoid membrane electron transport chain
Which of the following are products of the light reactions of photosynthesis that are utilized in the Calvin cycle?
ATP and NADPH
Water passes quickly through cell membranes because
it moves through aquaporins in the membrane.
What kinds of molecules pass through a cell membrane most easily?
small and hydrophobic
Which of the following factors would tend to increase membrane fluidity?
a greater proportion of unsaturated phospholipids
Which of the following is a characteristic feature of a carrier protein in a plasma membrane?
It exhibits a specificity for a particular type of molecule.
Which of the following is a reasonable explanation for why unsaturated fatty acids help keep any membrane more fluid at lower temperatures?
The double bonds form kinks in the fatty acid tails, preventing adjacent lipids from packing tightly.
Which of the following membrane activities require energy from ATP hydrolysis?
Na+ ions moving out of a mammalian cell bathed in physiological saline
Which of the following statements is correct about diffusion?
It is a passive process in which molecules move from a region of higher concentration to a region of lower concentration.
Which of the following would likely move through the lipid bilayer of a plasma membrane most rapidly?
CO2
Which of these are not embedded / inserted within in the hydrophobic portion of the lipid bilayer?
peripheral proteins
White blood cells engulf bacteria through what process?
endocytosis
Why are lipids and proteins free to move laterally in membranes?
There are only weak hydrophobic interactions in the interior of the membrane.
A membrane's fluidity is largely determined by the percentage of
unsaturated fatty acids
What will happen to the opening of the sodium-potassium pump if no ATP is present in a cell?
It will remain facing the cytoplasm, with sodium ions bound.
The sodium-potassium pump is called an electrogenic pump because it
contributes to the membrane potential.
The double bonds in naturally occurring fatty acids are usually _______ isomers.
Cis
The functions of fat include:
Insulation
Energy storage
Water-repellent properties
All of the above are correct
all the above are correct
Which of the following molecules are lipids?
Fatty acids
Steroids
Waxes
Triacylglyderols
Fatty acids
Steroids
Waxes
Triacylglyderols
Decomposers convert organic nitrogen to ________.
Ammonia
Nitrate
Nitrite
Dinitrogen
All of the above are correct
all the above are correct
The ammonia that is incorporated into urea molecules is produced in reactions catalyzed by all of the following enzymes except:
Urease activity possessed by intestinal bacteria
Serine dehydratase
Glutamate dehydrogenase
Serine hydroxymethyltransferase
L-Amino acid transferase
Serine hydroxymethyltransferase
Amino acids whose degradation yields α-ketoglutarate include all of the following except _________.
Leucine
Amino acids whose degradation yields succinyl-CoA includes all of the following except ________.
Methionine
Isoleucine
Valine
Histidine
None of the above is correct
Histidine
The end product of uracil degradation is ________.
β-Alanine
In general, catabolism of amino acids begins with
removal of the amino group
Urea is synthesized in which organ?
liver
What are the seven metabolic products produced by the degradation of amino acids?
acetoacetyl-CoA
acetyl-CoA
oxaloacetate
fumarate
succinyl-CoA
α-ketoglutarate
Ammonia
Malate
Purine
Phosphoenol pyruvate
acetoacetyl-CoA
acetyl-CoA
oxaloacetate
fumarate
succinyl-CoA
α-ketoglutarate
Ammonia
In humans the purine ring cannot be degraded. How is it excreted?
uric acid
The glutamate family of amino acids includes all of the following except
lysine
The bonds that link the base pairs in the DNA double helix are ________.
hydrogen bonds
Adenine is a purine.
True
False
true
A nucleotide consists of only a base and a sugar.
True
False
false
One of the differences between DNA and RNA is the sugar it contains.
True
False
true
Which of the following is not a component of nucleic acid?
Nucleotides
Glucose
Phosphate group
Purines
Pyrimidines
glucose
Which of the following statements describes the eukaryotic chromosome?
It consists of a linear molecule of double-stranded DNA plus proteins.
Why do histones bind tightly to DNA?
Histones are composed predominantly of positively charged amino acids , and DNA is negatively charged.
Which of the following is/are differences between DNA and RNA
DNA is single stranded
RNA is double stranded
RNA contains uracil
RNA is single stranded
Both D and E are correct
both D and E are correct
The DNA "backbone" is composed of
A phosphodiester bond between 3! and 5! hydroxyl groups of adjacent deoxyribose residues
Nucleosomes are composed of all of the following except ________.
H1
H2
H3
H4
H5
H5
Protein synthesis, using the mRNA template is called _____.
translation
In nucleosome structure the histone ________ aids in stabilizing the wrapping of DNA around the protein octomer.
H1
The melting point of a DNA molecule increases as the GC content increases.
True
False
true
Chargaff's rule apply to DNA but not RNA.
True
False
true
Which of the following sequences of DNA is most likely to form Z-DNA?
5'-ATCTACATCTACATAGATAT-3' |
5'-AAAAAAAAAAAAAAAAA-3'
3'-TTTTTTTTTTTTTTTTTTTT-5'
5'-GCGCGCGCGCGCCGCGCGCG-3'
3'-CGCGCGCGCGCGCGCGCGCG-5'
5'-GGGGGGGGGGGGGGGGGGGG-3'
3'-CCCCCCCCCCCCCCCCCCCCCC-5'
5'-GCGCGCGCGCGCCGCGCGCG-3'
3'-CGCGCGCGCGCGCGCGCGCG-5'
Topoisomerases are associated with
supercoiling of DNA
The Tm for melting the double helix is:
The midpoint of the range over which the helix denatures.
The hydrogen bonding interactions in a Watson-Crick AT base-pair involve what units in the adenine base?
N-1 and the amino group on C-6