chemistry ch 7
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
small particles such as atoms, molecules, and ions are counted using this
mole (avogradros number)

what is avogadros number?
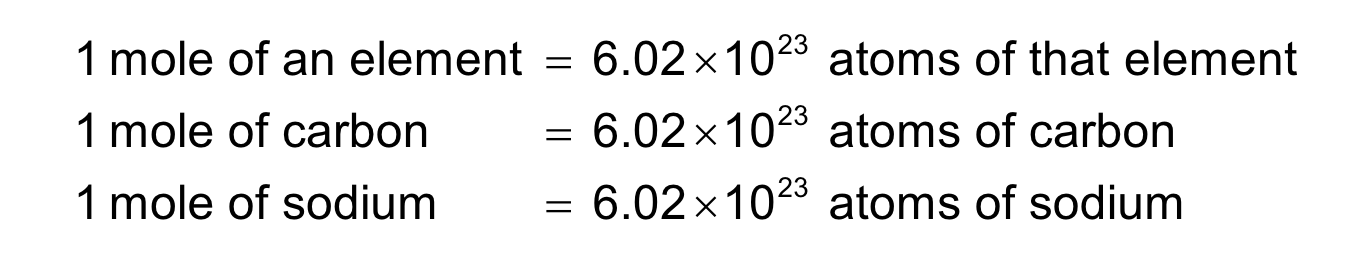
1 mole of any element =
avogadros number
what is used to convert moles to molecules
• combination reactions
• decomposition reactions
• single replacement reactions
• double replacement reactions
• combustion reactions
types of chemical reactions
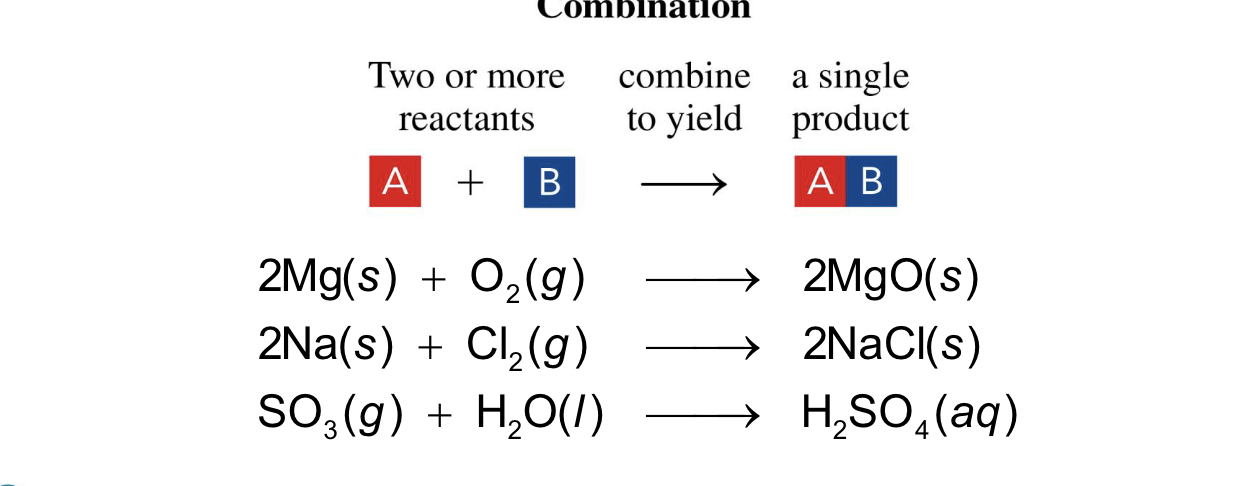
two or more elements form one product
combination reaction
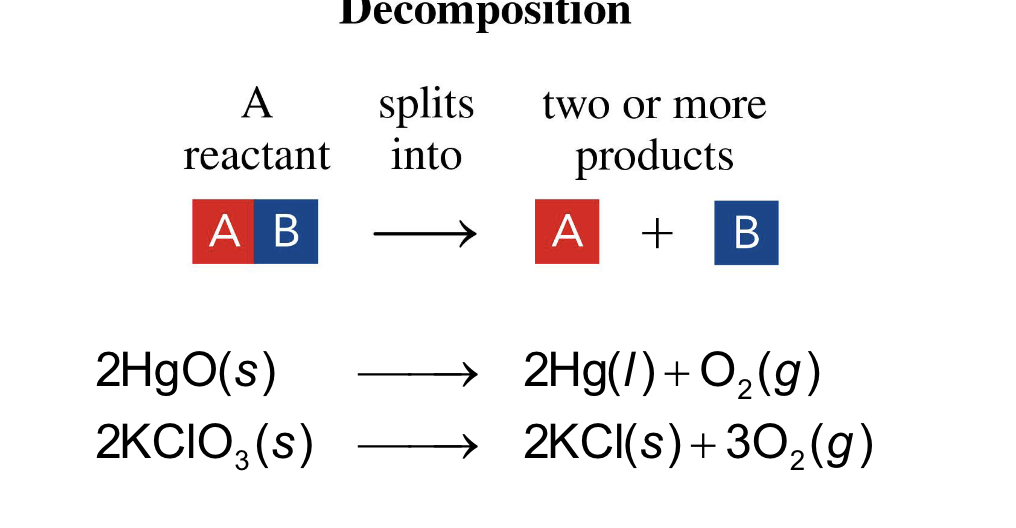
one substance splits into two or more simpler substances
decomposition reaction
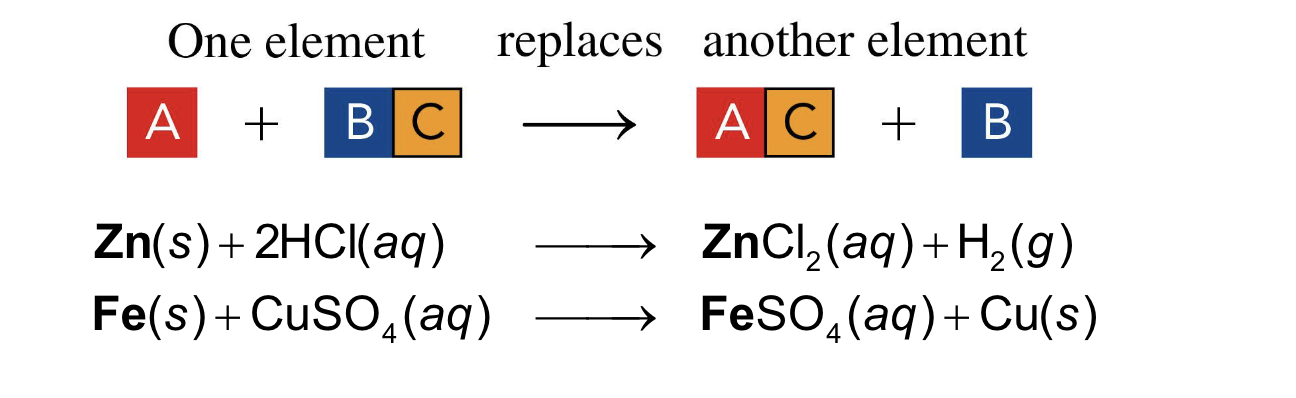
one element takes the place of a different element in another reacting compound
single replacement
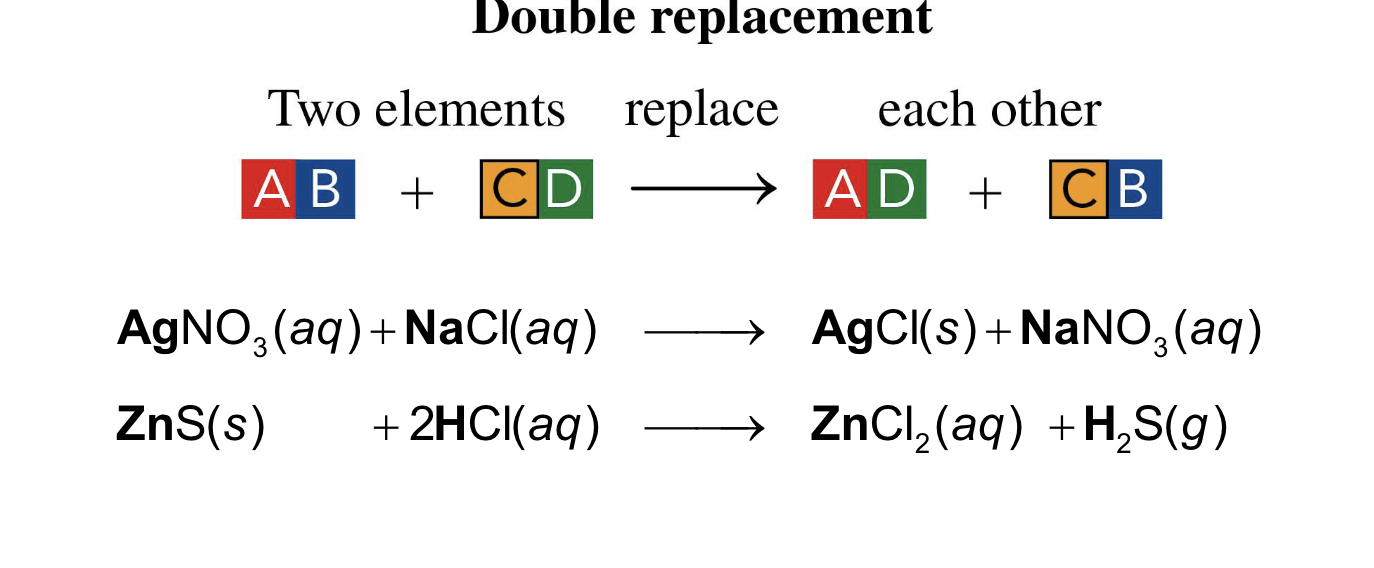
positive ions in the reactant compounds switch places.
double replacement
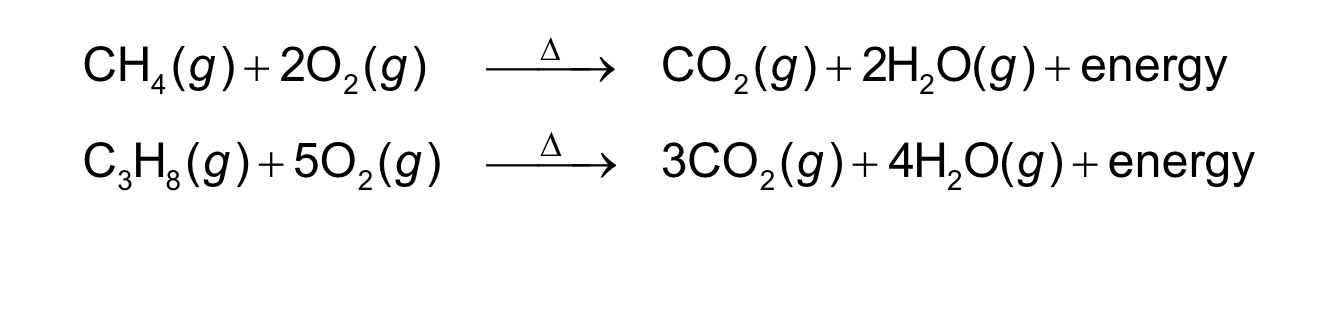
a carbon-containing compound burns in oxygen gas to
form carbon dioxide2(CO ) and water2(H O)
• energy is released as a product in the form of heat
combustion reaction
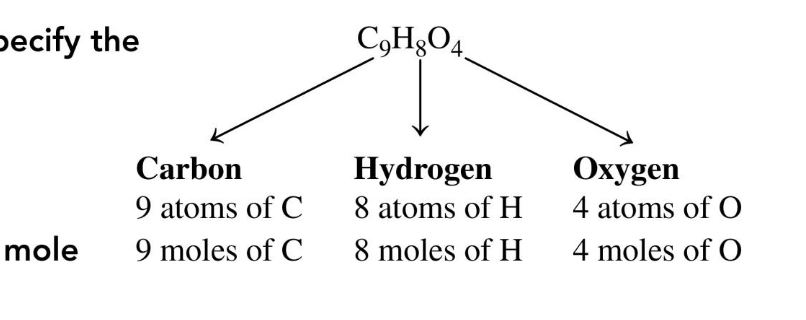
the moles/atoms of each element in 1 mole of compound
the subscripts in a chemical formula show…
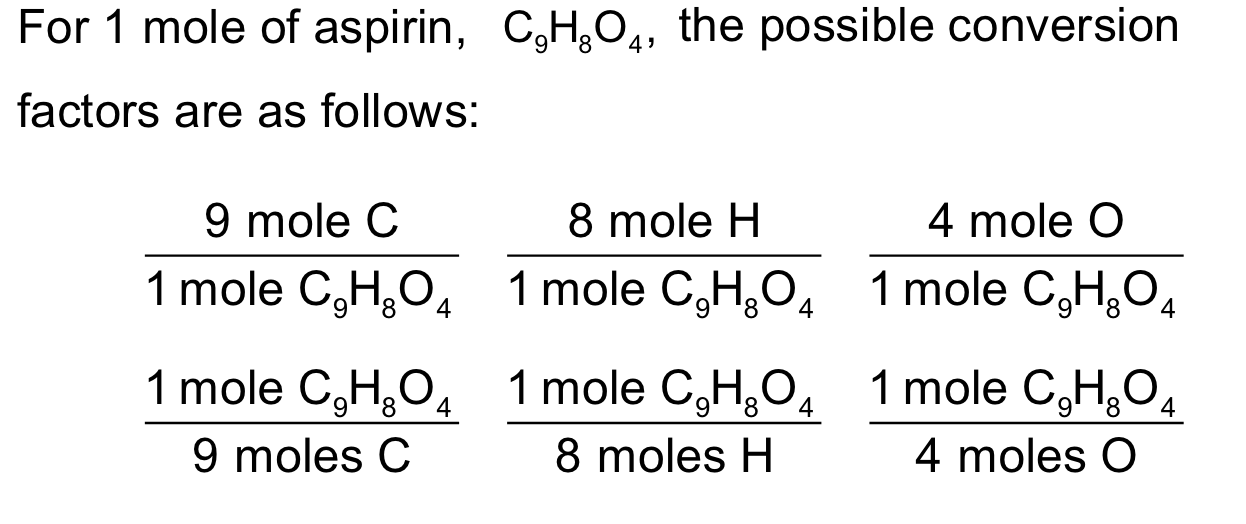
conversion factors
subscripts can be use to make…
loss of electrons (or H)
oxidation
The Gain of electrons (or the gain of Hydrogen)
reduction
The minimum amount of energy required to "jumpstart" a reaction
lower required energy of activation
activation energy
and catalysts
mass of 1 mole of an element
atomic mass expressed in grams
1 mole of C atoms= 12.01 g C atoms —> 1 mole C atoms/12.01 g C atoms
molar mass
molar mass conversion
what can be used to go from moles to grams or grams to moles
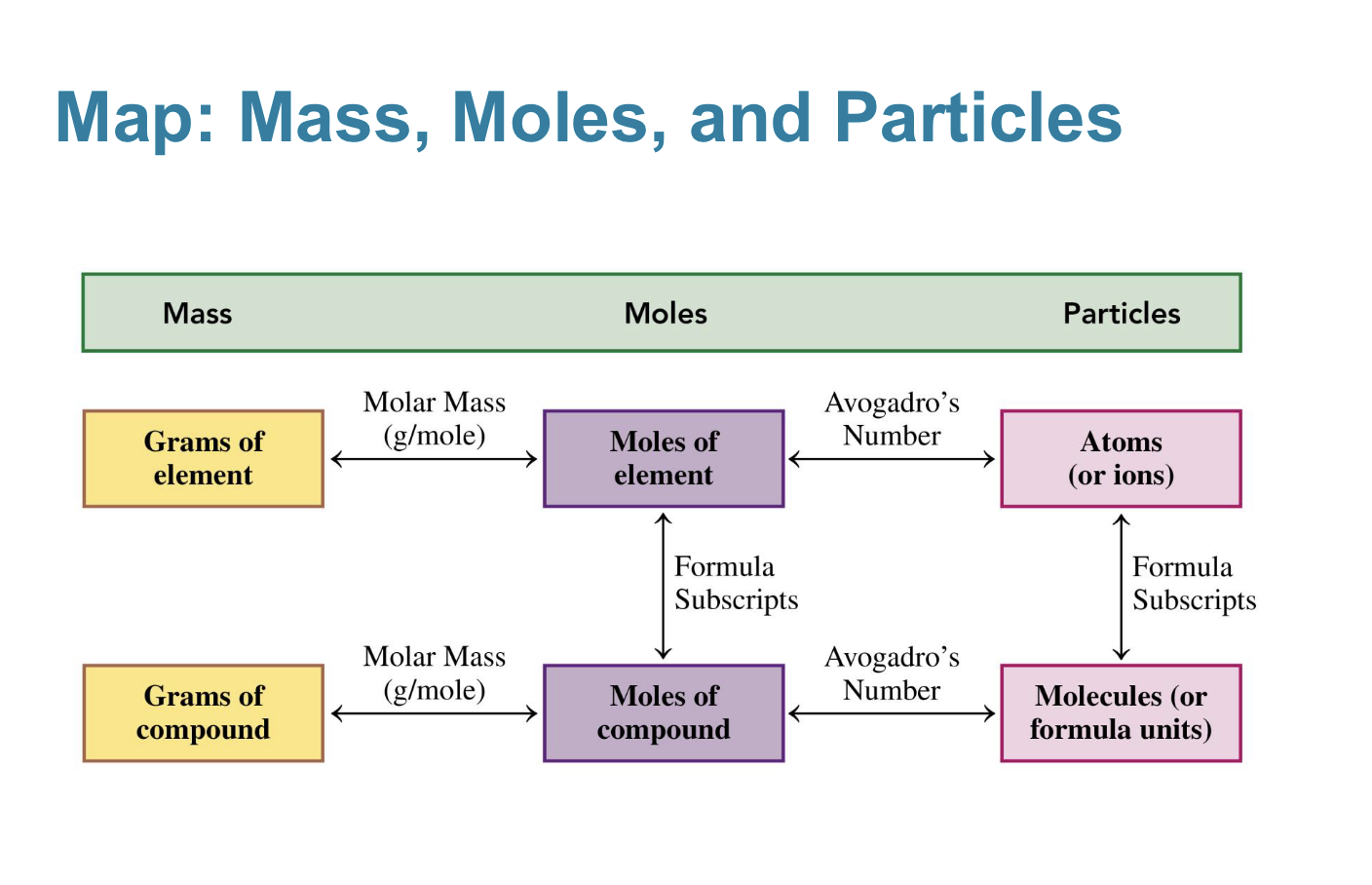
1) how to get from grams to moles
2) how to get from moles to atoms
3) how to get from atoms to molecules
1) heat
2) liquid
3) solid
4) dissolce in water
5) gas
what does a - indicate in chemical reactions
1) delta
2) l
3) s
4) aq
5) g
no atoms lost or gained
in a balanced chemical equation…
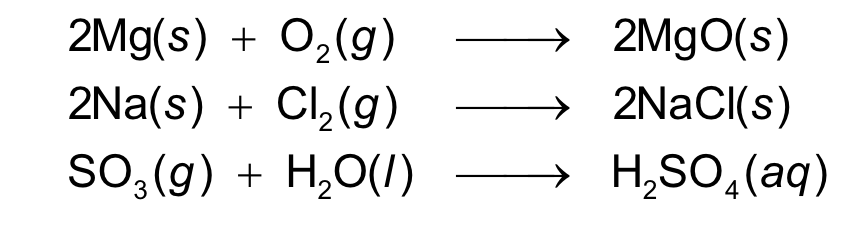
two or more elements form one product
combination reaction
one substance splits into two
or more simpler substances
decomposition reaction
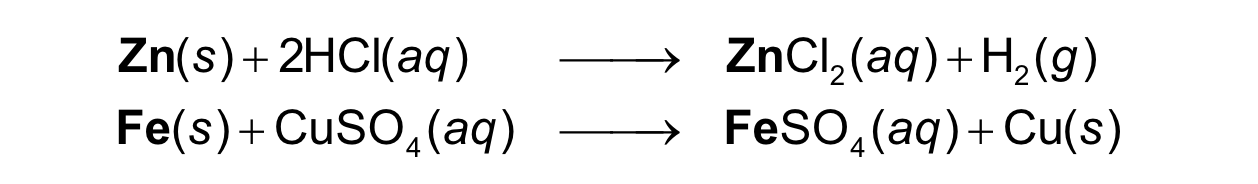
In a single replacement reaction, one element takes the
place of a different element in another reacting compound
A+ BC —> AC+ B
single replacement reaction
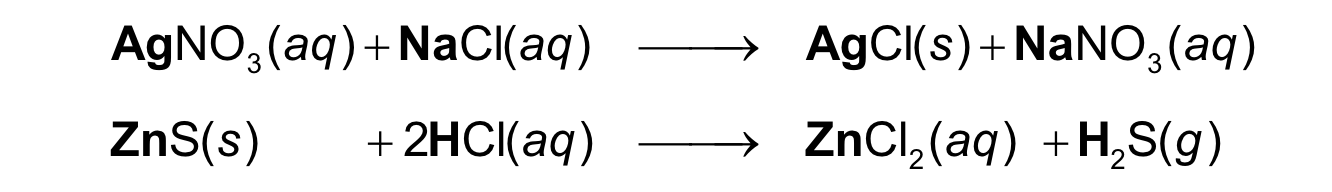
In a double replacement, the positive ions in the reactant
compounds switch places
double replacement reaction
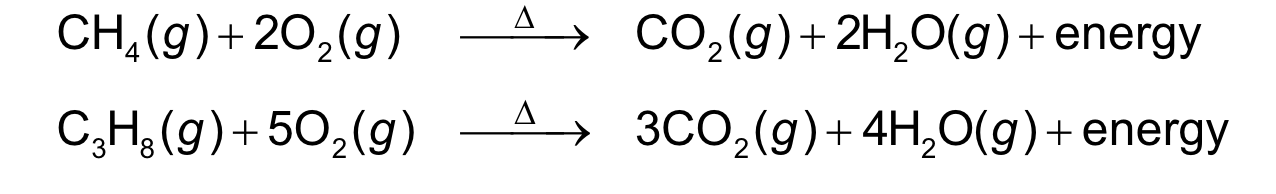
Co2 and H2O are always products
energy released in form of heat
combustion reaction
mass of products
mass of reactants =
The rate of the forward reaction equals the rate of the reverse reaction.
What does it mean for a reaction to be at Equilibrium?

Le chateliers principal
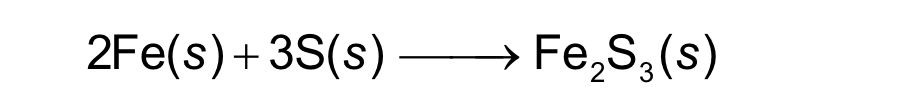
2 moles of Fe + 3 moles S = 1 mole Fe2S3
how can you read this
convert the mass of one substance to another substance
given a balanced chemical equation we can …

1) conver mass of substance A to moles using molar mass of A
2) converting moles of substance A to substance B (using mole ratio of B to A)
3) convert moles of substance b into grams (molar mass)
how do you use a balanced chemical equation to go from mass a to mass b
1) collision- reactants must collide
2) orientation- reactants must align properly to break and form bonds
3) energy - collision must provide the energy of activation
What are the three conditions that must be present for a reaction to occur?
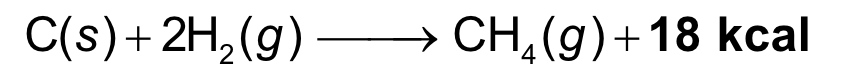
heat is released
the energy of the products is less than the energy of the reactants
heat= product
exothermic reactions

heat= absorbed
energy of the products is greater than energy of reactants
heat is a reactant (added)
endothermic reactions
speed at which reactant is used up
speed at which product forms
increases when temperature rises —> reacting molecules move faster—> more collisions between molecules with energy of activation
increases w conentration of reactants
reaction rate
increase rate of reaction
lowers energy of activation
not used up during reaction
catalyst
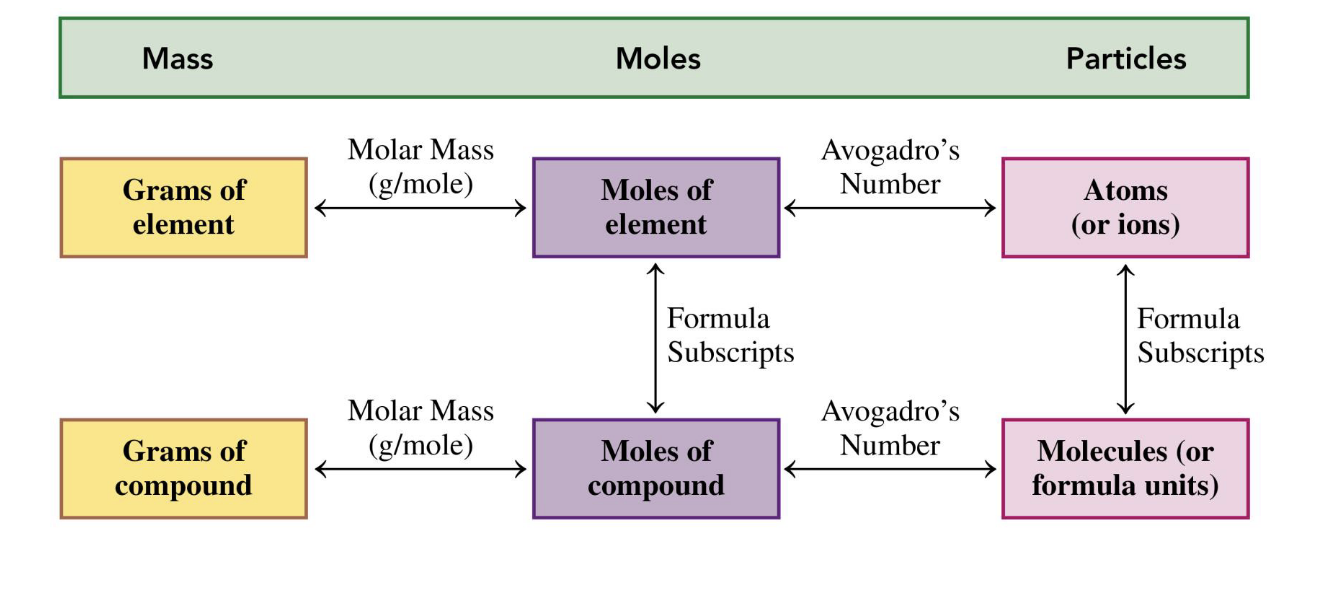
amount of atoms
charges
in a formula, the subscript descibes … and the high script describes…
1) brackets
2) add charge
when drawing a lewis structure for a polyatomic ion dont forget ..
dipoles
Ionic and nonpolar covalent bonds will not have..
"Like Dissolves Like"
Polar solvents dissolve polar or ionic solutes.
Nonpolar solvents dissolve nonpolar solutes.