Biochemistry Exam 4- Portage
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
B.
which of the following monosaccharides is not an aldose?
a. erythrose
b. dihydroxyacetone
c. glucose
d. glyceraldehyde
e. ribose
a.
Which of the following monosaccharides is a ketose?
a. dihydroxyacetone
b. galactose
c. glucose
d. glyceraldehyde
e. dose
d
When two carbohydrates are epimers:
A. One is a pyranose, the other a furanose
B.One is an aldose, the other a ketose
C. They differ in length by one carbon
D. They differ only in the configuration about one carbon atom E. They react differently with iodine
C.
Lactose is made from linking galactose and glucose. When this happens, _____
A. .Gas is eliminated
B. An anomeric carbon is formed on carbon-1
C. Water is released
D. An epimer is formed
E. An aldose is formed
E.
which pair is anomeric?
a. D-glucose and D-fructose
b. D-glucose and L-fructose
c. D-glucose and L-glucose
d. alpha-D-glucose and beta-D-fructose
e. alpha-D-glucose and beta-D-glucose
A.
Amylose and Amylopectin are both polymers of:
A. alpha-d-glucose
B. beta-d-glucose
C. Galactose
D. Idose
E. Maltose
A.
Which statement about starch and glycogen is false?
A) Both have β-D-glucose monomers
B) Both starch and glycogen are energy storage polysaccharides
C) Both are homopolymers of glucose
D) Glycogen is more extensively branched than starch
E) Both have O-glycosidic linkages
E.
lactose is made from linking galactose and glucose. when this happens:
a. a polysaccharide is formed
b. gas is eliminated
c. an epimer is formed
d. an anomeric carbon is formed on carbon-1
e. lactose forms by a condensation reaction
B.
Which monosaccharide is not a six-carbon monosaccharide?
A. Fructose
B. Ribose
C. Mannose
D. Galactose
E. Glucose
c.
chitin and cellulose are both polymers of:
a. alpha-D-glucose
b. galactose
c. beta-D-glucose
d. idose
e. maltose
C.
Which statement about cellulose is true?
A. Cellulose is the primary storage polysaccharide of animal cells
B. Cellulose is a highly branched polysaccharide
C. Cellulose is a homopolysaccharide
D. The glucose residues of cellulose contain the same configuration as the glucose residues of amylose
E. Cellulose is an anomer of chitin
D.
The polysaccharide cellulose is most chemically similar to:
A. Glycogen
B. Dextrose
C. Glycogen
D. Chitin
E. Starch
B.
which statement about starch and glycogen is false?
a. amylose is unbranched; amylopectin and glycogen contain many branches
b. both are heteropolymers of glucose
c. both have many -OH groups in their structures
d. both starch and glycogen are energy storage polysaccharides
e. glycogen is more extensively branched than starch
D.
which monosaccharide is not a 6-carbon monosaccharide?
a. idose
b. glucose
c. galactose
d. dihydroxyacetone
e. allose
The anomeric carbon is the reduced, or reacted, carbon ring structure. The only difference in these structures is the position of the –OH group on the anomeric carbon. The beta form has the –OH in the “up” position, while the –OH is “down” in the alpha form.
what is an anomeric carbon?
E.
Which of the following is the most common monosaccharide on earth?
A. Ribose
B. Cellulose
C. Chitin
D. Sucrose
E. Glucose
A.
which statement about cellulose is true?
a. cellulose is found in plants
b. cellulose in the primary storage polysaccharide of animal cells
c. cellulose is found in DNA
d. the glucose residues of cellulose contain the same configuration as the glucose residues of amylose
e. cellulose is an epimer of chitin
B.
the "D" in D-carbohydrates stands for:
a. 3-dimensional
b. dextrorotatory
c. derivative
d. design
e. double
B.
Which compound does not fit the formal definition of a carbohydrate?
A. C6H12O6
B. C7H10O7
C. C8H16O8
D. C3H6O3
E. C4H8O4
A.
Table sugar is also known as:
A. Sucrose
B. Dextrose
C. Glucose
D. Galactose
E. Glycogen
D.
Amylopectin has α1 → 6 branching about every________ residues.
A. 1 to 10
B. 8 to 12
C. 10 to 30
D. 24 to 30
E. No branching
alpha = -OH below the anomeric carbon
beta = -OH above the anomeric carbon
D = -OH on right of penultimate (second to last) carbon
L= -OH on left of penultimate (second to last) carbon
what is the difference between alpha/beta and D/L configurations?
B.
What element besides C, H, and O is found in the chitin?
A. Br
B. N
C. P
D. S
E. Cl
E.
Some carbohydrates form a(n)___________ structure, which resembles a structure in proteins.
A) α form
B) β-turn
C) βeta-sheet
D) D-configuration
E) α-helix
D.
sucrose is also known as:
a. lactose
b. milk sugar
c. chitin
d. table sugar
e. glycogen
c.
amylose folds into which of the following structures?
a. beta sheet
b. beta turn
c. alpha helix
d. D-configuration
e. alpha form
B.
the equilibrium between ___ structures is called mutarotation.
a. anomer and epimer
b. alpha and beta
c. L and D
d. helix and beta sheet
e. none of the above
c.
consider the functions of carbohydrates. which of the following is not a function of these molecules?
a. structure
b. energy storage
c. enzyme structure
d. both A and B
e. both A and C
A.
The word saccharide means ___________.
A. Sugar
B. Carbohydrate
C. Energy molecule
D. Sucrose
E. Starch
Surcrose contains -OH groups which can form hydrogen bonds with H2O. these non-covalent interactions allow solubility by permitting carbs to interact with H2O and become an aqueous solution.
Table sugar dissolves easily in water. Given its structure, explain why this is.
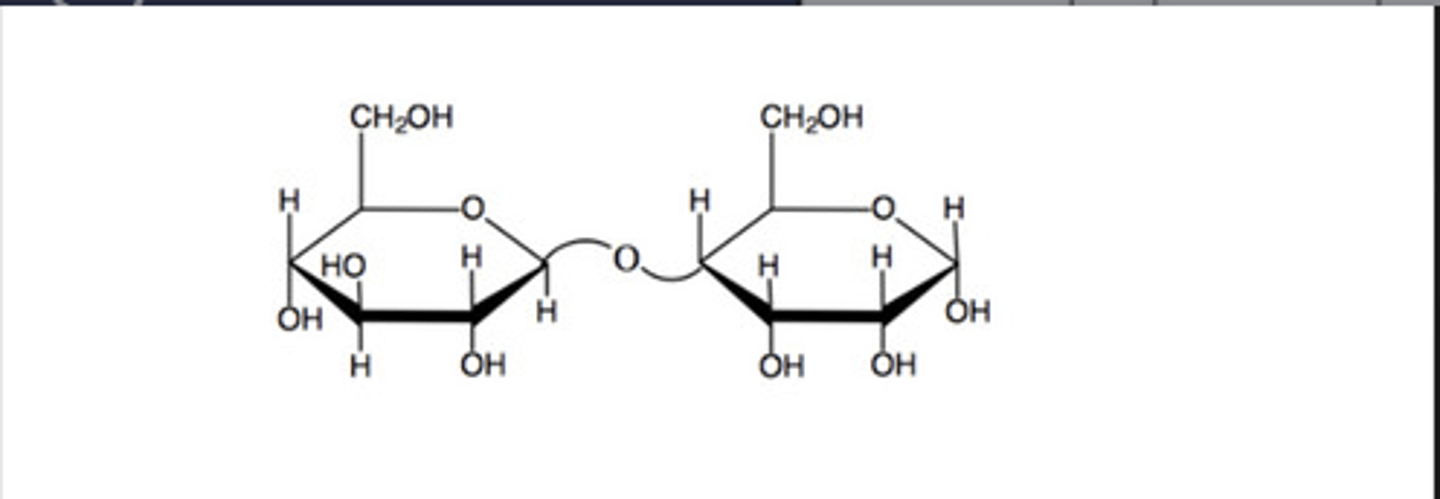
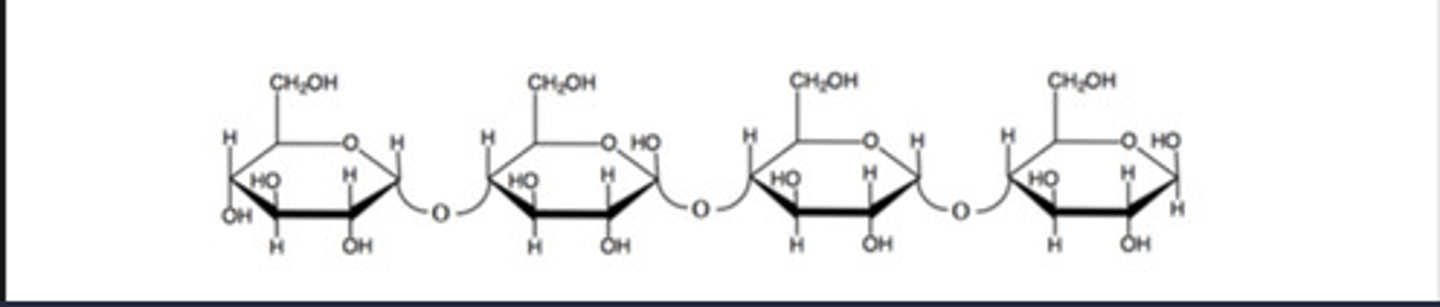
a.) 2 pyranoses and 0 furanoses
b.) beta 1 to 4
c.) yes because it has a free anomeric carbon
A) In the above structure, how many monosaccharide units are furanoses and how many are pyranoses?
B) What is the linkage between the monosaccharides?
C) Is this a reducing sugar?
The exoskeleton of beetles are composed of chitin. Chitin is made of glucose residues with beta 1-->4 linkage, however, human enzymes cannot breakdown this type of linkage. Human enzymes are able to breakdown alpha 1-->4 linkages only. Therefore, the hiker will not intake glucose or get nutrition from the beetle and should not eat it.
A hiker is lost in the wilderness and without food. He runs across several beetles with hard chitin exoskeletons. Would he get nutrition in the form of glucose from eating these beetles? Explain.
Amylose is a linear chain of glucose joined together in alpha 1-->4 bonds in one continuous chain with no branching. These chains have a few thousand to a million glucose molecules bonded together and are referred to as the main chain.
Like amylose, amylopectin has a long chain of glucose bonded together, but it also has branches every 24 to 30 glucose molecules apart from the main chain.
What is the difference between amylose and amylopectin?
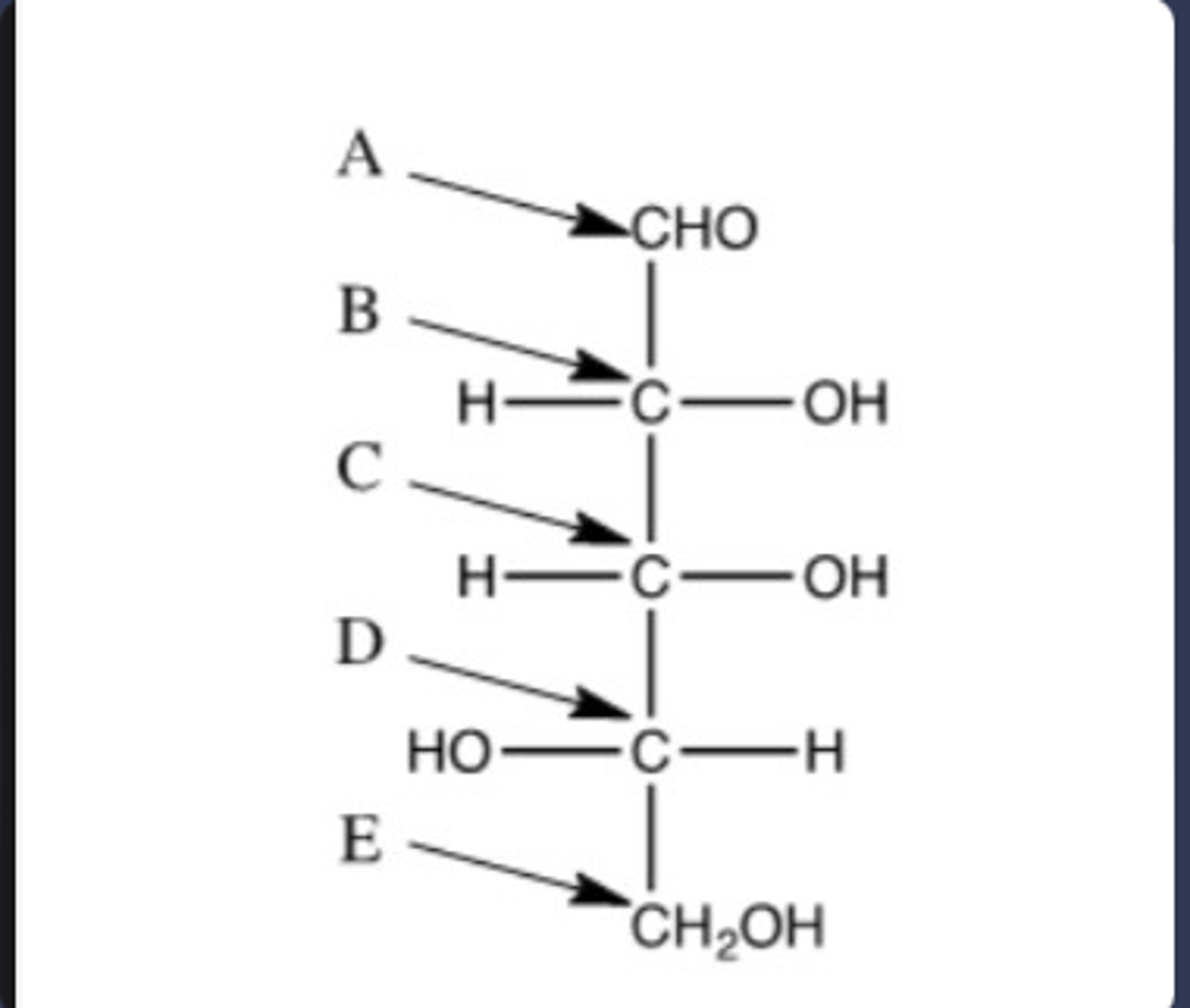
A) Carbon D, the penultimate carbon, determines if the molecules is D or L. In the case of glucose, this would be carbon-5.
B) The monosaccharide is L because the -OH group is on the left side.
A) Which carbon in the following molecule determines if the molecule is D or L? Indicate the letter corresponding the correct carbon ("A","B", etc.) and explain.
B) Is this molecule D or L?
Humans cannot use cellulose as a nutrient because human enzymes cannot breakdown the bonds associated with it. Cellulose contains a beta 1-->4 linkage, and human enzymes can only split alpha 1-->4 linkages. Because cellulose linkages cannot be broken down by the human body, it cannot be used as a nutrient.
Explain in molecular terms why humans cannot use cellulose as a nutrient.
Oligosaccharides and polysaccharides are both composed of monosaccharides, which are the functional unit of carbohydrates. However, oligosaccharides consist of shorts chains of monosaccharides joined together, and polysaccharides consists of long chains of 20 or more monosaccharides joined together. Polysaccharides form linear structures as well as highly branched structures.
How are oligosaccharides and polysaccharides similar in their structure? How are they different?
the penultimate carbon
on a Fischer projection, what is the carbon that is the second furthest away from the carbonyl group is termed?
Epimers are monosaccharides that differ in the orientation at only one carbon position.
Anomers are the alpha and beta forms that differ only in the -OH orientation about the anomeric carbon that forms the hemiacetal group.
what is the difference between epimer and anomer?
2D structures that represent 3D carbohydrates or other organic molecules
what are Fischer projections?
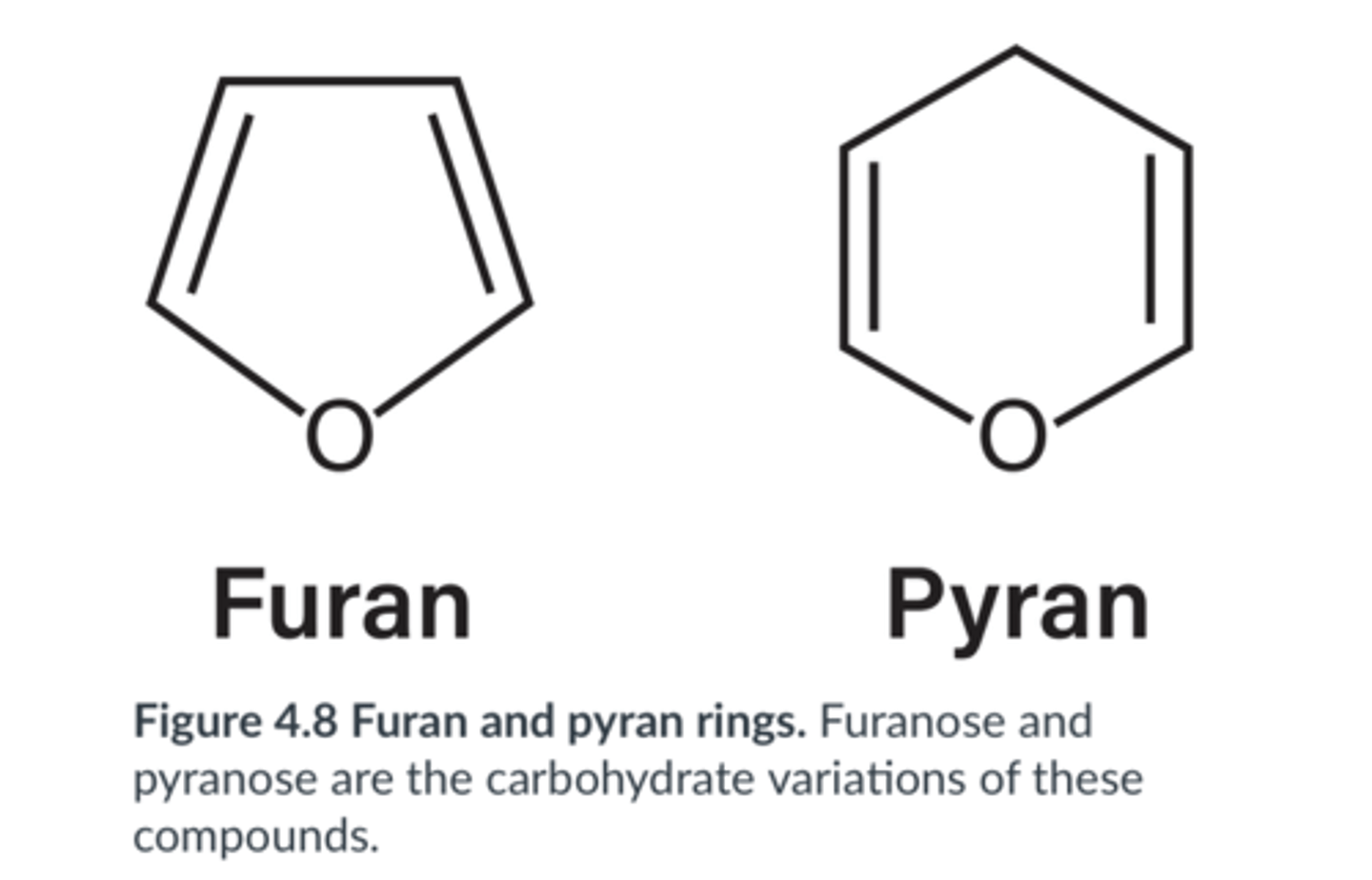
A furanose ring is a 5-atom structure consisting of 4 carbons and 1 oxygen.
A pyranose ring is a 6-atom structure consisting of 5 carbons and 1 oxygen.
furanose vs. pyranose
starch, glycogen, and cellulose
3 homopolysaccharides of glucose
a free anomeric carbon within a hemiacetal group due to its lack of an o-glycosidic linkage
can undergo chemical changes easily because the carbon is not connected to another monosaccharide
what is a reducing end?
A nonreducing end of a sugar is one that contains an acetal group
what is a non-reducing end?
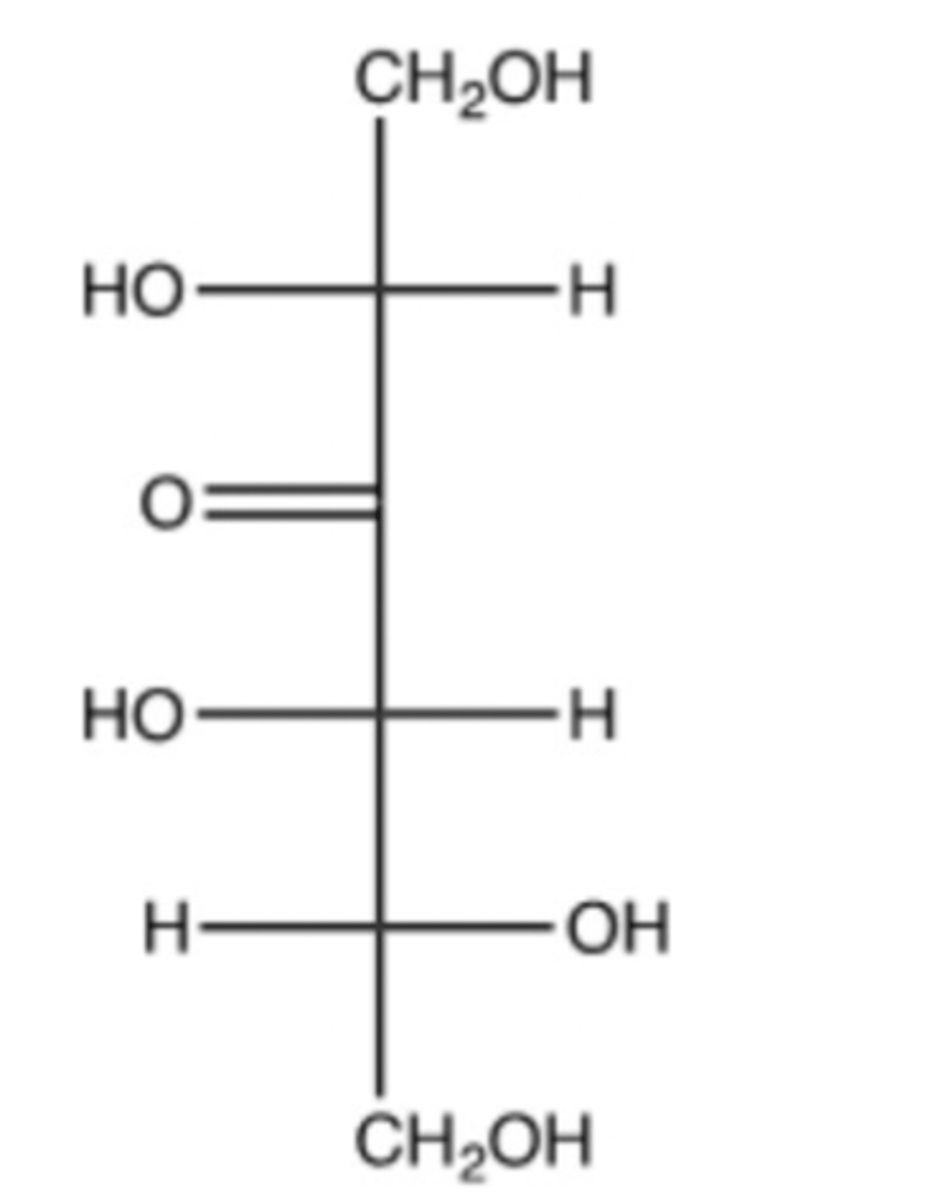
Ketose
There is a C - O double bond on the third carbon from the top
Is the following molecule an aldose or ketose? Briefly explain.
On carbon number 3, which is a penultimate carbon
Which carbon in erythrose would tell you if the monosaccharide is D or L? Give the carbon's number in your answer.
Galactose.
Glucose and mannose are epimers. Name another epimer of glucose besides mannose.
L. The –OH group is on the left side on the penultimate carbon
Is the following structure a D or L carbohydrate? Briefly explain.
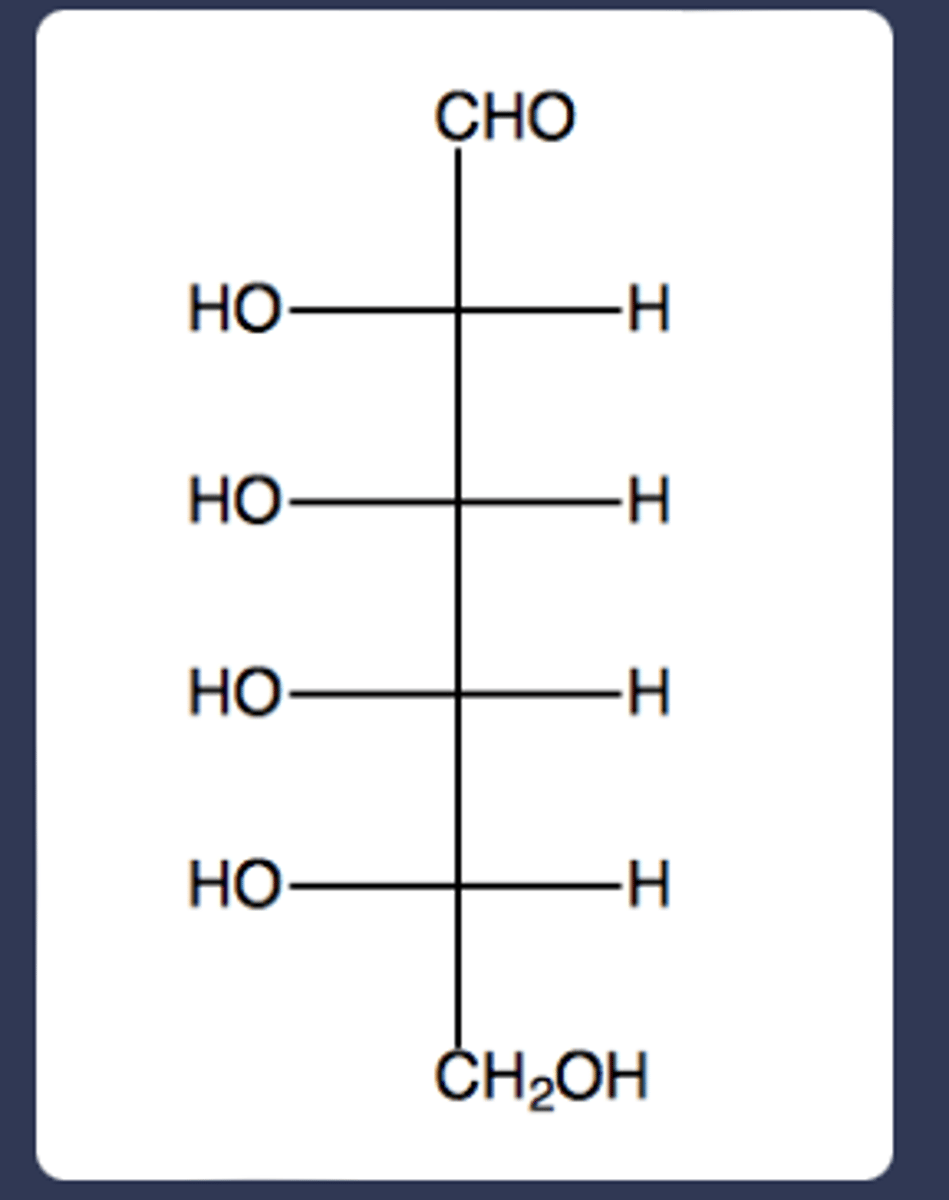
No. This compound has does not fit the formula CnH2nOn as indicated in the module. There is one additional oxygen in this compound.
Does the compound C7H14O8 fit the formal definition of a carbohydrate? Explain.
The α 1→6 bond is found in polysaccharides when branching occurs.
When is the α 1→6 connection typically found in carbohydrates?
Similarities: Both of are homopolysaccharides of glucose. They each have α 1 → 4 connections between glucose. They each are part of starch.
Differences: Amylopectin has branching with α 1→6 linkages. Amylose does not. Amylopectin is typically larger than amylose.
What are the similarities and differences between amylose and amylopectin? Explain in at least three complete sentences.
Chitin has glucose with derivatives attached. -NH-CO-CH3 is connected to each glucose.
How is chitin different from cellulose?
C.
The two monosaccharides shown below could best be described as:
A. Anomers
B. Ketoses
C. Epimers
D. Rotamers
E. Pentoses
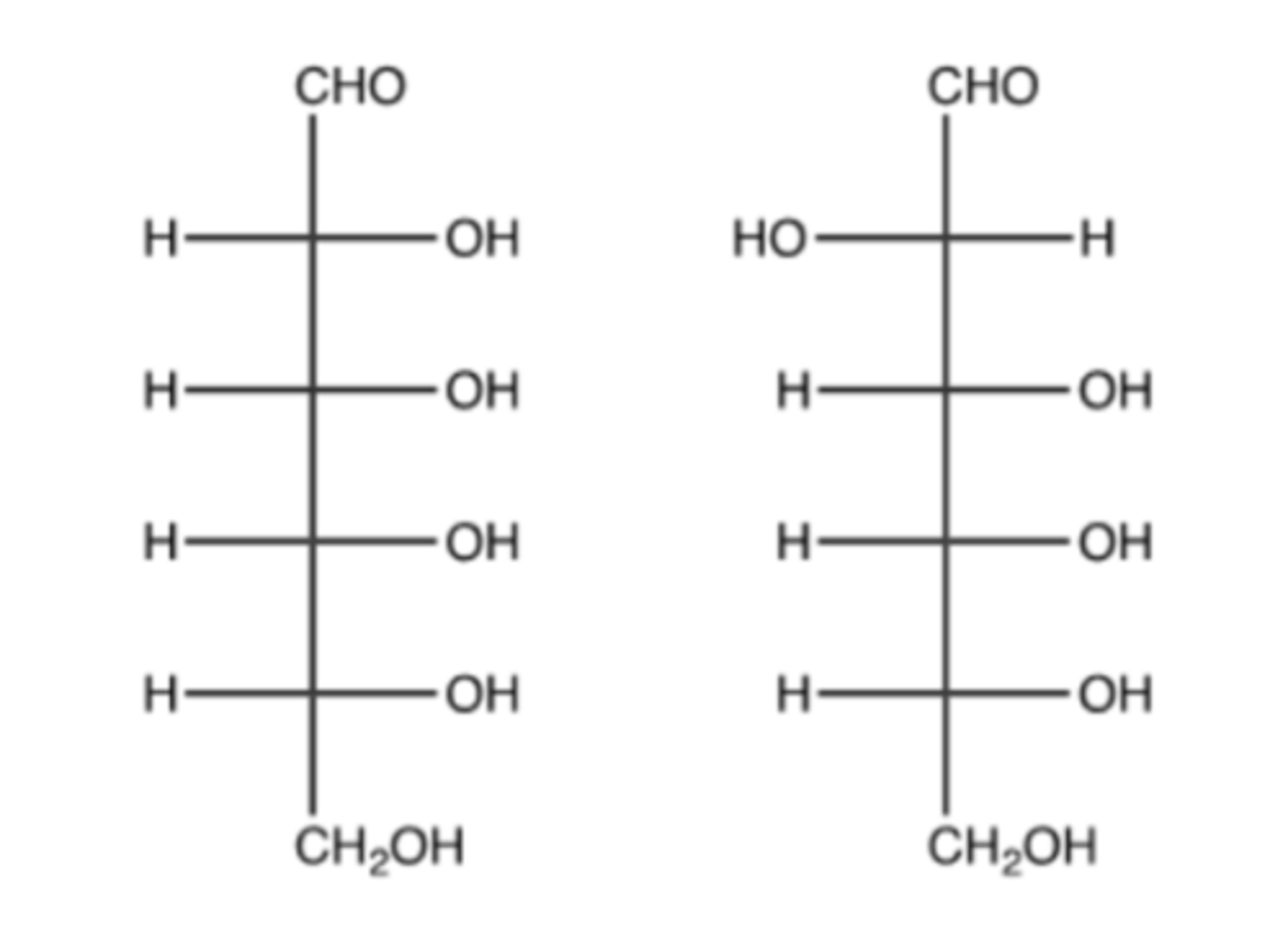
A.
Which arrow correctly identifies the atom that will become the anomeric carbon?
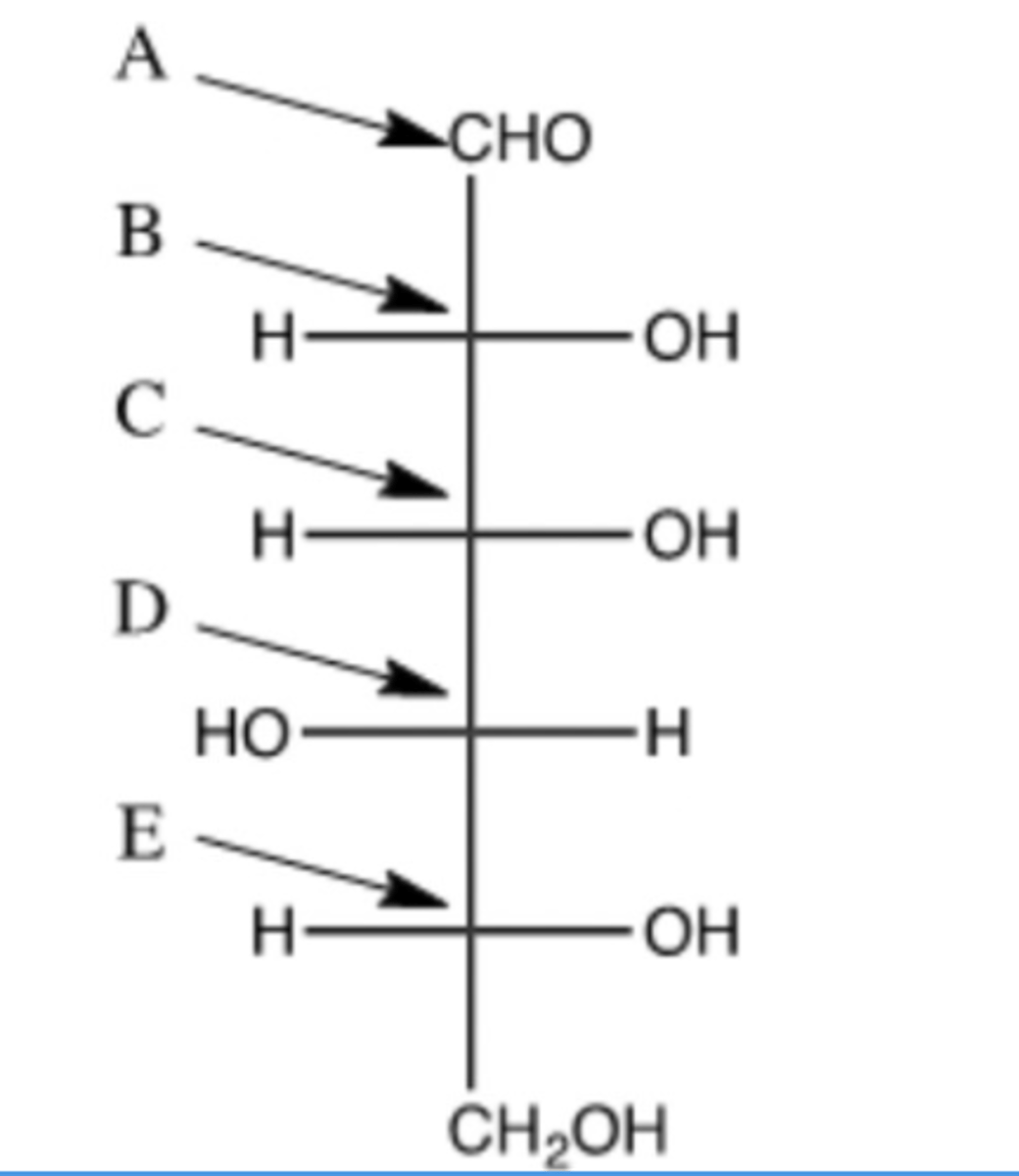
B.
Which of the following monosaccharides is not an aldose?
A) erythrose
B) fructose
C) glucose
D) glyceraldehyde
E) ribose
C
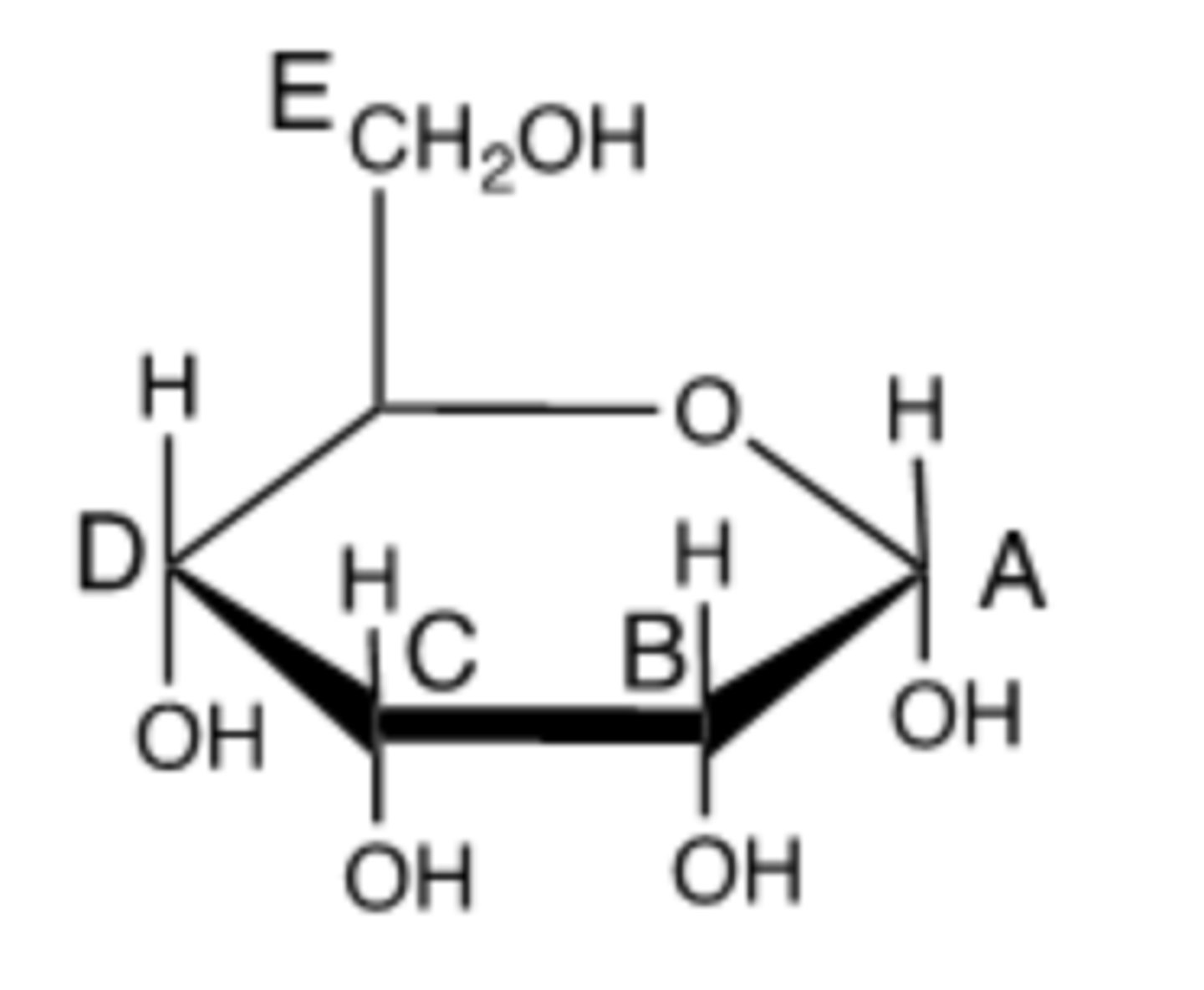
Which of the following is carbon number 3? (Indicate by the appropriate letter.)
A
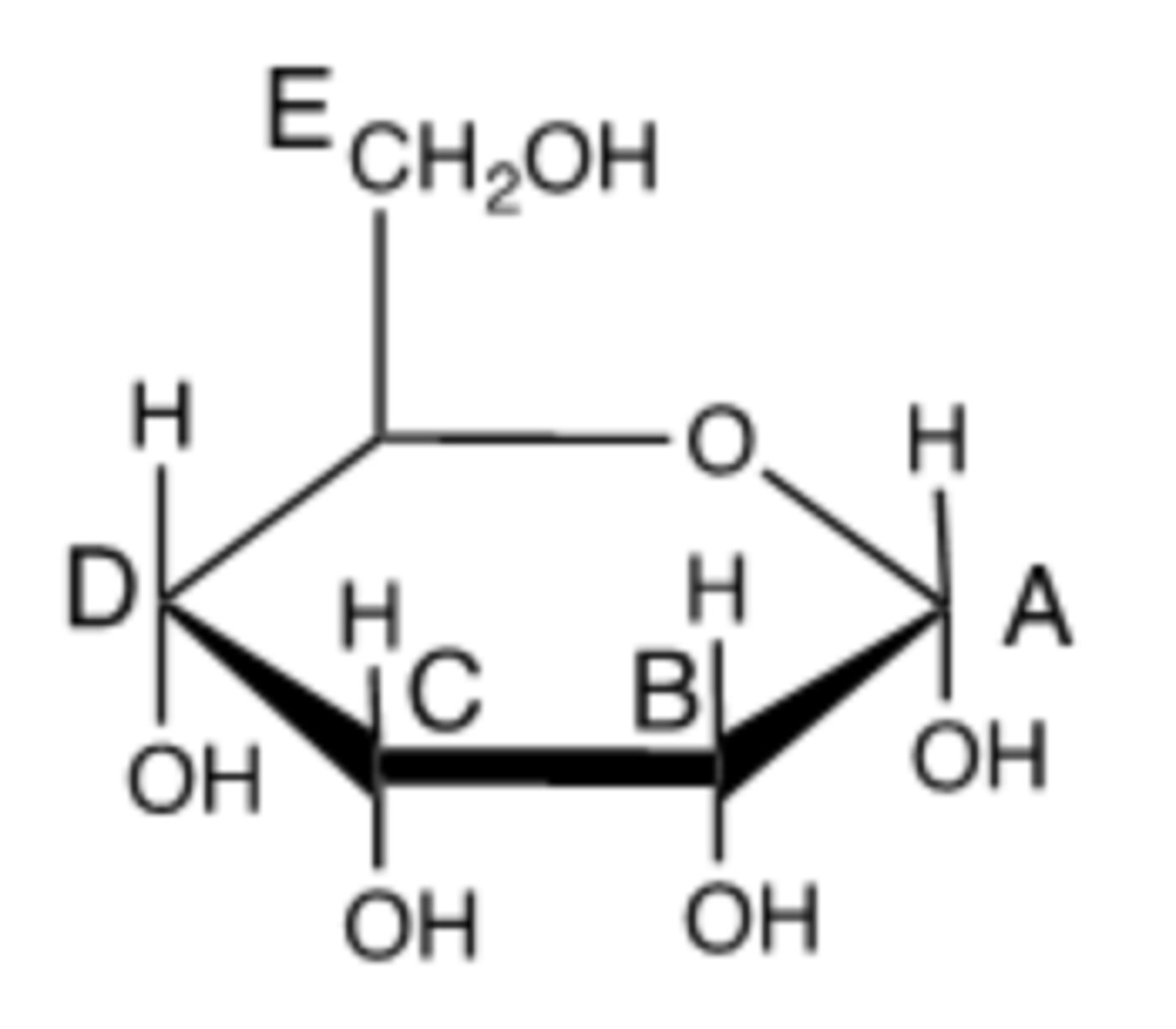
Which carbon in this molecule is the anomeric carbon? (Indicate by the appropriate letter.)
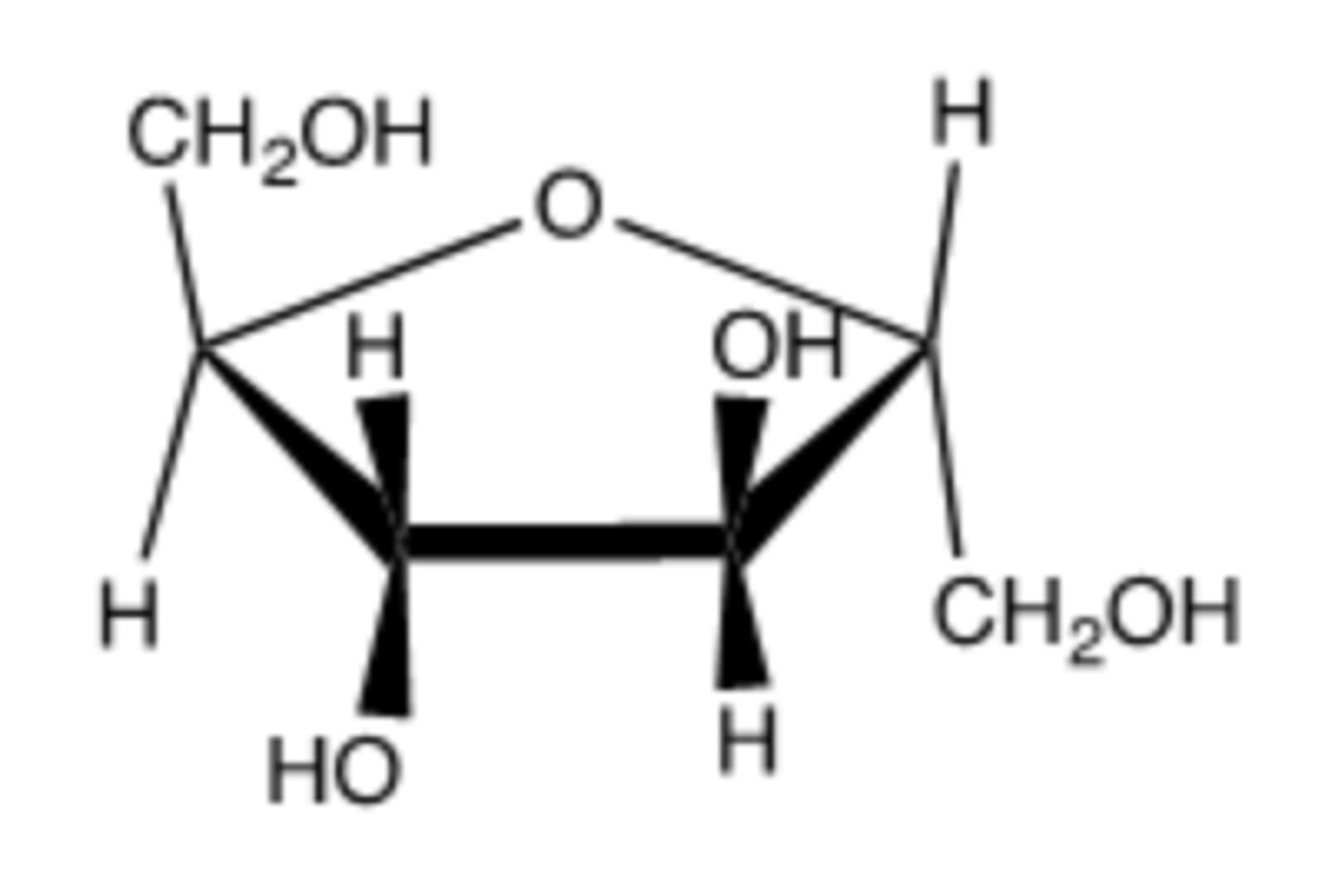
furan. There are only five atoms in the ring: one oxygen and four carbons.
Is the following a furan or pyran ring? Briefly explain.
They can both form alpha helices
In what ways are amylose and a protein alike?
chitin
What is the name of the carbohydrate found in beetle exoskeleton?
Yes.
Is D-glyceraldehyde an aldose?
D
The following monosaccharide are covalently bonded. What type of bond unties them
a. Alpha1->4
b. Alpha1-> Beta4
c. 1, 4
d. Alpha1->6
e. Beta1->6
Glucose
The organic molecule that is produced abundantly by photosynthesis is
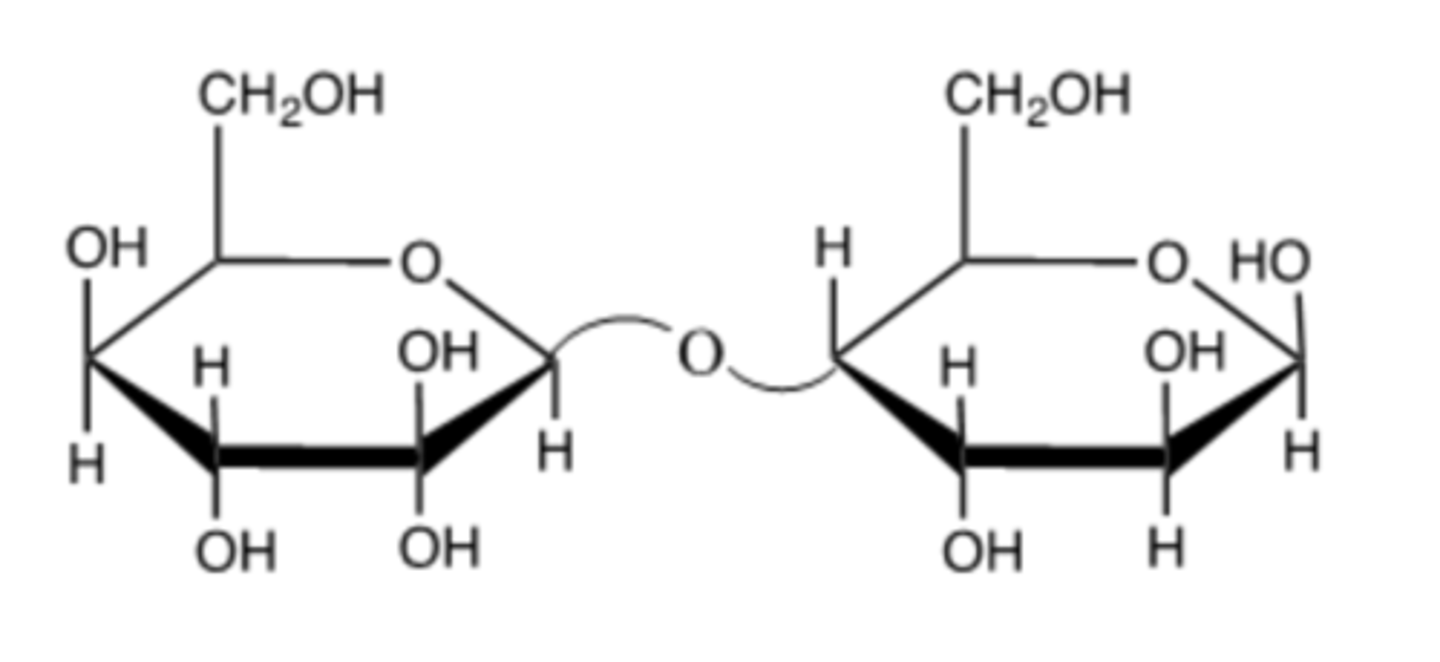
This is a beta1----->4 because the molecule on the left has the oxygen in the up, or beta, position.
Is the following an alpha1---->4 or beta1---->4 connection? Briefly explain.
D
Which compound does not fit the formal definition of a carbohydrate?
A.C6H12O6
B.C9H18O9
C.C3H6O3
D.C8H14O8
E.C5H10O5
D
Which monosaccharide is not a six-carbon monosaccharide?
A. Idose
B. Glucose
C. Galactose
D. Dihydroxyacetone
E. Allose
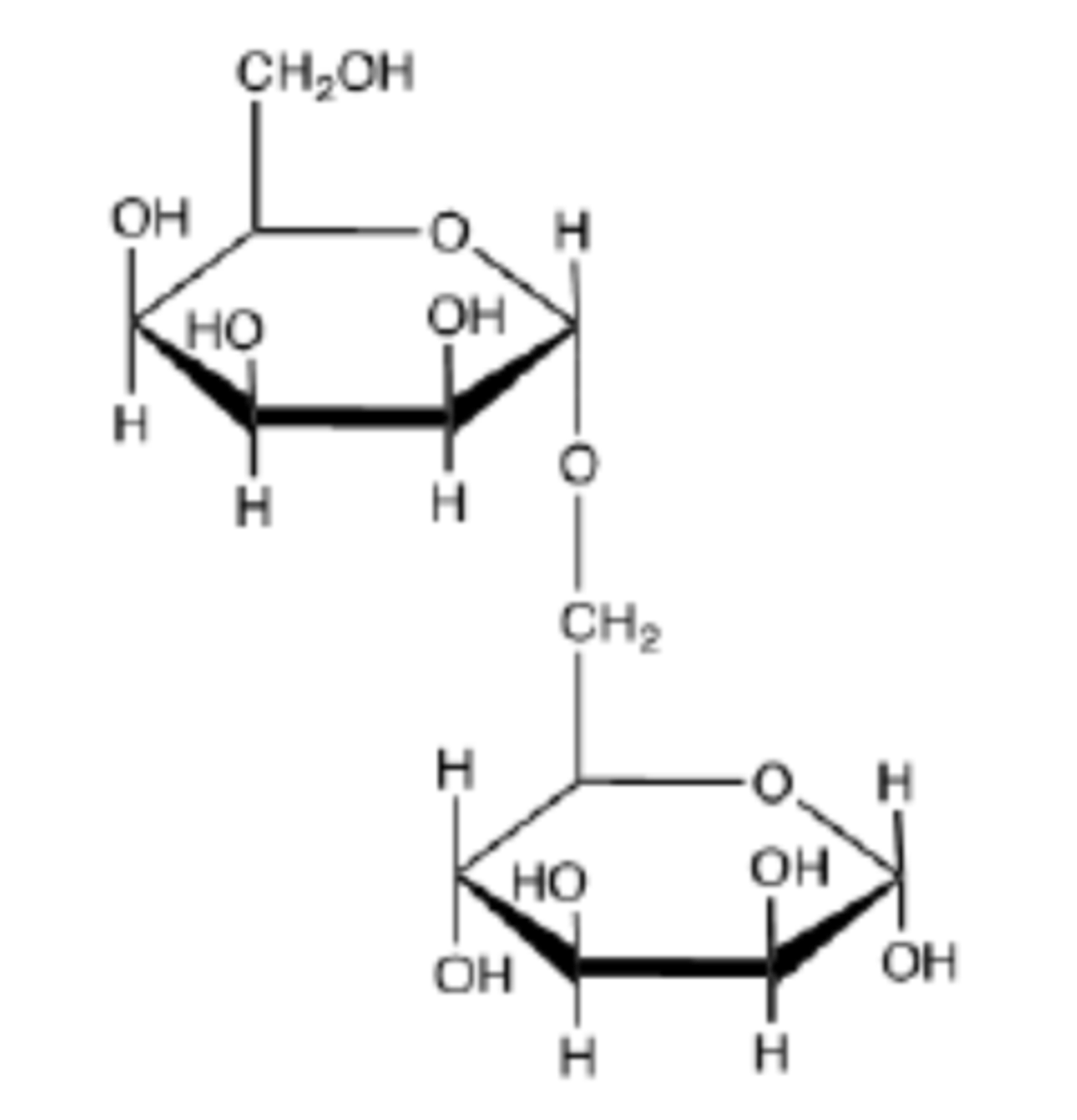
The OH groups on the oligosaccharide forms hydrogen bonds with water molecules. This forms an aqueous solution.
The following oligosaccharide interacts favorably with water. What is the source of the ability of the interaction between H2O and this carbohydrate?
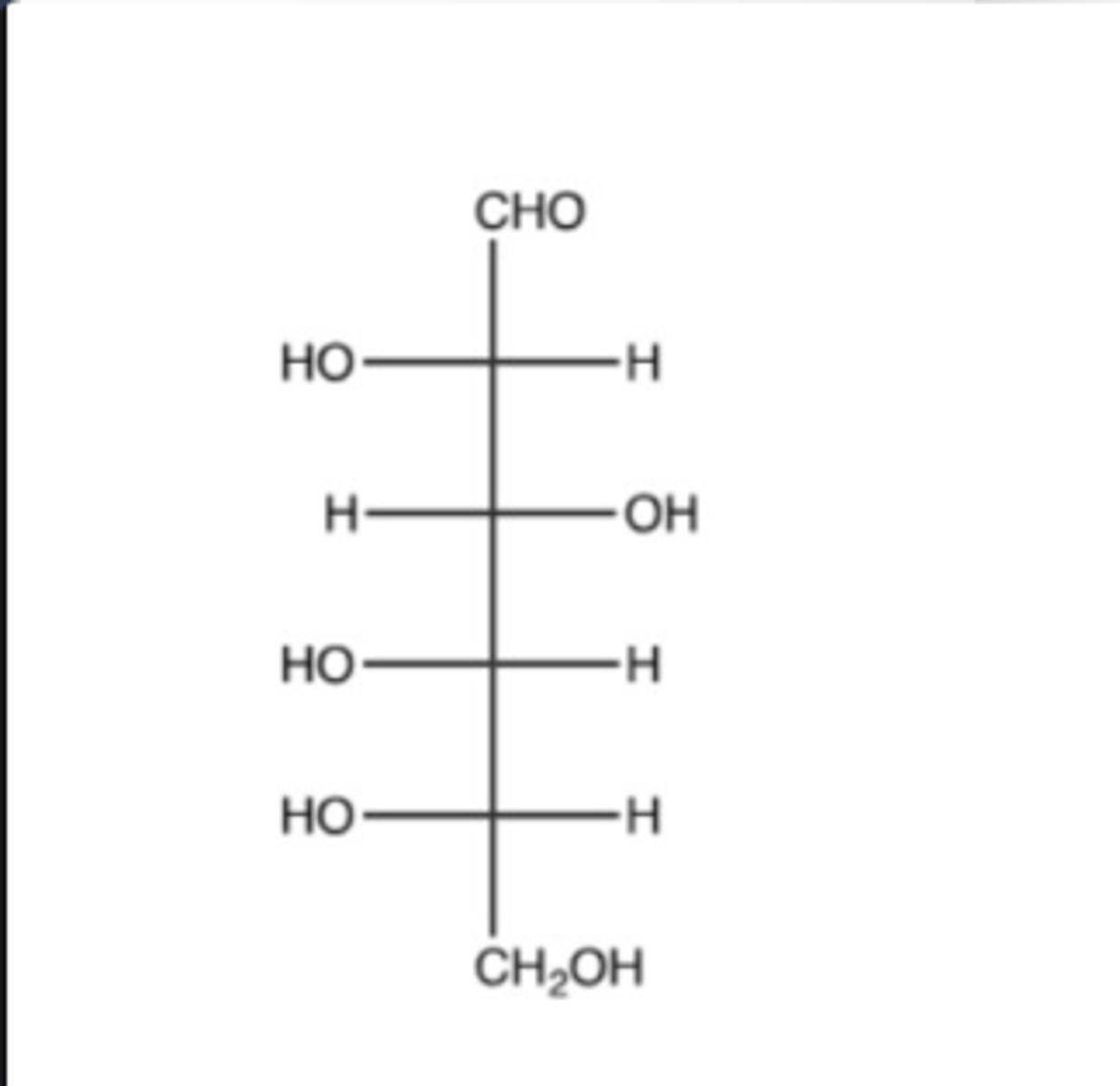
L
What is the configuration of the following compound?
C.
Which statement about starch and glycogen is false?
A) Amylose is unbranched; amylopectin and glycogen contain many (16) branches
B) Both are homopolymers of glucose
C) Both serve primarily as structural elements in cell walls
D) Both starch and glycogen are energy storage polysaccharides
E) Glycogen is more extensively branched than starch
A.
Which letter represents the hemiacetal bond?
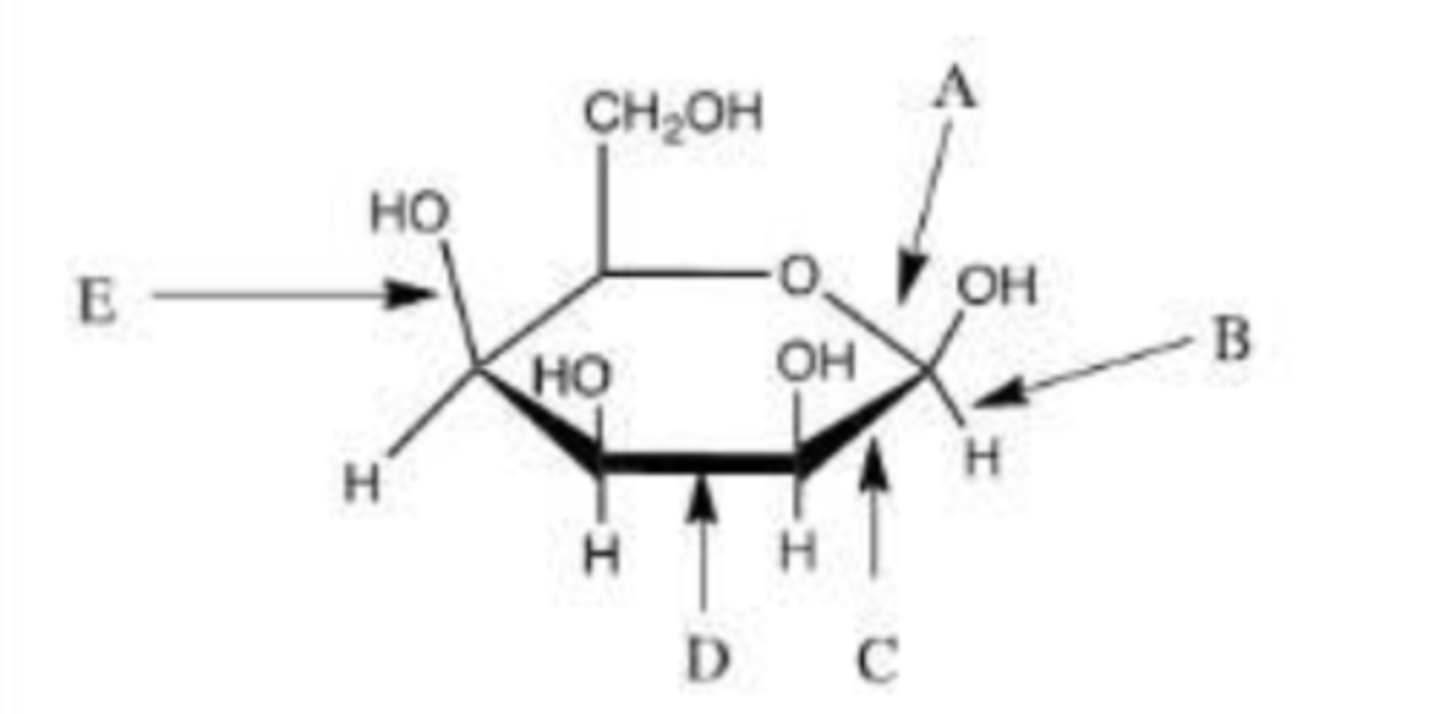
B.
Which of the following monosaccharides is a ketose?
a. glucose
b. fructose
c. galactose
d. mannose
No
Are starch and glycogen heteropolymers of glucose