Lecture 6: Polyamides, Polyimides
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
What are engineering plastics?
Plastics with better mechanical or thermal properties than commodity plastics. Examples include PA, PC, POM, PET
What is the key structural group in polyamides?
The amide group –CONH–, which enables strong intermolecular hydrogen bonding.
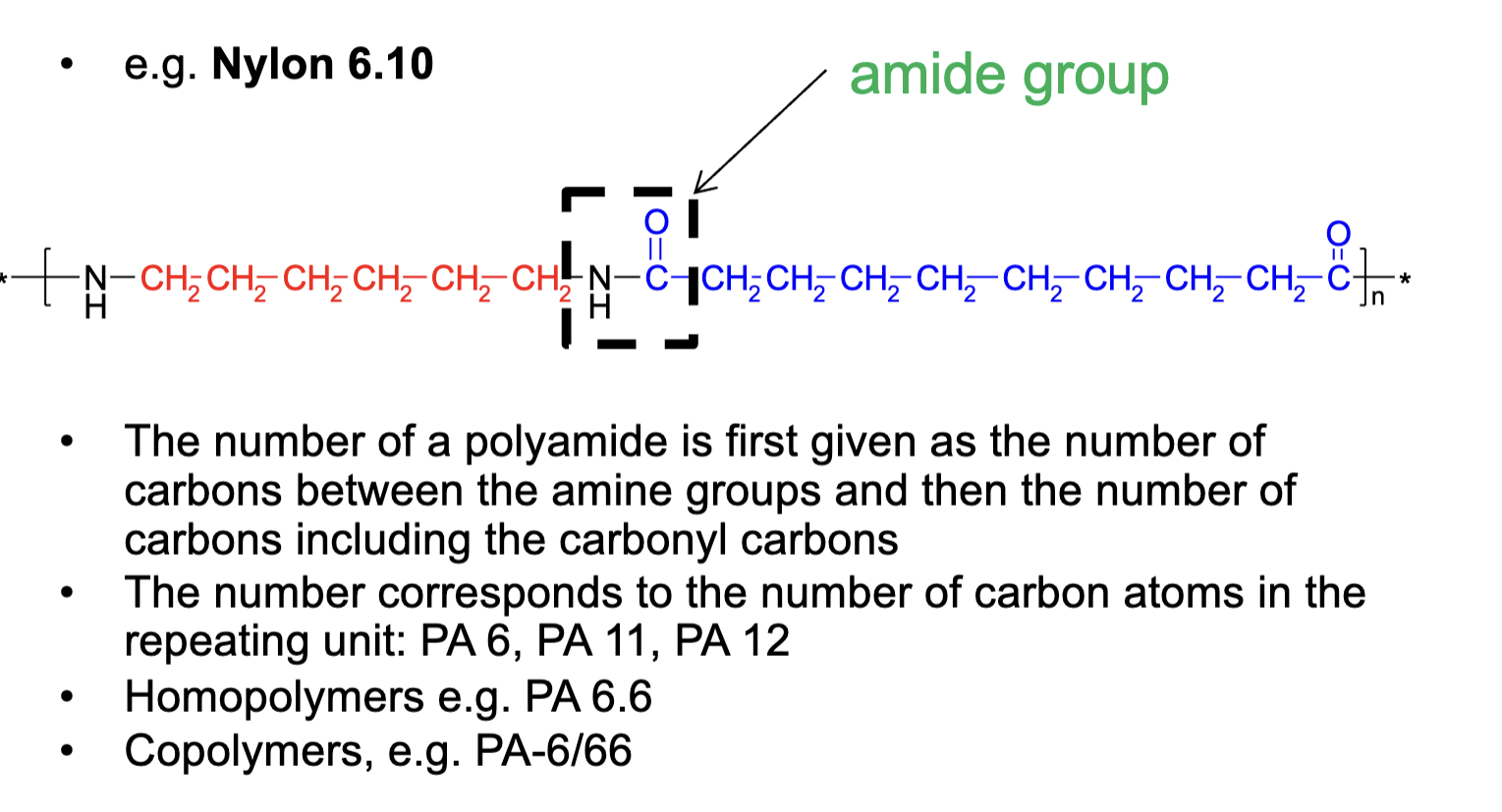
Mention 2 different synthesis methods of Polyamides
Step-growth reaction with diamines and dicarboxylic acids (PA 6.6)
Ring-opening polymerization of a lactam ring (PA 6)
How does the –CONH– group affect polyamide properties?
It increases:
interchain interactions
melting temperature (Tm)
mechanical strength
resistance to swelling and solvents.
How does the distance between amide groups affect properties?
Shorter distance → more hydrogen bonding → higher:
density
stiffness (because its getting more difficult to separate the chains)
strength (because its getting more difficult to separate the chains)
Tm (because its getting more difficult to separate the chains)
creep resistance (because its getting more difficult to separate the chains)
Water absorption (because its getting mooore polar)
Why do polyamides with an even number of methylene groups have higher Tm?
Even numbers allow better chain packing and more regular hydrogen bonding, giving higher crystallinity and Tm.
Why do polyamides absorb water?
Because the amide groups are polar and hydrogen-bond with water.
How does water affect polyamide properties?
Water acts as a plasticizer, which:
lowers Tg
reduces strength and modulus
increases impact strength.
What effect does copolymerization have on polyamides?
It reduces crystallinity and increases the amorphous fraction, giving more flexible and transparent materials.
Why are heat stabilizers added to PA?
To prevent thermal oxidation during processing and use
Why are nucleating agents used in polyamides?
They:
improve mechanical properties
speed up crystallization
enable faster injection molding.
Why are glass fibers added to polyamides?
To increase:
strength
stiffness
dimensional stability
reduce moisture uptake.
What must be considered when processing PA?
water absorption
high melting temperature
oxidation because of aliphatic CH2 groups
crystallization rate
drying before processing.
What are common applications of polyamides?
fibers (textiles)
automotive components
What are aramids?
Aromatic polyamides with very high strength and heat resistance.
Examples:
Kevlar
Nomex
Why is Kevlar extremely strong?
Because of:
rigid aromatic backbone
strong hydrogen bonding
highly oriented chains.
What properties characterize polyimides?
very high thermal stability
solvent resistance
excellent electrical insulation
flame resistance.
Why are polyimides difficult to process?
Because their rigid aromatic structure gives very high Tg and thermal stability.
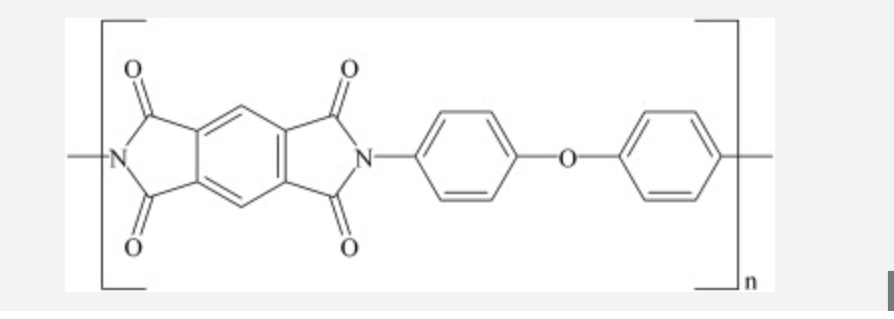
Why are modified polyimides developed?
To improve processability while retaining thermal stability, like Polyamide- imide (PAI)
What are the main advantages of polyamides?
high strength
high wear resistance
good creep resistance
good chemical durability.
What are the main disadvantages of polyamides?
moisture absorption
sensitivity to UV
limited thermal stability because of aliphatic CH2 chains
Expensive