(9) Pharmaceutical Calculations
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Viscosity
Definition:
resistance to flow
Viscosity
Examples:
Toothpaste → high viscosity
Mouthwash → low viscosity
Types of fluids:
Newtonian
Viscosity = constant
Does NOT change with shear
Non-Newtonian
Viscosity changes with force/shear
What type of viscosity we need?
Need optimal viscosity:
Too high → drug release ↓
Too low → unstable / melts
Example of not having optimal viscosity
Suppositories melting in package = ❌ bad
Arrhenius equation relates:
Viscosity ↔ Temperature
Temperature ↑ → Viscosity ↓
Temperature ↓ → Viscosity ↑
VISCOSITY vs TEMPERATURE (ARRHENIUS)
Example:
Heat suppository → melts → viscosity ↓
Refrigerate → becomes thicker
VISCOSITY vs TEMPERATURE (ARRHENIUS)
APPLICATION
Used for:
Shelf life determination
Stability testing
DRUG RELEASE FROM OINTMENT
equation
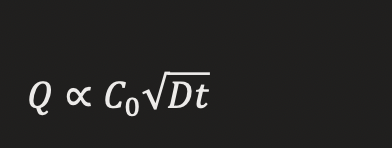
Variables:
Q = drug released
C₀ = initial concentration
D = diffusion coefficient
t = time
💡 KEY RELATIONSHIPS ✅⭐
C₀ ↑ → Drug release ↑
Time ↑ → Drug release ↑
D ↑ → Drug release ↑
SUPPOSITORIES DRUG RELEASE
Depends on:
Viscosity
Type of base
SUPPOSITORIES DRUG RELEASE
Base types:
Base | Mechanism |
Cocoa butter | melts |
PEG | dissolves |
SUPPOSITORIES DRUG RELEASE
KEY IDEA
High viscosity → slow release
Low viscosity → unstable
DRUG RELEASE MEASUREMENT
Need to measure:
How much drug released
DRUG RELEASE MEASUREMENT
Methods:
UV spectroscopy (common)
HPLC (more sensitive)
BEER-LAMBERT LAW
calculations
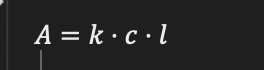
Variables:
A = absorbance
c = concentration
k = constant
l = path length
BEER-LAMBERT LAW
KEY RELATIONSHIP
Concentration ↑ → Absorbance ↑ (linear)
💡 EXAM LOGIC
If A doubles → C doubles
CHARACTERIZATION TECHNIQUES - overview
Qualitative:
Quantitative:
CHARACTERIZATION TECHNIQUES
Qualitative:
IR
Raman
X-ray
DSC
Microscopy
👉 Used for:
Structure
Impurities
CHARACTERIZATION TECHNIQUES
Quantitative:
UV spectroscopy
HPLC
👉 Used for:
Drug concentration
SUPPOSITORY CALCULATIONS
WHEN TO IGNORE CALCULATIONS
Drug < 100 mg
Suppository ≈ 2 g
👉 Volume negligible
SUPPOSITORY CALCULATIONS
WHEN TO USE FACTORS
Drug > 100 mg
OR suppository < 2 g
DOSAGE REPLACEMENT FACTOR (f)
MATH:
ON EQUATION SHEET
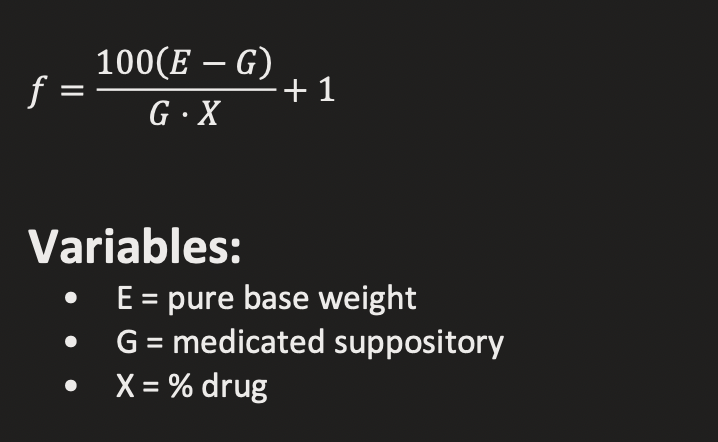
DOSAGE REPLACEMENT FACTOR (f)
EXAMPLE STEPS
Convert drug to %
Plug into equation
Solve for G
DENSITY FACTOR
MATH:
ON EUQATION SHEET
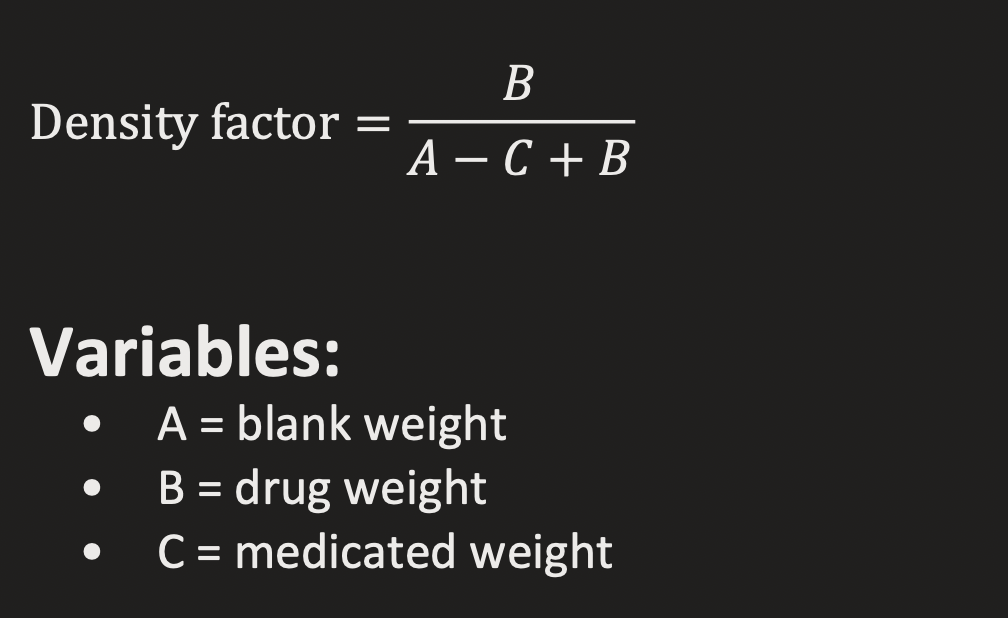
PULMONARY (AEROSOL CALCULATIONS)
MATH
RAOULT’S LAW
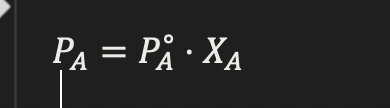
Total pressure = sum of partial pressures