insulin and hypoglycemic agent - lecture 30
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
diabetes mellitus
insulin is not secreted adequately or tissues are resistant to its effects
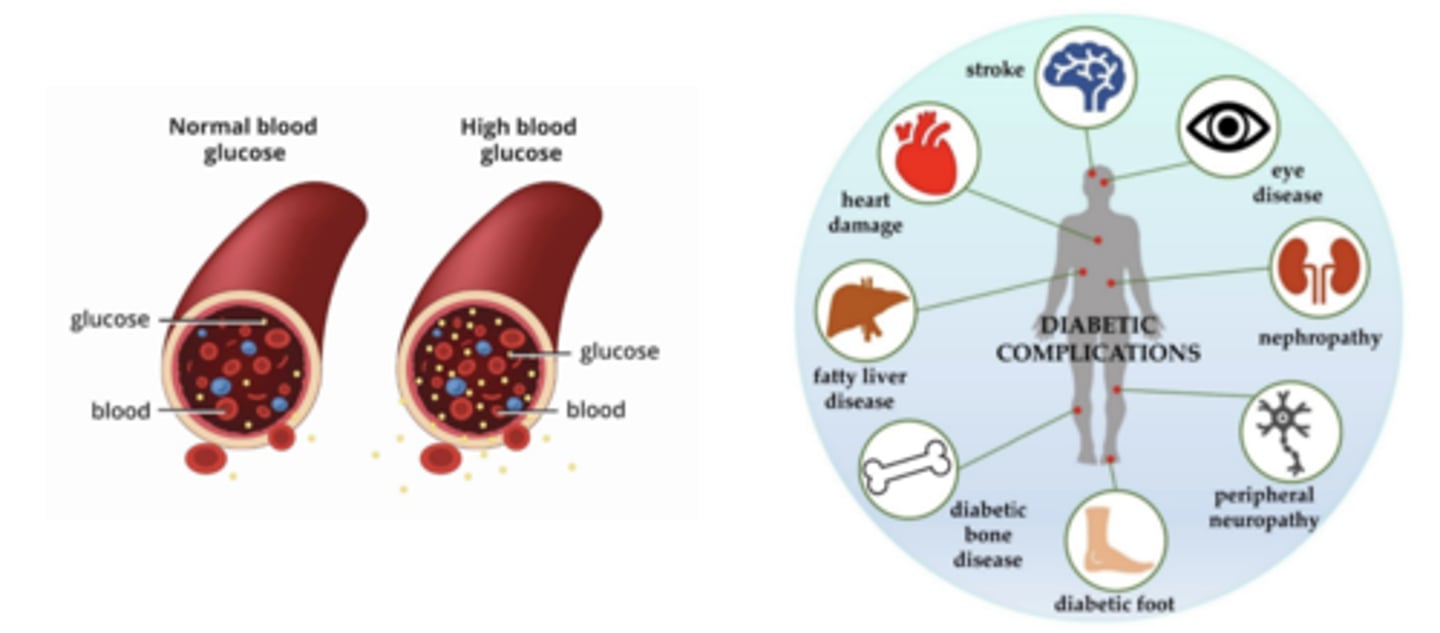
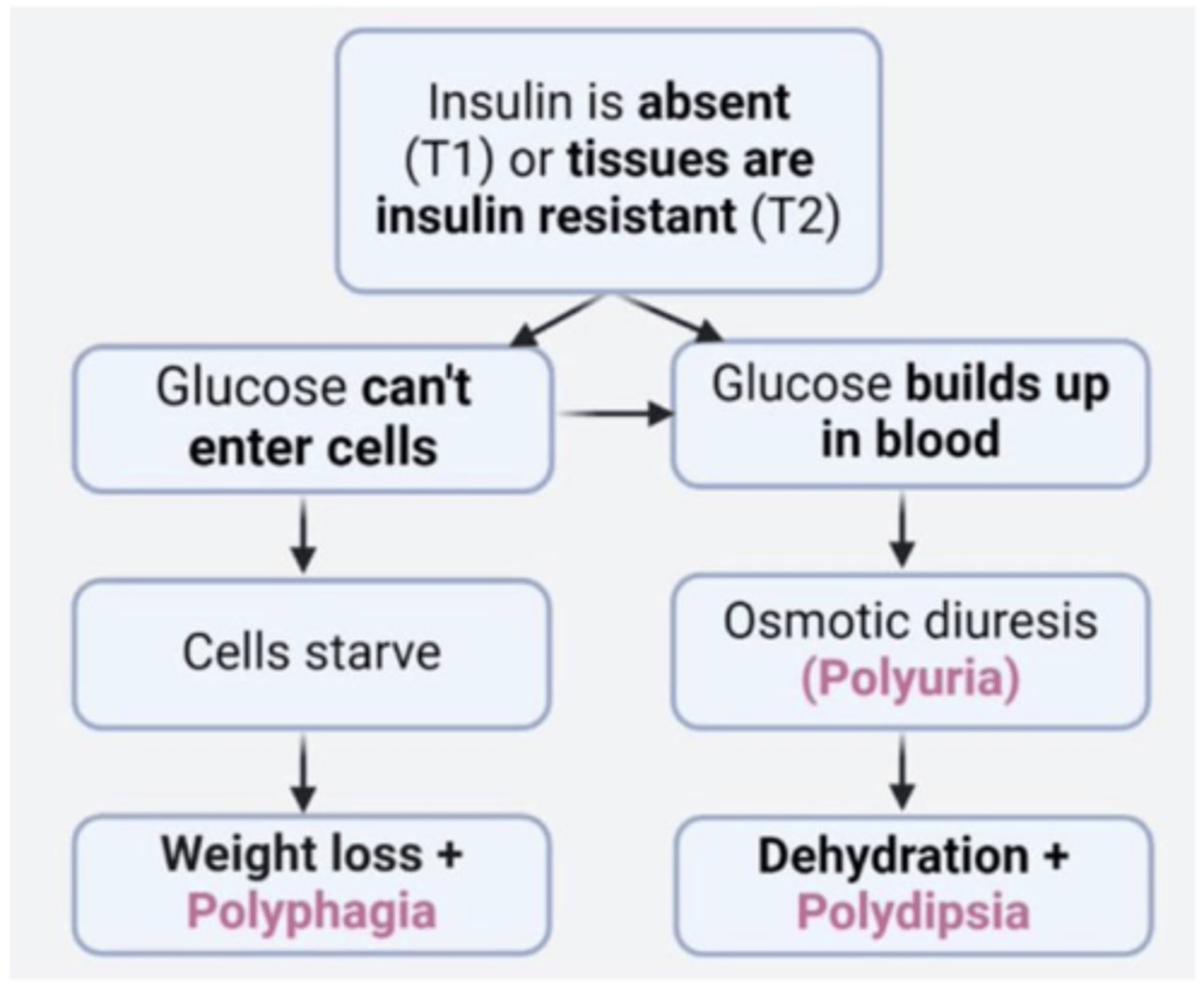
acute manifestations of diabetes mellitus
the 3 Ps
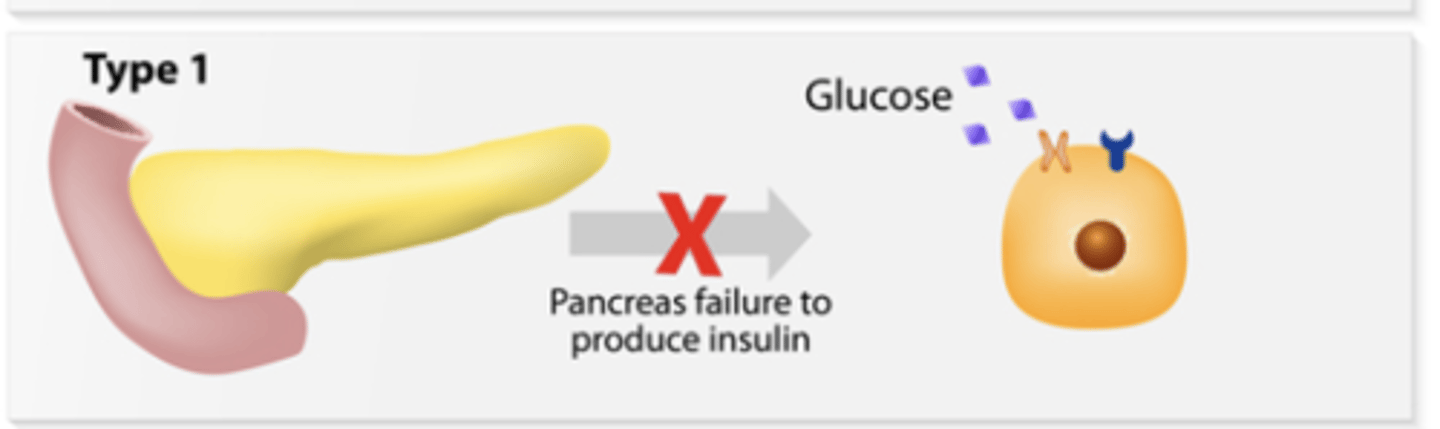
type 1 diabetes
occurs when the pancreas is unable is unable to produce enough insulin
tends to develop at a young age
cannot be prevented
required insulin therapy
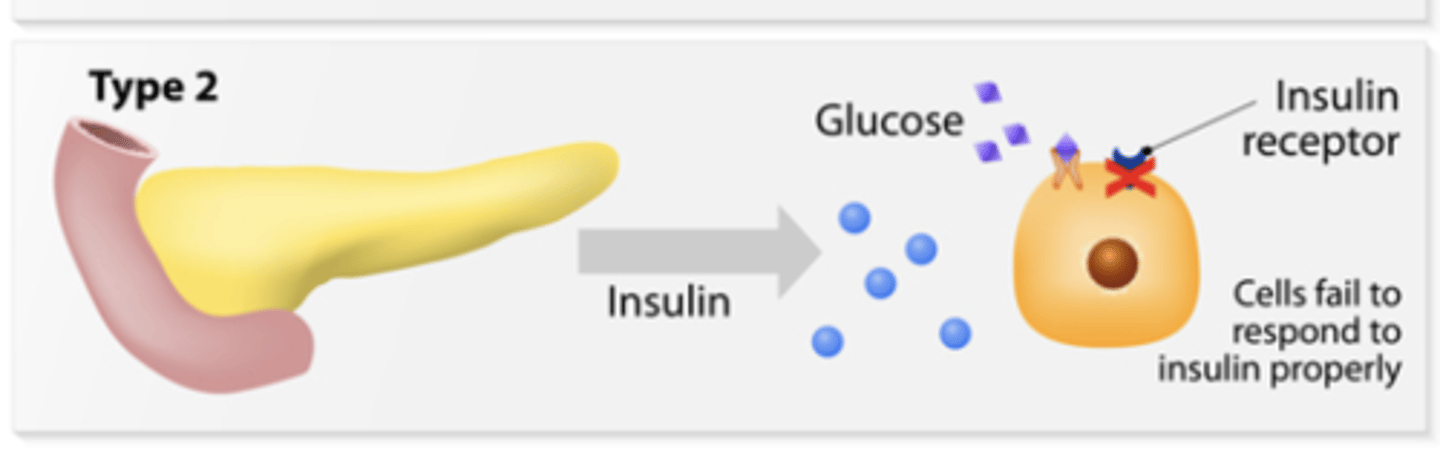
type 2 diabetes
occurs due to insulin resistance (when the body does not respond well to insulin)
tends to develop at an older age
can be prevented with lifestyle changes
can be managed with lifestyle modifications alone if diagnosed early
Type 1 DM pharmacotherapy
insulin replacement
Type 2 DM pharmacology
oral agents (metformin is first line for most; GLP-1 agonist and SGLT2 inhibitors can be first line for certain patient populations), non-insulin injectables, insulin replacement; weight loss particularly helpful in lowering blood glucose
Gestational DM pharmacology
insulin replacement if nutrition therapy and exercise alone fail; regular insulin is preferred
Rapid-acting - T1DM
insulin lispro, aspart, glulisine
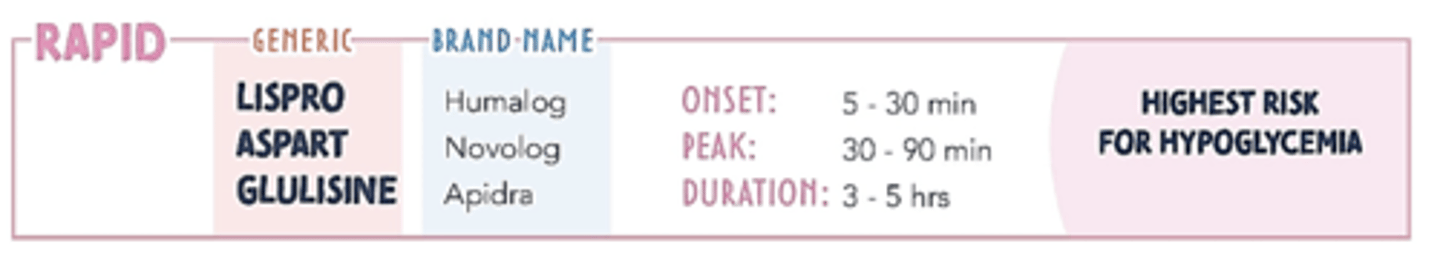
Short-acting - T1DM
regular insulin

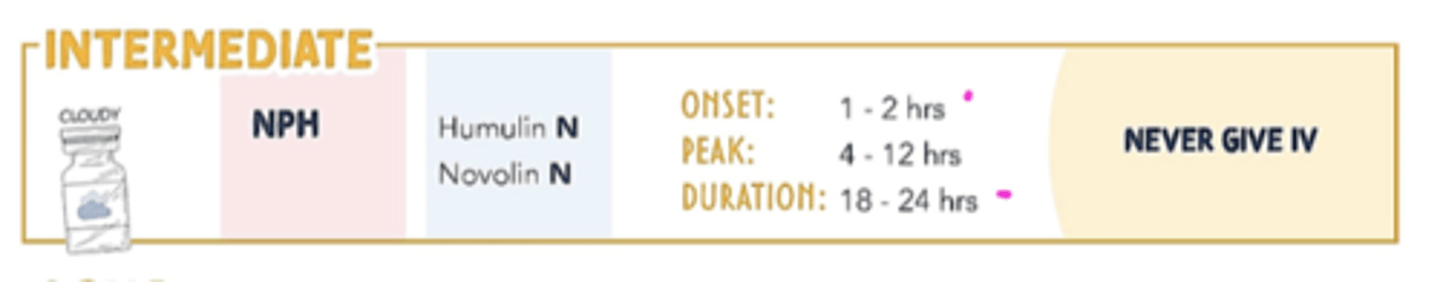
Intermediate-acting - T1DM
NPH (neutral protamine hagedorn) insulin
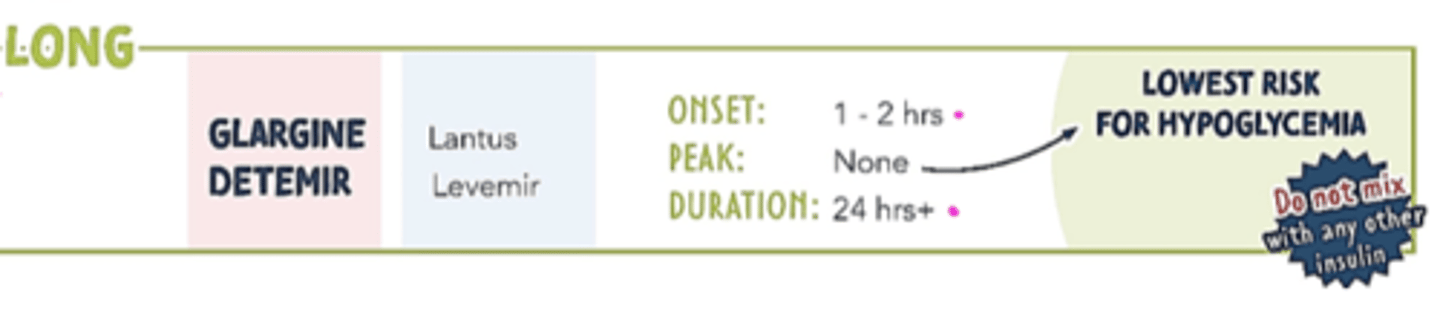
Long-acting - T1DM
insulin glargine, detemir
Ultra Long-acting (not pictured) - T1DM
insulin degludec
administration - T1DM
must be given subcutaneously or IV (short acting: regular insulin - only insulin given IV)
insulin is destroyed in the GI tract so it cant be given PO
remove all air bubbles
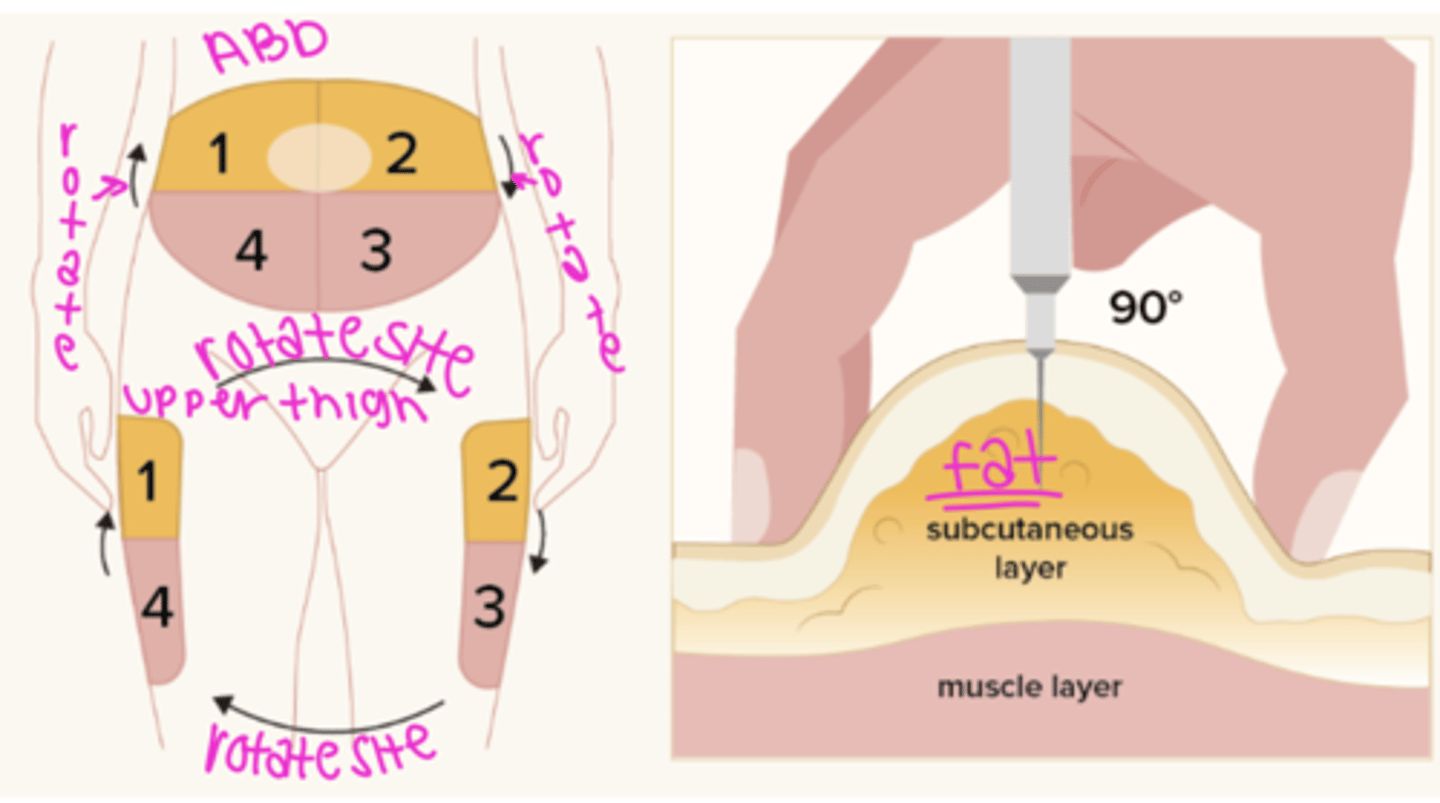
rotate site 1 inch from previous site
common sites: back of arms, thigh, abdomen (at least 2 inches away from belly button)
complications - T1DM
hypoglycemia (especially with rapid insulin)
weight gain
--insulin growth hormone
lipoatrophy (loss of subcut fat - bad ways)
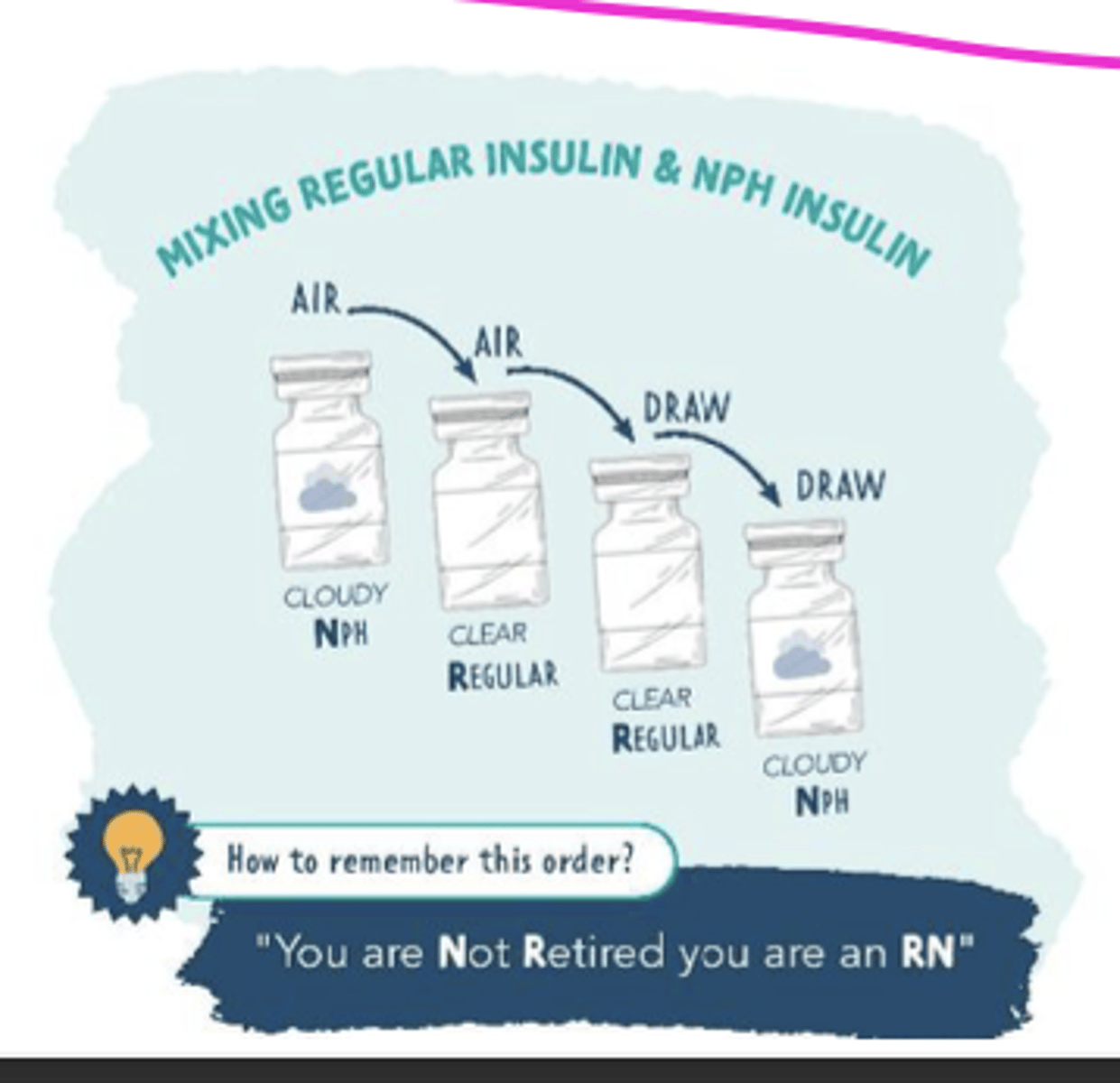
mixing insulin
insulin
A protein hormone synthesized in the pancreas that regulates blood sugar levels by facilitating the uptake of glucose into tissues
insulin administration (SQ)

monitor glucose levels + insulin pump
insulin therapy
Clinical indications: type 1 and type 2 DM
Adverse effects: hypoglycemia, weight gain (rare for type 1 DM), injection site pain (glargine), lipatrophy, lipohypertrophy
CONS: high risk of hypoglycemia; causes weight gain (only a con in Type 2 DM – discussed later)
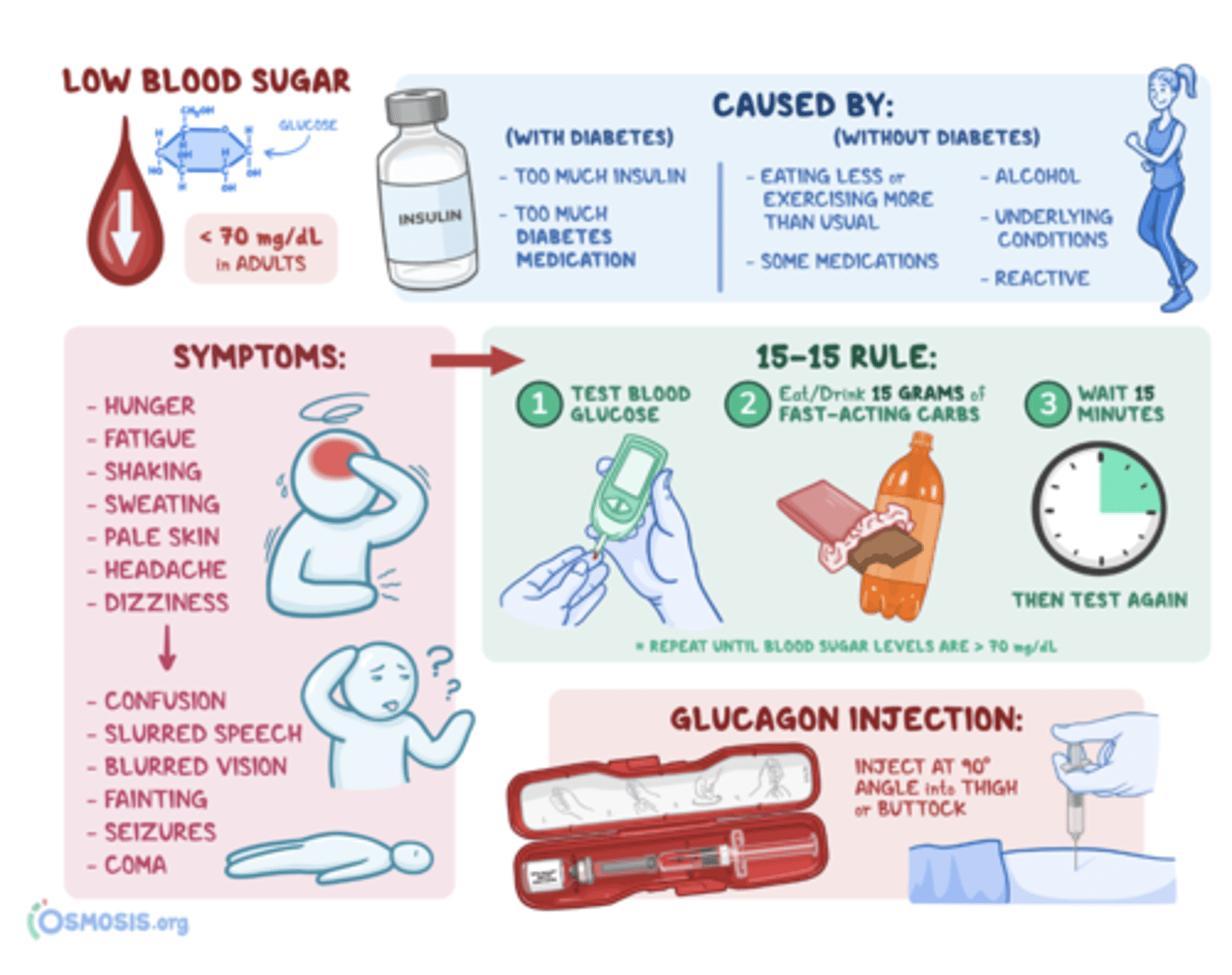
monitoring for glycemic control and hypoglycemia (note symptoms, 15-15 rule, and glucagon injection)
glucagon injection
inject at 90 degree angle into thigh or buttocks if patient is unresponsive
Glucagon (injection, nasal spray) - always have injection just in case patient is unresponsive
clinical indications: hypoglycemia
adverse effects: GI symptoms, headaches
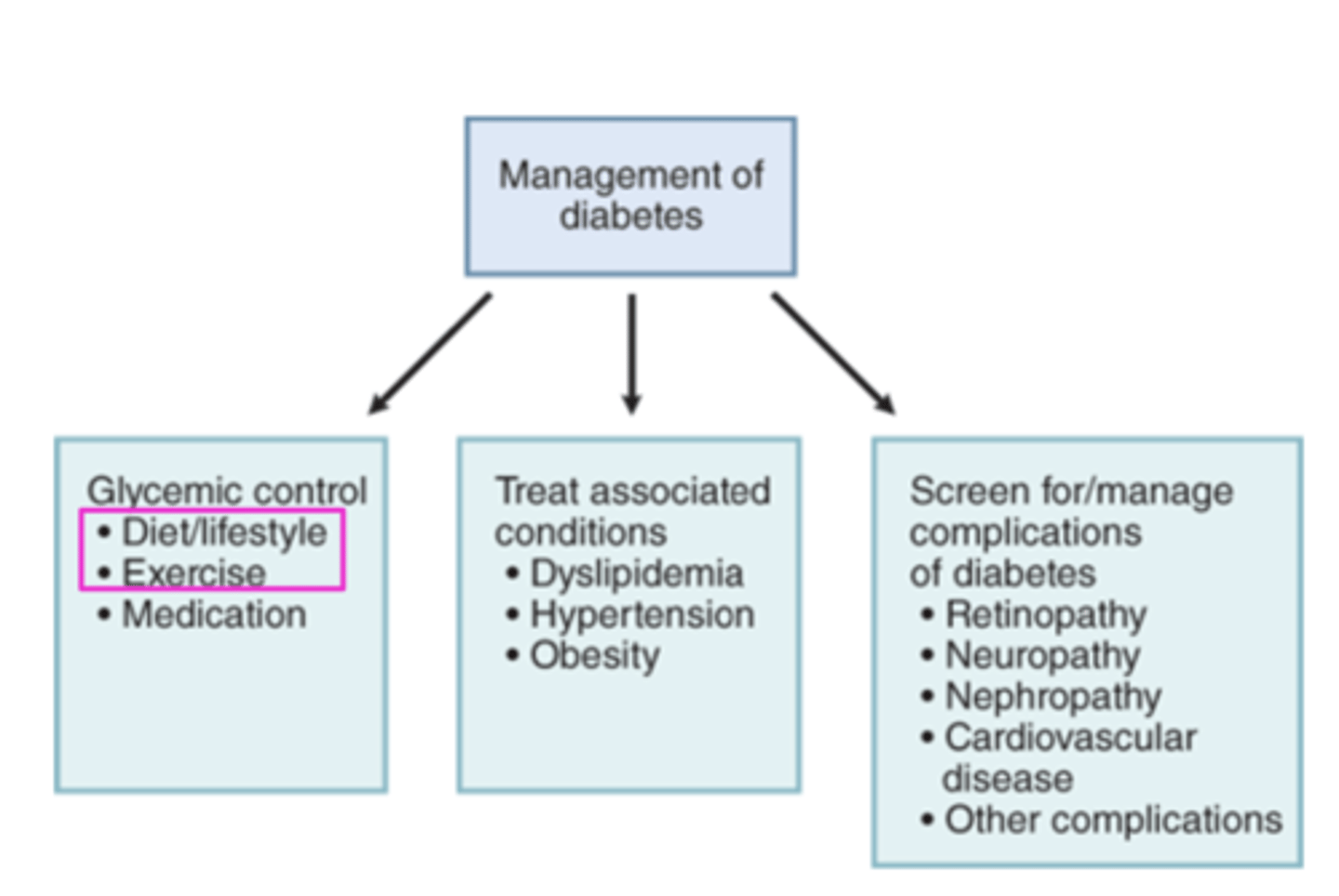
goals of therapy for Type 2 DM: #1 is diet and exercise
goat's rue for diabetes
Previous biguanides were too toxic (lactic acidosis), but the synthesis of metformin (and its structural differences from prior biguanides) led to the discovery of the most prescribed glucose-lowering medication worldwide.
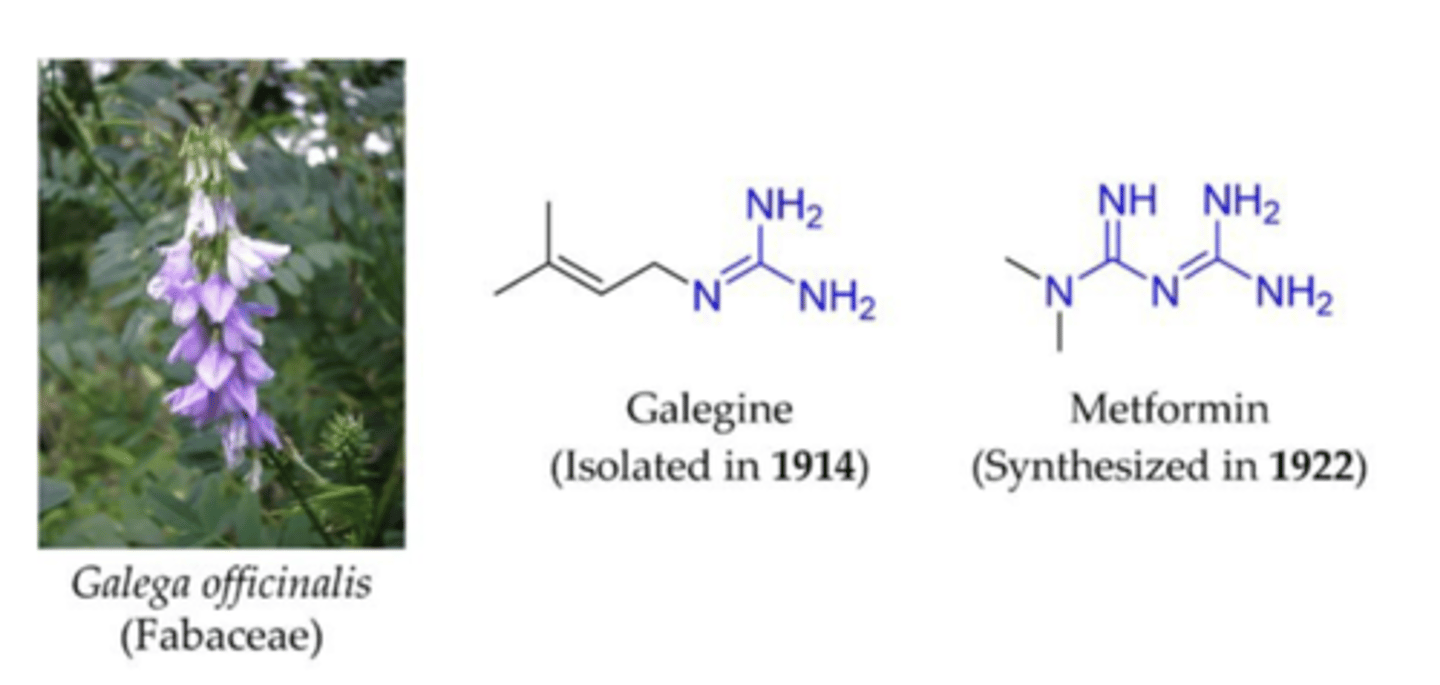
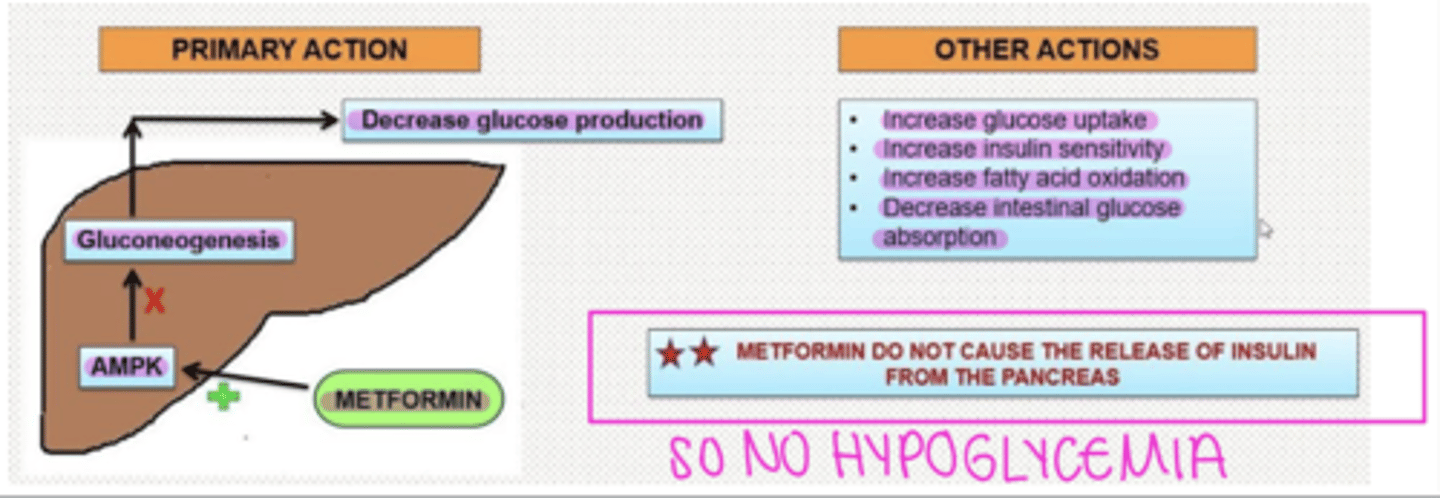
Metformin (biguanide)
MOA: inhibits mitochondrial complex I = prevents the production of mitochondrial ATP = activates AMPK (AMP-activated protein kinase), an enzyme that plays an important role in the regulation of glucose metabolism = decreases hepatic glucose production, decreases intestinal absorption of glucose, increases insulin sensitivity, and increases glucose uptake and use in fat and skeletal muscles
Clinical indications: type 2 DM
PRO: Does NOT present with increased risk of hypoglycemia since it does not increase insulin levels
Adverse effects: GI upset, lactic acidosis, vitamin B12 and folic acid deficiency,
Metformin is substantially excreted by the kidney, so the dose must be reduced in patients with renal impairment.
For most patients, metformin should be stopped at the time of contrast administration. Why? Because there have been case reports of patients developing lactic acidosis after receiving iodinated contrast material while on metformin.
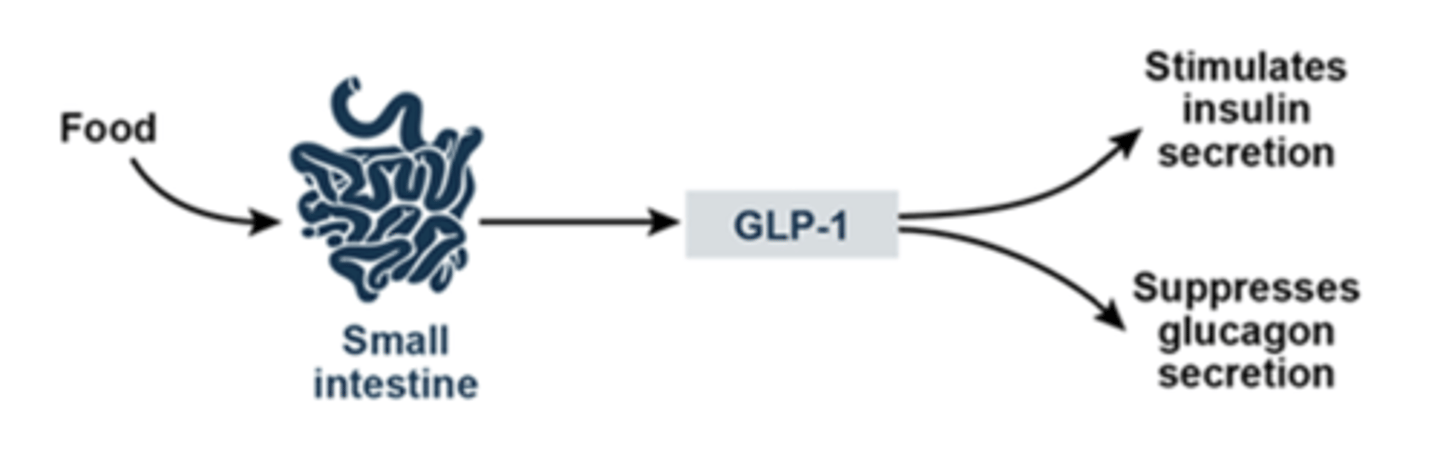
Glucagon Like Peptide 1 (GLP-1) Agonists (oral, SQ)
semaglutide, exenatide, liraglutide

Glucagon Like Peptide 1 (GLP-1) Agonists (oral, SQ) semaglutide, exenatide, liraglutide
GLP-1 stimulates glucose-dependent insulin release, inhibits glucagon release, and stimulates b-cell proliferation
MOA: bind to GLP-1 receptors = increased release of insulin + b-cell proliferation and inhibited glucagon release
Glucagon Like Peptide 1 (GLP-1) Agonists (oral, SQ) semaglutide, exenatide, liraglutide (2)
Administration:
- Once daily SQ: liraglutide
- Once weekly SQ: exenatide ER, semaglutide
- PO: semaglutide
Clinical indication: type 2 DM
Therapeutic effects: Enhances glucose-dependent insulin release, inhibits glucagon secretion, delays gastric emptying, delaying/reducing post-meal increases in glycemia, suppress appetite, increases Beta cell number
Adverse effects: GI (nausea, loss of appetite), hypersensitivity reactions, acute pancreatitis, AKI, gallbladder disease
PROS: Low hypoglycemia risk when administered alone (However, does have increased risk when administered with insulin or sulfonylureas); reduces appetite = weight loss; beneficial for atherosclerotic disease
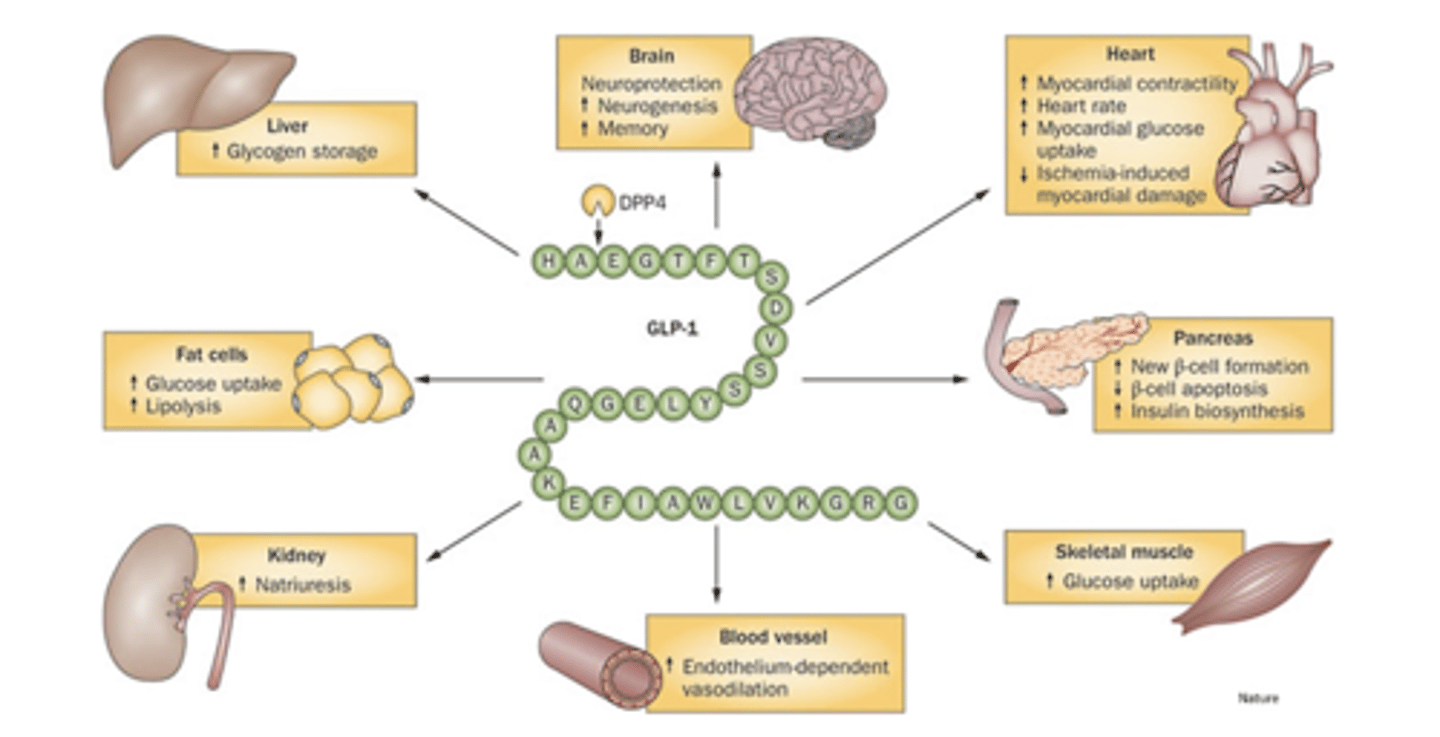
Pleiotropic Effects of GLP-1 Agonists
semaglutide, exenatide, liraglutide
note: when these first came out you had to inject subcutaneously once a day, now there are formulations where you can inject once a week or take it orally
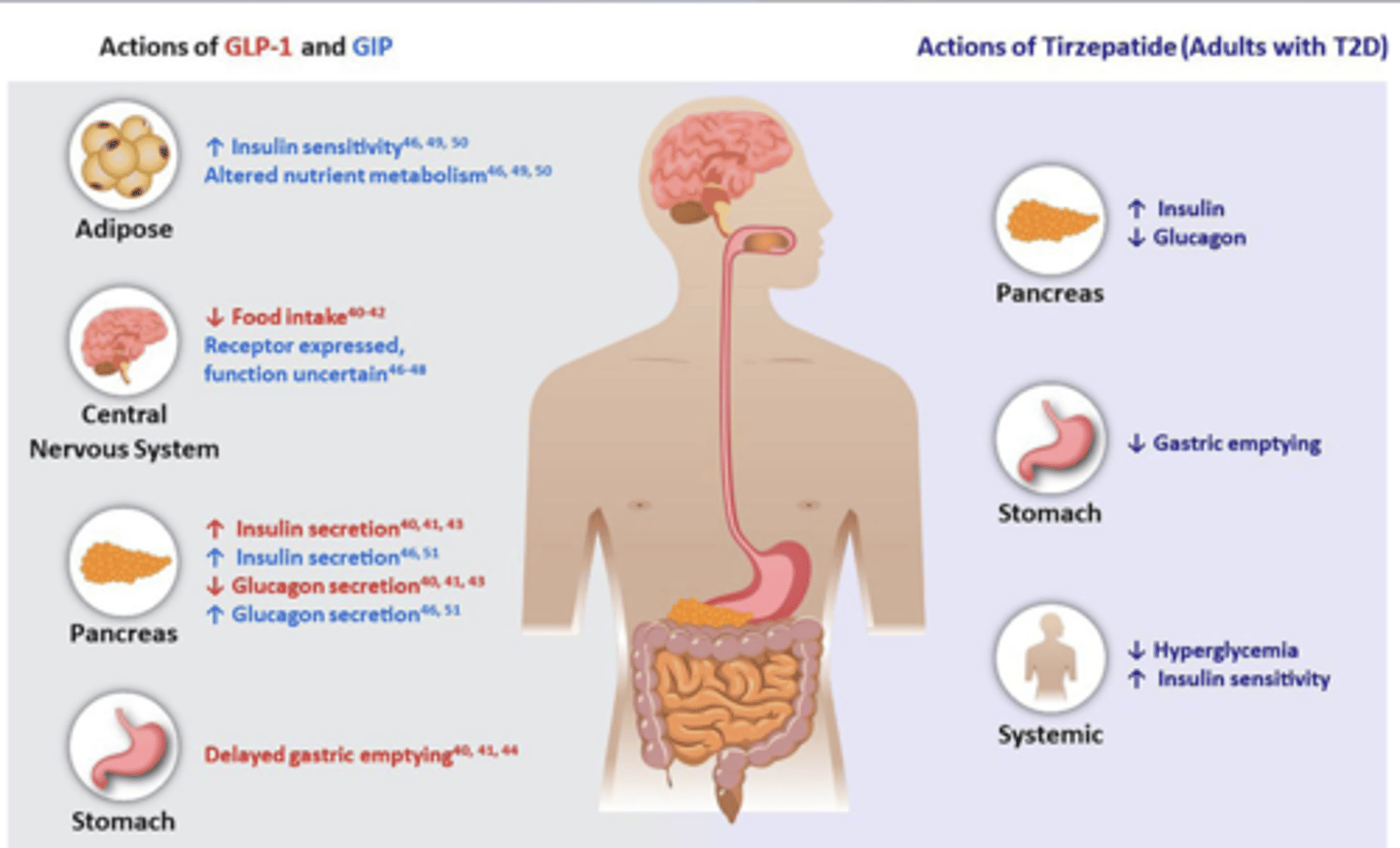
Glucose-Dependent Insulinotropic Polypeptide (GIP) + Glucagon-Like Peptide (GLP-1) Receptor Agonist Tirzepatide (SQ) - UNIQUE FROM OTHERS
MOA: both a GLP-1 and a GIP receptor agonist that increases glucose-dependent insulin secretion, decreases inappropriate glucagon secretion, and slows gastric emptying
Clinical indication: type 2 DM; obesity (Zepbound®)
Adverse effects: GI, hypersensitivity reactions
Due to lack of additive glycemic benefit, avoid
concomitant use with a DPP-4 inhibitor.
May require a dose reduction of insulin and/or insulin secretagogues (sulfonylureas, meglitinides) to avoid hypoglycemia.
PRO: Significant weight loss
CON: Use is not recommended in patients with severe GI disease.
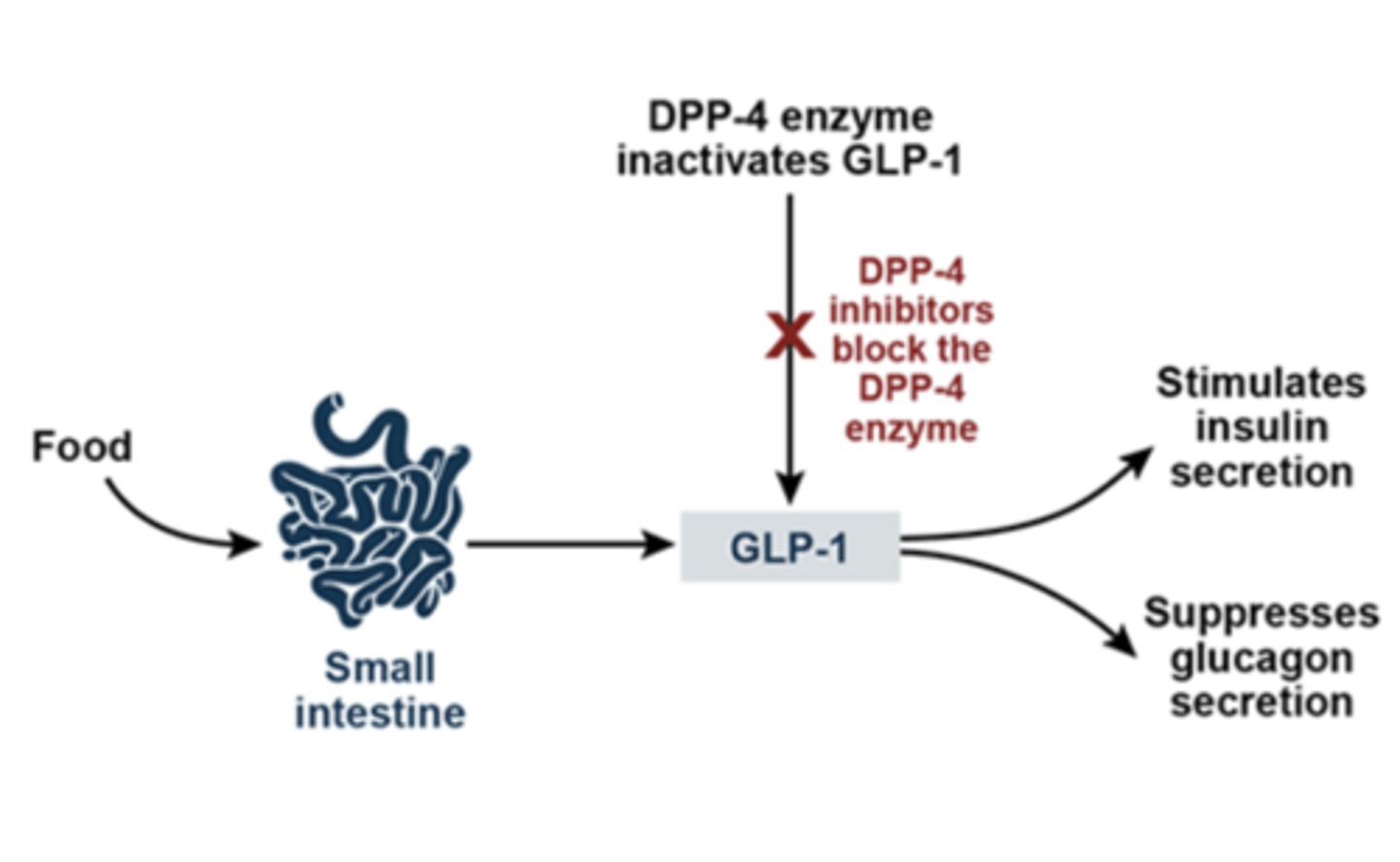
Dipeptidyl Peptidase-4 (DPP4) Inhibitors (Gliptins) saxagliptin, sitagliptin, linagliptin
GLP-1 stimulates insulin release, inhibits glucagon release, and stimulates b-cell proliferation
DPP4 degrades GLP-1
MOA: inhibits DPP4 = inhibits degradation of GLP-1 = subsequent increase in GLP-1 = increased release of insulin + b-cell proliferation and inhibited glucagon release
NOT AS EFFECTIVE SO NOT GIVEN MUCH
Clinical indications: type 2 DM
Adverse effects: (FYI only, as these are rare) respiratory and urinary infections, arthralgia (joint pain), dermatologic reactions, heart failure (only saxagliptin), hypersensitivity reactions (anaphylaxis and angioedema), acute pancreatitis – generally well tolerated
CONS: Not used as frequently for type 2 DM as they are not as effective as GLP-1 agonists; does NOT reduce appetite or slow gastric emptying like GLP-1 agonists
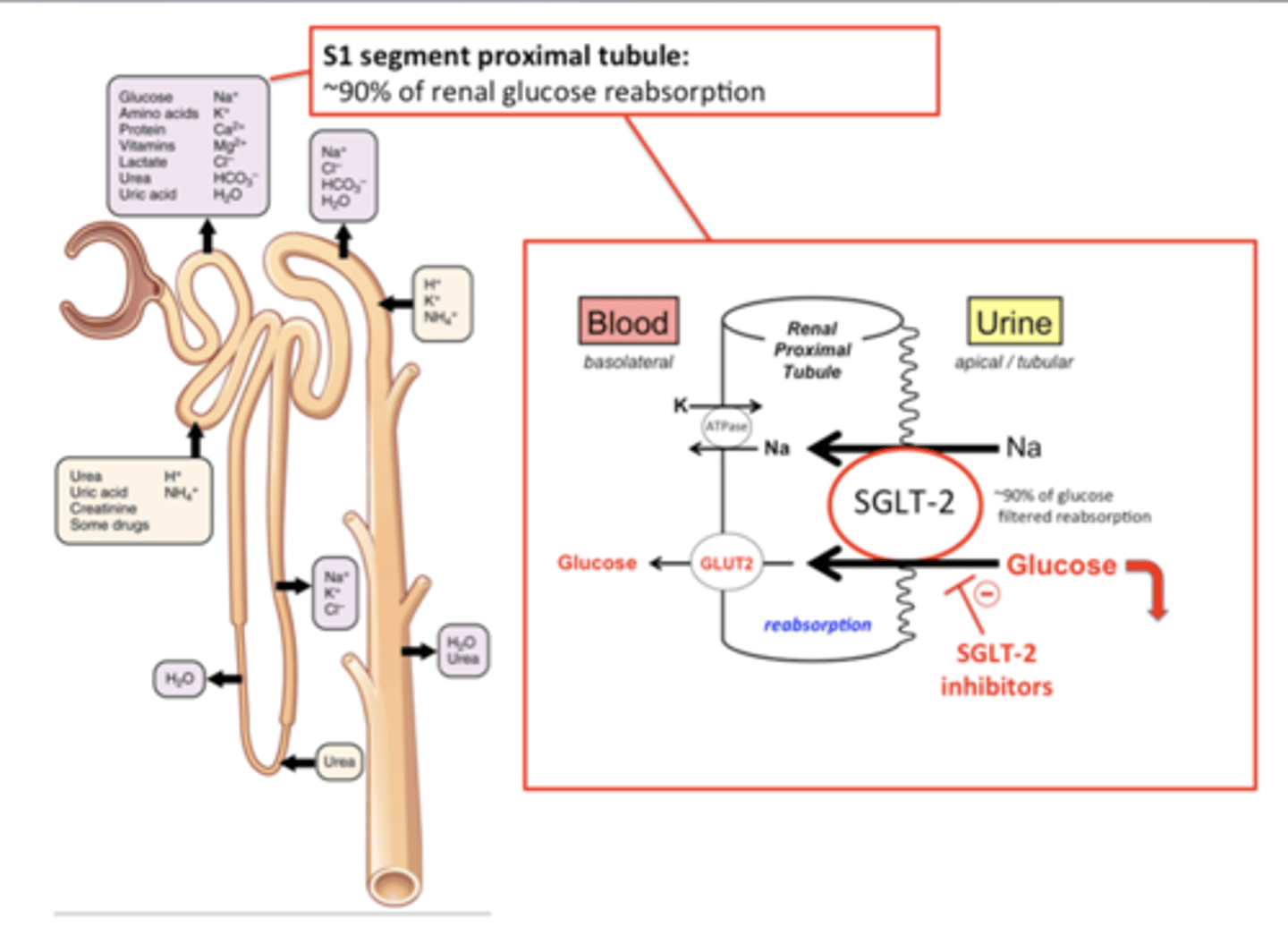
Sodium-Glucose Cotransporter-2 (SGLT2) Inhibitors canagliflozin, dapagliflozin, empagliflozin, ertugliflozin, bexagliflozin
SGLT2 is responsible for reabsorbing ~90% of filtered glucose in the proximal renal tubules
MOA: inhibits SGLT2 = reduce absorption of filtered glucose + lower renal threshold for glucose = increases urinary excretion of glucose = reduces plasma glucose concentration
Clinical indications: type 2 DM (also used to treat HF)
Adverse effects: AKI (acute kidney injury), hyperkalemia, hypersensitivity, hypovolemia, hypotension, infections (especially UTI), ketoacidosis
PROS: Low hypoglycemia risk; reduces appetite = weight loss; beneficial for CHF (especially HFpEF for which it is the only FDA-approved pharmacological agent)
CONS: Not used as frequently for type 2 DM as they are not as effective as GLP1 agonists
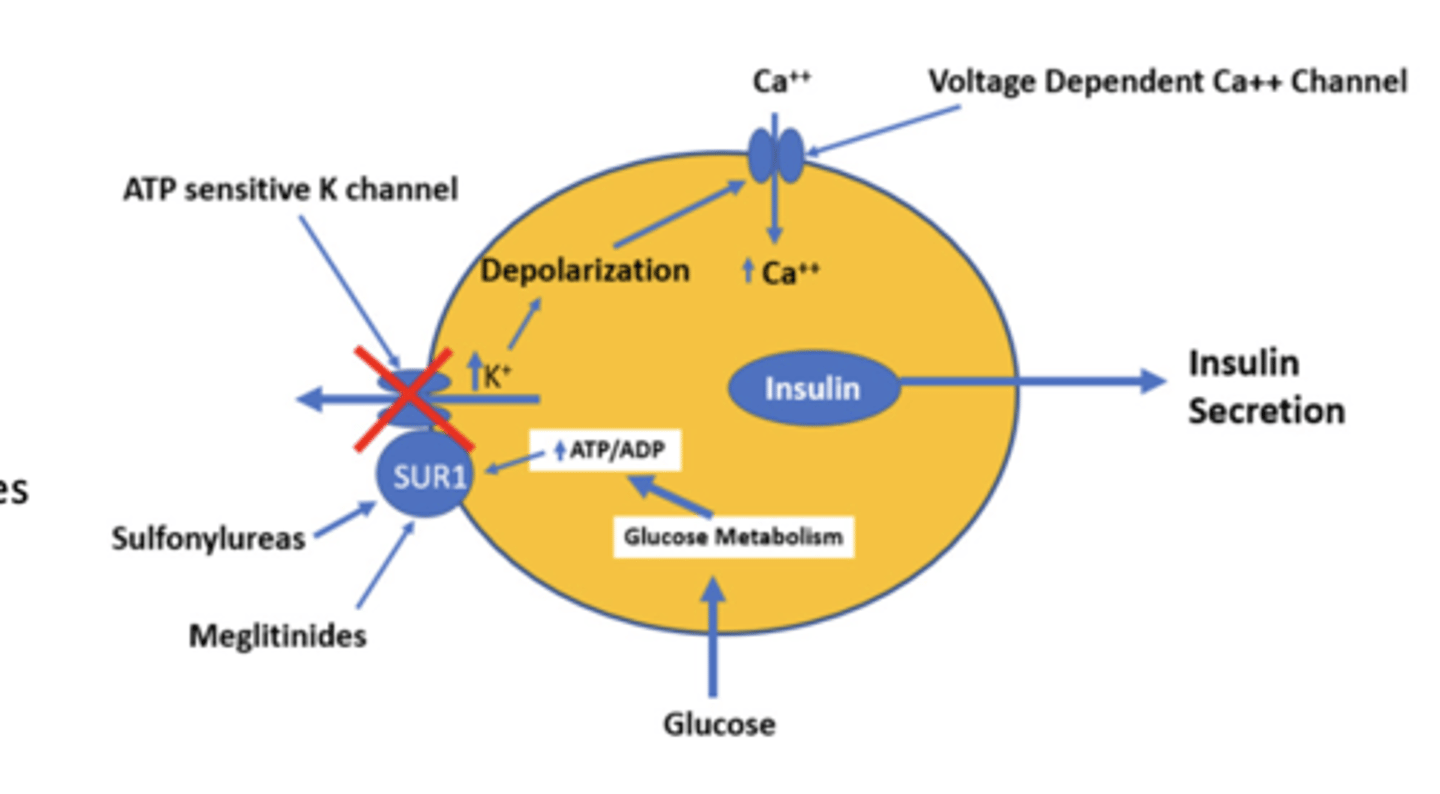
Sulfonylureas (2nd generation) glipizide, glyburide, glimepiride
MOA: bind to SUR1 (ATP-sensitive K+ channel) on β-cells = prevent K+ efflux =
β-cell depolarization = Ca2+ influx = stimulates insulin secretion
Clinical indications: type 2 DM
Adverse effects: hypoglycemia, weight gain, allergic reaction (sulfonamides)
*Significant risk of hypoglycemia, but still commonly used because cheap and effective
PROS: low cost
CONS: high risk of hypoglycemia; causes weight gain
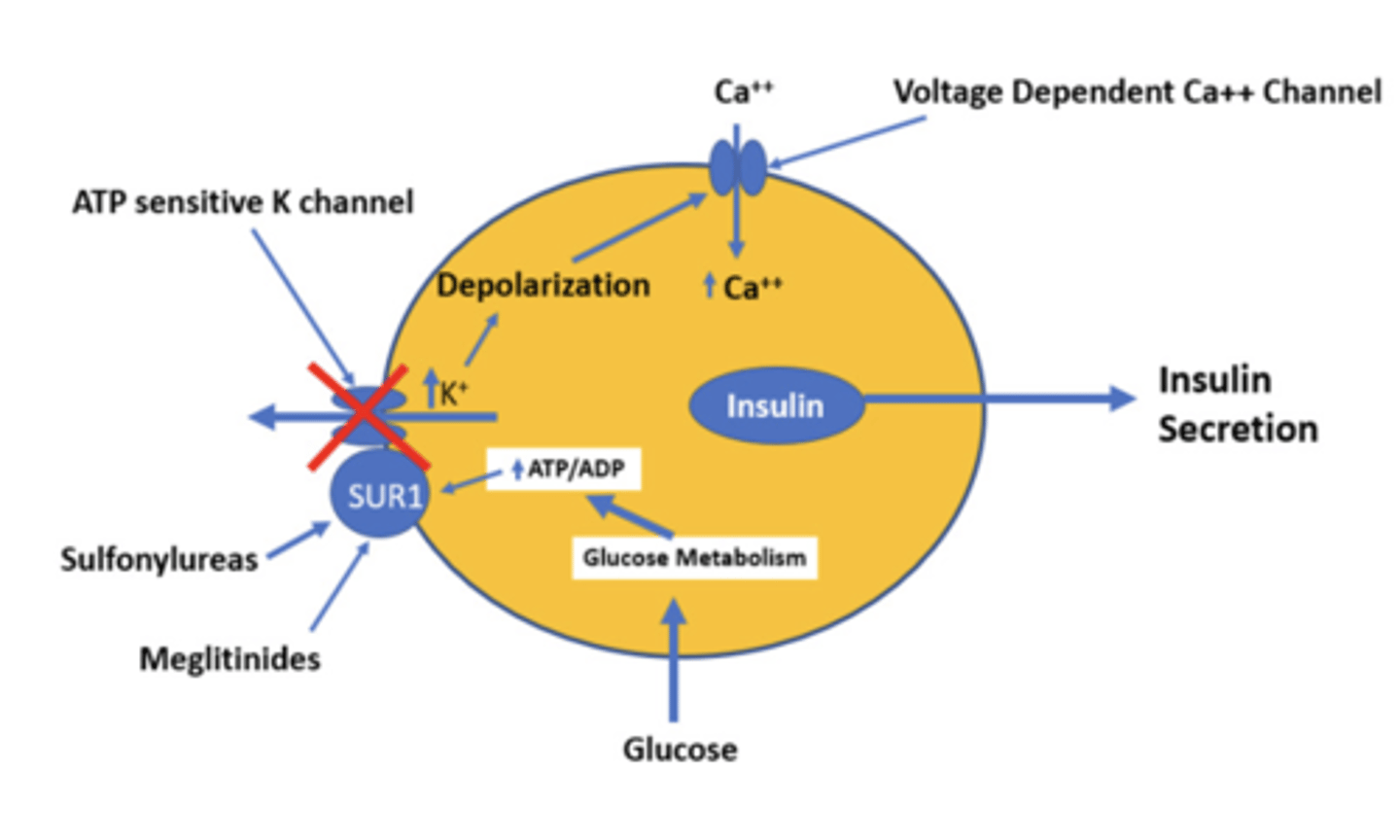
Meglitinides repaglinide, nateglinide
MOA: bind to SUR1 (ATP-sensitive K+ channel) on β-cells = prevent K+ efflux =
β-cell depolarization = Ca2+ influx = stimulates insulin secretion
same moa as Sulfonylureas (2nd generation) glipizide, glyburide, glimepiride but it has a shorter T1/2 life to lower risk of hypoglycemia
Clinical indications: type 2 DM
Adverse effects: hypoglycemia, weight gain, upper respiratory tract infection
PRO: Although meglitinides have the same MOA as sulfonylureas, they have a reduced risk of hypoglycemia as they have a shorter half-life.
CON: Frequent dosing (three times/day prior to meals)
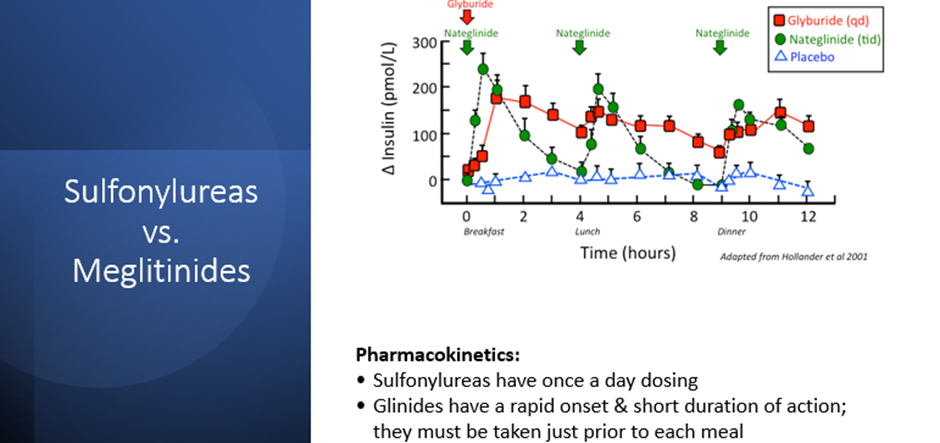
sulfonylureas vs. meglitinides
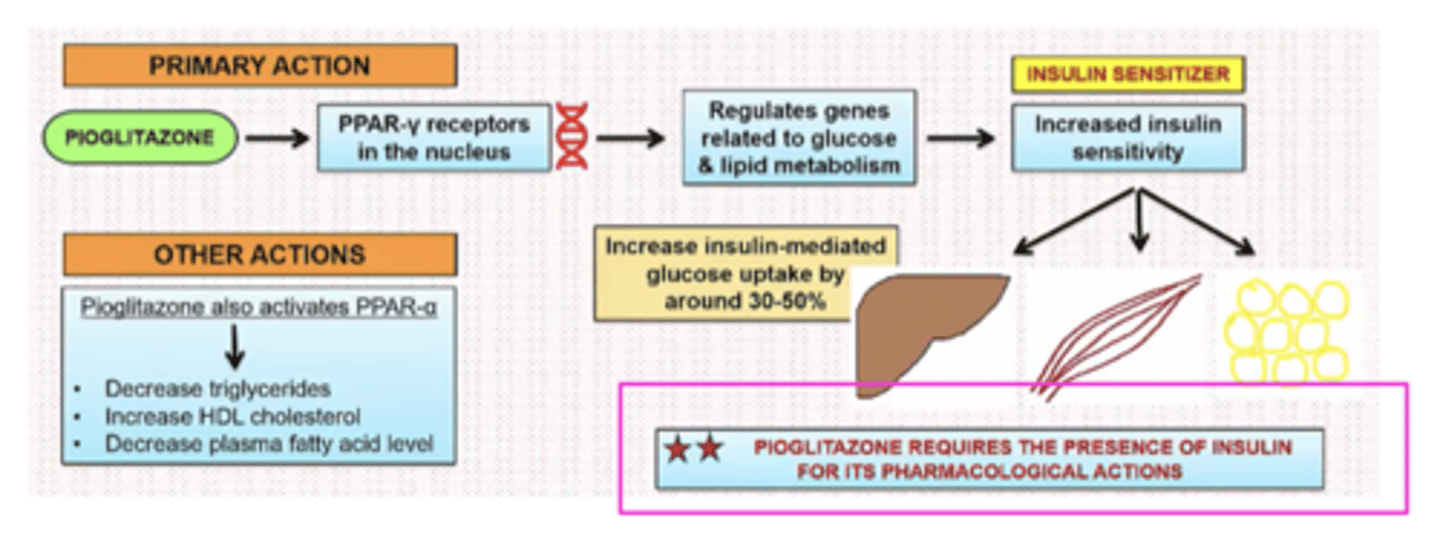
Pioglitazone (NOT TESTED)
PPARγ increases the transcription of insulin-sensitive genes
MOA: stimulate PPARγ = Increase in glucose uptake (adipose, muscle, and liver), lipogenesis (adipose, liver), glycolysis and glucose oxidation (muscle), and decrease gluconeogenesis and glycogenolysis
requires the presence of insulin for its pharmacological actions
Clinical indications: type 2 DM
Adverse effects: fluid retention, edema, and CHF; bladder carcinoma, bone fractures, hepatoxicity
Boxed warnings: CHF
Contraindications: patients with class III or IV HF
PROS: reduces blood pressure, enhances vascular function, good for atherosclerotic disease, low risk of hypoglycemia
CONS: adverse effects in CHF; causes weight gain
For the initial treatment of type 2 diabetes, some patients are candidates for insulin as their initial therapy:
Severe hyperglycemia on presentation
Difficulty distinguishing type of diabetes
Pancreatic insufficiency
Other

balancing risks and benefits for personalized goals
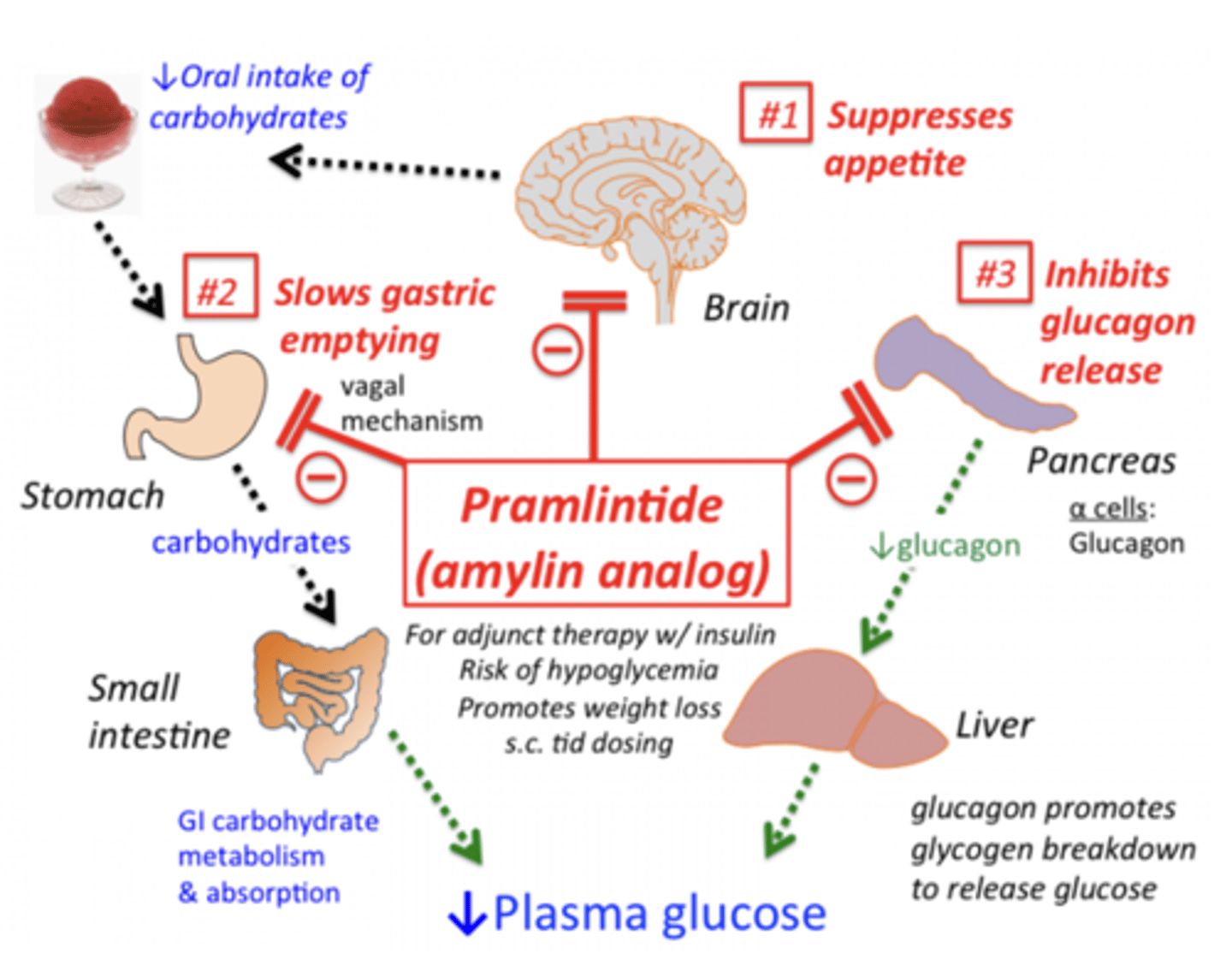
amylin analog (SQ) pramlintide (NOT TESTED)
Amylin is cosecreted with insulin from b-cells after mealtime to slow gastric emptying, suppress appetite, and reduce blood glucose by decreasing glucagon release.
MOA: amylin analogue = same effects as amylin (delayed gastric emptying, appetite suppression, and inhibited glucagon release)
Clinical indications: adjunct treatment in patients with type 1 or type 2 DM who use mealtime insulin therapy and who have failed to achieve desired glucose control despite optimal insulin therapy
Administered in area distinct from insulin
CON: injected at every meal – rarely used because of injection frequency - Falling out of favor to the GLP-1 agonists or tirzepatide which can be administered once per week
Adverse effects: GI (nausea), hypoglycemia, injection site reactions
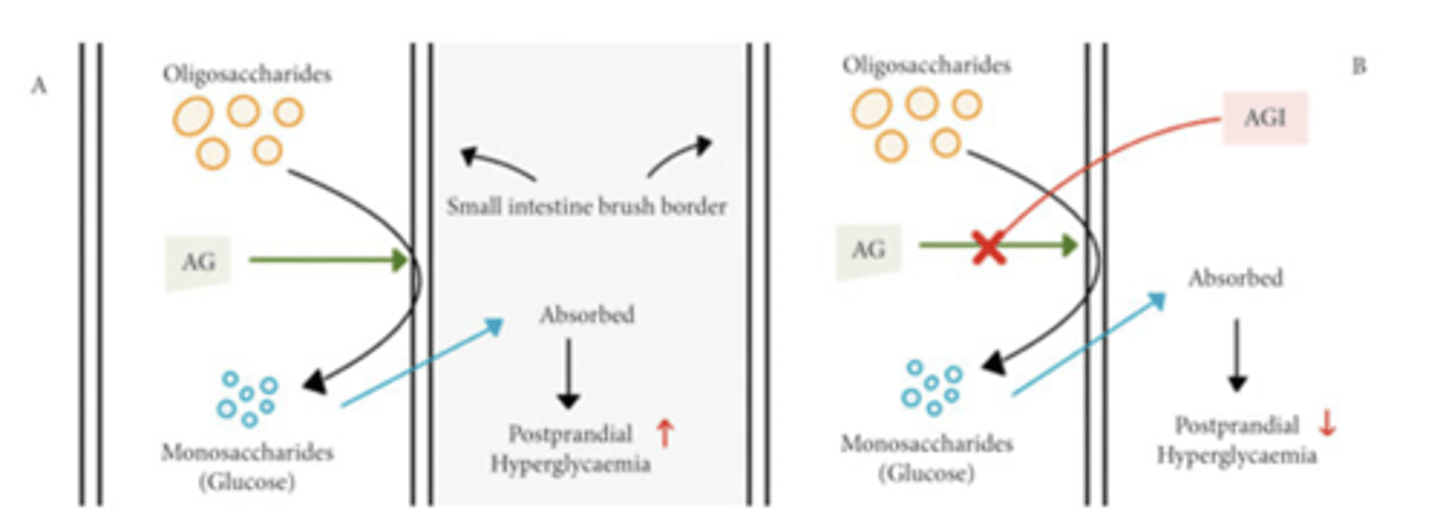
α-Glucosidase Inhibitors acarbose, miglitol (NOT TESTED)
α-glucosidase: small intestine enzyme that hydrolyzes dietary carbs into monosaccharides
MOA: inhibit α-glucosidase = competitively inhibit the hydrolysis and absorption of starch, dextrin, and disaccharides (AKA inhibitors of intestinal glucose absorption)
Should be administered with the first bite of a meal
Clinical indications: type 2 DM
Adverse effects: GI (flatulence, diarrhea, abdominal pain) – significant!
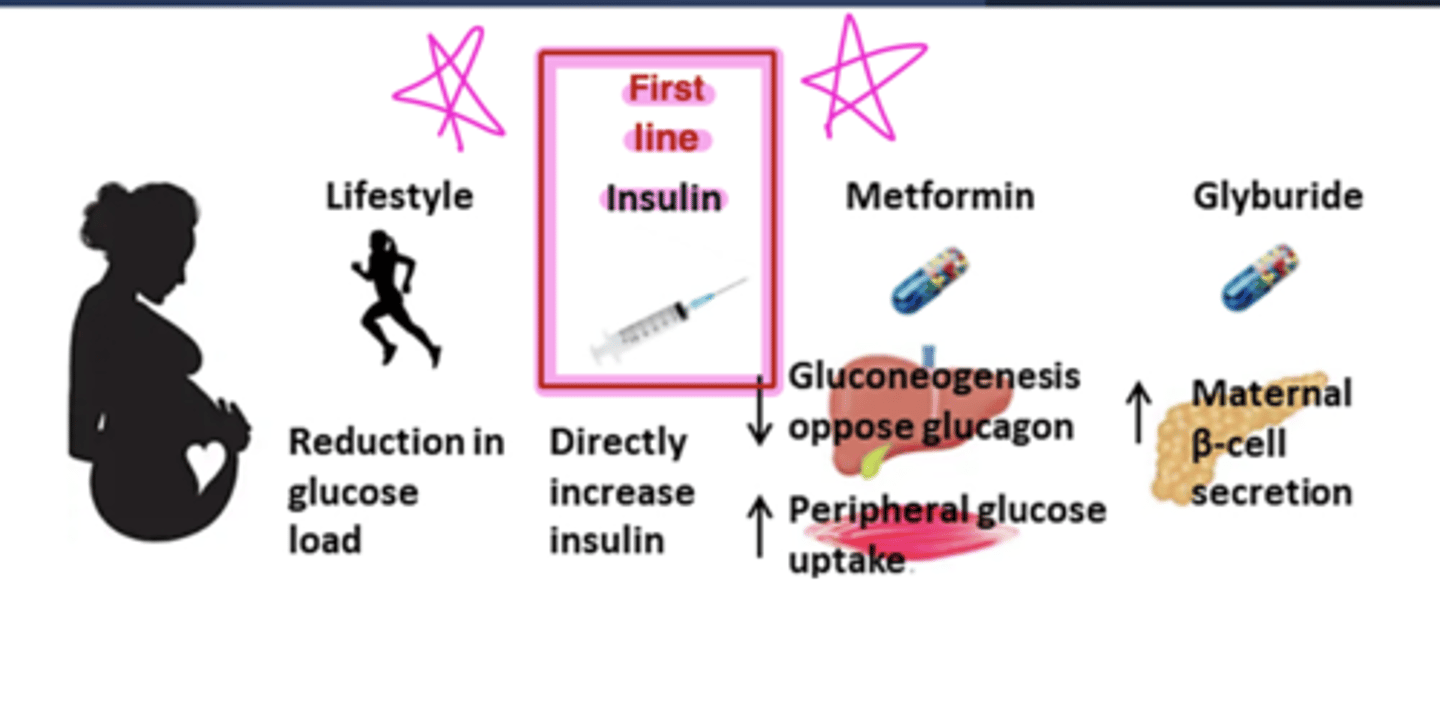
glucose management in gestational diabetes
atherosclerotic cardiovascular disease (ASCVD)
Benefit:
SGLT2 inhibitors (canagliflozin and empagliflozin)
GLP1-agonists (semaglutide, liraglutide, and dulaglutide)
Potential benefit:
Metformin, pioglitazone
Neutral:
DPP4 Inhibitors, Insulin, newer generation sulfonylurea (glimeperide)
congestive heart failure
Benefit:
SGLT2 inhibitors (canagliflozin, empagliflozin, dapagliflozin)
Neutral:
Metformin, GLP-1 agonists, sulfonylurea, insulin
Increased Risk:
Pioglitazone
Potential Risk:
DPP4 inhibitors (saxagliptin)
weight change
Loss:
SGLT2 inhibitors - less effective
GLP1 agonists - more effective
GIP+GLP1 agonist - more effective
Neutral:
Metformin (potential modest loss)
DPP4 inhibitors
Gain:
Pioglitazone
Sulfonylureas
Insulin
hypoglycemia risk
High risk:
Sulfonylurea
Insulin
Low risk:
Metformin
SGLT2 inhibitors
GLP1 agonists
Pioglitazone