Organic Chemistry II: Exam 2 Material
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is a diene?
Molecule with 2 double bonds
Isolated Diene
alkenes are separated by one sp3 carbon
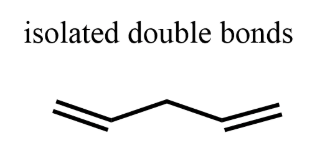
Conjugated Diene
alkenes that are not separated by an sp3 carbon, all are delocalized
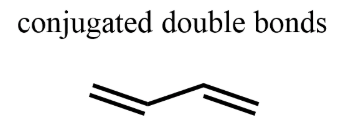
Why are conjugated dienes more stable than isolated?
Conjugated dienes release less energy when bonds are broken in a reduction reaction
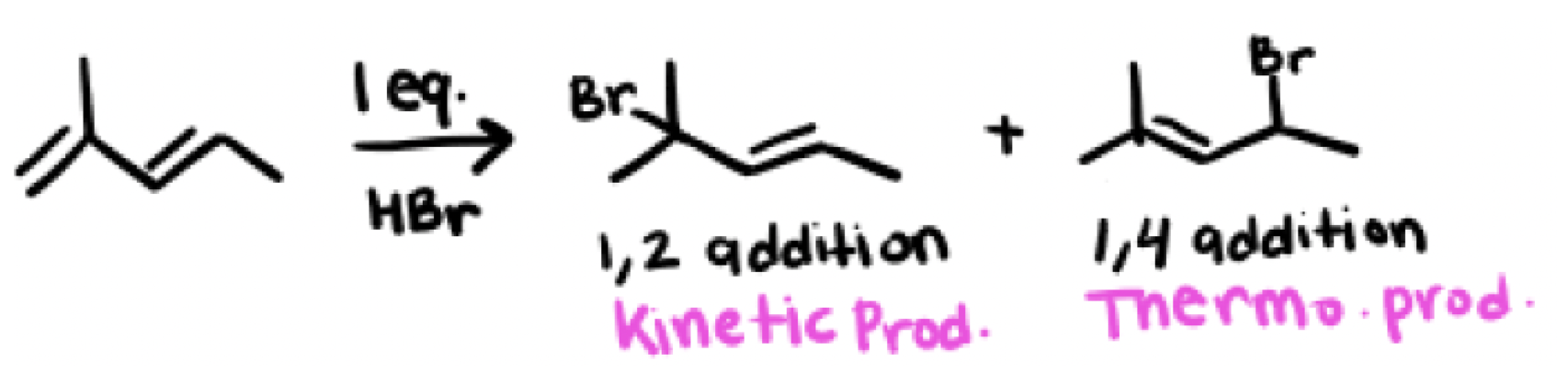
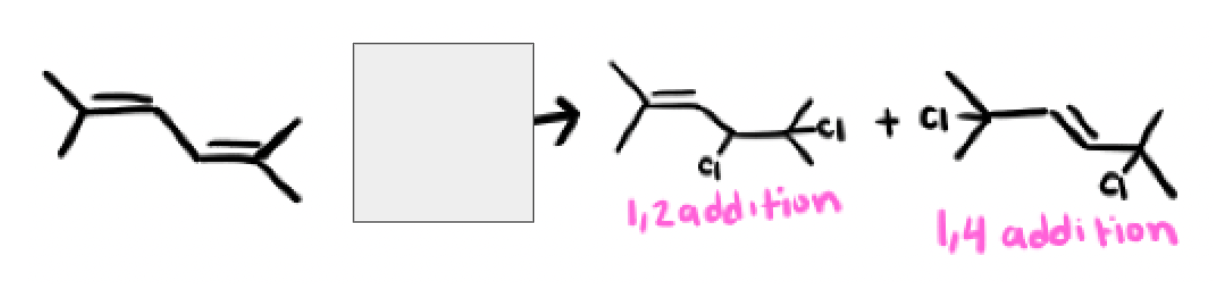
In an hydrohalogenation reaction with dienes (HX) what are the two products you get? Which one is major and which one is minor?
Kinetic product and Thermodynamic product
Major and minor products depend on the temperature, cold environments favor kinetic products while hot environments favor the thermodynamic product, room temperature environments gives a 1:1 of both products
Why do hot environments promote the thermodynamic product?
The heat gives the system enough energy activation to rearrange the carbocation and form a more stable alkene
The kinetic product forms via the ______
proximity effect
in most cases the 1,4 product (usually thermodynamic product) will be the ____
most substituted alkene, most stable (zaitsev)
In a free energy diagram, how can you tell what the thermo and kinetic product are?
The thermo product will have a slightly higher activation energy bump than the kinetic product after the carbocation formation. At the end of the graph, the thermo product will have a lower position than the kinetic product
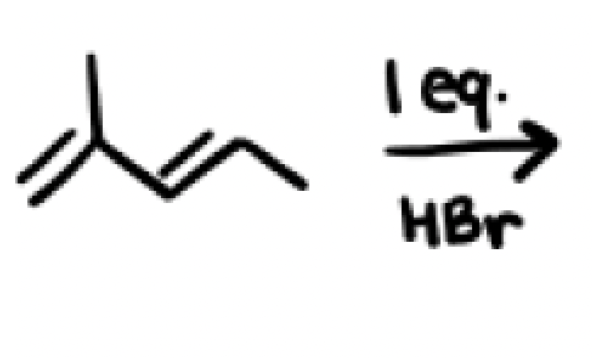
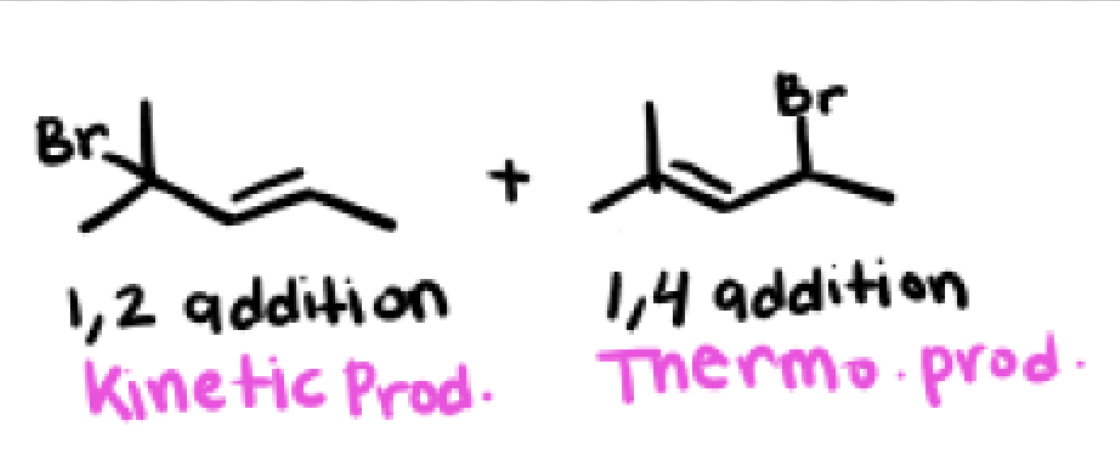
Can a product be both kinetic and thermodynamic?
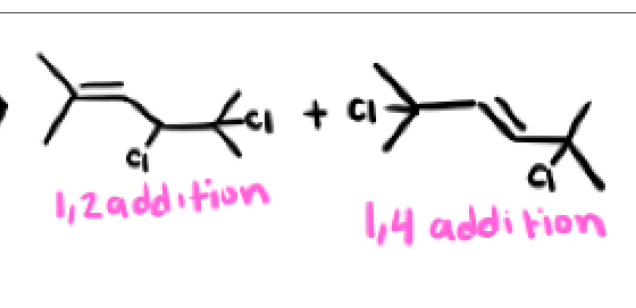
Yes, there are exceptions, as long as the 1,2 addition product has the most stable (most substituted) alkene than the 1,4 addition product, it can be thermodynamic
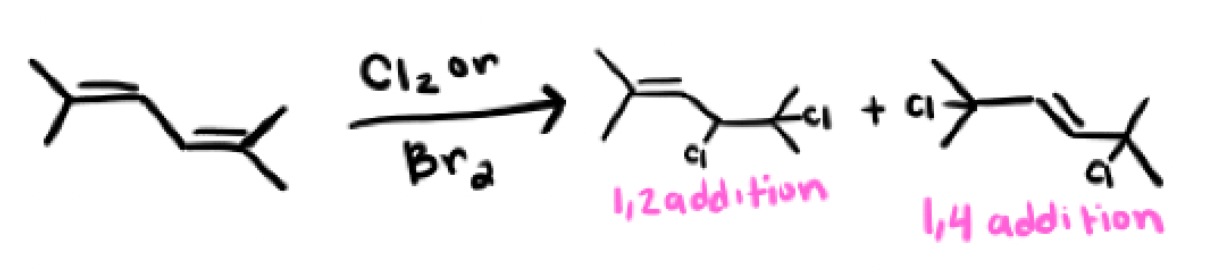
In a halogenation reaction, where do you start and why?
Always start at the most substituted area for the most stable carbocation intermediate
In a halogenation reaction, where would the halide attack?
the halide would attack the most substituted carbon to open up the intermediate
What is another way to open up the intermediate in a halogenation reaction?
Pushing the double bond
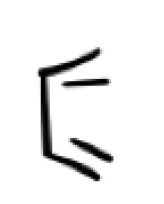
what is this? what does it act as?
Diene in an s-cis conformer position, it acts as the nucleophile in a diels alder reaction
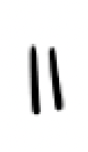
what is this? what does it act as?
dienophile, it acts as the electrophile in a diels alder reaction
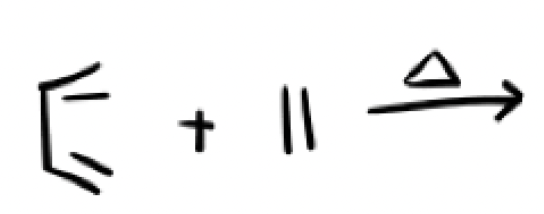
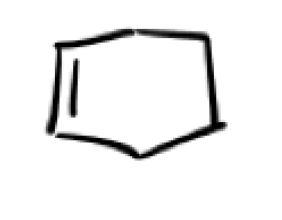
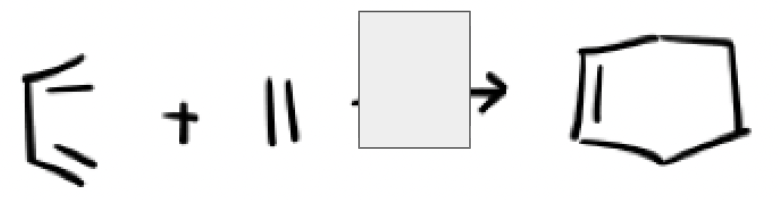
heat (triangle)
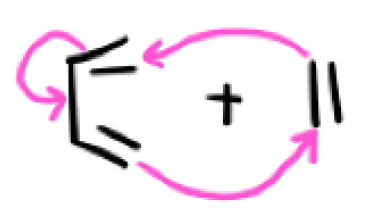
What are the mechanism arrows here?
If you want your dienophile to be reactive, what do you need?
Electron Withdrawing Groups
If you want a reactive diene, what do you need?
Electron Donating Groups, a locked diene, and more outside groups (due to less steric hindrance, inside groups experience a lot of repulsion)
what is a locked diene? why is it good?
A diene that has no free rotation to move, it works really well in a diels alder reaction because you don’t need any additional rotation (no extra energy needed for it since it cant move)
Electron Withdrawing Groups (EWG)
groups that withdraw electron density at positions 5 and 6 of the dienophile
Electron Donating Groups (EDG)
atom that has a lone pair directly connected to the diene/dienophile
Why do EWG increase reactivity?
It puts a partial positive charge on the dienophile
What are some EWG?
Ketones, esters, carboxylic acids, amides, cyano groups, and nitro groups
What are some EDG?
hydroxyl groups, ethers, amines, alkyl groups (via hyperconjugation)
Why are EDG not the greatest for a dienophile?
it makes a less electrophilic site, which is not desirable for an electrophile
Why do we use heat in a diels alder reaction?
Because the s-cis conformer is less stable than s-trans conformer, heat allows us to push the equilibrium to the less stable conformer
Stereochemistry of ___ can affect reactivity
diene
List in increasing order of reactivity: normal diene, locked diene, diene with EDG
normal diene < diene with EDG < locked diene