Patho II Exam 2
1/110
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
111 Terms
What is the function of the kidneys?
Filtering waste products (urea → protein metabolism, creatinine → muscle metabolism)
Fluid balance (release renin when low blood volume, BP; RAAS)
Electrolyte balance (Maintains serum Na⁺, K⁺, Ca²⁺, phosphate, and
bicarbonate)
Acid-base regulation (maintains pH by excreting H+, reabsorbs bicarbonate; kidney fails = metabolic acidosis)
Produces / activates hormones:
Erythropoietin
Vitamin D activation
How does Erythropoietin relate to the kidneys?
Produced by cells in kidney → triggered by low oxygen → stimulates bone marrow → increase RBC production
If kidneys fail → ↓ EPO → ↓ RBCs → anemia
How does the kidney activate Vitamin D?
Kidneys activate Vitamin D → Calcitriol (active form) → ↑ Calcium absorption in intestines + maintains bone strength
What labs indicate Kidney failure?
↑ BUN
↑ Creatinine
Low GFR
What is the normal GFR value?
~125 mL / min
Low = kidney failure
You can lose >75% of nephrons before symptoms→ why CKD is silent early
What is the normal urine output value?
30 to 60 mL / hr
What is Glomerulopathy?
A broad term of disease affecting the kidneys glomeruli (filter)
Ex. Glomerulonephritis
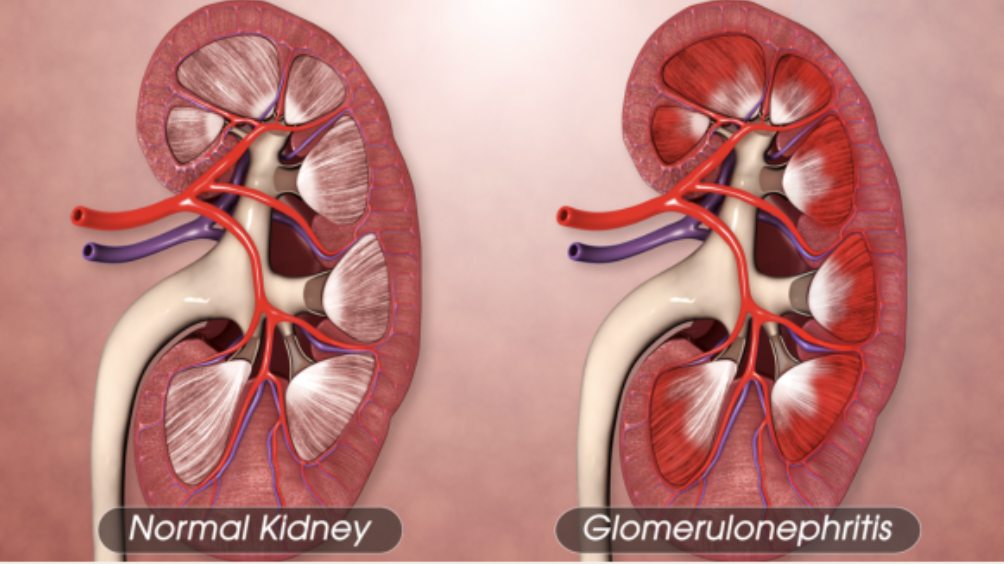
What is Glomerulonephritis?
Immune-mediated inflammation of glomerulus, impairing the kidneys’ ability to filter waste and fluids effectively
What are symptoms of Glomerulonephritis?
Hematuria and RBC casts found in urine
Proteinuria (protein in urine)
Decrease GFR
Hypertension (retention of fluid → increase BP)
What is the treatment of Glomerulonephritis?
Corticosteroids and immunosuppressants (prevent inflammation + damage to glomeruli)
BP control
Dietary and fluid restriction
Chronic form → risk of ESRD → dialysis or transplant
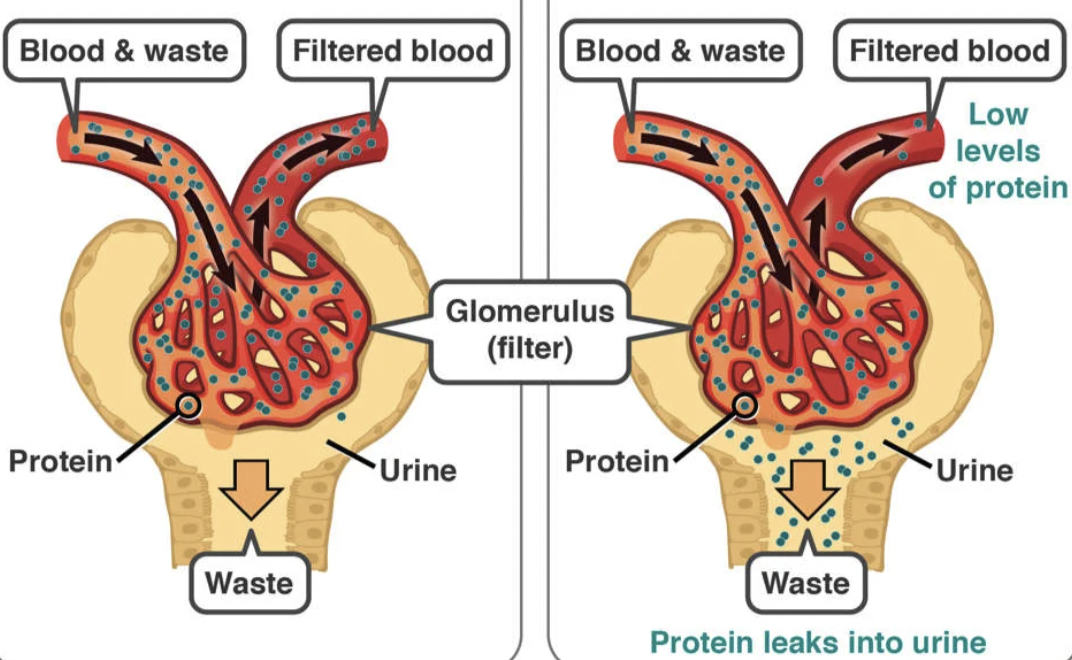
What is Nephrotic Syndrome?
Damage to glomerular barrier causing massive protein loss
What is the cascade of Nephrotic Syndrome?
The filtration barrier is broken down → large proteins escape in urine (albumin)
Albumin loss in urine → hypoalbuminemia
Fluid leaks from blood vessels to tissues → edema
Liver detects low protein levels → release albumin + lipoproteins
What are signs of Nephrotic Syndrome?
Edema
Hyperlipidemia (from overproduction of lipoproteins)
Hypercoagulability → risk of DVT / PE (losing antithrombin III, clotting factor)
Infection risk (lost antibodies)
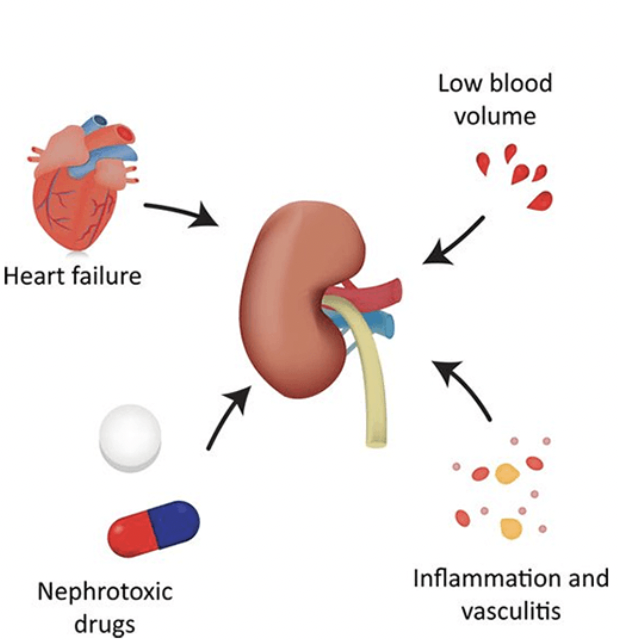
What is Acute Kidney Injury (AKI)?
Sudden episodes of kidney failure or damage occuring within hours or days
What are the common types of AKI?
Prerenal (before the kidneys / before entering the kidneys)
Intrarenal (within the kidneys)
Postrenal (after the kidneys)
Prerenal AKI
Decrease blood flow into to the kidneys, caused by:
Dehydration
Hemorrhage
Heart failure
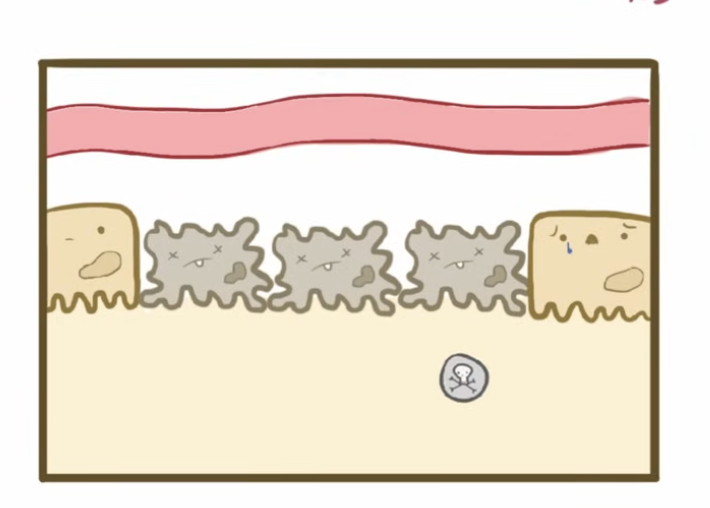
Intrarenal AKI
Direct damage to kidney tissue, commonly caused by acute tubular necrosis or nephrotoxins
Glomerulonephritis also a type
What is acute tubular necrosis (ATN)?
Epithelium cells that line the tubules die from ischemia → kidney damage
What is Oliguric Phase of ATN?
Decrease in urine output → accumulation of K+
K⁺ = 7.8 → can cause fatal arrhythmias
How is Oliguric Phase treated?
Sodium Polystyrene Sulfonate (Kayexalate) → removes potassium through GI tract
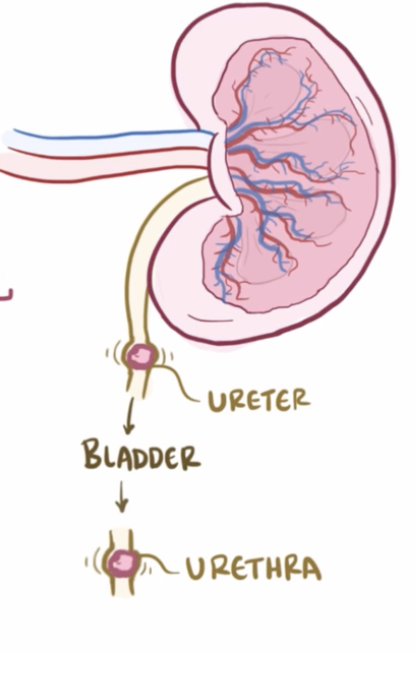
Postrenal AKI
Obstruction below the kidneys such as BPH, stones, tumors
What are the priority interventions for AKI?
Identify high-risk patients early: Dehydration, sepsis, rhabdomyolysis, contrast dye exposure
Maintain fluid volume & cardiac output (Adequate perfusion protects tubular cells)
Avoid nephrotoxins (NSAIDs, aminoglycosides, IV contrast — use with caution or avoid)
What is Chronic Kidney Disease (CKD)?
Progressive, irreversible kidney damage staged 1-4; damaged kidneys cannot filter blood properly, causing waste to build up over three or more months
What kills CKD patients?
Cardiovascular disease → fluid overload, heart strain, accumulation of toxins
How can the progression of CKD be slowed?
Glycemic control (diabetic nephropathy → high blood sugar damages nephrons)
ACE inhibitors + ARBs (reduce proteinuria and slow nephron loss)
Control BP (hypertension accelerates CKD progression)
Reduce CVD risk
What are complications of CKD?
Anemia (↓ EPO → ↓ RBCs)
Metabolic Acidosis (can’t excrete H+ or reabsorb bicarbonate) → ↓ pH, ↓ HCO₃⁻
Drug clearance (dose adjustments required, especially in older adults)
What should you increase in CKD diet?
Calories → prevent muscle breakdown
Calcium → bone health
Vitamin D → the kidneys can’t activate it anymore
What should you restrict in a CKD patient?
Potassium → body can’t excrete anymore; prevents arrhythmias
Phosphorus → high phosphorus pulls calcium from bones; prevents bone disease
Sodium → controls BP + fluid
Fluids → prevent overload
Protein → reduces BUN load + accumulation of urea (waste)
What treatment is used for End-Stage Renal Disease (ESRD)?
Dialysis (hemo or peritoneal)
Kidney transplant
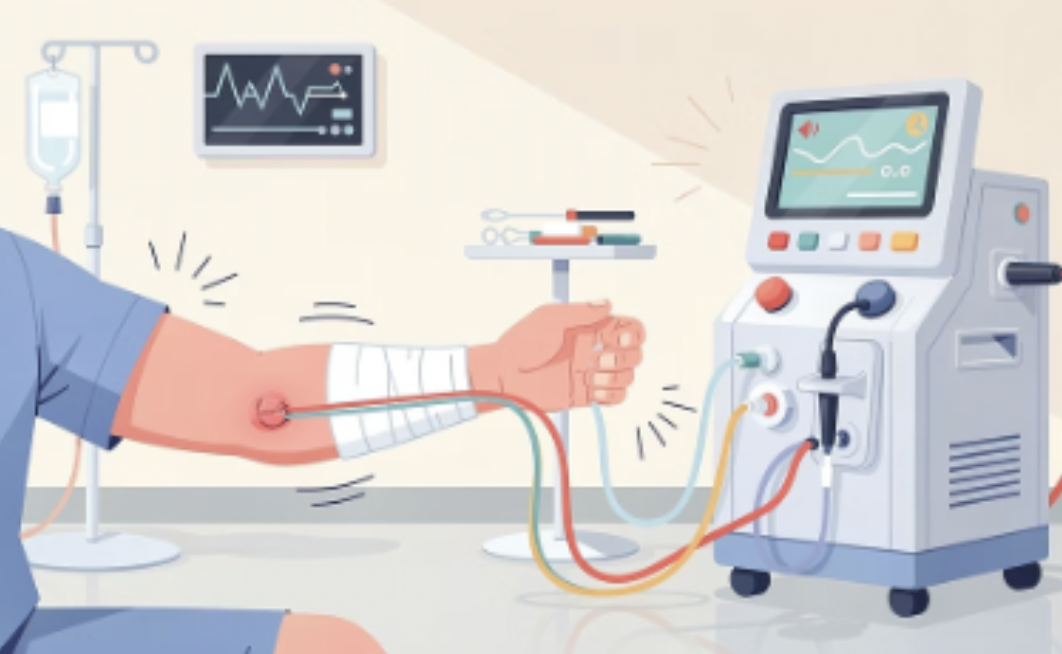
Hemodialysis
Uses AV fistula or graft and done 3x / week
What are nursing priorities for hemodiaylsis?
Assess for bruit & thrill (whoosing sound → indicates bloackage)
No BP or IV on that arm with fistula
What is post-treatment after hemodialysis?
Watch for hypotension, dizziness, and nausea
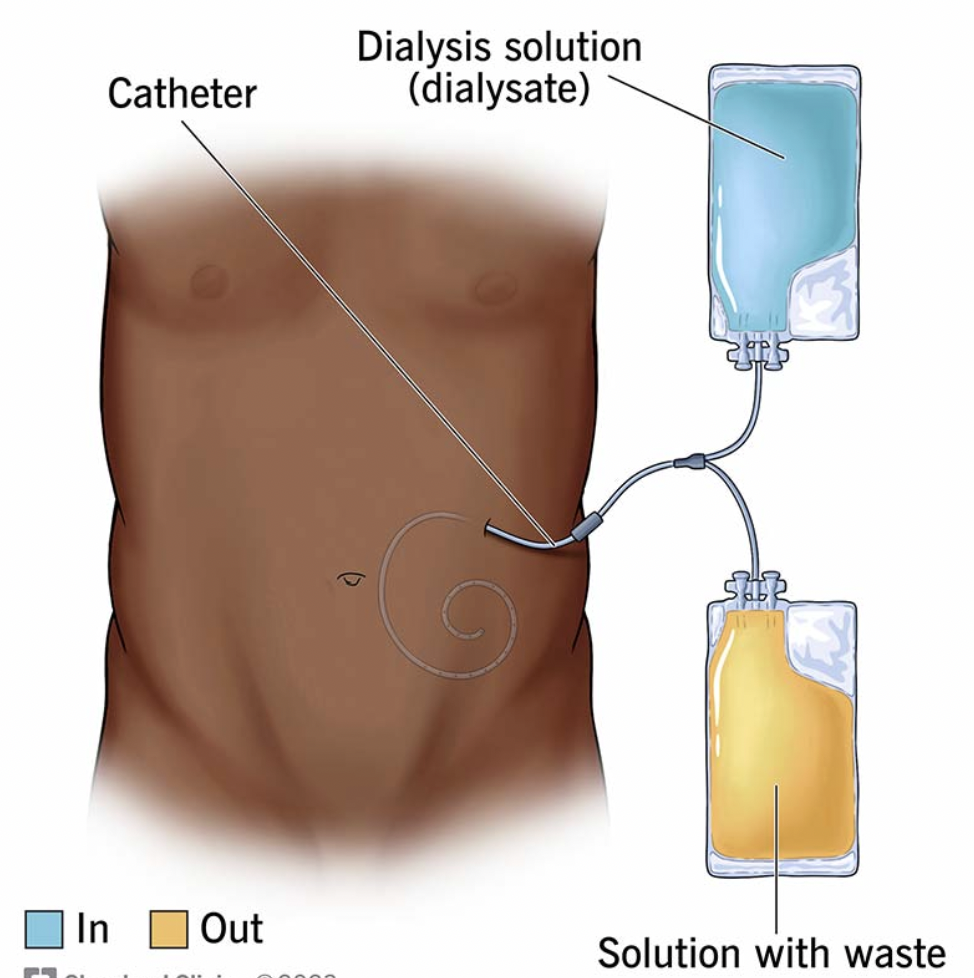
Peritoneal Dialysis
Uses abdominal lining to filter waste and excess fluid; Fluid removed via osmosis (solution high in glucose → draws fluid out)
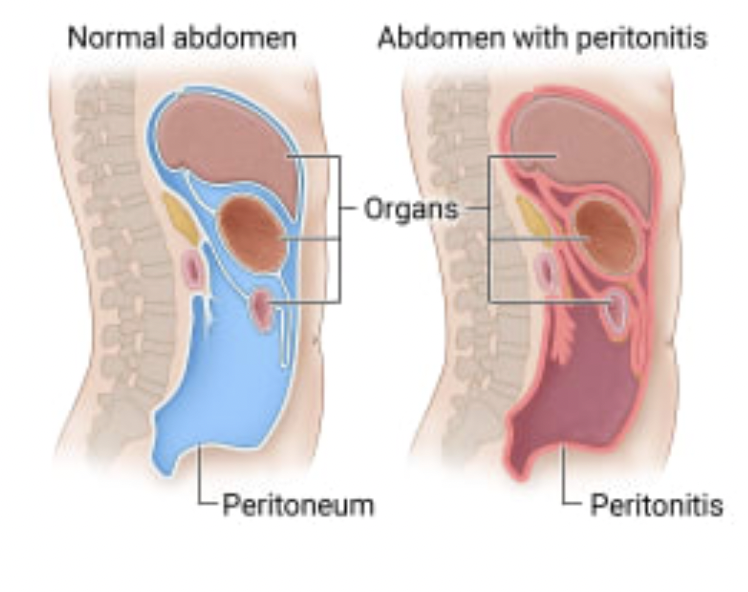
What are complications of Peritoneal Dialysis
Peritonitis → redness + swelling of lining of abdomen; will manifest as cloudy dialysate drainage
What medication is given to a patient with a Kidney Transplant?
Lifelong Tacrolimus (Prograf) → monitor for hypertension and renal insufficiency
What are signs of acute rejection of Kidney Transplant?
Fever, jaundice, dark urine
May also see Cirrhosis (liver scarring)
What are diuretics?
Medications that increase sodium excretion; “Water follows sodium”
What indications for using diuretics?
Edema from heart failure, liver disease/cirrhosis, renal disease
Acute pulmonary edema
Hypertension
Glaucoma (reduces intraocular pressure)
What are the classes of diuretics?
Thiazide & Thiazide-Like Diuretics
Loop Diuretics
Carbonic Anhydrase Inhibitors
Potassium-Sparing Diuretics
Osmotic Diuretics
Thiazide Diuretics
Blocks chloride pump in kidney distal convoluted tubule; First line therapy for essential hypertension
Ex. Hydrochlorothiazide, Chlorothiazide, Metolazone
What are side effects of Thiazide Diuretics?
Hypokalemia (↓ K⁺ → digoxin toxicity if too low)
Hypercalcemia (↑ Ca²⁺)
Hyperuricemia (excess uric acid in blood, gout)
Hyperglycemia
What is the onset, duration, and peak of Thiazide Diuretics?
Oral onset: 2 hr
Peak: 4–6 hr
Duration: 6–12 hr
Loop Diuretics
Powerful diuretic that blocks chloride pump in ascending loop of Henle; drug of choice for acute heart failure and pulmonary edema
Ex. Furosemide/Lasix, Bumetanide, Torsemide, Ethacrynic Acid
What are side effects of Loop Diuretics?
Hypokalemia (increased digoxin toxicity risk)
Hypocalcemia, Hypomagnesemia, Hypotension
Hyperglycemia, Hyperuricemia
Alkalosis
Ototoxicity (never IV push rapidly; avoid with aminoglycosides or cisplatin)
What are important nursing interventions when giving Loop Diuretics?
Administer early in the day to avoid nighttime diuresis
Monitor weight daily (report gain of 3 lbs/day or 5 lbs/week)
NSAIDs reduce diuretic effectiveness
Interstitial pulmonary edema post-hypovolemic shock: Furosemide + Albumin
Carbonic Anhydrase Inhibitors
Block carbonic anhydrase at the proximal tubule → sodium bicarbonate and H⁺ lost in urine; primarily used for Glaucoma, mountain sickness, epilepsy, heart failure, and edema
Ex. Acetazolamide, Methazolamide
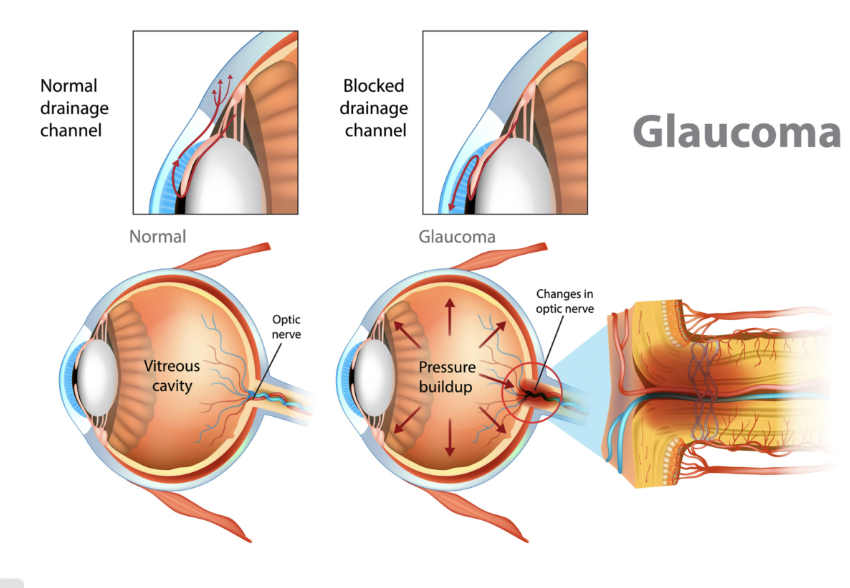
How does Carbonic Anhydrase Inhibitors work on Glaucoma?
Reduce intraocular pressure by decreasing aqueous humor production
What are side effects of Carbonic Anhydrase Inhibitors?
Metabolic acidosis (loss of bicarbonate in urine)
Hypokalemia
Neuro Effects:
Paresthesia
Confusion
Drowsiness
What are contraindications for Carbonic Anhydrase Inhibitors?
High-dose aspirin (additive acidosis)
Sulfonamide or thiazide allergy (cross-sensitivity)
Potassium-Sparing Diuretic
Prevent body from losing too much potassium (hypokalemia). Used for patients on digoxin, antiarrhythmics, or with arrhythmias; also 1st choice for hyperaldosteronism (cirrhosis, nephrotic syndrome)
Ex. Spironolactone/Aldactone, Eplerenone, Amiloride, Triamterene
What are side effects of Potassium-Sparing Diuretics?
Hyperkalemia: lethargy, confusion, muscle cramps, fatal arrhythmias.
Hormonal effects for Spironolactone:
gynecomastia (enlarged male breasts), decreased libido, hirsutism (excessive dark hair growth in women), menstrual changes
Eplerenone does NOT cause these
What contraindications of Potassium-Sparing Diuretics?
Avoid K⁺-rich foods if levels elevated
Do not give with ACE inhibitors ARBs w/o checking K+ levels
Severe renal disease
Anuria
What organ are Potassium-Sparing diuretics good for?
Spironolactone and eplerenone improve survival in Heart failure
Osmotic Diuretics
Pulls water into bloodstream → excreted; IV administration only. Used for increased intracranial pressure, acute glaucoma, acute renal failure (prevents oliguric phase), and toxic substance clearance
Ex. Mannitol
What are side effects of Osmotic Diuretics?
Rapid fluid shifts can cause acute heart failure or pulmonary edema
What are contraindications for Osmotic Diuretics?
Severe renal disease
Pulmonary congestion
Heart failure
Dehydration
Intracranial bleeding
NOT used in heart failure or cirrhosis.
What are core nursing actions across all diuretics?
1. Daily weights
Best indicator of fluid status
2. Monitor labs
K⁺ (most important)
BUN/Creatinine
3. Give in morning
Prevents nocturia
4. Watch for orthostatic hypotension
Due to volume loss
What is the connection of potassium and diuretics?
Thiazides, Loop Diuretics: decrease K+
monitor K + closely + encourage dietary potassium
Potassium-Sparing Diuretics: increase K+
limit K+ rich foods, avoid K+ supplementation unless directed by provider
Digoxin + loop/thiazide:
increases the risk of digoxin toxicity
Level of 2.4 ng/mL → hold dose, notify provider
What is pain?
Unpleasant sensory and emotional experience triggered by actual or potential tissue damage; subjective
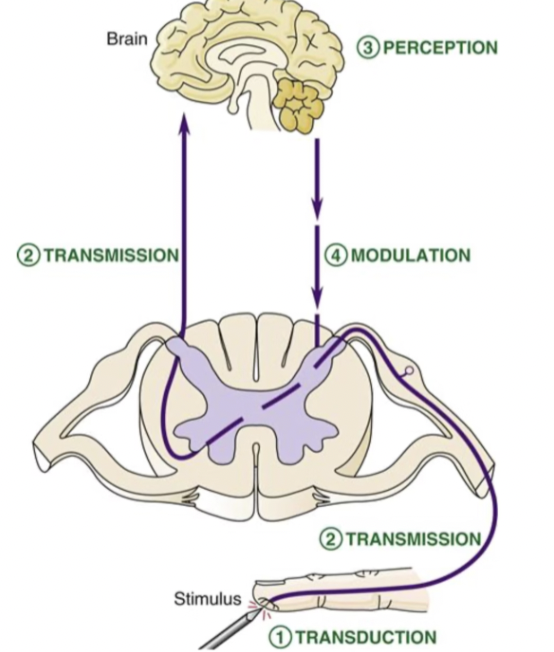
What are the 4 steps of pain?
Transduction (Where pain STARTS)
Transmission (Signal traveling)
Perception (Pain becomes “real”)
Modulation (Pain control system)
Transduction
Happens at site of injury:
Tissue is damaged → cells release arachidonic acid + kinins
Enzyme cyclooxygenase (COX) converts arachidonic acid → prostaglandins
Prostaglandins sensitize nociceptors → lower pain threshold (even light stimuli can feel painful now)
How do NSAIDs work on transduction?
Block COX → ↓ prostaglandins → stops pain before it even starts
Ex. Ibuprofen
Transmission
Pain signal travels:
Nerve fibers (A-delta and C fibers) → enter spinal cord (dorsal horn) → travels up the spinothalamic tract → brain (thalamus then cortex)
What is the difference between A-delta fibers and C fibers?
A Delta Fibers:
Fast (myelinated)
Sharp, stabbing pain (felt immediately after injury)
Well localized
Ex. Needle stick = sharp pain
C Fibers:
Slow (unmyelinated)
Dull, aching, burning
Diffuse (more numerous)
Ex. Needle stick = lingering soreness
Perception
The brain recognizes the signal as pain
Assigns meaning and adds emotional response
Clinical example: morphine changes how brain perceives pain, though it still exist
Modulation
The brain sends signals to spinal cord (descending pathway) to either inhibit pain or enhance it
What is the Gate Control Theory?
Pain is regulated in the spinal cord (dorsal horn) like a gate, can either be closed or open
What happens when the Gate is closed?
Pain is blocked / lessened from activation of large A fibers through pressure or vibration
Ex, Touch, massage, heat, relaxation, positive emotion
What happens when the Gate is open?
Pain is amplified; Small fiber dominate
Ex. Anxiety, fatigue, fear, focus on pain
What are the different categories of pain?
Duration:
Acute
Chronic
Source:
Nociceptive
Neuropathic
Psychogenic
Location:
Somatic
Visceral
Referred
What are the types of duration pain?
Acute: Short-term pain from recent injury or illness with clear cause and resolves as healing occurs
Chronic: Pain lasting beyond normal healing (months) and may persist without clear injury
What are the types of source pain?
Nociceptive: caused by actual tissue damage or inflammation
Neuropathic: caused by nerve damage or dysfunction
Psychogenic: pain influenced by psychological factors; brain amplifies pain without clear physical cause, but is real to the patient
What are the types of location pain?
Somatic: Well localized in skin, muscle, bones, joints
Visceral: Hard to localize, internal organs
Referred: Felt somewhere else than the source b/c different organs share the same nerve pathways in the spinal cord
What is an example of Referred pain?
Heart attack → arm/jaw pain
Diaphragm irritation → shoulder pain
What are Endogenous opioids?
Our body’s natural painkillers such as endorphins and enkephalins that regulate pain, stress, and reward
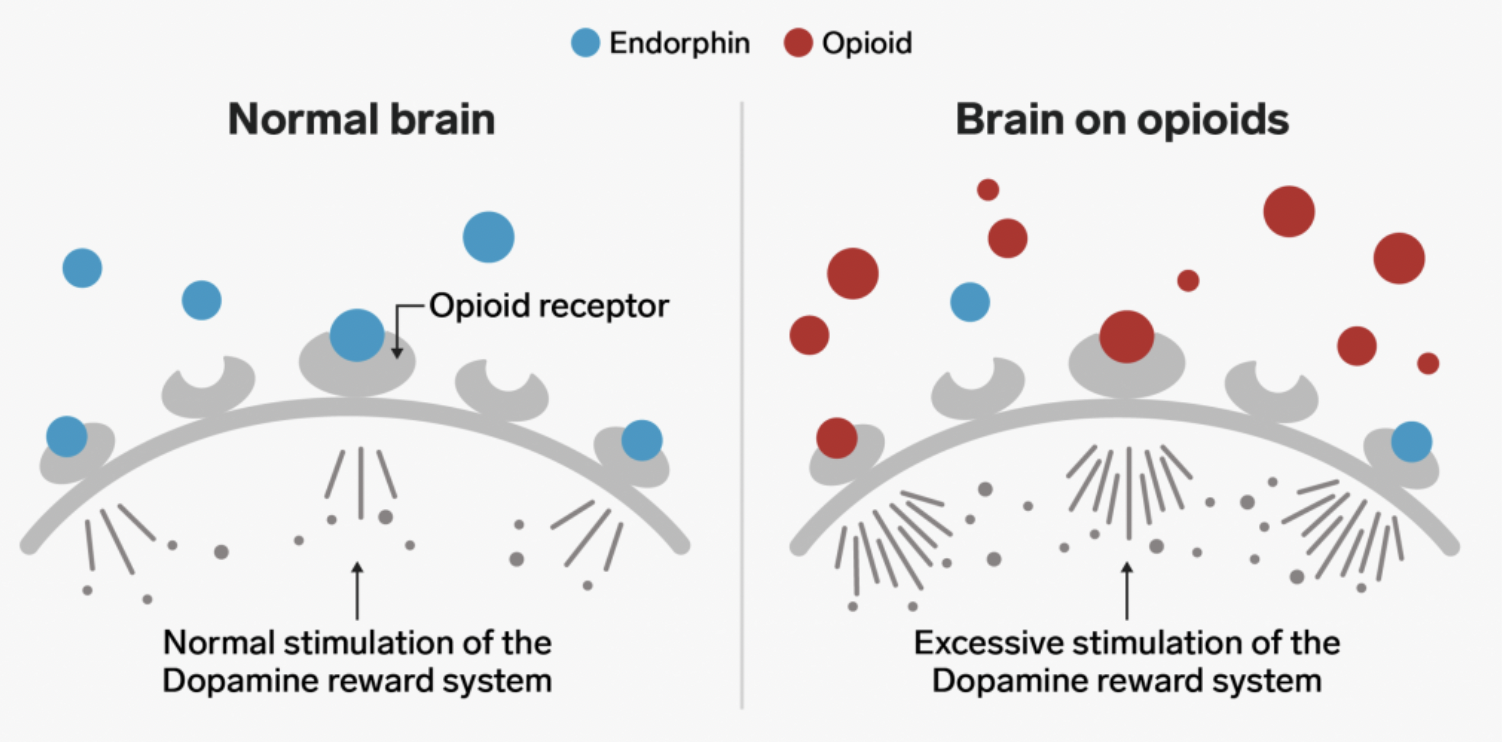
How do Endogenous opioids work?
Bind to opioid receptors (Mu (μ) and Kappa (κ) Receptors) on G protein-coupled receptors to reduce pain
What are Opioid Agonists?
Exogenous opioids: External substances introduced to the body to treat severe pain → activate the same receptors but carry a higher risk of addiction

What is the effect of activating Mu (μ) receptor?
Potent analgesia (pain relief)
Respiratory depressants (dangerous)
Euphoria and sedation
Decreased GI motility → constipation
Pupil constriction (miosis)
Physical dependence with prolonged use
What is the effect of activating Kappa (κ) receptors?
Secondary analgesia (sedation)
Dysphoria (unpleasant mood — opposite of euphoria)
Pupil constriction (miosis)
What are the common Opioid Agonist medications?
Morphine
Fentanyl
Hydromorphone (Dilaudid)
Hydrocodone / Oxycodone
Methadone
Tramadol
Morphine
Used for moderate to severe pain, post op, and MI; IV onset is immediate
Baseline opioid everything is compared to
What are risk to assess for using Morphine?
RR before IV dose → Respiratory depression
BP → Hypotension (vasodilation)
Constipation
Fentanyl
Very potent opioid
Chronic severe pain → patch
Surgery → IV (fast, potent)
What are risk to assess for using Fentanyl?
RR VERY closely → Extreme respiratory depression
Patch location & timing
Signs of overdose → Patch overdose risk (heat increases absorption!)
RR = 8 → emergency
Hydromorphone
Used for:
Severe pain
When morphine isn’t enough
What are risk to assess for using Hydromorphone?
ALWAYS check RR before every IV dose
Pain level
Sedation
Hydrocodone / Oxycodone (Perc)
Use for moderate → severe pain (oral use); has high abuse potential
What are risk to assess for using Hydrocodone / Oxycodone?
Pain relief
Signs of misuse
Bowel function → constipation
Methadone
Chronic severe pain
Opioid use disorder (withdrawal management)
What are risk to assess for using Methadone?
Cardiac rhythm (ECG if long-term)
Sedation over time
Signs of toxicity
Tramadol
↑ serotonin & norepinephrine, and has a low dependence risk:
Moderate pain
When trying to avoid stronger opioids
What are risk to assess for using Tramadol?
Seizures
Serotonin syndrome (with antidepressants)
Less respiratory depression (but still possi
What are common Opioid Adverse Effects?
Respiratory Depression (most dangerous)
Sedation & Drowsiness (fall risk due to impaired alertness and coordination)
Dizziness & Anxiety
Hallucinations (more common in higher doses or renal impairment)
Miosis (can indicate toxicity)
Cough Suppression (concern for post-surgical patients who need to deep breathe and cough)
How does opioid use cause constipation?
↓ gut motility
↑ water absorption
Not caused from tolerance developing
What nursing actions should be implemented for constipation?
Fluids
Fiber
Stool softener
How does opioid use cause Nausea & Vomiting?
Stimulate chemoreceptor trigger zone (CTZ) in medulla → N/V (common in initial doses)
Don’t give in pt with GI obstruction → further motility suppression
What effect does opioid use have Cardiovascular?
Causes Orthostatic hypotension → Instruct patients to rise slowly; fall prevention is a priority
What effect does opioid use have on Genitourinary?
Urinary retention: Monitor urine output; bladder scan as indicated
Decreased libido with chronic use
What is tolerance?
Body becomes less sensitive to analgesic effect → higher dose is needed over time
NORMAL physiological adaptation
What is Physical dependence?
When the body adapts physiologically to the presence of opioids → Withdrawal if drug stopped