Carboxylic Acid Reactions
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Oxidation (1° alcohol → acid)
Starting Material: Primary alcohol; Reagents / Conditions: Strong oxidant (KMnO₄, Na₂Cr₂O₇); Product: Carboxylic acid; Key Notes: Goes past aldehyde
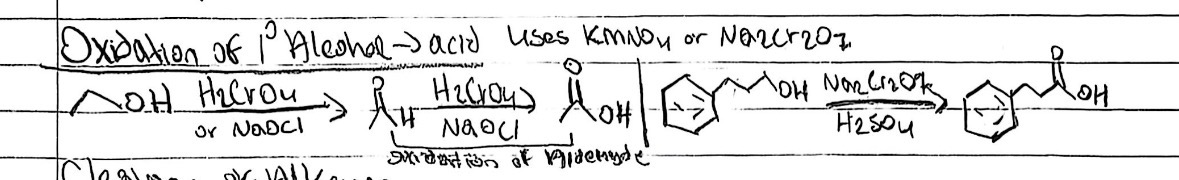
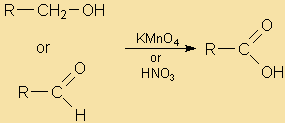
Oxidation of aldehyde
Starting Material: Aldehyde; Reagents / Conditions: Oxidizing agent; Product: Carboxylic acid; Key Notes: Very easy oxidation
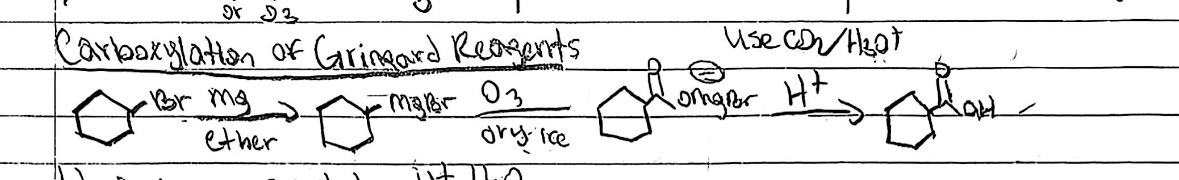
Grignard carboxylation
Starting Material: RMgX; Reagents / Conditions: CO₂, then H₃O⁺; Product: Carboxylic acid; Key Notes: Adds one carbon
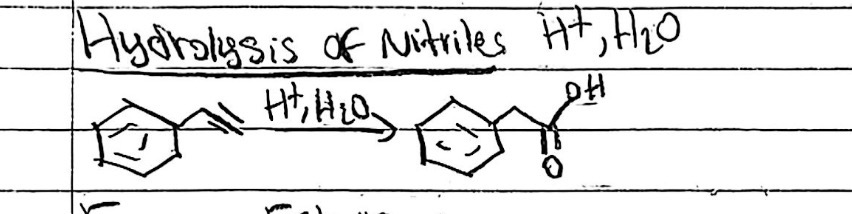
Nitrile hydrolysis
Starting Material: Nitrile; Reagents / Conditions: H₃O⁺, heat; Product: Carboxylic acid; Key Notes: Via amide intermediate
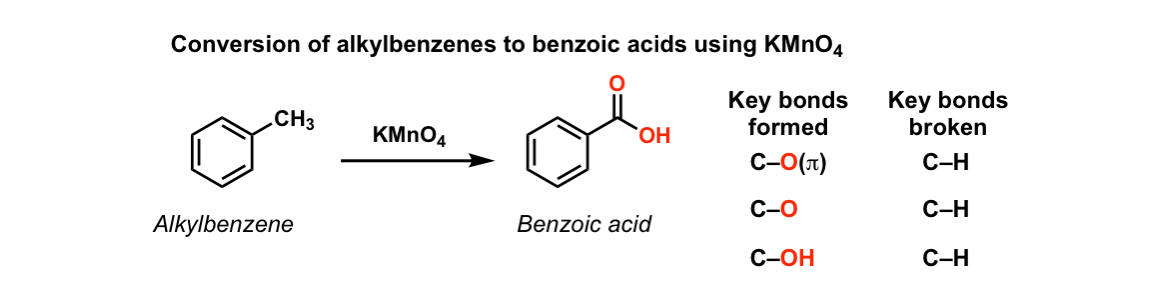
Oxidation of alkylbenzene
Starting Material: Alkylbenzene; Reagents / Conditions: KMnO₄, heat; Product: Benzoic acid; Key Notes: Needs benzylic H
Acid-base reaction
Starting Material: Carboxylic acid; Reagents / Conditions: Base (NaOH, etc.); Product: Carboxylate ion; Key Notes: Strong acidity
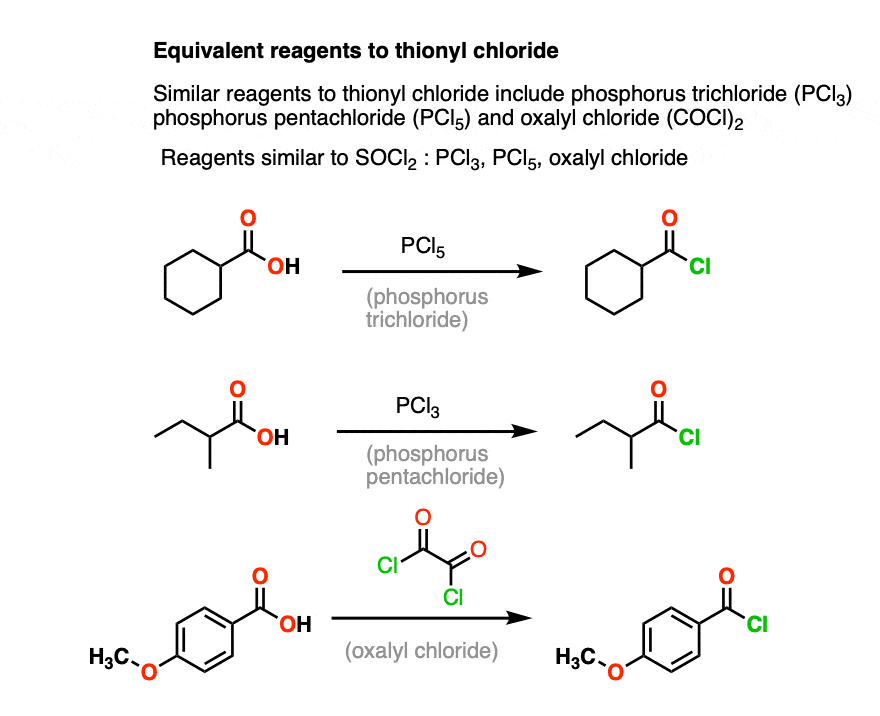
Formation of acid chloride
Starting Material: Carboxylic acid; Reagents / Conditions: SOCl₂ or PCl₅; Product: Acid chloride; Key Notes: Activates acid
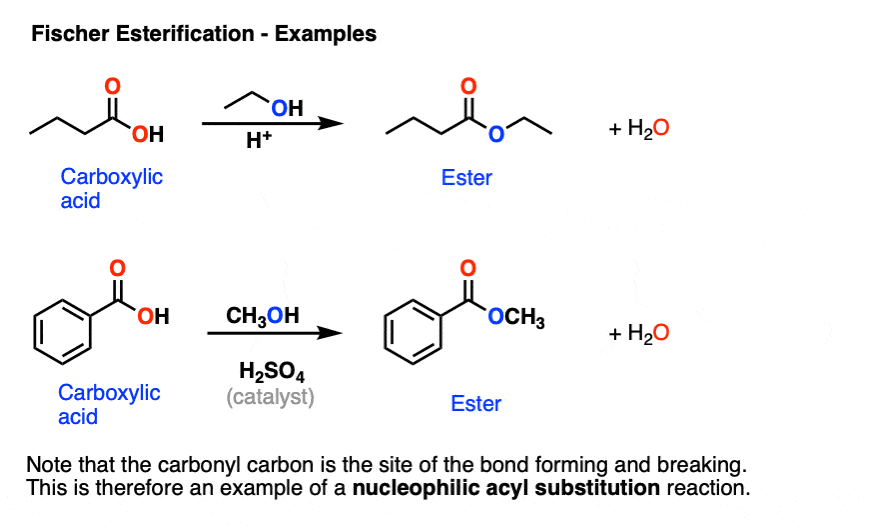
Formation of ester
Starting Material: Carboxylic acid; Reagents / Conditions: ROH, H⁺; Product: Ester; Key Notes: Fischer esterification
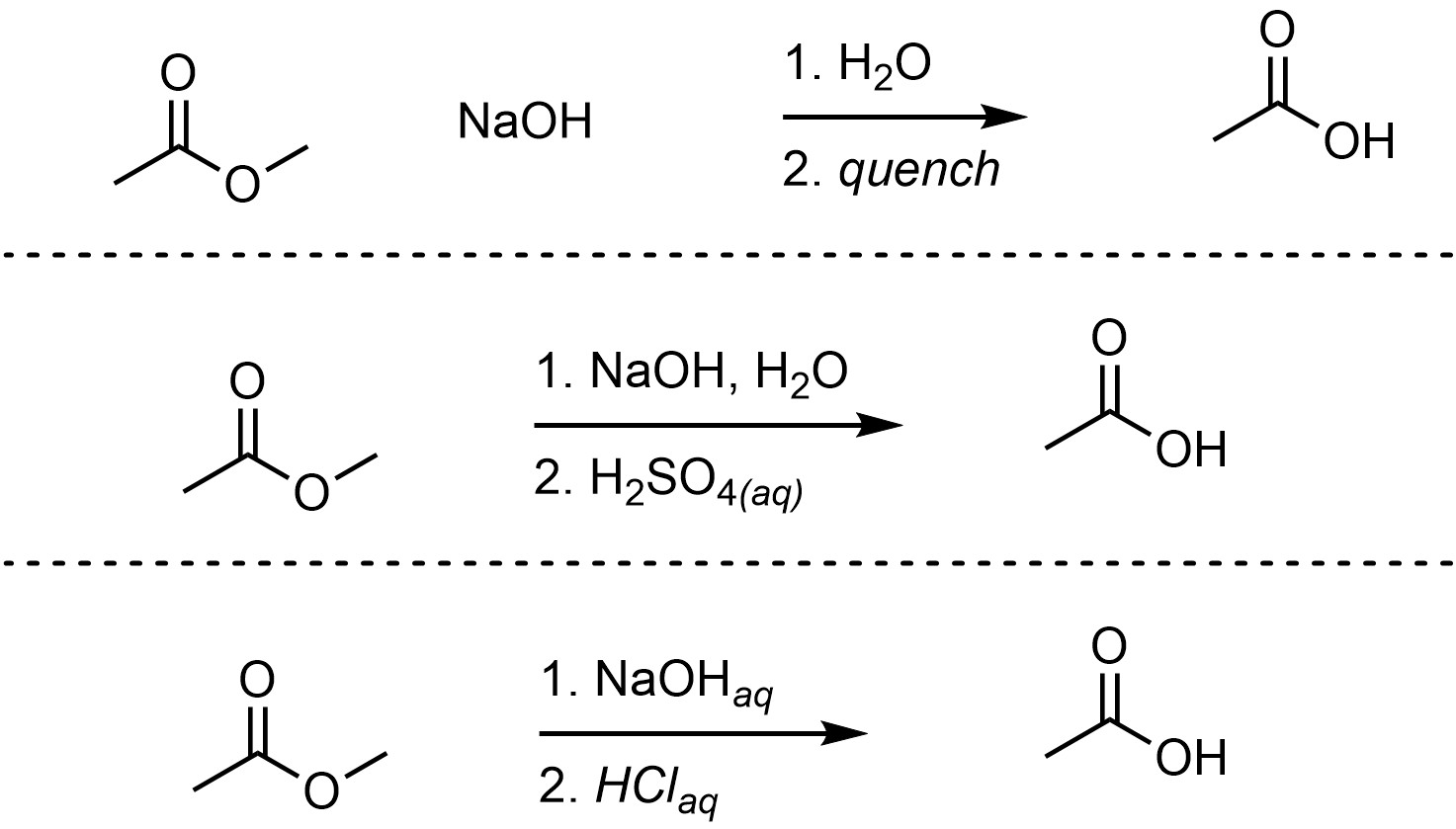
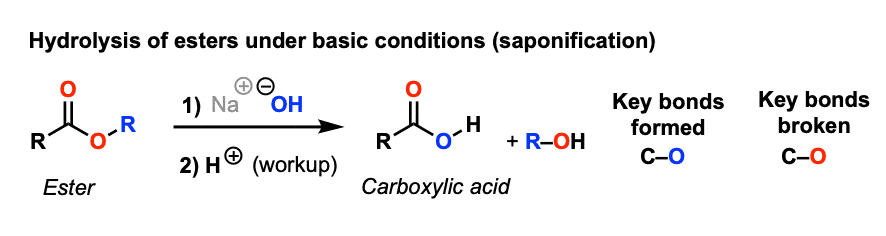
Hydrolysis of ester
Starting Material: Ester; Reagents / Conditions: H₃O⁺ or OH⁻; Product: Carboxylic acid; Key Notes: Reverse reaction
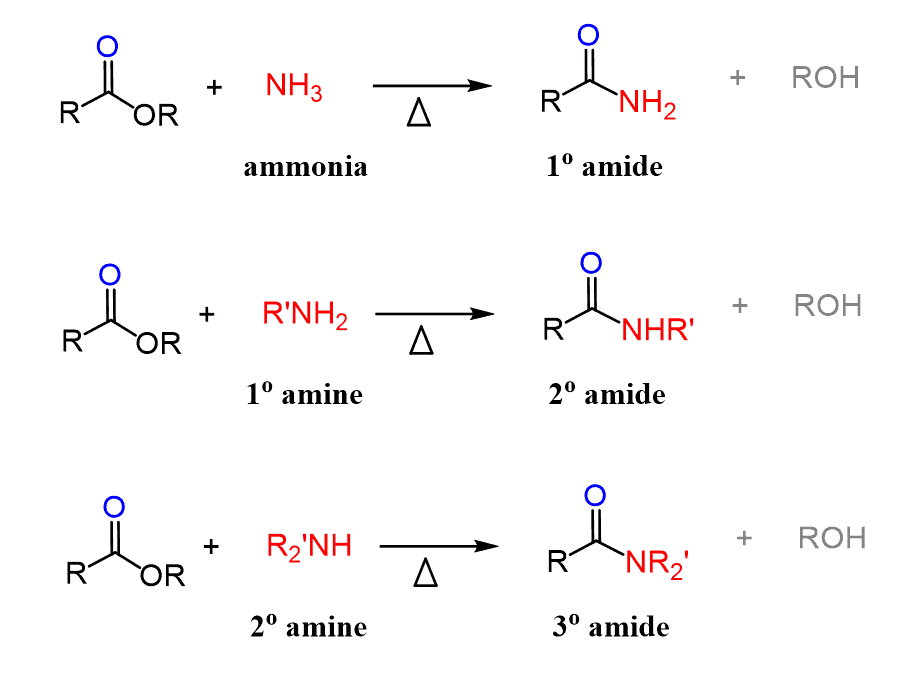
Formation of amide
Starting Material: Carboxylic acid; Reagents / Conditions: NH₃ or amine; Product: Amide; Key Notes: Often via acid chloride
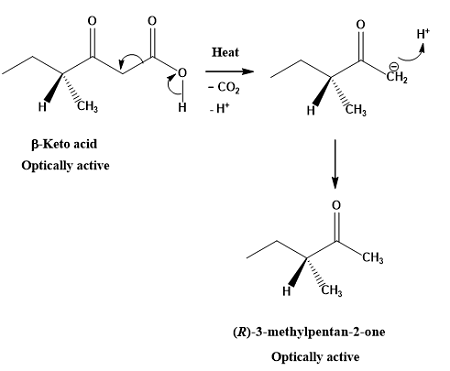
Decarboxylation
Starting Material: β-keto acid; Reagents / Conditions: Heat; Product: CO₂ + ketone; Key Notes: Loses CO₂
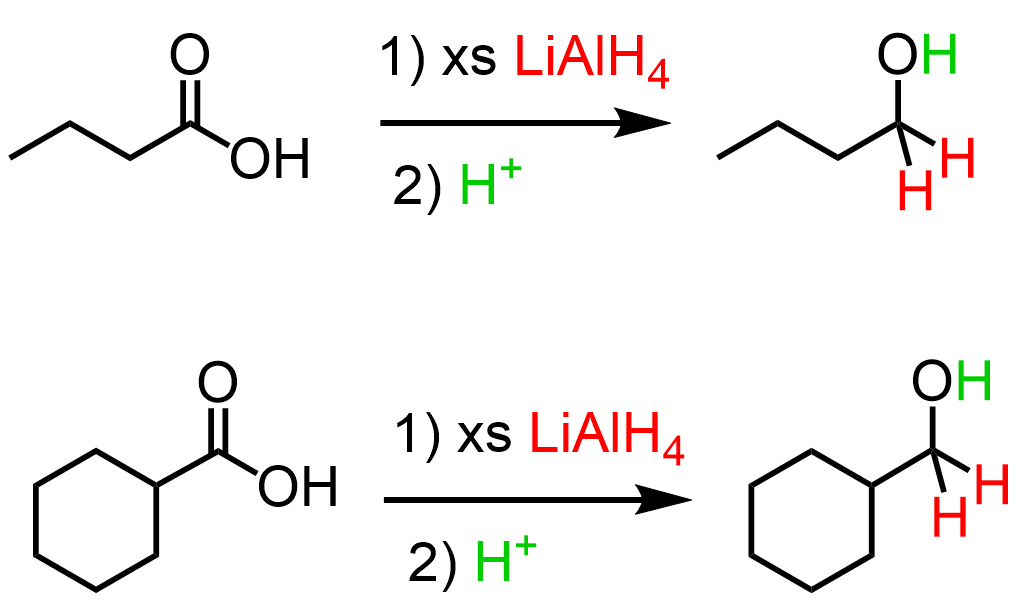
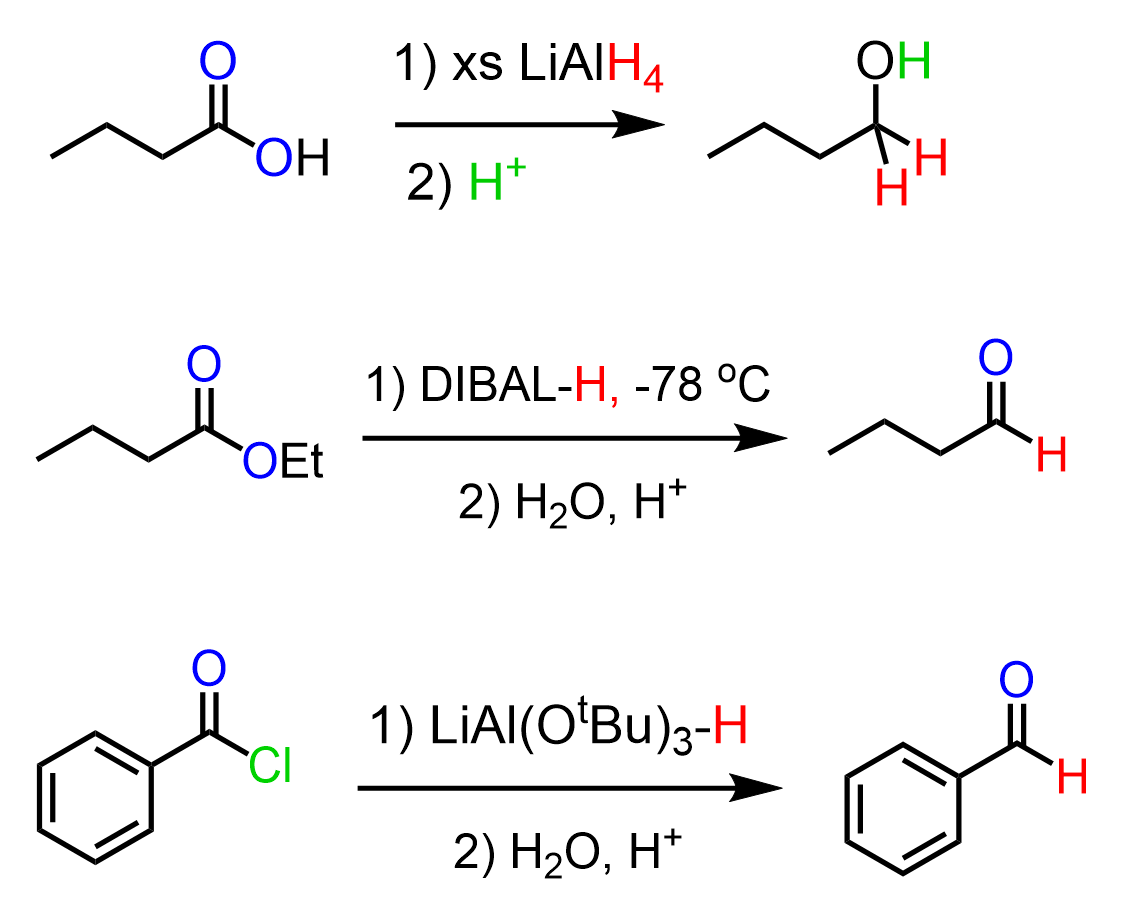
Reduction to alcohol
Starting Material: Carboxylic acid; Reagents / Conditions: LiAlH₄; Product: Primary alcohol; Key Notes: Strong reduction
Reduction to aldehyde (indirect)
Starting Material: Acid derivative; Reagents / Conditions: Controlled reduction; Product: Aldehyde; Key Notes: Not direct from acid
Formation of carboxylate salt
Starting Material: Carboxylic acid; Reagents / Conditions: NaHCO₃; Product: Carboxylate + CO₂; Key Notes: Gas evolution test