CHEM 2364, Final Exam (Ngo)
1/208
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
209 Terms
type of reaction that occurs when the OH group of an alcohol is removed, along with the proton on an adjacent carbon
dehydration reaction
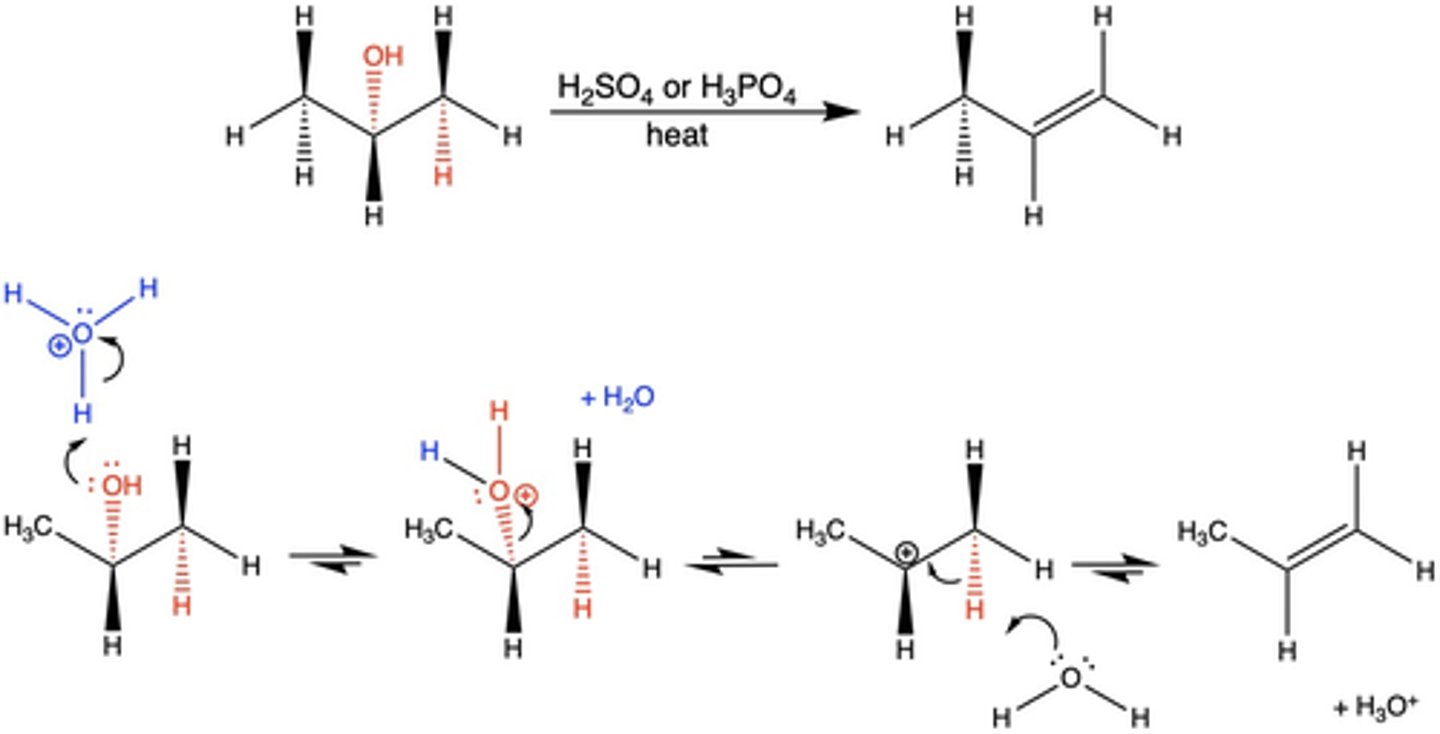
What occurs in a dehydration reaction?
a strong mineral acid (H2SO4 or H3PO4) and heat causes an OH group of an alcohol along with the proton on an adjacent carbon to be removed and form a double bond
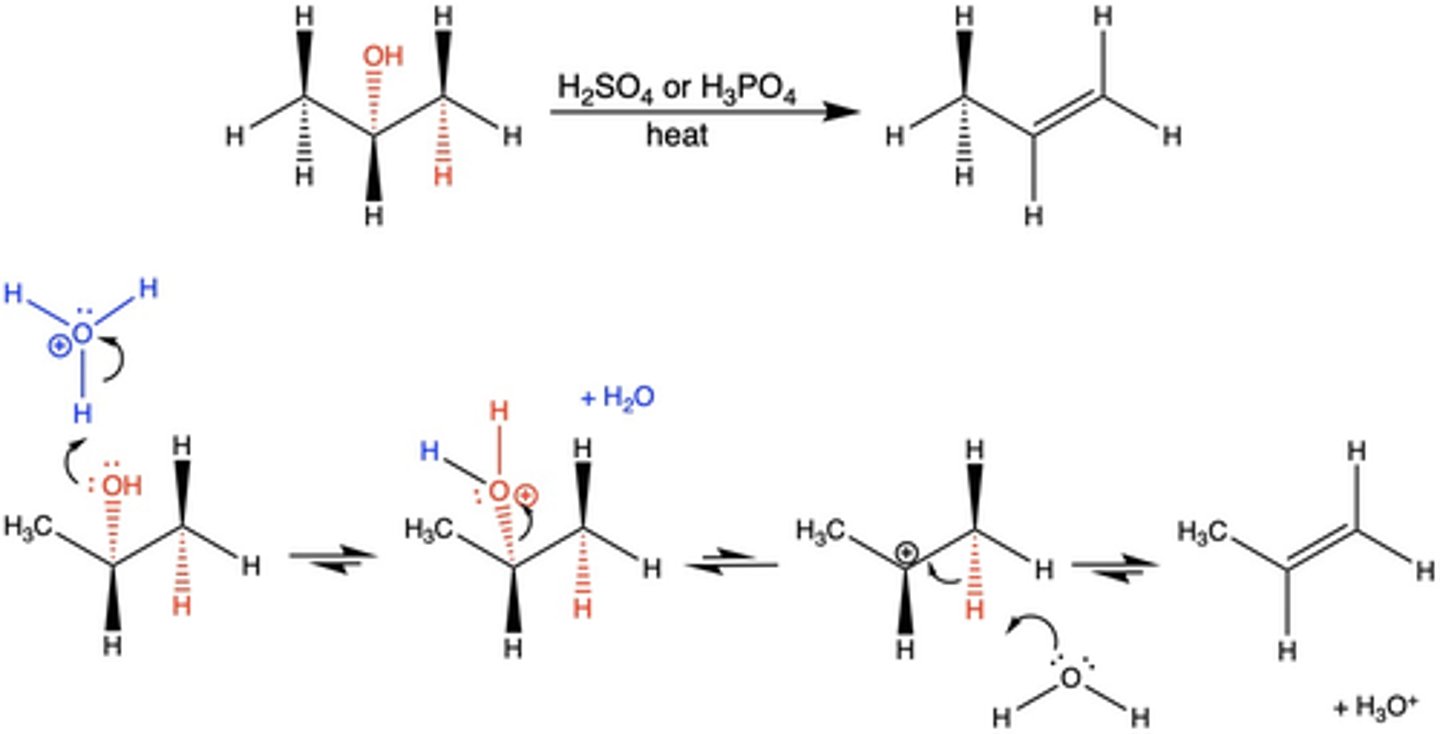
What reagents cause a dehydration reaction?
an alcohol AND a mineral acid (H2SO4 or H3PO4) + heat
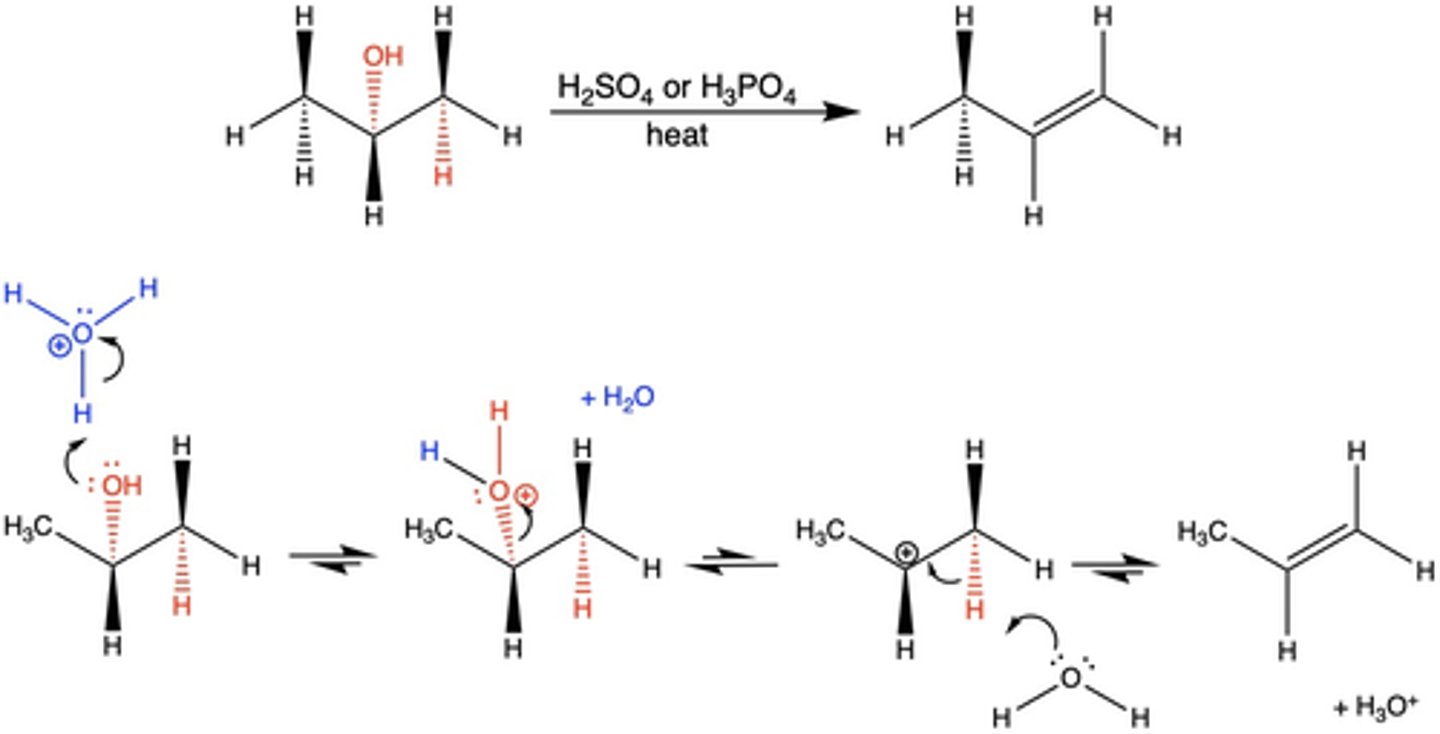
In the dehydration elimination reaction, why is there no stereoselectivity in the product?
the mechanism is multistep (E1)
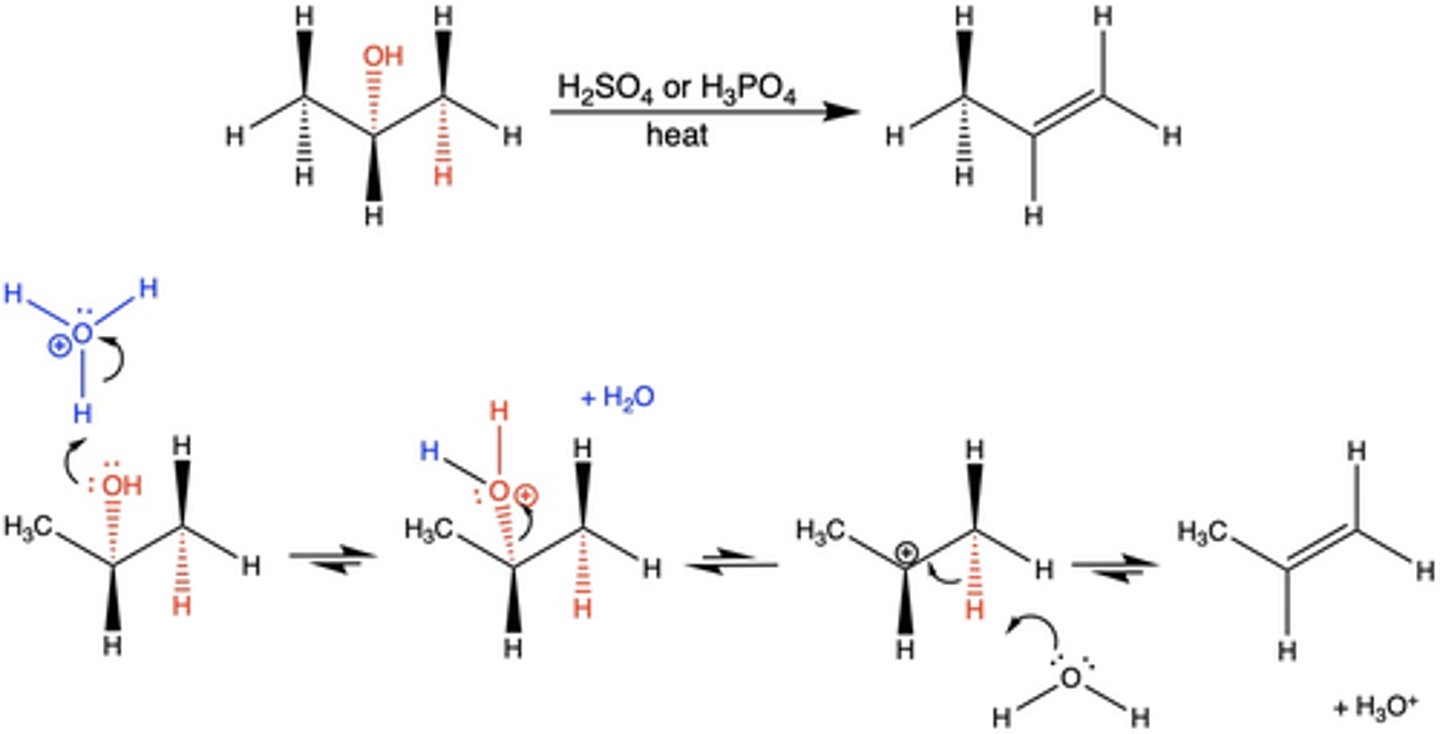
Because the elimination in the dehydration reaction is multi-step (E1), what effect does this have on the product?
generally no stereoselectivity (multiple products can be produced)
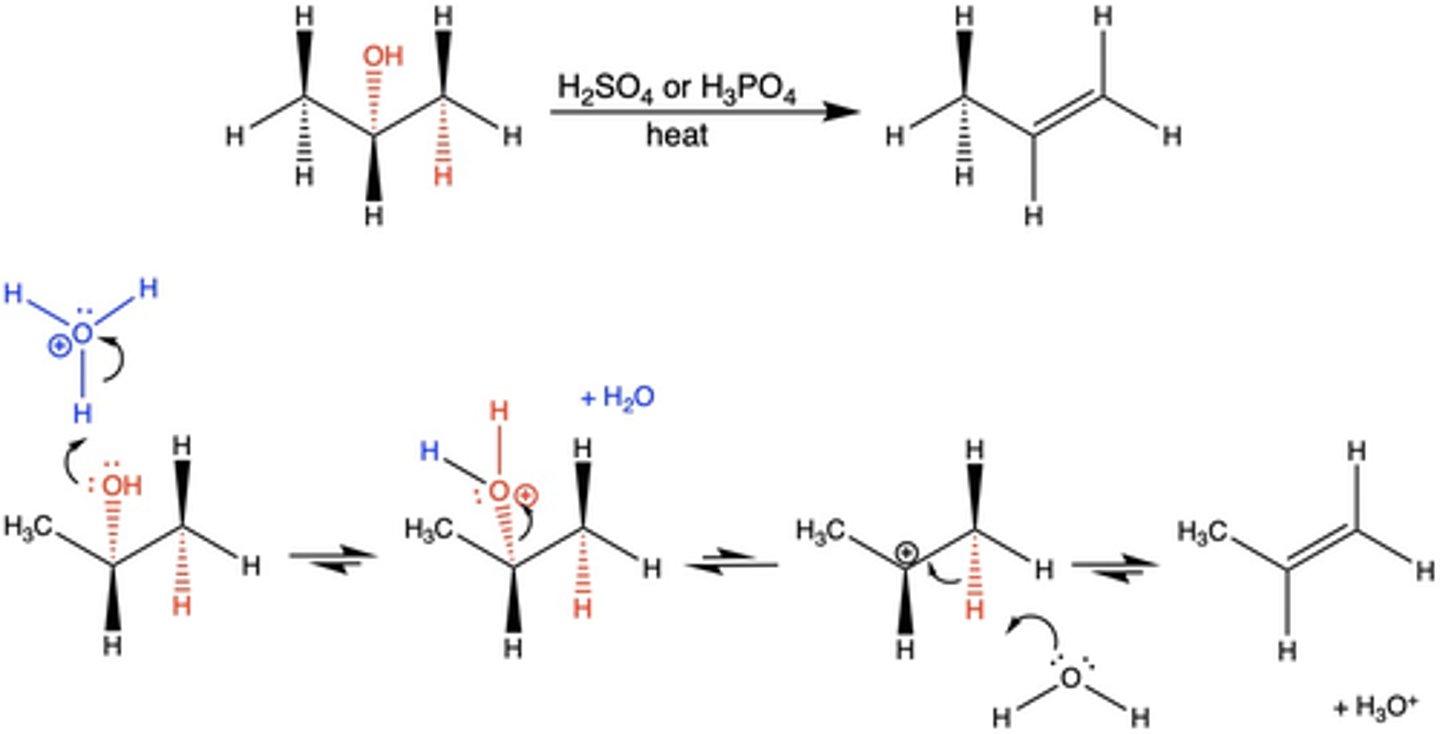
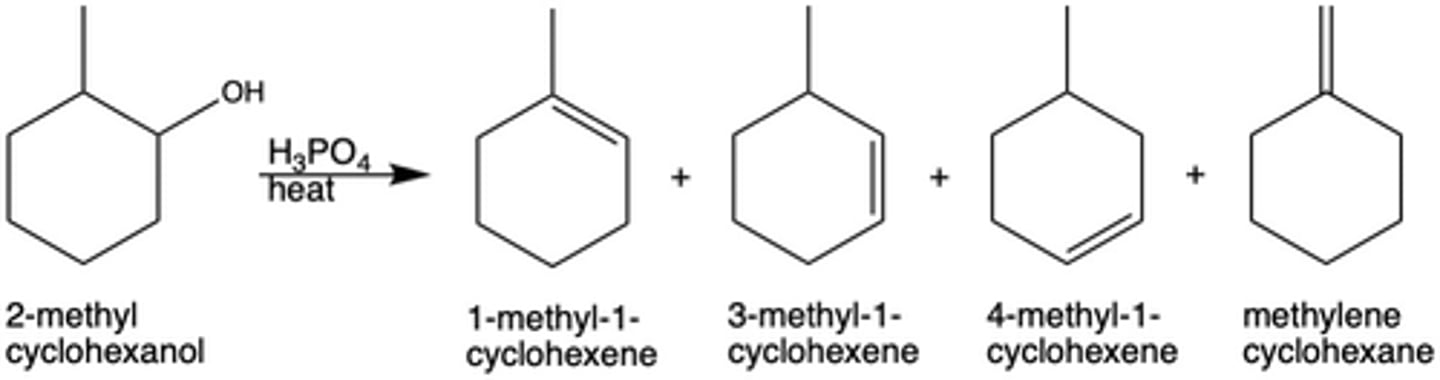
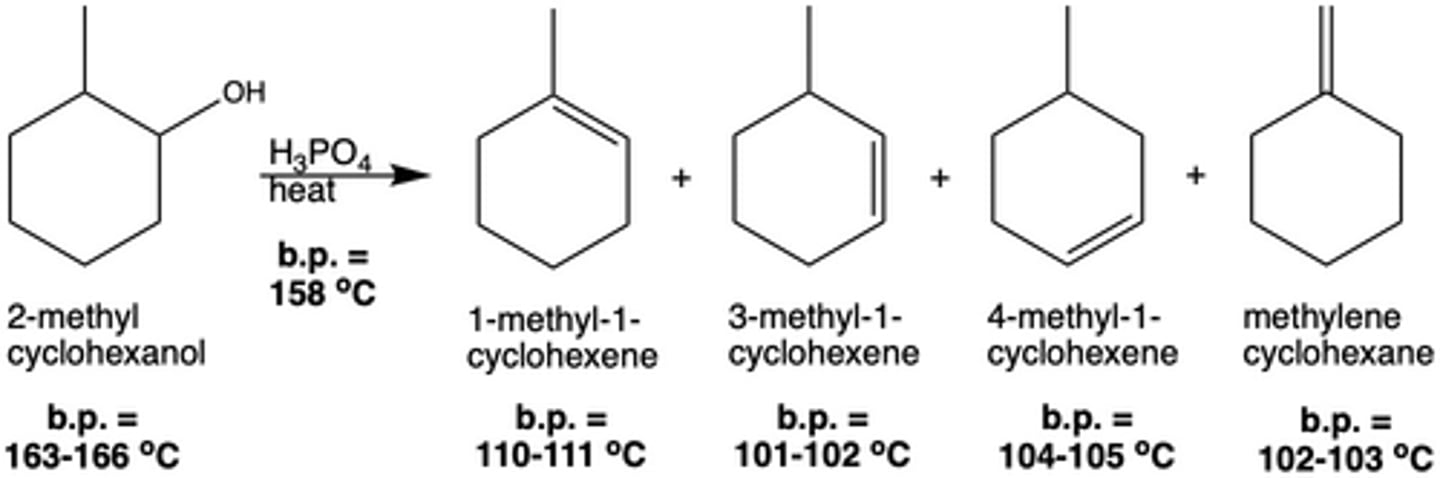
In Exp. 7, what were the reagents for the dehydration reaction?
2-methylcyclohexanol and H3PO4 + heat
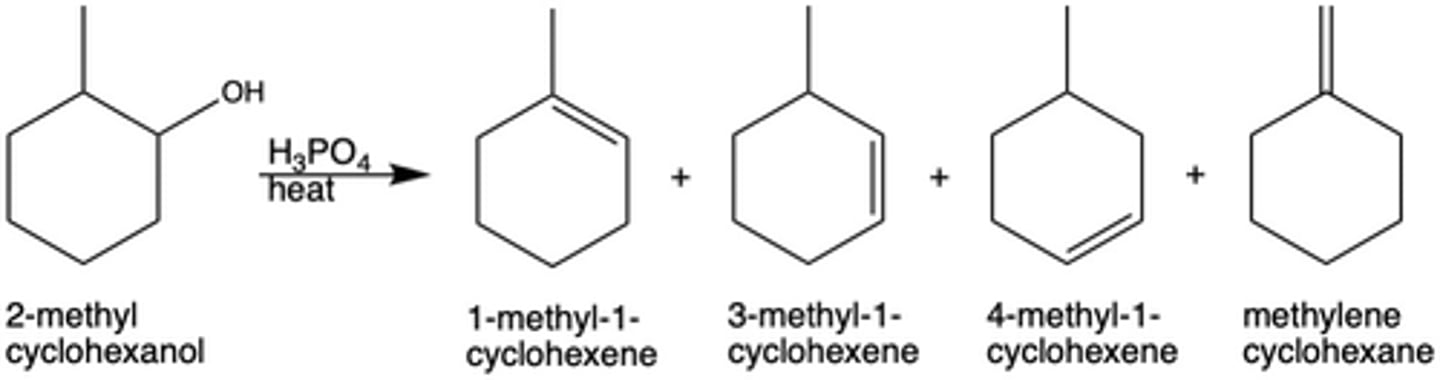
In Exp. 7, how many possible products were there for the dehydration reaction with 2-methylcyclohexanol? Why?
4 different products; the elimination reaction is E1 (multi-step), and thus is not stereoselective
a reaction that can in principle produce two or more stereoisomers but in fact produces one (or almost only one) stereoisomer product
stereoselective
What was the major product of the dehydration reaction in Exp. 7? Why?
1-methyl-1-cyclohexene; produces the most stable carbocation intermediate and can be formed in two ways
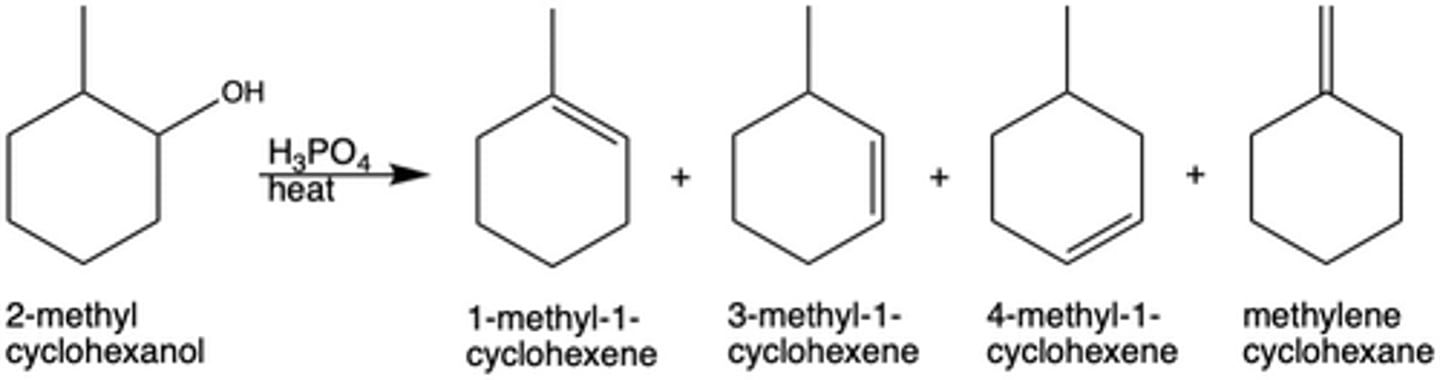
What is the major product of a dehydration reaction determined by (Exp. 7)?
the stability of the carbocation
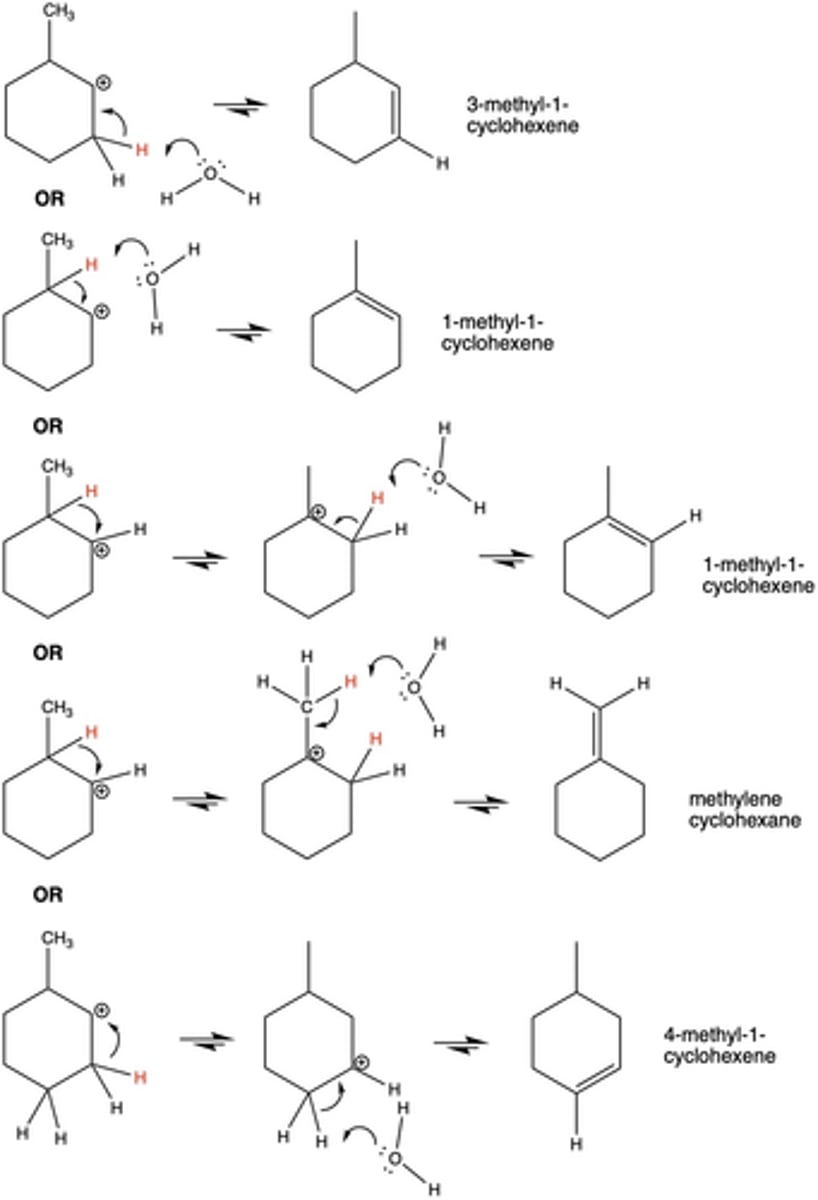
Explain the order of stability of carbocations.
A 3° carbocation is more stable than a 2°; and a 2° is more stable than a 1° carbocation.
What can happen to a 2° carbocation in order to become more stable and reactive (significant in dehydration/elimination reactions, Exp. 7)?
hydride shift (to become a 3° carbocation)
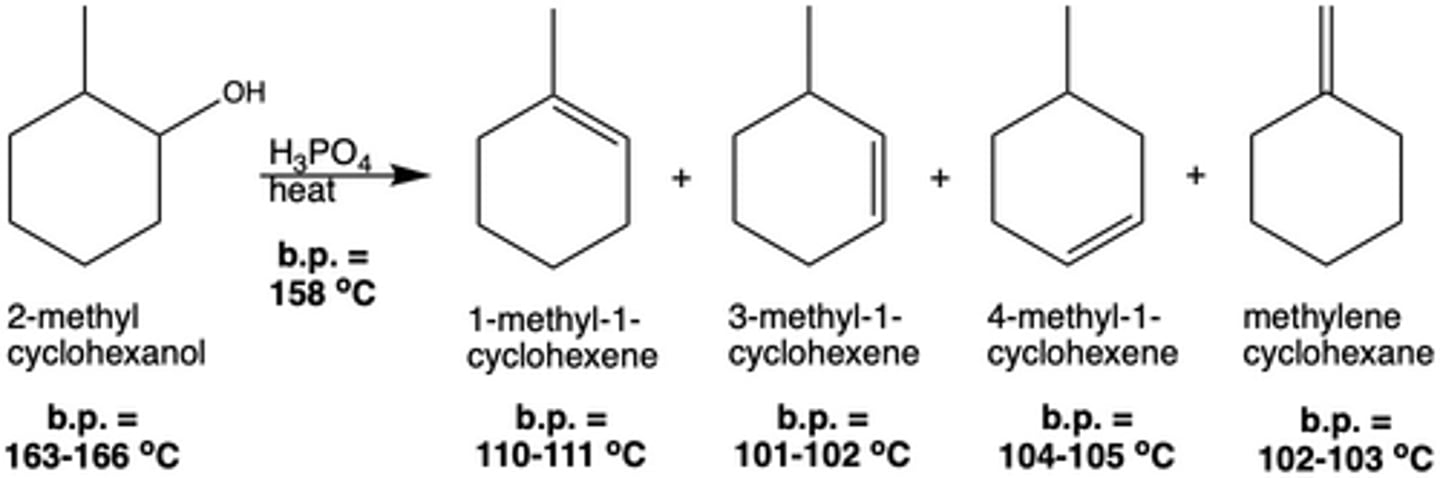
How were the products of Exp. 7 separated from the reactants? Why was it done this way?
A simple distillation is sufficient to separate the different products from the reactants since the boiling points are different enough. This distillation will collect the products in a round bottom flask (not in fractions), because the boiling points of the products are too similar.
What is the byproduct of a dehydration reaction (Exp. 7)?
water
each of two or more compounds with the same formula but a different arrangement of atoms in the molecule and different properties
isomers
a rearrangement of a hydrogen atom in a carbocation to make the intermediate structure more stable
hydride shift
What is a hydride shift and why does it occur?
a rearrangement of a hydrogen atom in a carbocation to make the intermediate structure more stable
a mixture of two liquids which has a constant boiling point and composition throughout distillation (can no longer separate the liquids via distillation) (ex: at 95% ethanol in water, no matter how much you distill the ethanol it will always remain around this percentage of ethanol and water--cannot separate the mixture any more)
azeotrope
an azeotrope (mixture of two liquids with a constant boiling point and cannot be distilled/purified further) where the boiling point of the azeotrope is lower than that of the individual components
positive azeotrope
an azeotrope (mixture of two liquids with a constant boiling point and cannot be distilled/purified further) where the boiling point of the azeotrope is higher than that of the individual components
negative azeotrope
After the distillation of the hydrocarbon products in Exp. 7 (dehydration reaction), what type of azeotrope is formed and why?
Water forms a positive azeotrope with the hydrocarbon products (boiling point of the azeotrope is lower than that of the individual components)
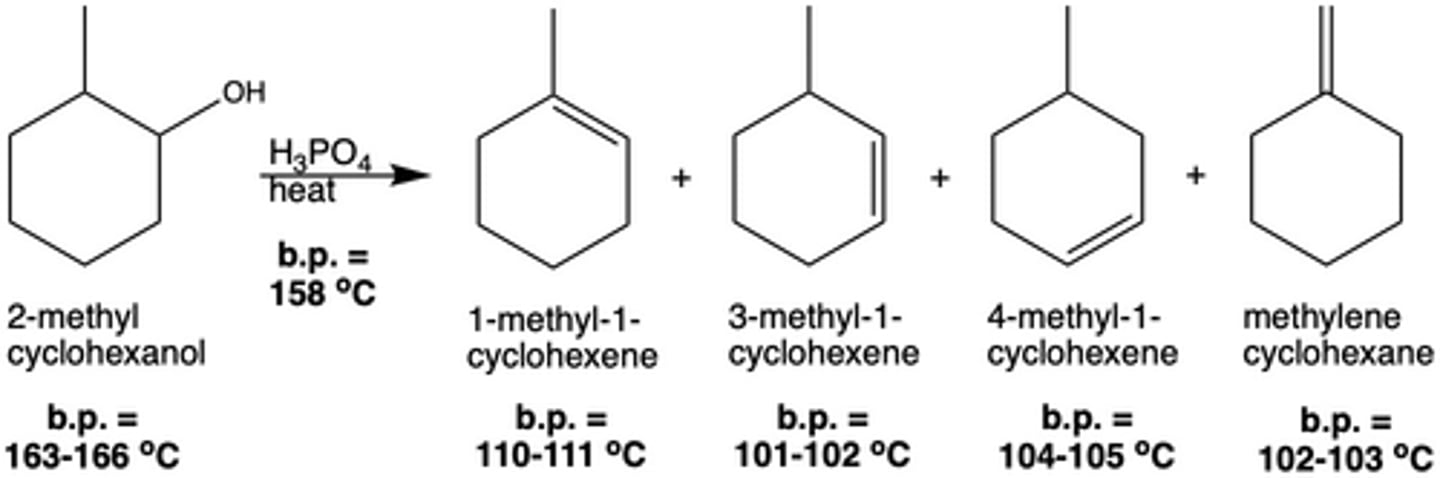
After distilled with the hydrocarbon products produced in the dehydration reaction in Exp. 7, how can the water be removed from the azeotrope?
water will form a second layer on the bottom of the solution because it is generally immiscible with non-polar hydrocarbons and wants to be on the bottom because of its greater density than that of the dehydration products
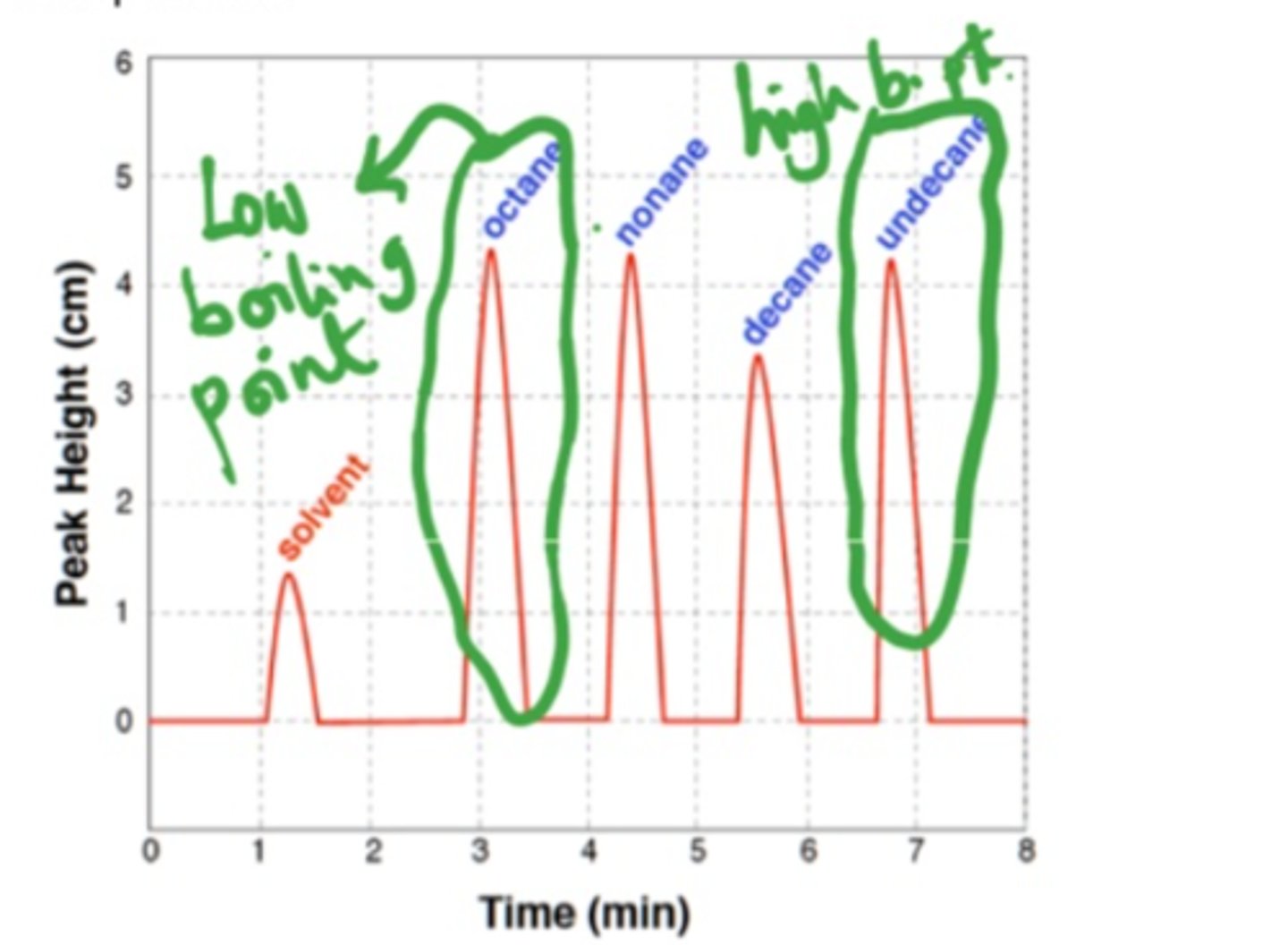
method of characterization that uses differences in relative polarity (differences in boiling points) to separate compounds
Gas Chromatography
What is the mobile phase in Gas Chromatography?
an inert gas
What is the stationary phase in gas chromatography?
hydrophobic substance coated on the inner walls of the tube
What does gas chromatography display about a solution (what does it characterize)?
the differences in relative polarity (as the compounds in the solution separate)
How does gas chromatography work and what do the results show?
The liquid sample that is injected into the GC is then heated until each molecule evaporates (i.e., becomes a gas), and so the first one to reach the detector will be the one with the lowest boiling point.
How can the relative amount of each compound present in a solution be measured in gas chromatography?
the area under the curve of a peak

When looking at a GC chromatogram, how can you tell which compound has the lowest boiling point?
the first peak that appears (pay attention to time on the x-axis), has the lowest boiling point (took the least amount of time and energy to break the IMFs and boil)
What formula can be used to find the relative amount of one compound in a solution from a gas chromatogram?
% P1 = aP1 / (aP1 + aP2 + aP3 ...) x 100%
a = area under curve
P = peak

Which qualitative tests in Exp. 7 were used to confirm the presence of an alkene group?
bromine test and potassium permanganate test
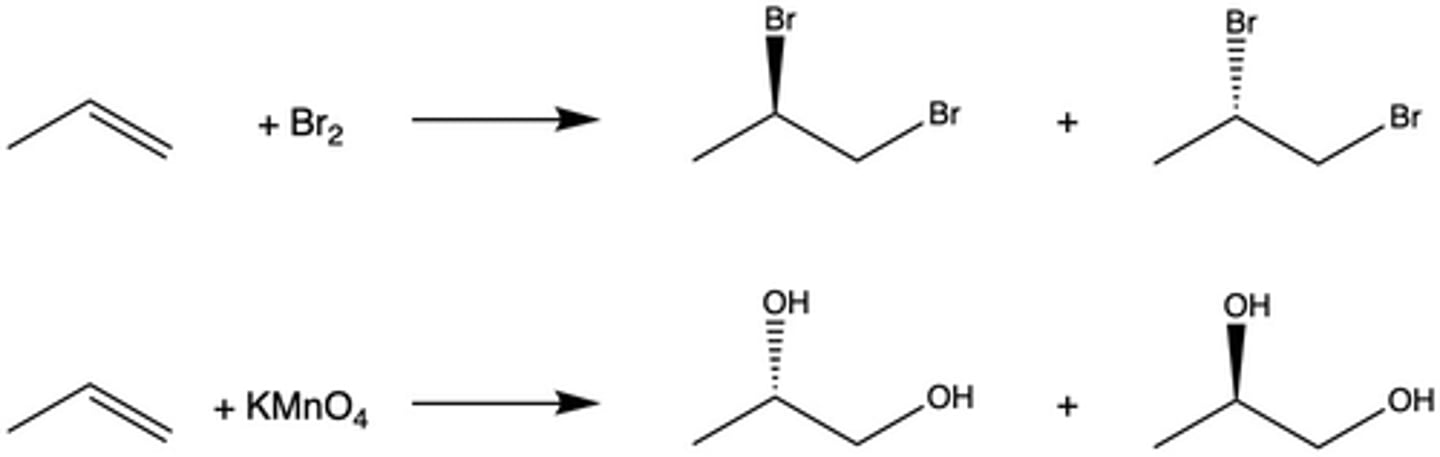
How is bromine used to test the presence of an alkene in a molecule?
When Br2 is mixed with an alkene, the dark reddish-brown color of the reagent disappears; if it does not disappear, there was not an alkene present
What type of addition occurs between Br2 and an alkene in the bromine test?
anti addition
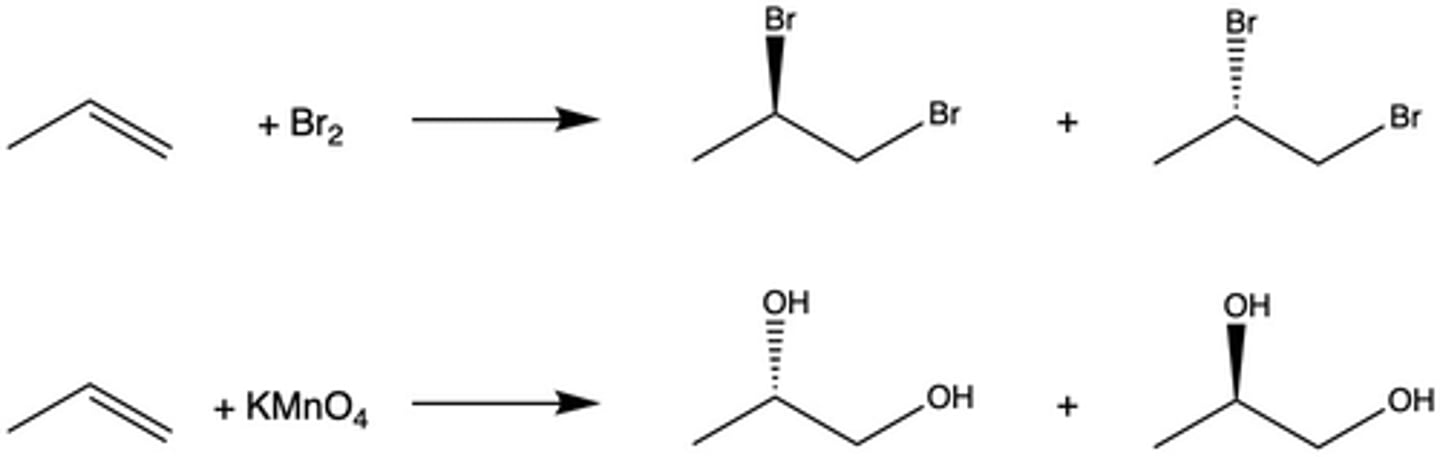
How is potassium permanganate used to test the presence of an alkene in a molecule?
When KMnO4 is added to an alkene, the dark purple color of the reagent disappears. Additionally, a dark brown solid appears (the byproduct MnO2).
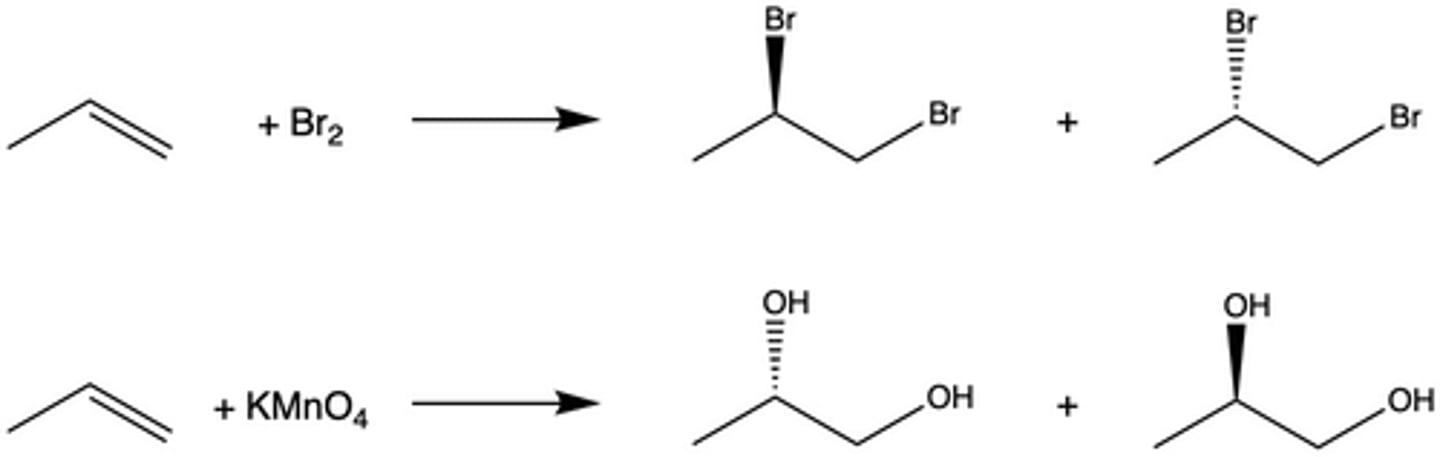
What is the stereochemistry of the oxidation of KMnO4 and an alkene (type of addition)?
syn
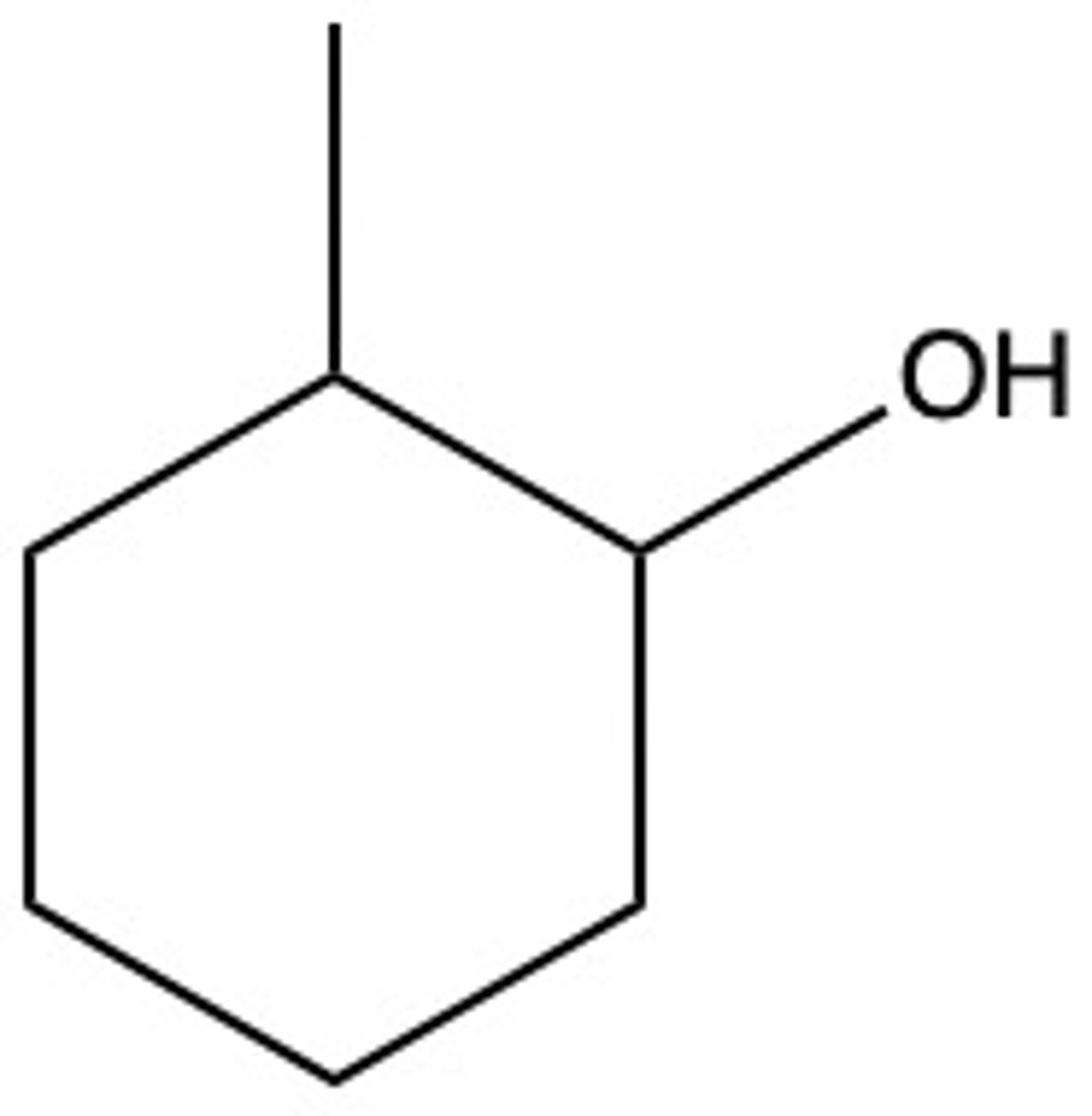
What are the chemical hazards of:
2-methylcyclohexanol
flammable, inhalation hazard
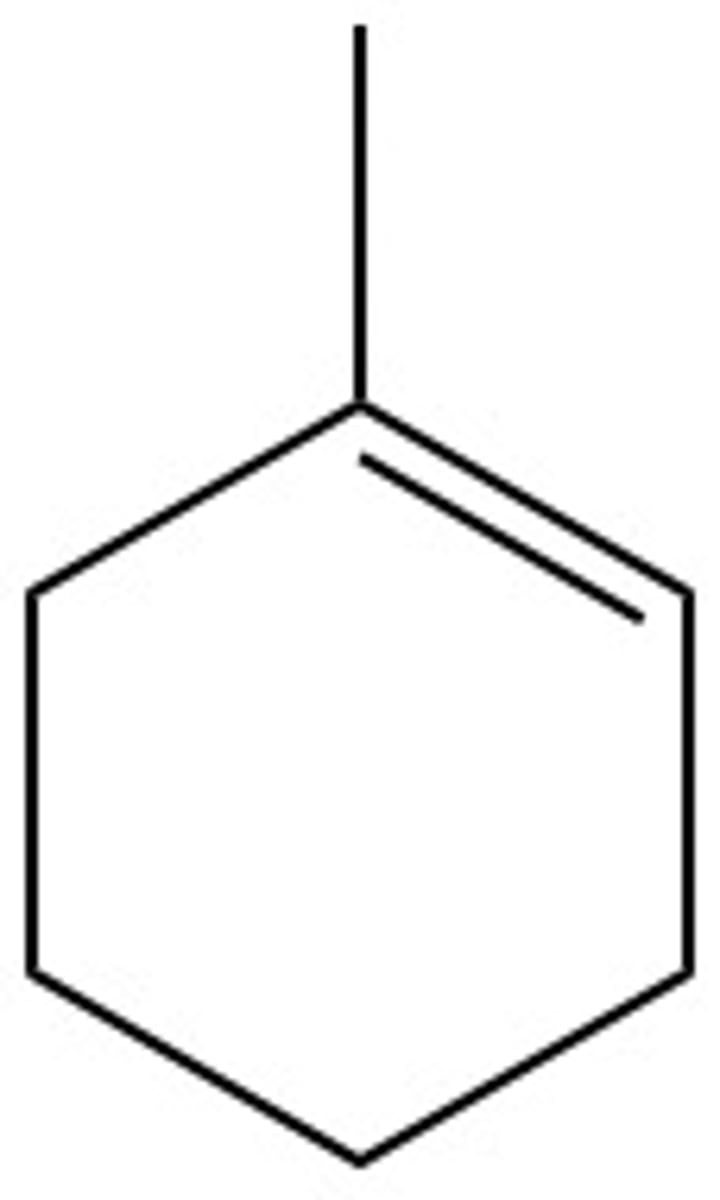
What are the chemical hazards of:
1-methyl-1-cyclohexene
flammable, toxic, irritant
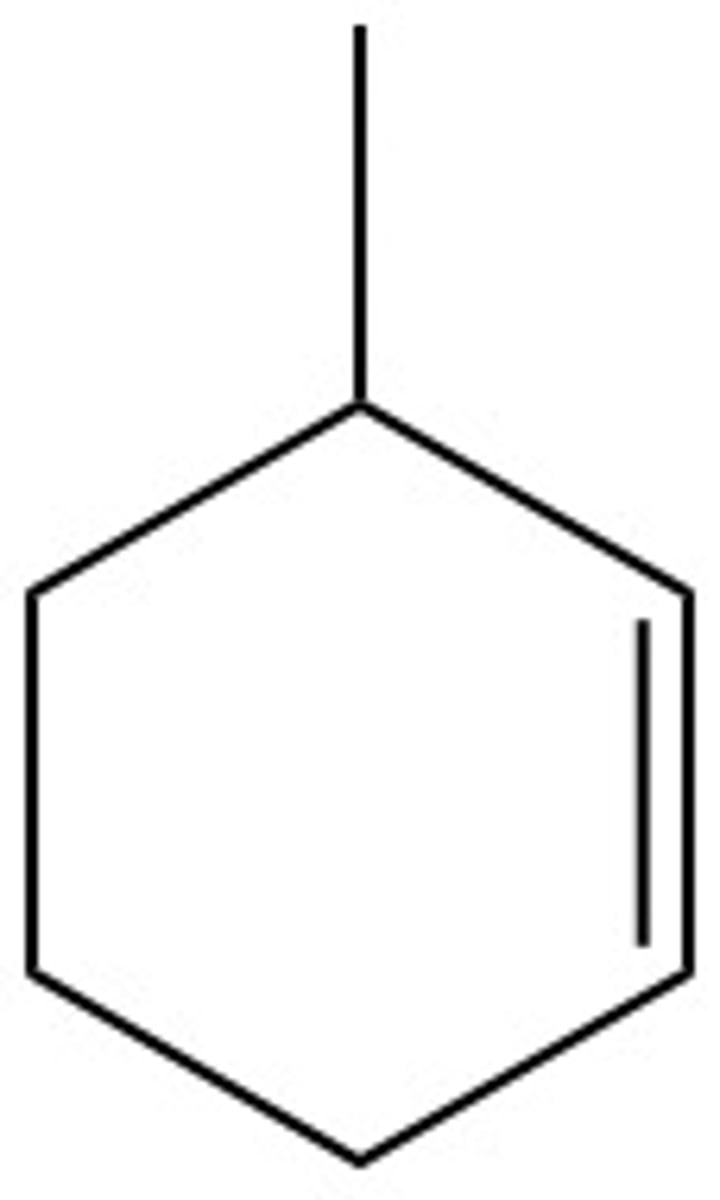
What are the chemical hazards of:
3-methyl-1-cyclohexene
flammable, toxic, irritant
What are the chemical hazards of:
4-methyl-1-cyclohexene
flammable, toxic, irritant
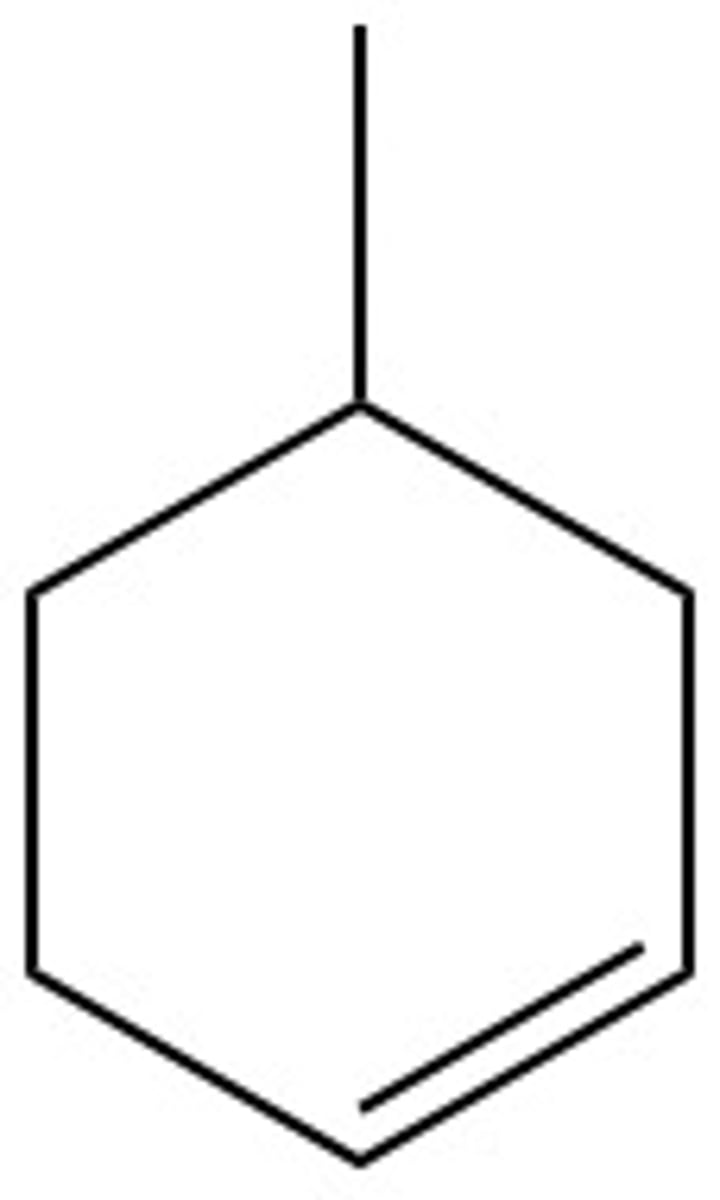
What are the chemical hazards of:
methylene cyclohexane
flammable, toxic
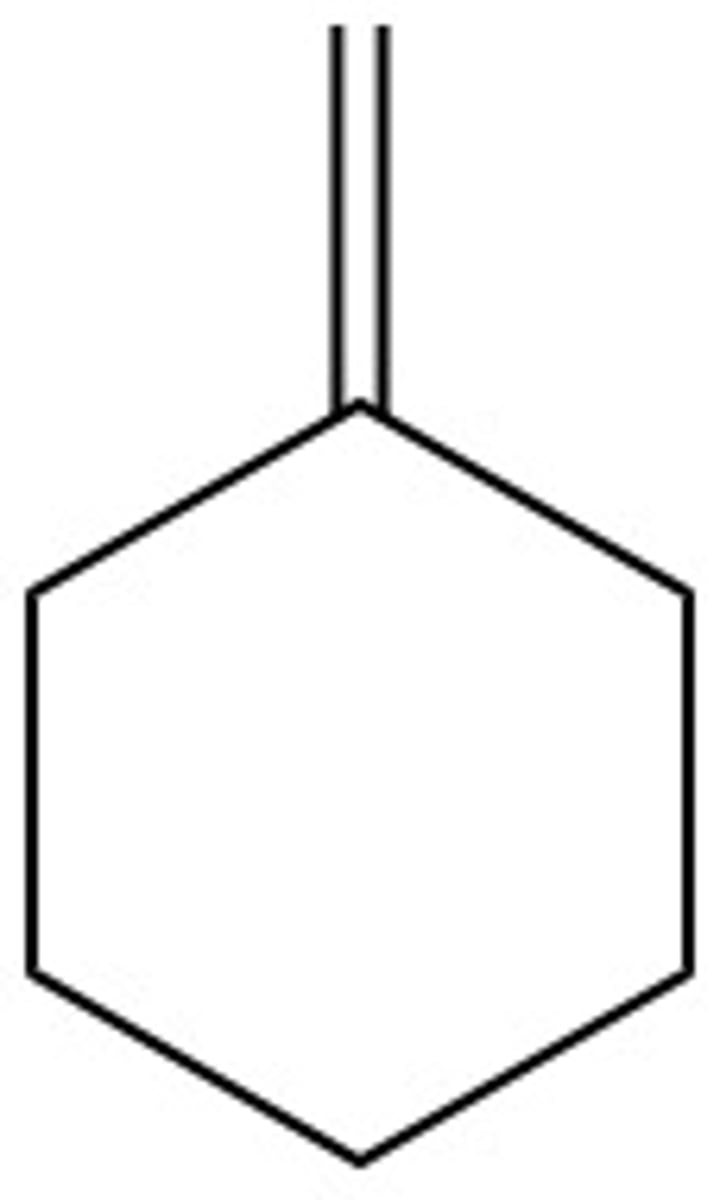
What are the chemical hazards of:
water (H2O)
none
What are the chemical hazards of:
saturated sodium bicarbonate (NaHCO3)
none
What are the chemical hazards of:
bromine (in DCM)
corrosive, toxic, environmental hazard
What are the chemical hazards of:
potassium permanganate (aq) (KMnO4)
oxidizer, toxic, irritant, environmental hazard
What are the chemical hazards of:
dichloromethane (CH2Cl2)
irritant, carcinogen, toxic
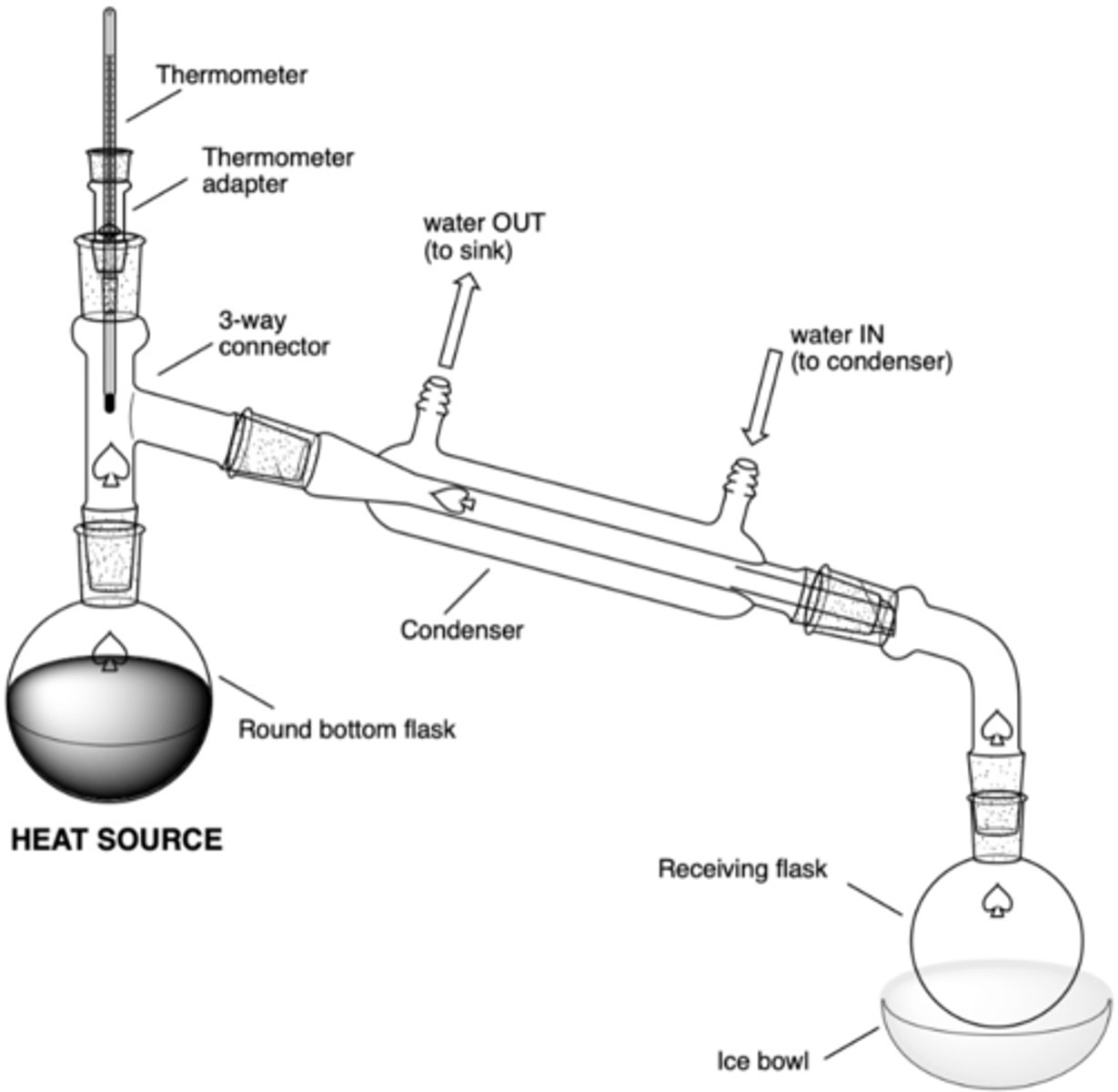
What are the significant components of a simple distillation set up?
- heat source (hot plate)
- RBF in sand bowl w/ boiling chips (rxn flask)
- 3-way connector
- thermometer adaptor/thermometer
- condenser (water in at bottom, water out at top)
- vacuum connecting tube (to connect to receiving flask)
- receiving RBF
- (possibly) ice bowl
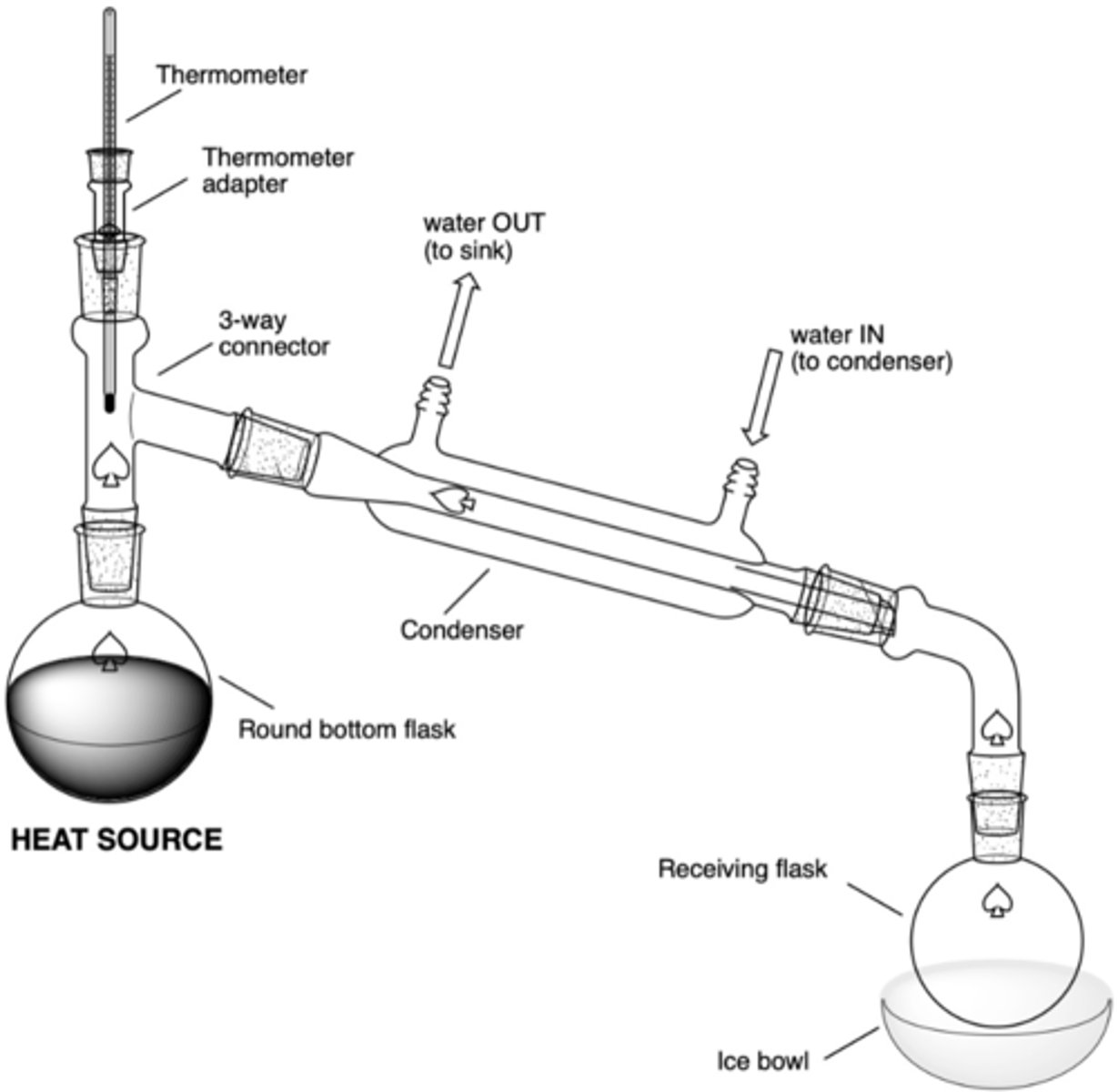
What are the chemical hazards of:
phosphoric acid (H3PO4)
corrosive
What precautions should you take when handling phosphoric acid? Why?
Handle with extreme care. Do not take the reagent bottles out the fume hood. Rinse the graduated cylinder with water (and NaHCO3) after use. Phosphoric acid is a strong acid.
What is the benefit of wrapping a distillation apparatus (reaction flask and tube) with foil?
the foil insulates the flask and keeps the temperature constant (keeps the system from loosing heat)
Why should you never distill to dryness?
NEVER distill the distillation flask to dryness as there is a risk of explosion and fire
Which compounds used in Exp. 7 are considered to be strong oxidizers? What does this mean?
Bromine and potassium permanganate are strong oxidizers. Handle these compounds with care (Oxidizers are a severe fire hazard)
In Exp. 7, why is the boiling point of the hydrocarbon products lower than that of the reactant (alcohol)? (Hint: think of intermolecular forces.)
Alcohol has H-bonding, the products cannot H-bond (Van Der Wals Forces), thus they have a lower IMF and lower boiling point.
During the distillation, why does the water inlet need to go in the bottom barb of the condenser and out of the top barb?
Allows for the coolest water to enter into the bottom, allowing for the greatest heat exchange to occur and for condensation to occur
In Exp. 7, why is 1-methyl-1-cyclohexene the major product? (Hint: there are two reasons.)
1-methyl-1-cyclohexene can form from two separate eliminations
Forms from the most stable carbocation (tertiary)
What is the purpose of washing a liquid product with NaHCO3 (usually in an extraction)?
remove residual acid
What is the purpose of adding MgSO4 to a liquid product (usually after an extraction)?
to remove residual H2O (dry the product)
What determines the ordering of the layers in the microscale extraction (or any extraction)?
the ordering of the layers is determined by the density of the liquids
What is the benefit of using distillation to separate the reactants from the products (as in Exp. 7: Dehydration Reaction), instead of using a separatory funnel?
Distillation can separate the reactants and products based on their boiling point, which makes it easier to separate two liquids with similar densities (and liquids that are miscible).
Select the answer that correctly explains the set up for the water flow in the condenser:
The water flows in at the bottom barb and out the top, since the water is coldest when coming from the tap and will heat up as it runs through the condenser
In a simple distillation set up, what will be the result of this error (why is this an error?):
all the exit points are sealed
it is extremely dangerous to heat a closed system
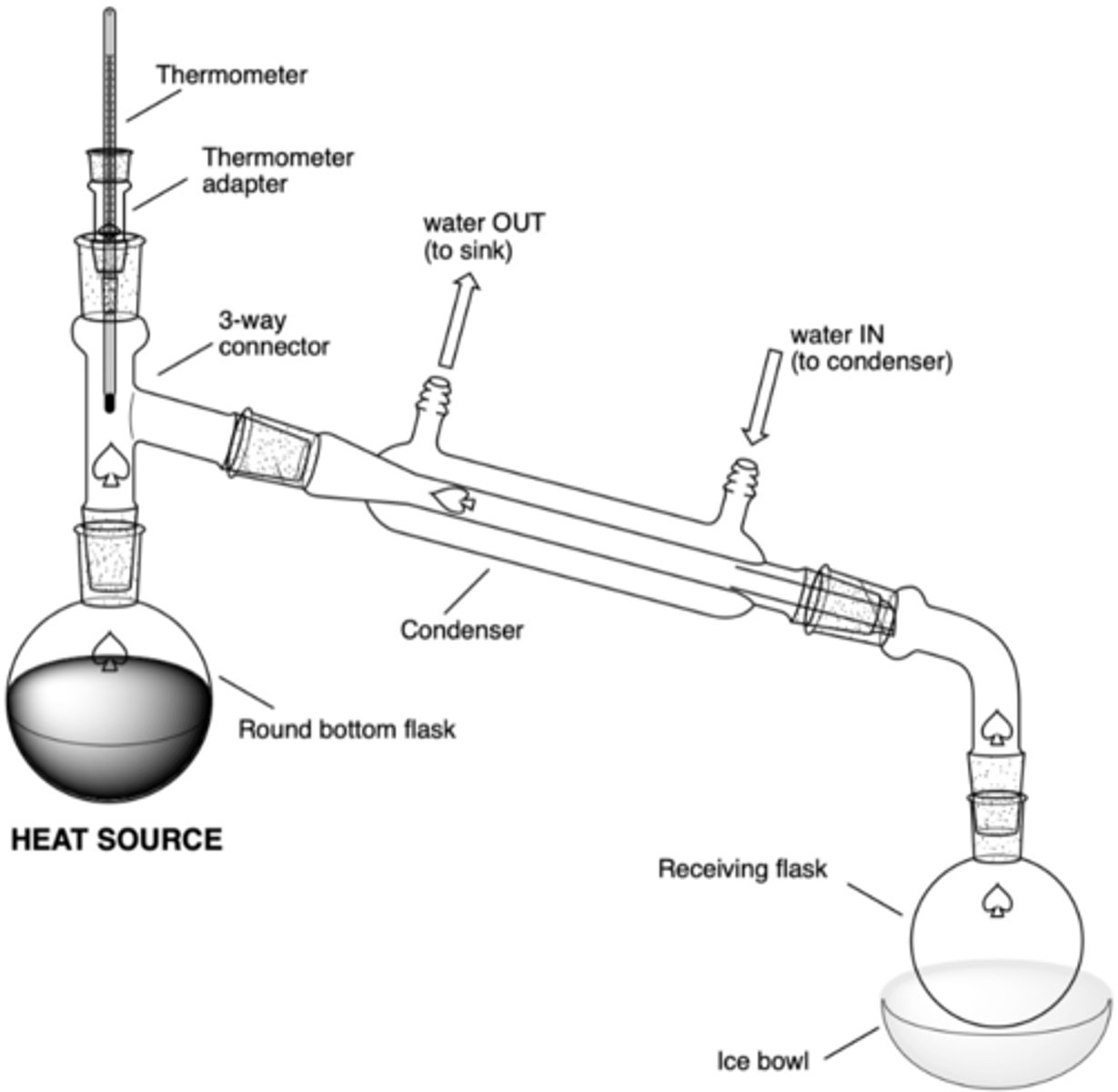
In a simple distillation set up, what will be the result of this error (why is this an error?):
there is an ice bath where there should be a hot plate and sand bath
the solution will not boil and thus will not distill
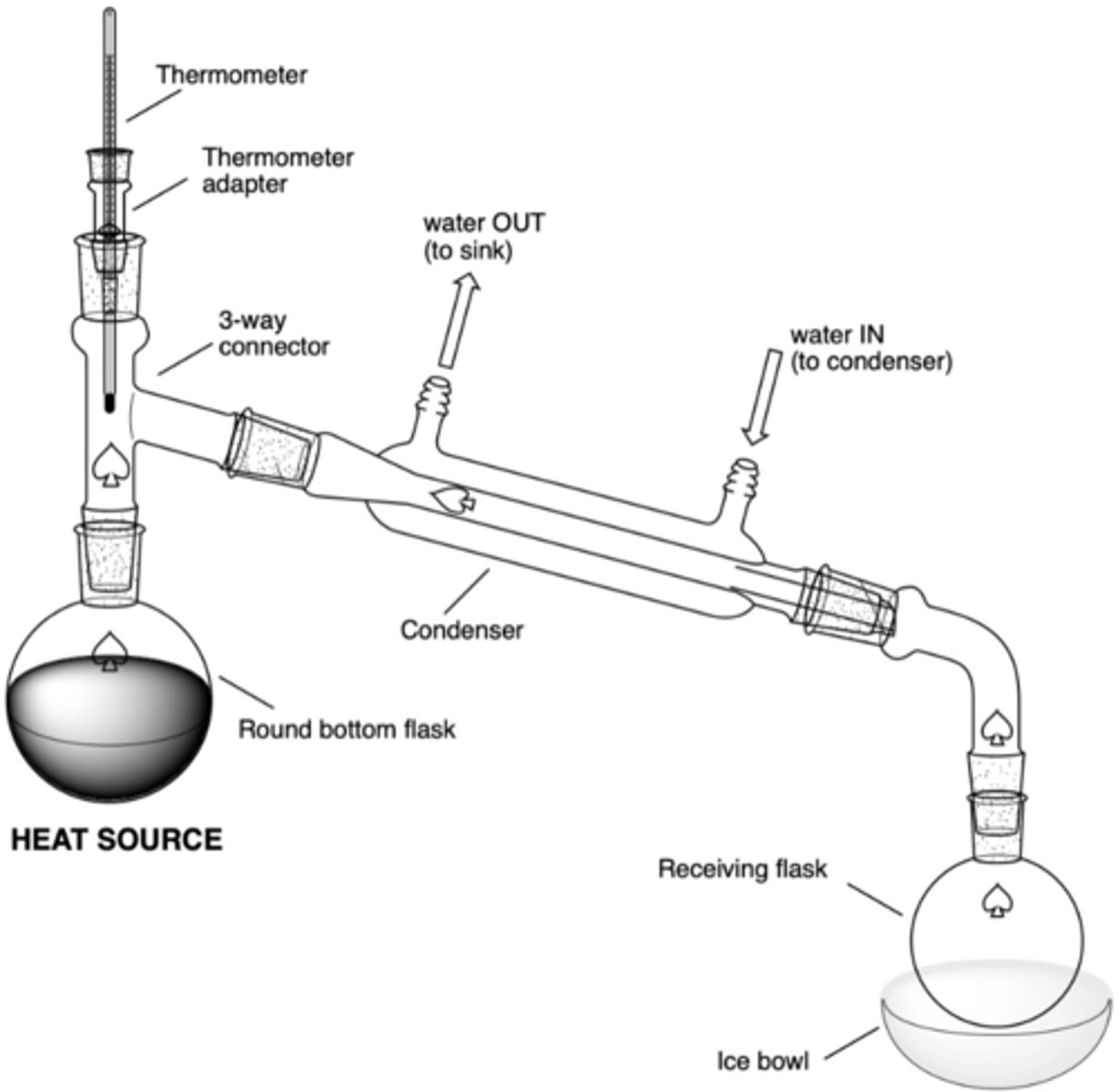
In a simple distillation set up, what will be the result of this error (why is this an error?):
incorrect clamping
the set up is not secure (and may fall)
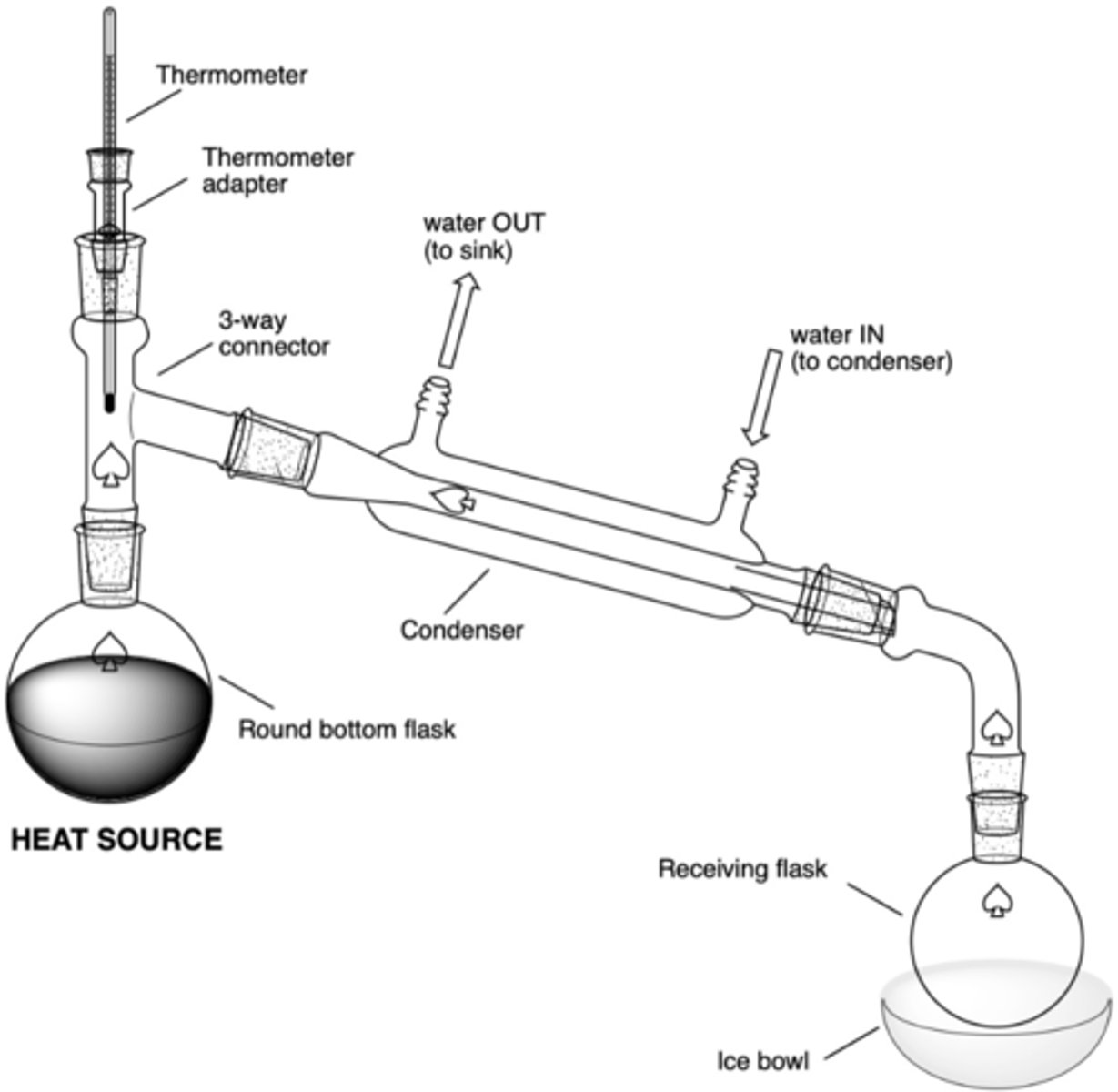
In a simple distillation set up, what will be the result of this error (why is this an error?):
there is no stir bar or boiling chips
The solution could bump or boil too vigorously
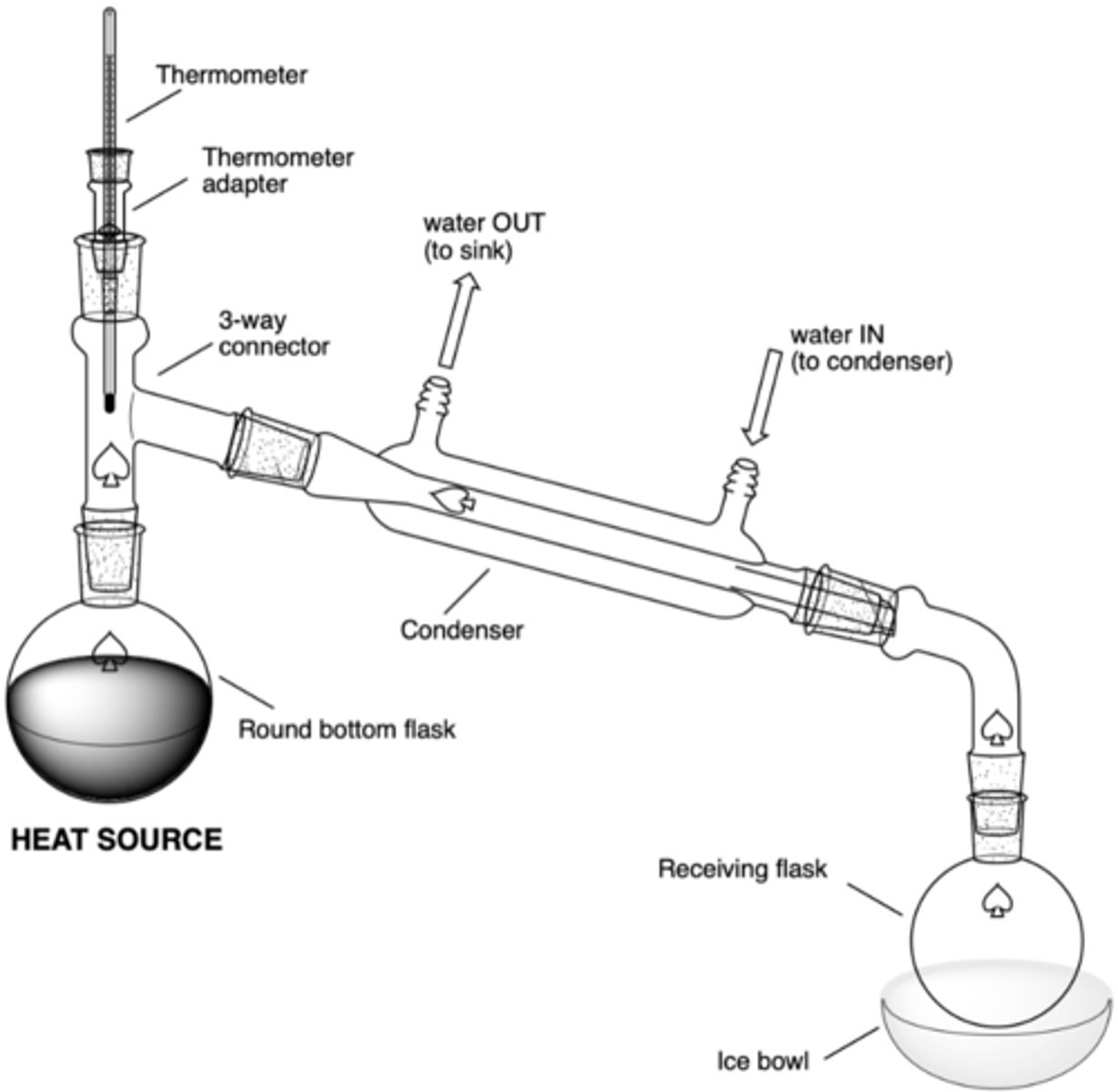
In a simple distillation set up, what will be the result of this error (why is this an error?):
the thermometer is missing
the boiling points cannot be measured
Which of the following is true regarding the microscale extraction at the end of Experiment 7 (dehydration of 2-methyl cyclohexanol)?
a. brine is added to help separate the aqueous and organic layer
b. the organic and aqueous layers are mixed with a metal spatula
c. solid MgSO4 is added to remove residual water from the organic layer
d. Saturated NaHCO3 is added to react with any residual acid
c. solid MgSO4 is added to remove residual water from the organic layer
d. Saturated NaHCO3 is added to react with any residual acid
After completing the dehydration of 2-methylcyclohexanol (Exp. 7) and measuring the IR spectrum of their product, some students found that they had a broad band at 3500 cm-1, several narrow bands around 3000-2800 cm -1, and more narrow bands at 1655 cm-1 and between 1150-1050 cm-1. What does this potentially mean?
Not all of the alcohol was dehydrated, so part of the reactants distilled over with the products
Why is benzene considered to be a "stable" molecule?
aromaticity
the reaction used to add a nitro (NO2) functional group to a benzene ring
nitration
What are the reagents of a nitration (of a benzene ring)?
Benzene and HNO3 + H2SO4
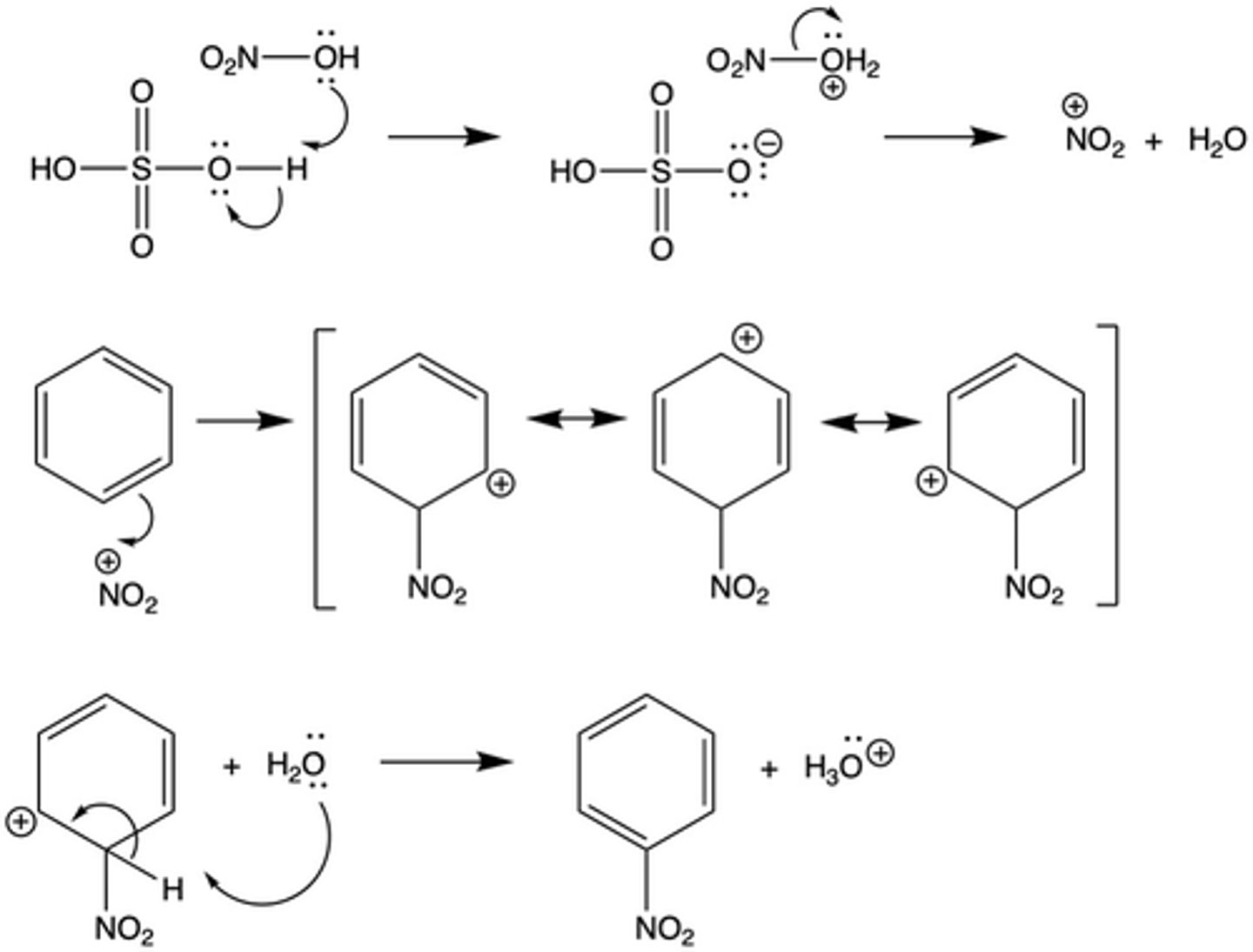
If there is already a functional group (i.e. the first substituent) on a benzene ring, what does this affect?
where the second substituent is added onto the ring
What factors control where the second substituent is added to a ring in electrophilic aromatic substitution?
chemical makeup as well as size of the substituents
Which group, electron donating or electron withdrawing, is considered to be ortho, para directors?
electron donating (activators)
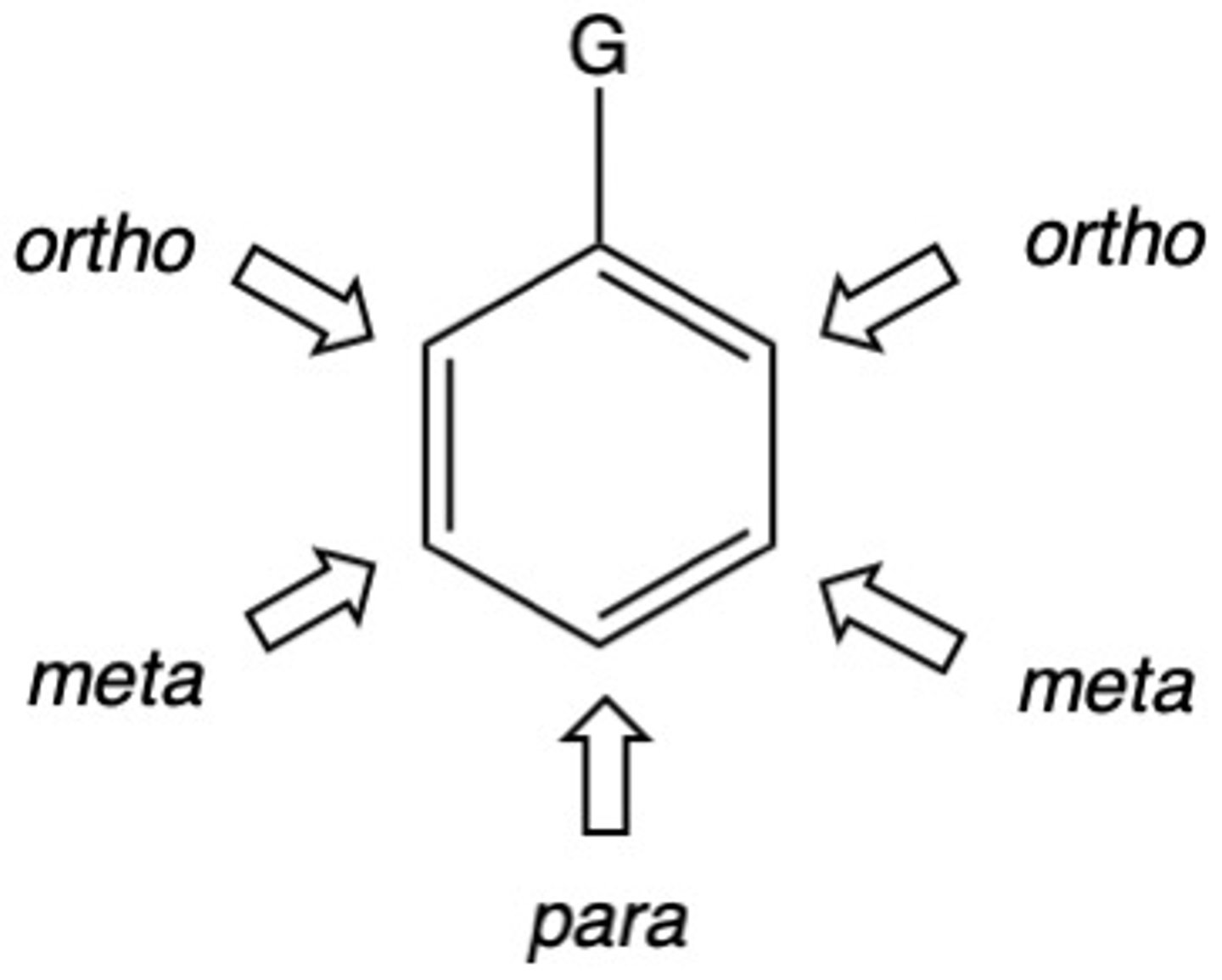
Which group, electron donating or electron withdrawing, is considered to be meta directors?
electron withdrawing groups
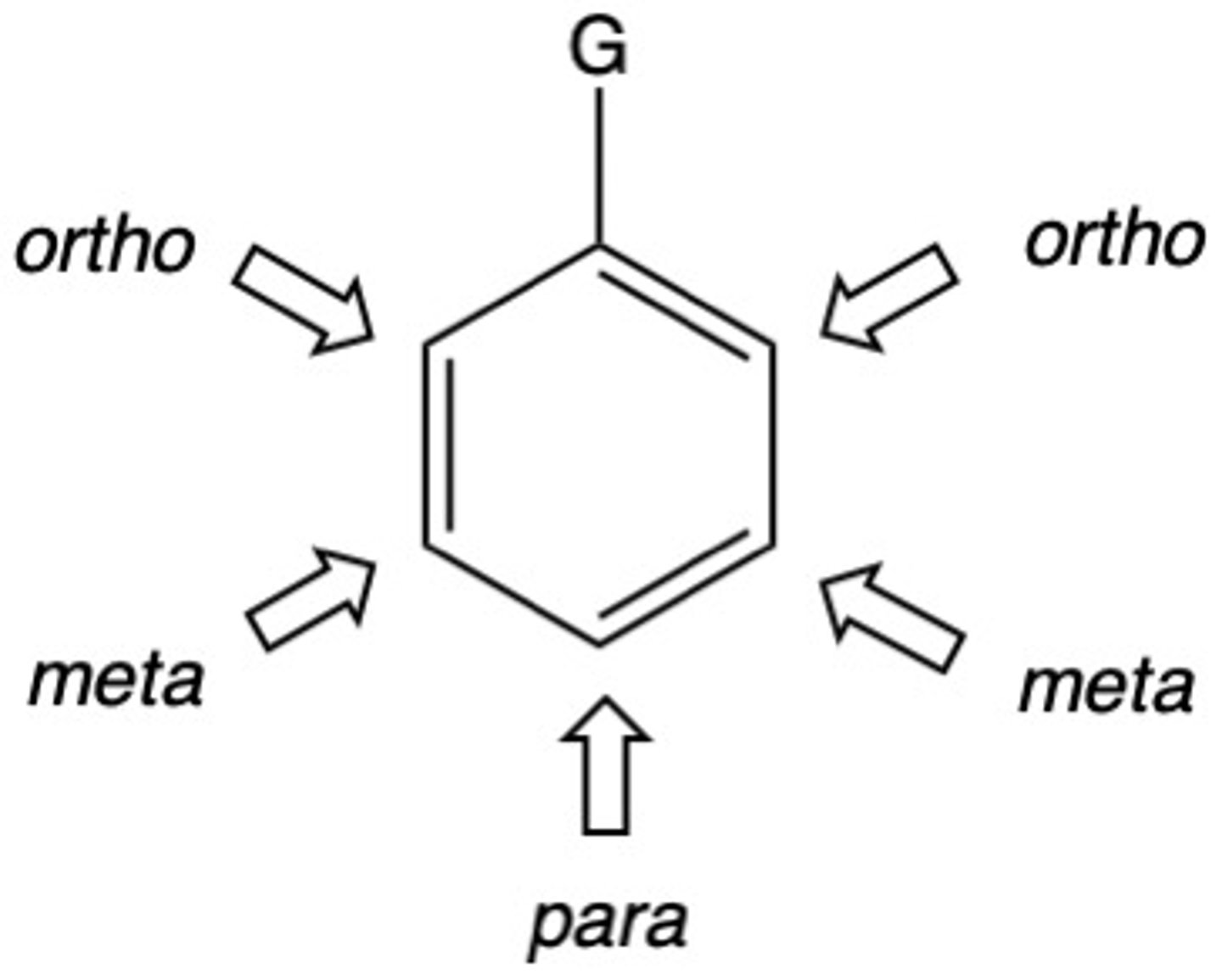
List the possible substituents on a benzene ring from most activating (most electron donating/ortho, para directors) to least activating (most electron withdrawing/meta directors).
ORTHO, PARA (activators/electron donating)
- OH, NH2
- OR, NHR, NR2
- OC=OR, NHC=OR
- R
- X
-H
META (deactivators/electron withdrawing)
- C=O
- NO2, NH3, NR3, C=N, SO3H, CX3
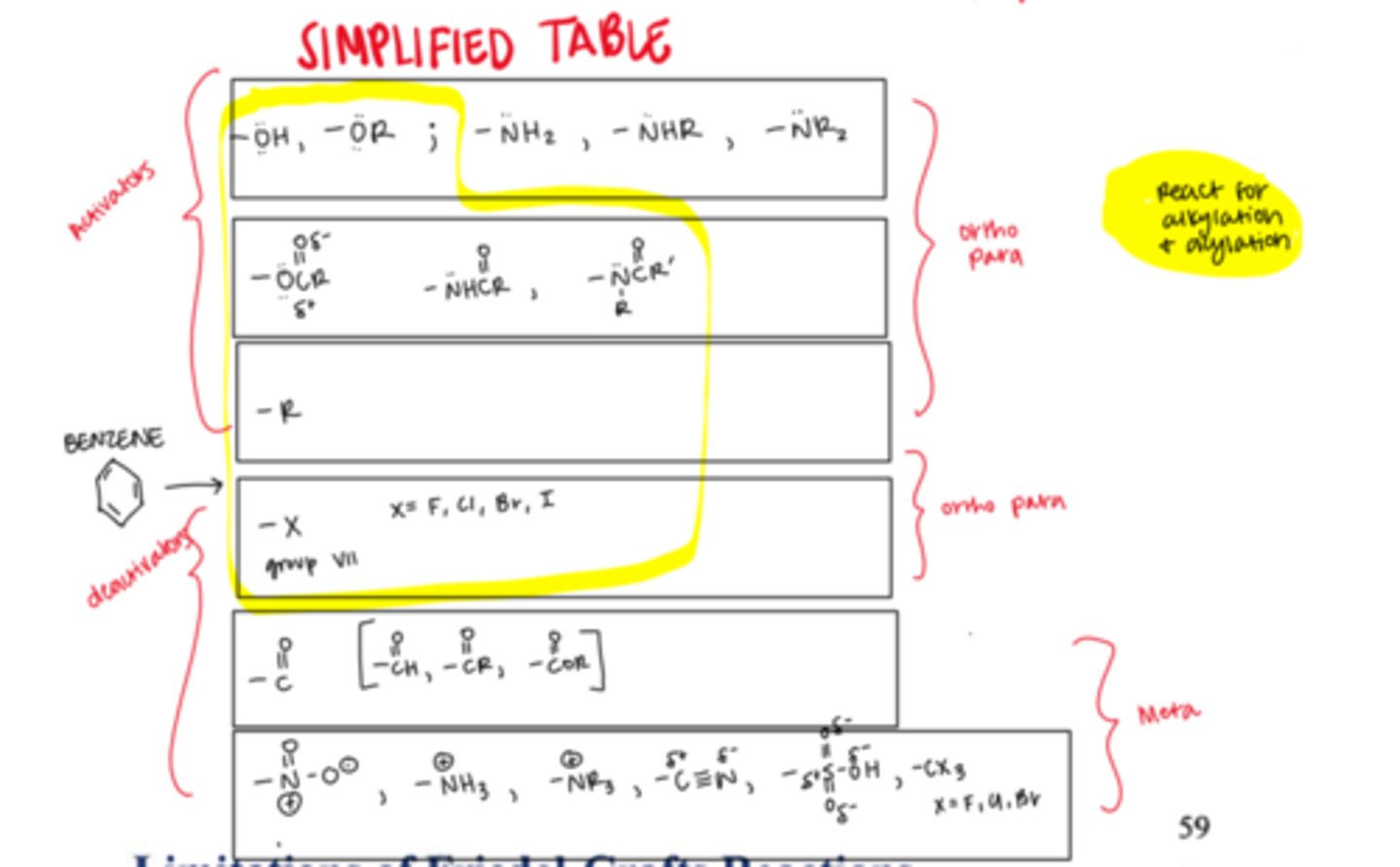
Because bromine is a halogen, is it an ortho, para director or a meta director?
ortho para director (despite being somewhat of a deactivator)
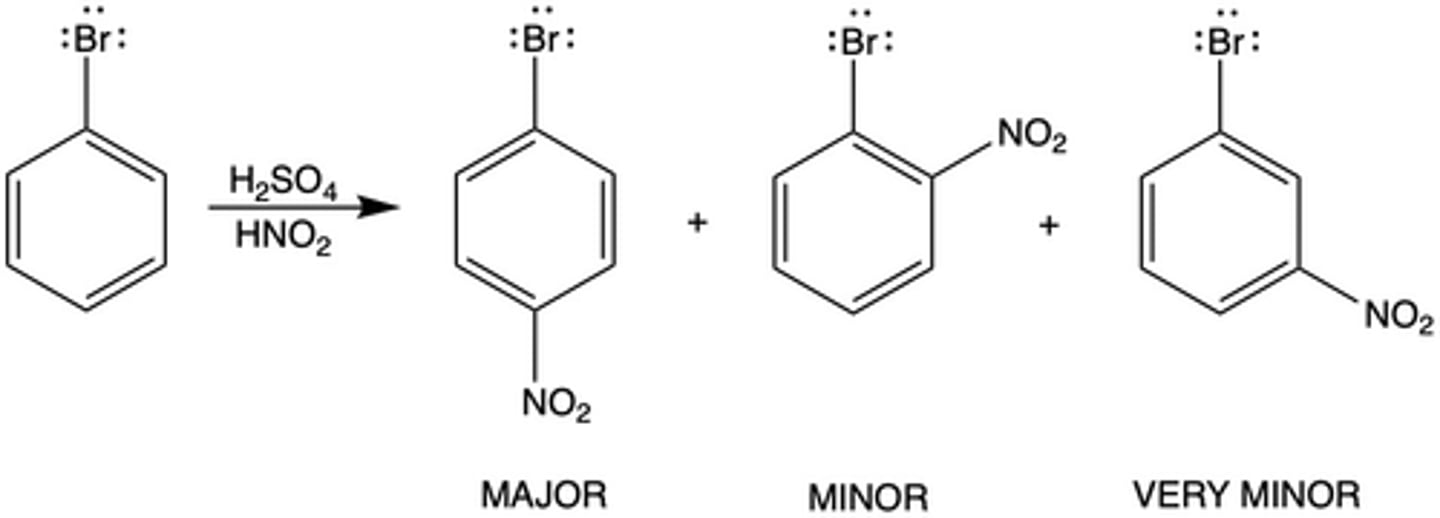
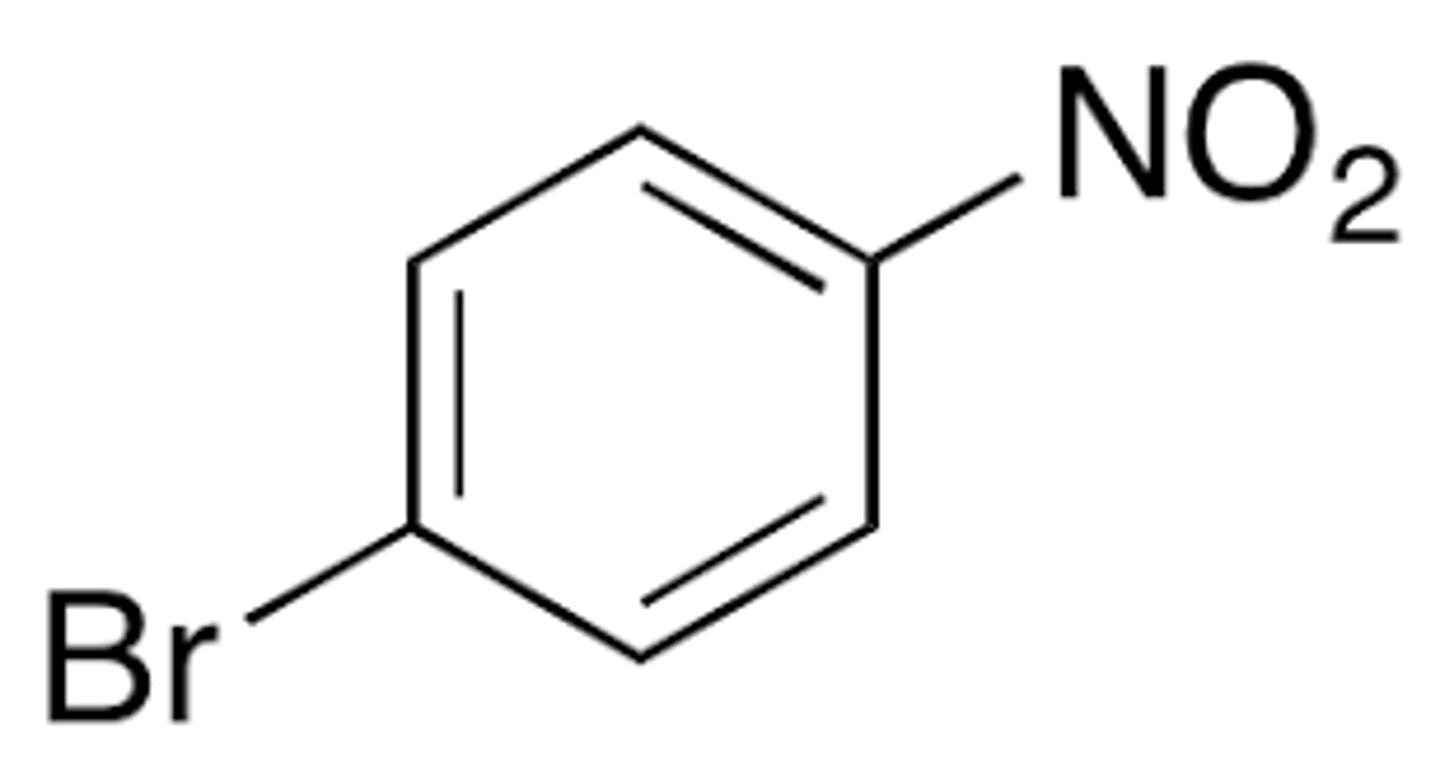
When the nitro group is added to bromobenzene through EAS (Exp. 8) what position is the nitro group added to that makes up the major product? Why?
para position; the ortho position is sterically hindered (blocked) due to the somewhat large BR atom and bulky nitro group
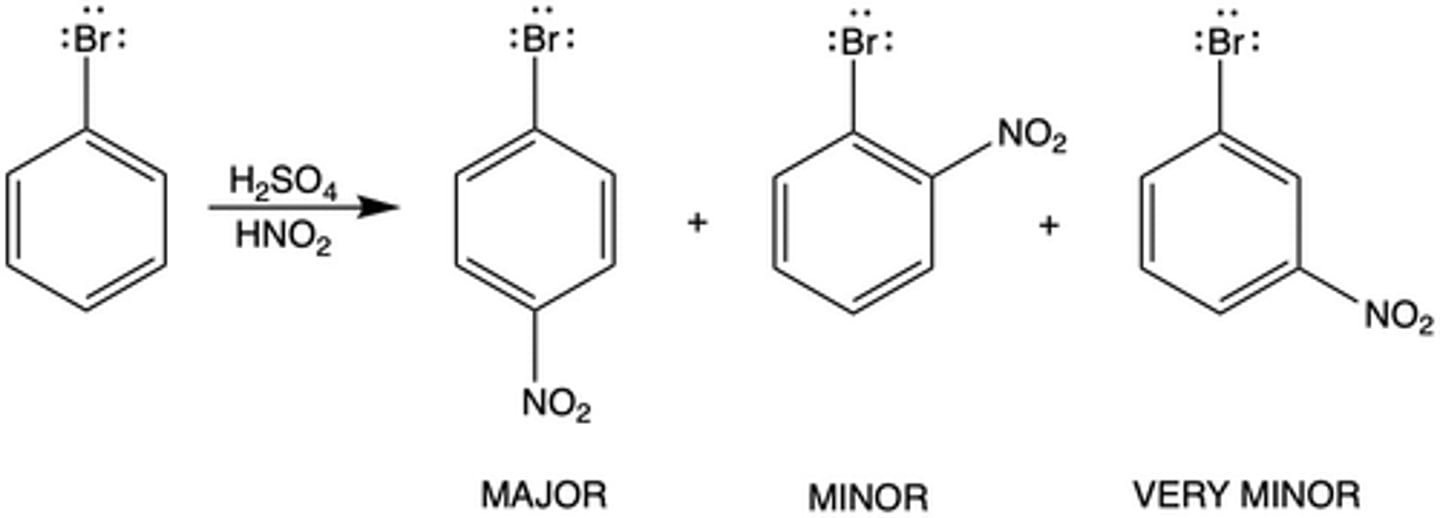
If nitrobenzene was reacted with bromine, what position would bromine be added to on the benzene ring (Exp. 8--EAS)?
meta position
What functional group is the most strongly deactivating meta director?
Nitro (NO2)
technique used to verify the structure of a molecule in organic chemistry; focuses on a signal generated from each H atom and can generally tell the number of H atoms in a molecule by integrating the area beneath the peak
NMR spectroscopy
What atom produces signals in NMR spectroscopy most commonly?
H1 (proton)
How can NMR provide the amount of hydrogens found in a molecule?
Because each H gives a signal (which is a peak), this technique can generally tell the number of H atoms in a molecule by integrating the area beneath the peak.
the location of the peak along the x-axis of the NMR spectrum
chemical shift
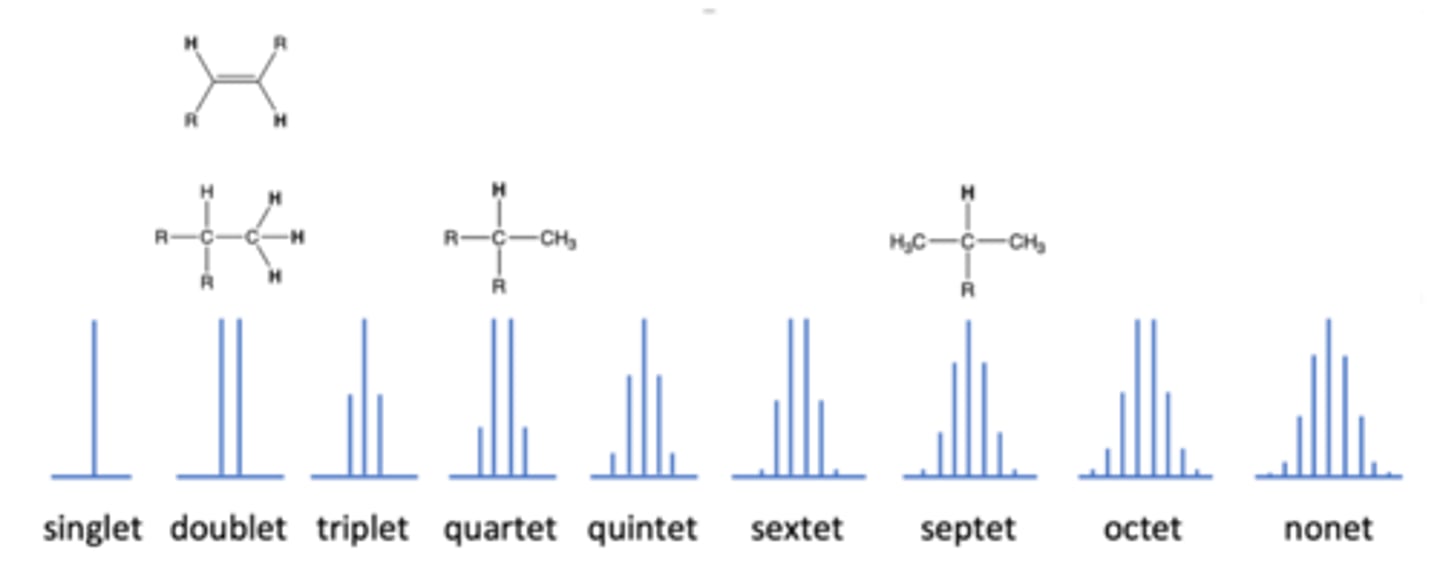
What causes a peak to "split" in NMR spectroscopy?
If there are protons on the atom adjacent to the H atom, the peak will split (splitting of peaks = n + 1, where n is number of neighboring H atoms)
occurs in NMR spectrum when peak splits in two because there is one adjacent H atom
doublet
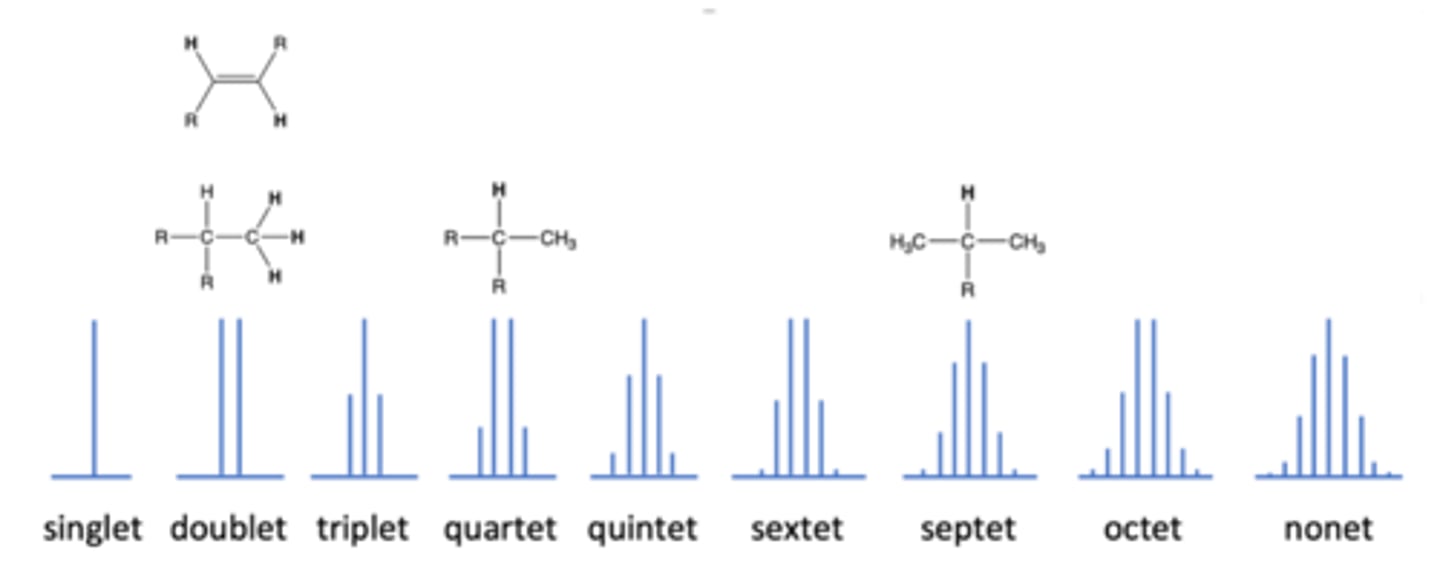
occurs in NMR when the peak splits in three because there are 2 adjacent H atoms
triplet
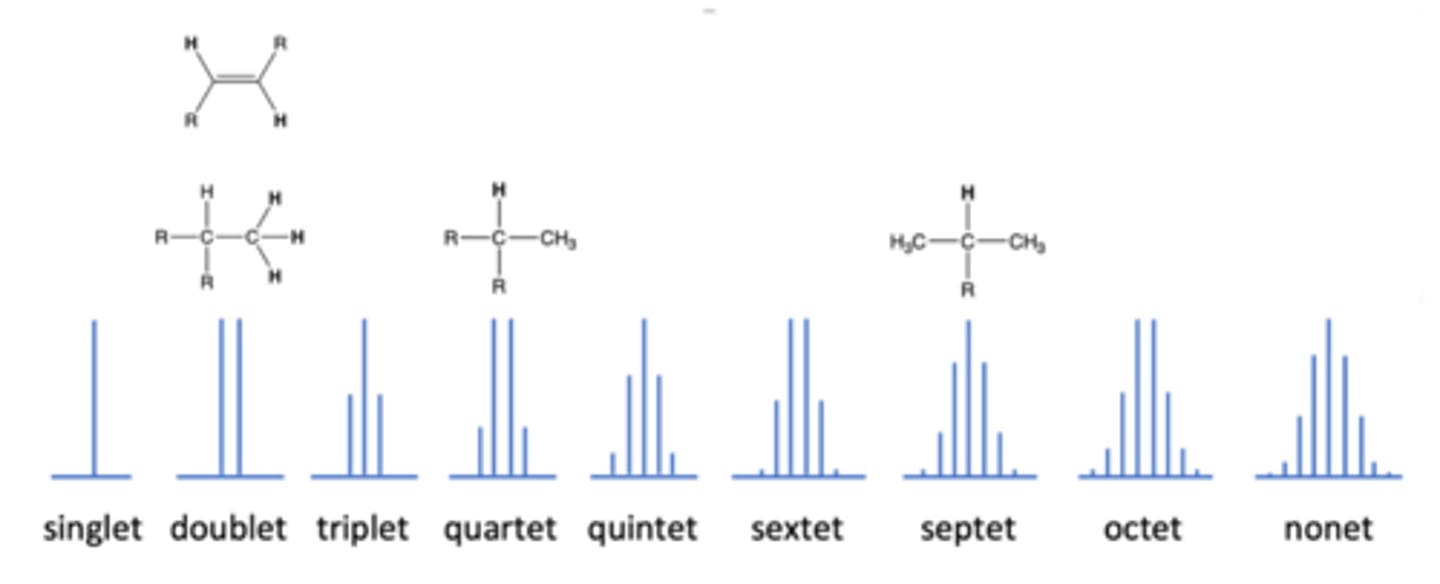
occurs in NMR when the peak splits in 4 because there are three adjacent H atoms
quartet
Explain why p-bromonitrobenzene is the major product in this experiment (Exp. 8), not both the ortho and para products.
Steric hindrance in the ortho position because of the large Br molecule near the nitro group causes the para position to be favored because it is less sterically hindered.
What are important safety considerations to have in place before working with strong acids (such as with sulfuric acid or nitric acid)?
- Sulfuric acid and nitric acids are extremely corrosive and must be handled with extreme care.
- Make sure that your gloves are in good condition before handling acids to prevent any acid getting to your skin.
- Pour all acids under the fume hoods
- After putting the acid in your Erlenmeyer flask, put HNaCO3 in the sink and rinse the flask immediately with water.
- Check if any spills on the bench are acids by pouring HNaCO3 over it, seeing if it fizzes, THEN disposing of properly.
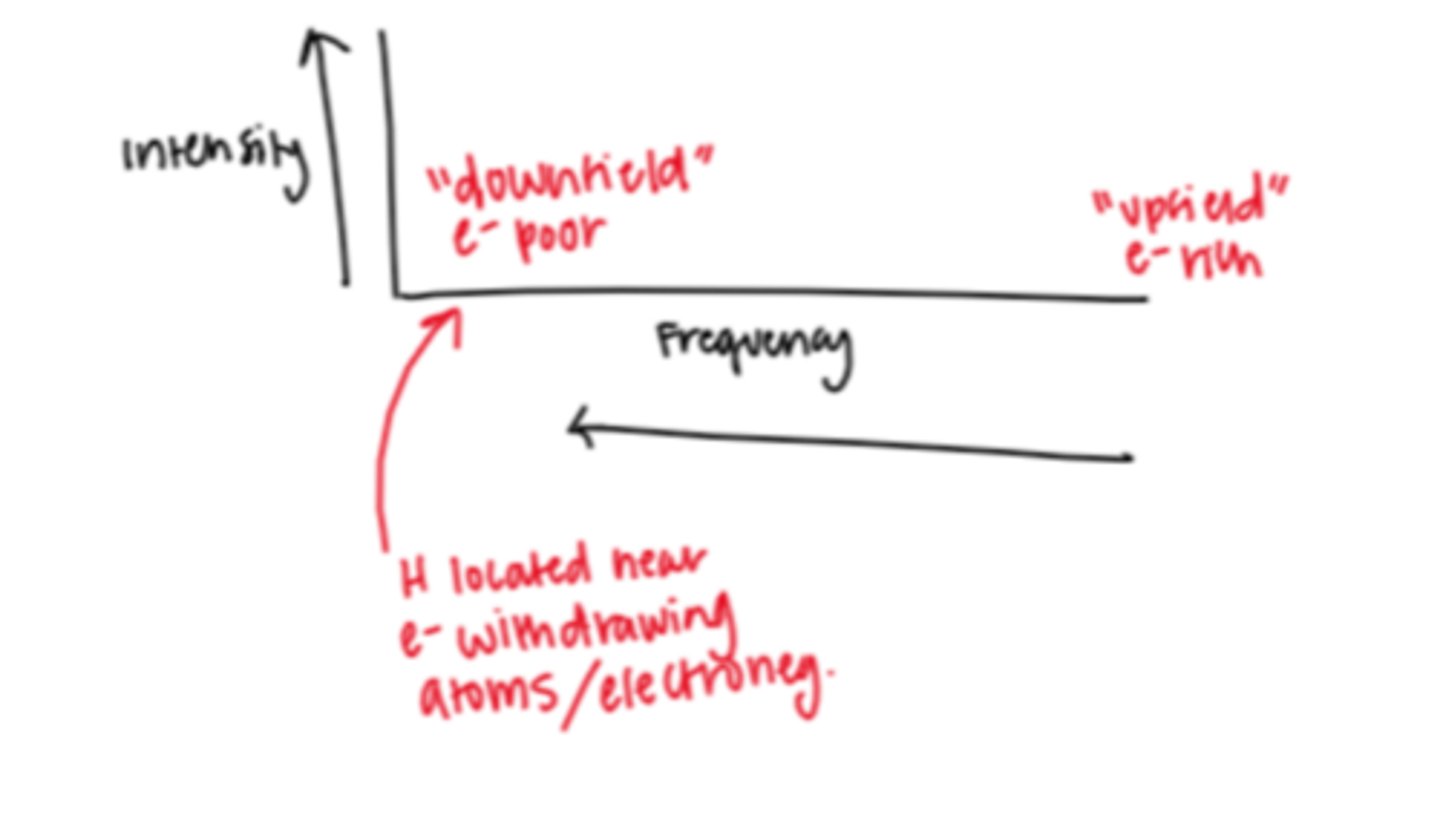
If a peak is located farther left in an H-NMR spectrum what does this mean about the H in the signal?
located "downfield" so they are more electron poor (located near e- withdrawing atoms)
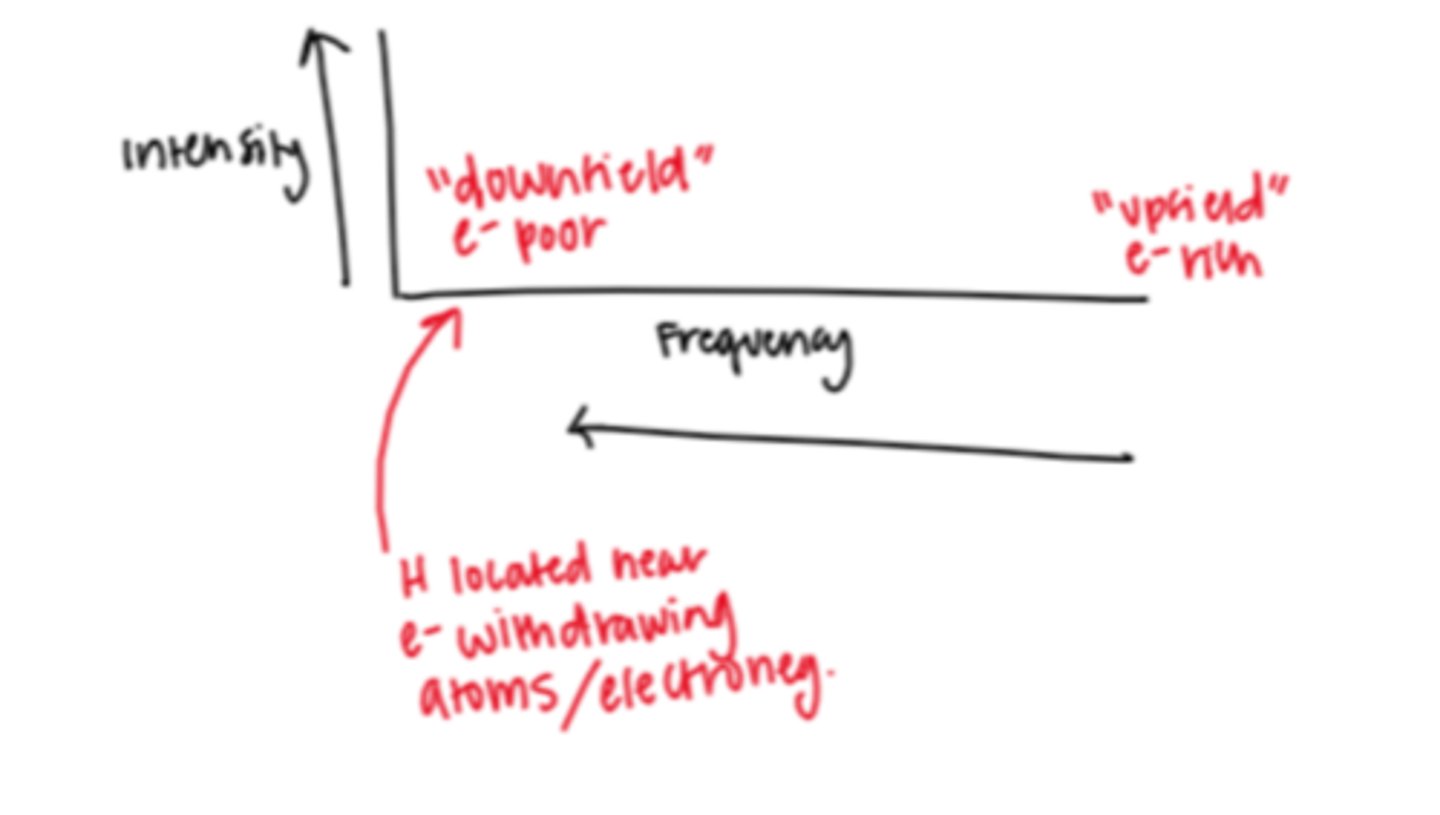
If a peak is located farther right in an H-NMR spectrum what does this mean about the H in the signal?
located "upfield" so they are more electron rich (NOT next to e- withdrawing molecules)
What is the formula to determine the number of adjacent hydrogens when examining splitting in an NMR spectrum?
N + 1
What is the formula for the unsaturation index of a compound?
UI = 2C + 2 - H - X + N / 2
What information does the unsaturation index of a compound give you?
the number of double bonds and/or rings in the molecule (ex: benzene has an UI of 4)
In Exp. 8, why is the product first vacuum filtered with water? What is the purpose of the ethanol rinse?
The first vacuum filtration is to filter out the acid from the original reaction; the purpose of the ethanol rinse is to remove water from the product.
What benefit is there by analyzing a product by both IR spectroscopy and NMR spectroscopy?
better understanding of which functional groups are in the molecule, as well as the C-H framework of the molecule
How would the NMR spectrum of o-bromonitrobenzene appear?
The "equivalent hydrogens" of a benzene ring would no longer be equivalent; each hydrogen on the benzene ring would produce its own signal
What are the chemical hazards of:
bromobenzene
flammable, irritant, environmental hazard
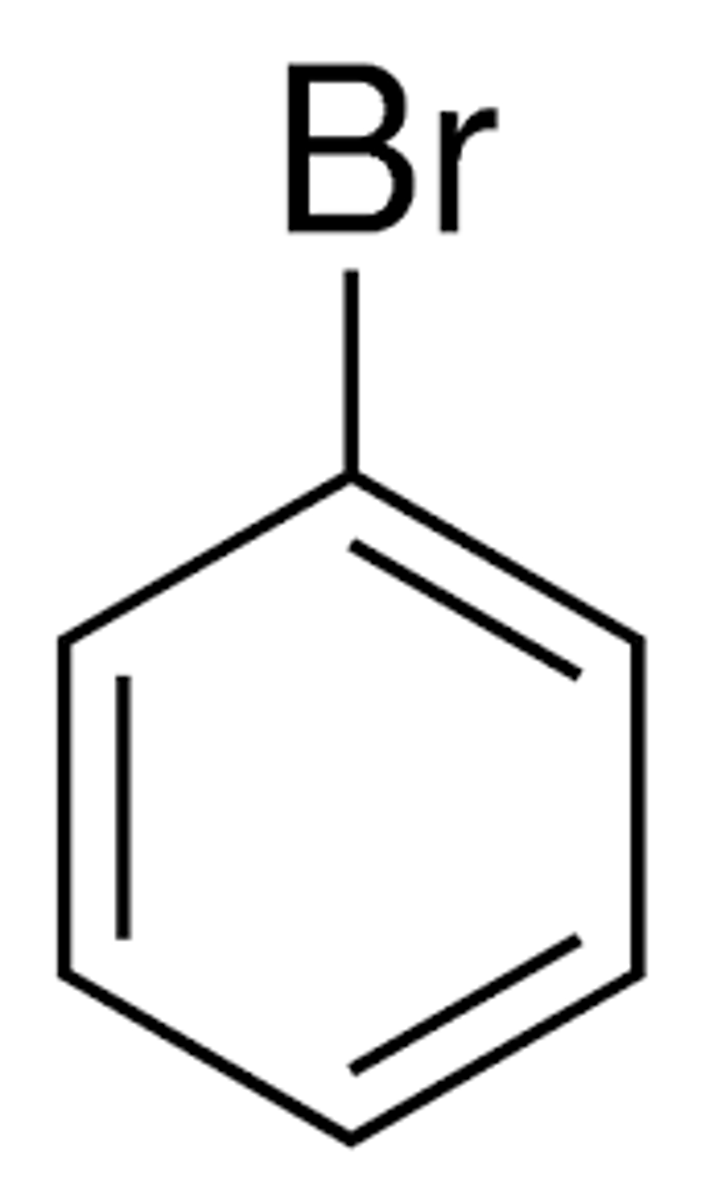
What are the chemical hazards of:
sulfuric acid (H2SO4)
corrosive
What are the chemical hazards of:
nitric acid (HNO3)
strong oxidizer, toxic, corrosive
What are the chemical hazards of:
p-bromonitrobenzene
irritant, harmful if swallowed