Chem117 Exam 4 Reactions
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
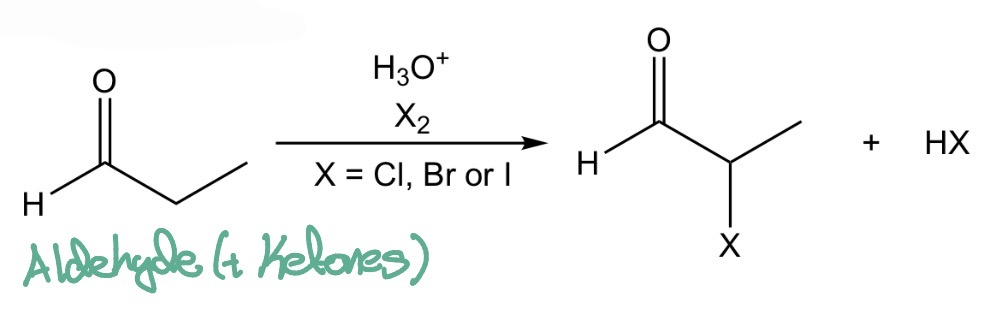
Aledyhyde/Ketone —> α-halo aldehyde/ketone
Acid-Catalysed α-halogenation
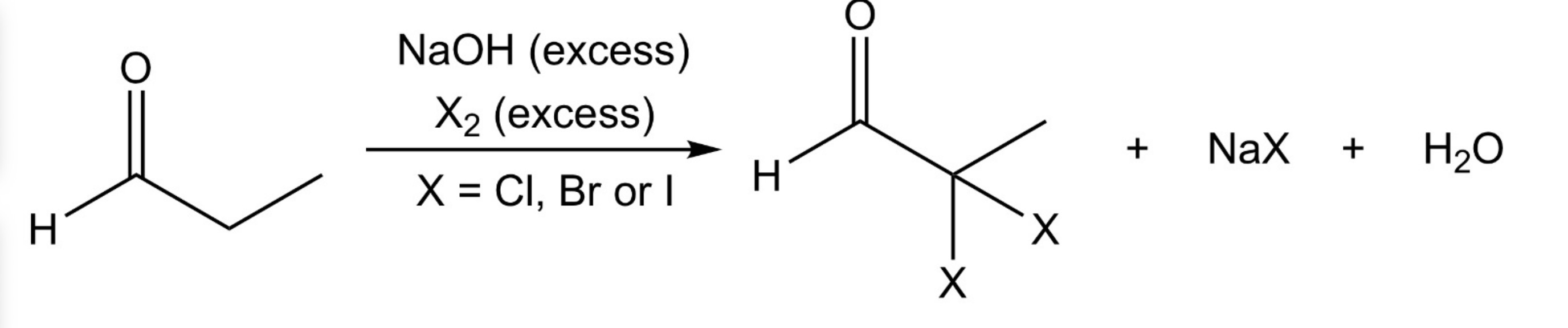
Aldehyde/Ketone —> α,α dihalo aldehyde/ketone
Base-promoted α-halogenation
Aldehyde/Ketone —> Carboxylic Acid + Haloform
HVZ Reaction
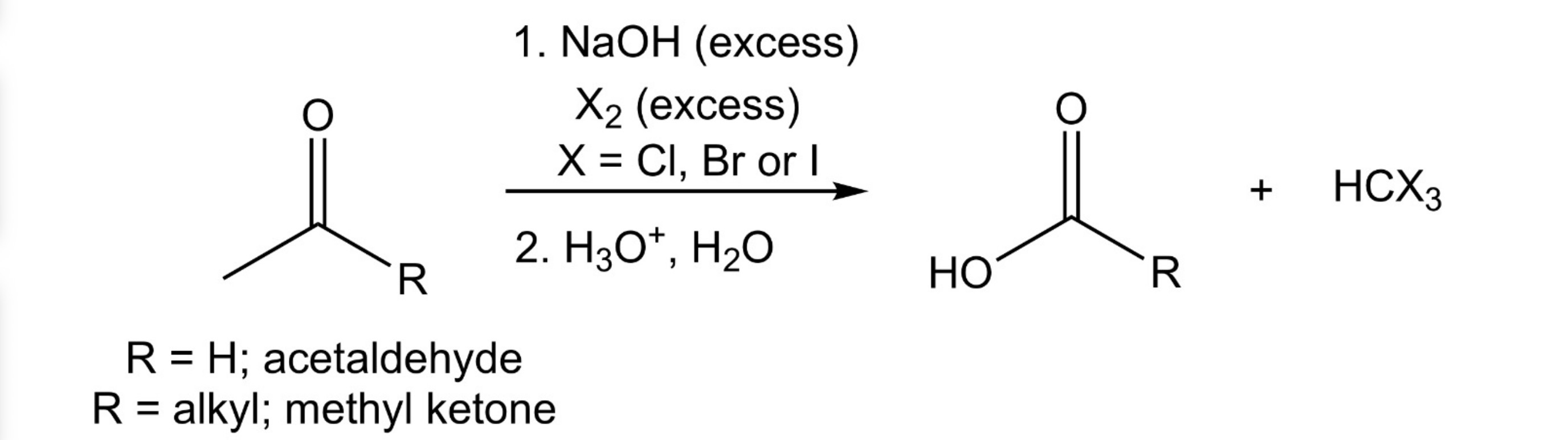
Carboxylic Acid —> α-bromo acid halide
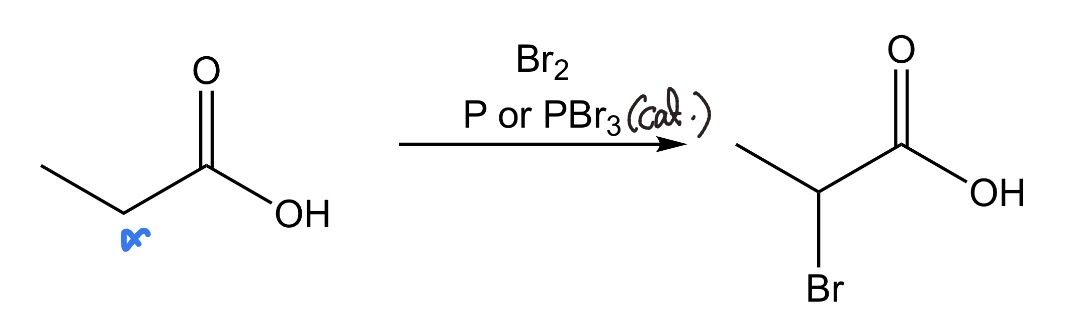
2 Aldehydes/Ketones —> β-hydroxyaldehyde/ketone
Adol Reaction
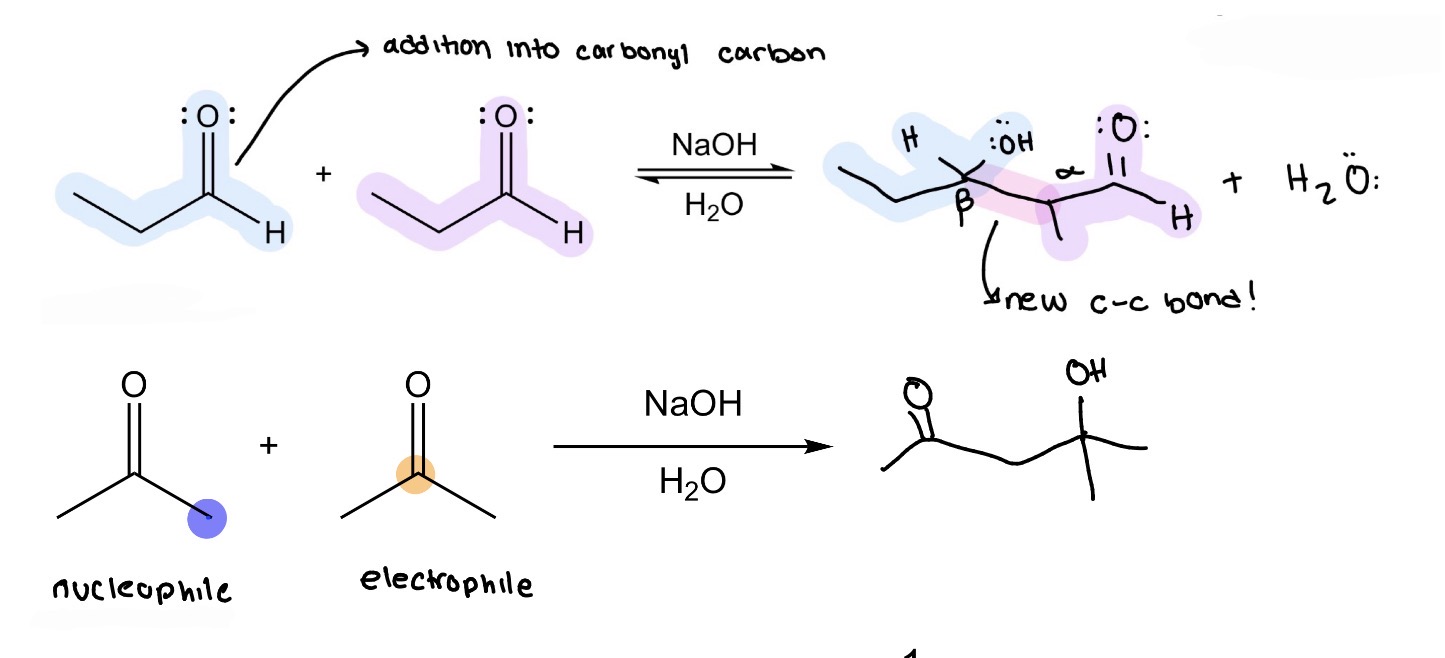
β hydroxy aldehyde —> α,β unsaturated aldehyde
Adol Condensation
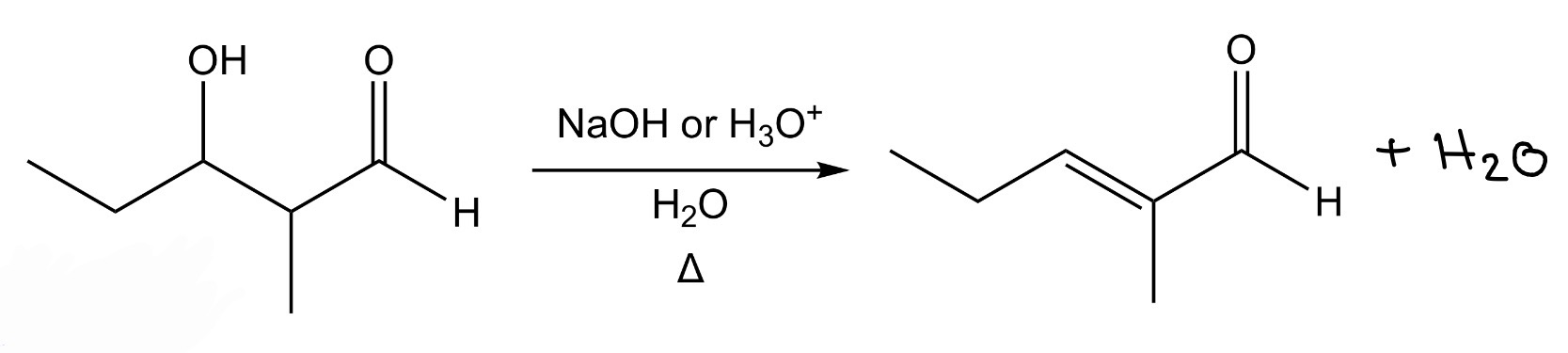
2 diff ketones/aldehydes —> β-hydroxy ketone/aldehyde mixture of products
Crossed Adol Addition
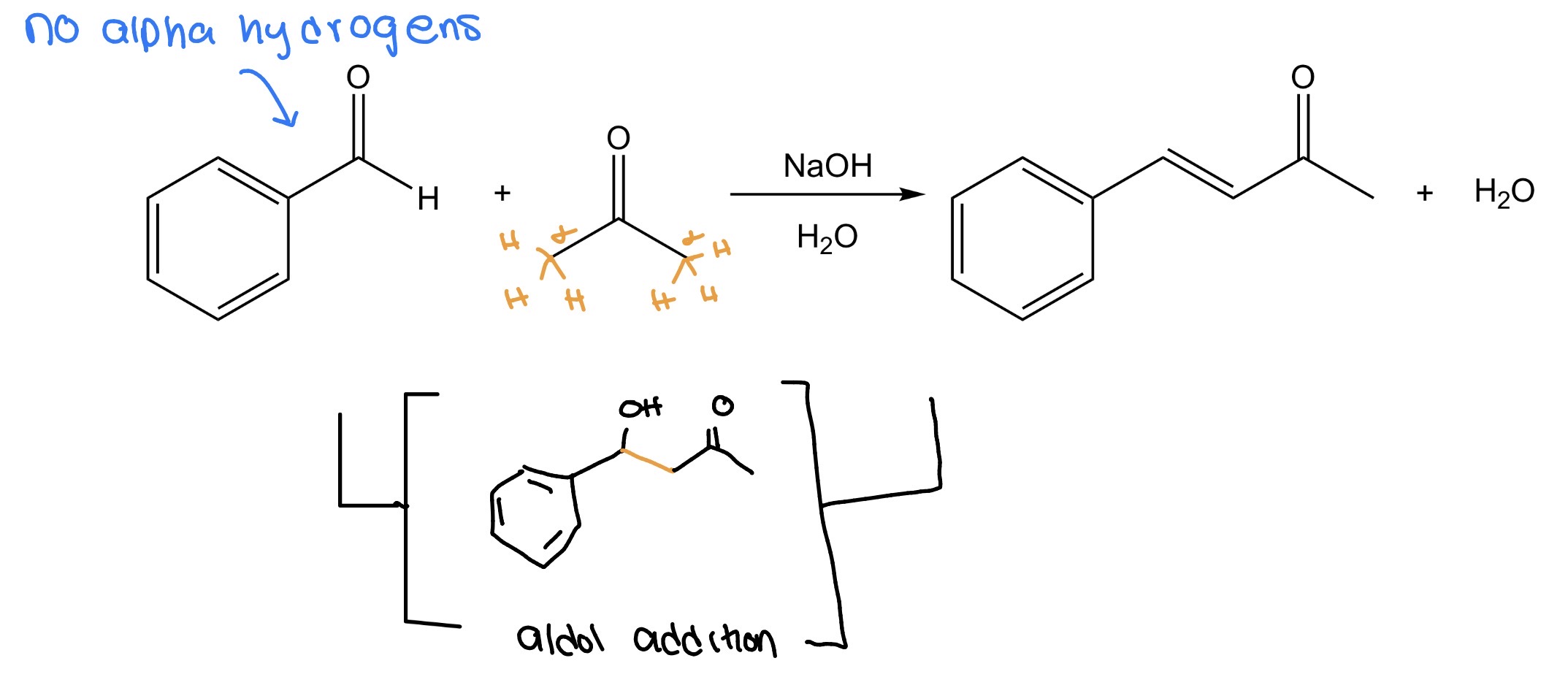
Aldehydes more reactive electrophilic bc less sterically hindered + less e- donating
ketone typically enolate donor
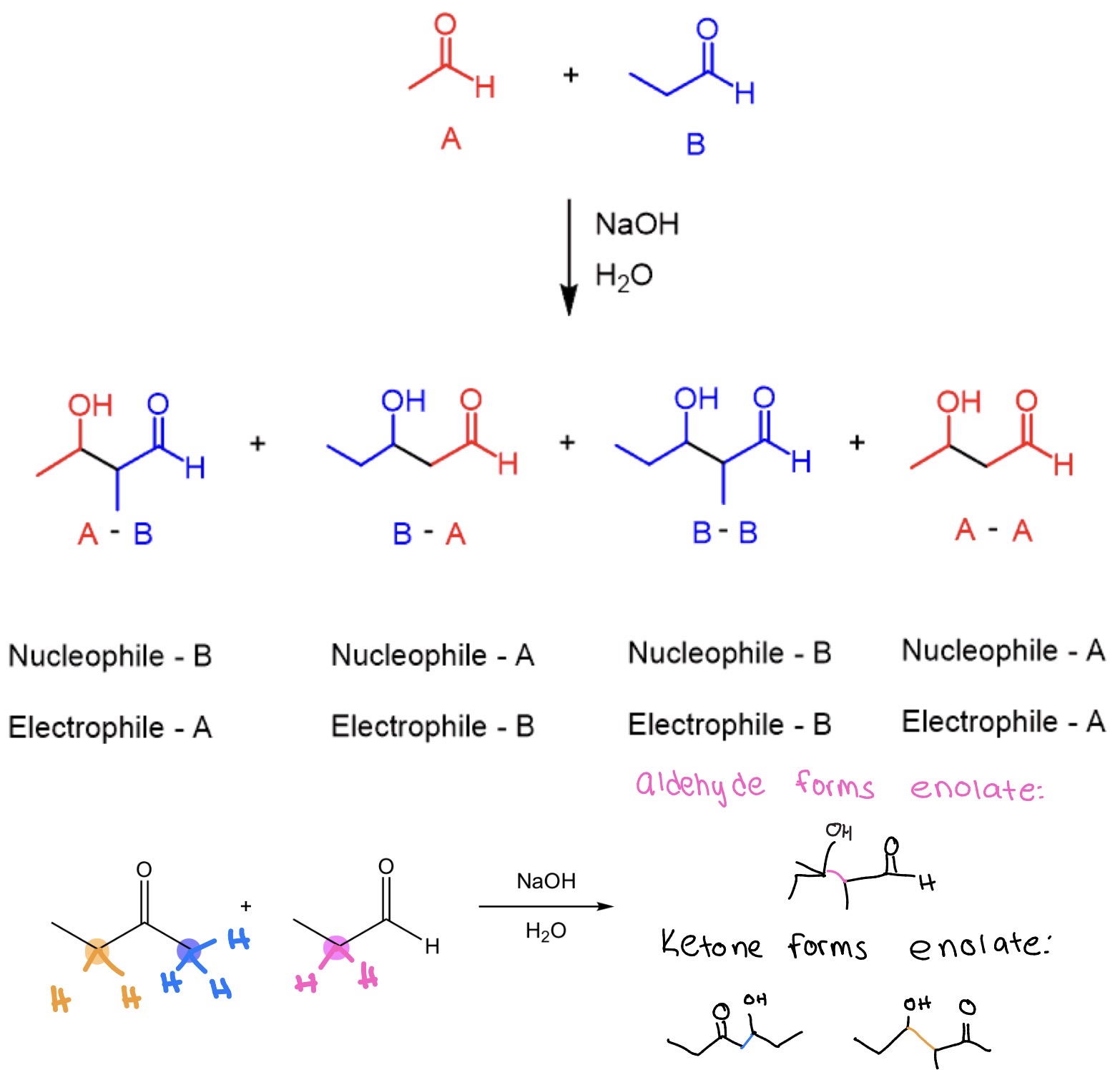
Ketone/Aldehyde w/ α hydrogens + w/ out—> α,β unsaturated ketone/aldehyde
Claisen-Schmidt Condensation
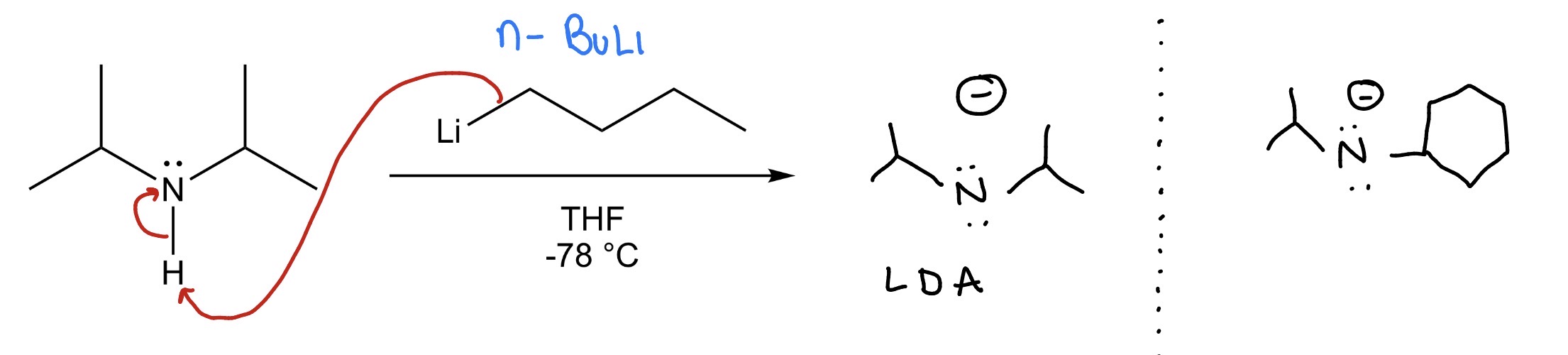
secondary amine —> LDA
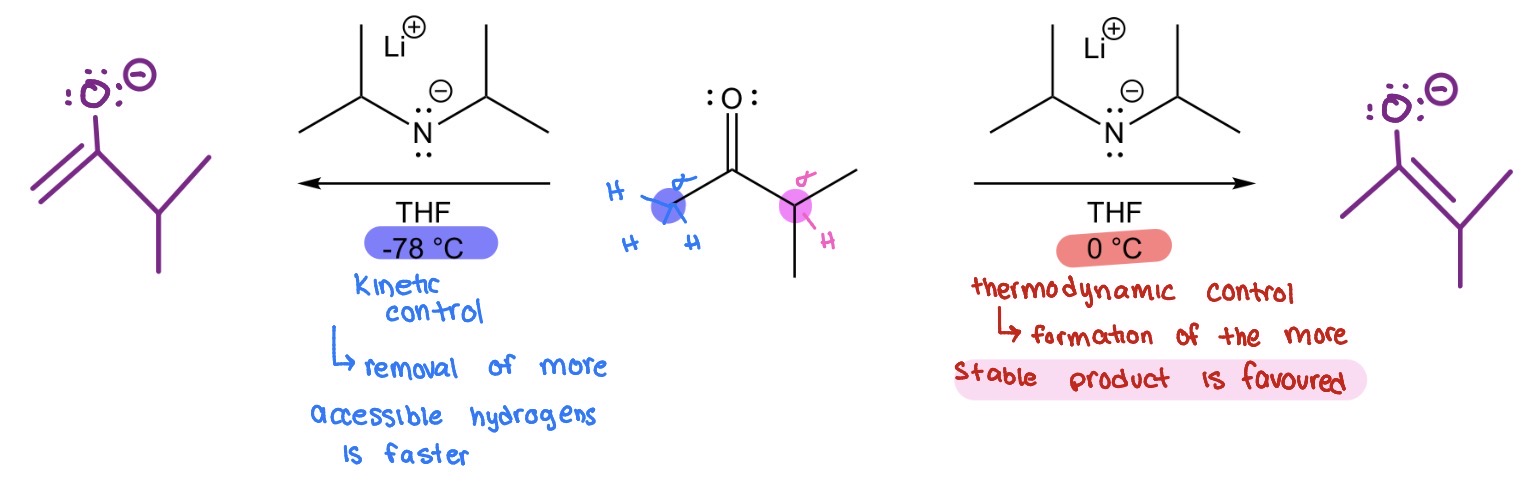
Ketone —> enolate (2 conditions)
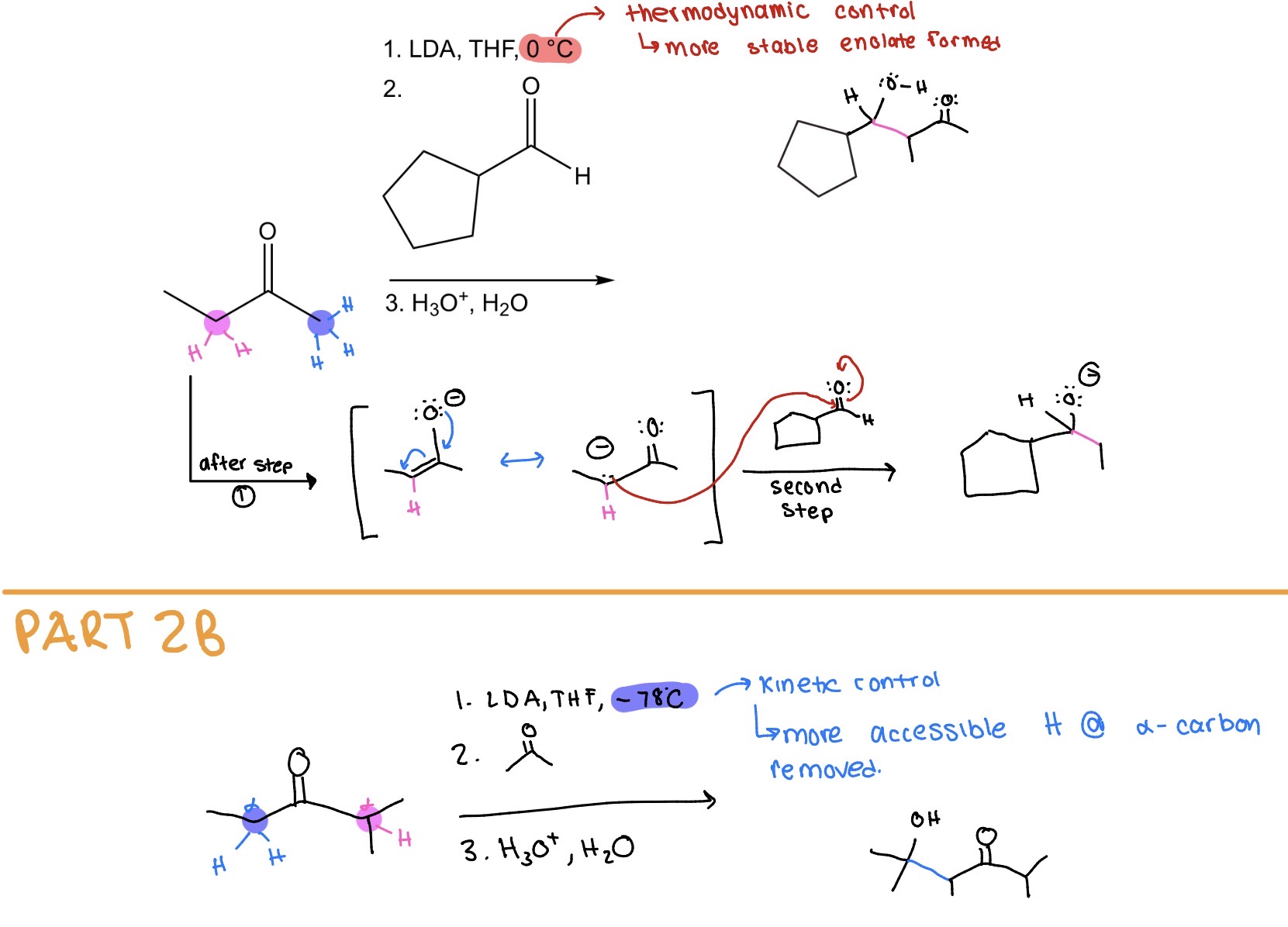
Ketone —> β-hydroxyketone +Cs (2 conditions)
cmpd w 2+ aldehyde/ketone —> 5/6-membered ring
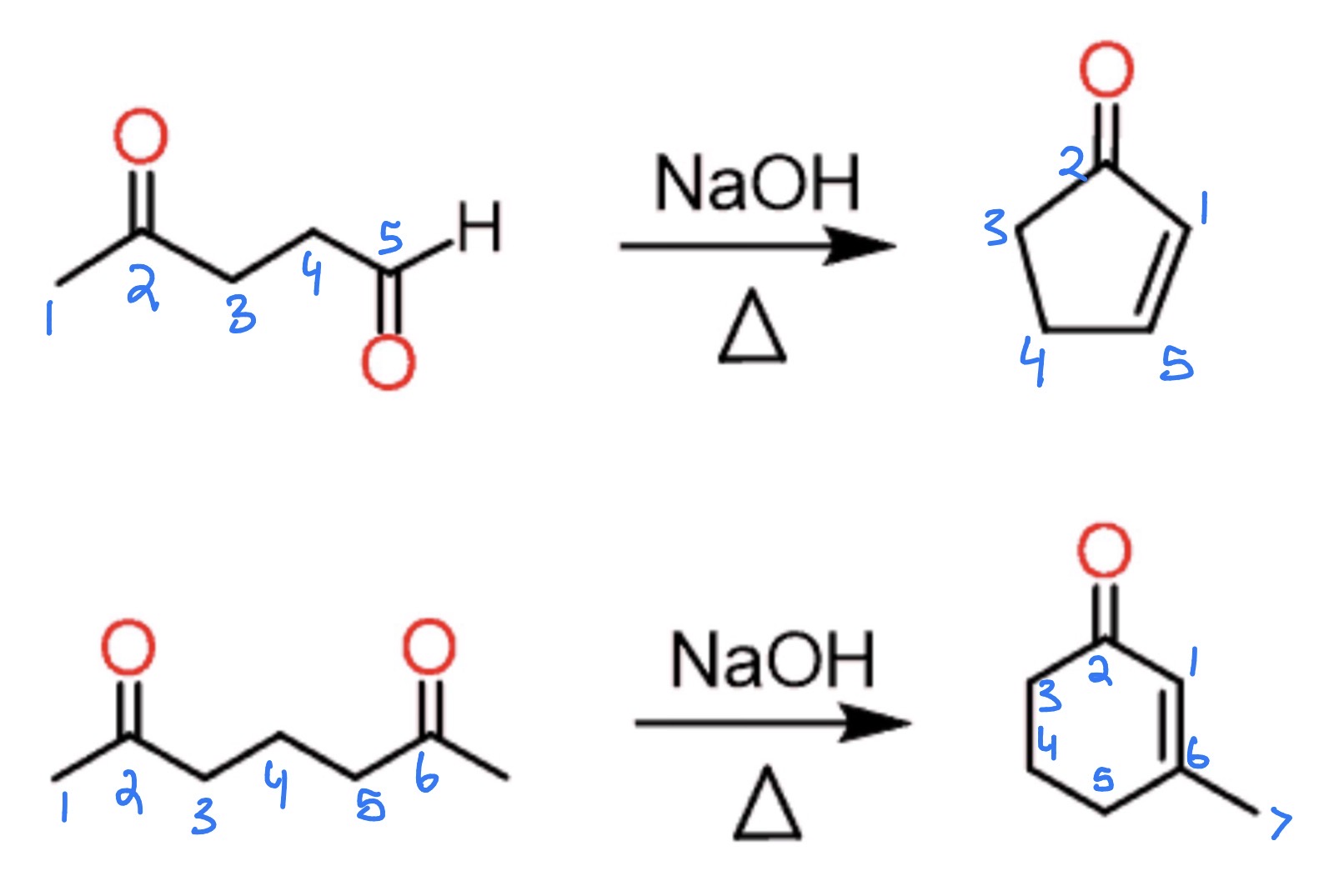
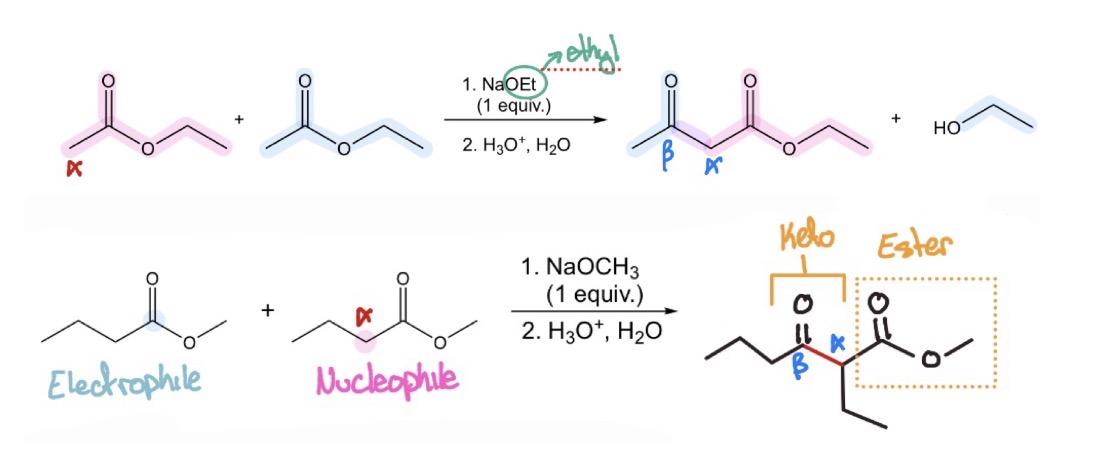
2 esters —> β-keto ester
Claisen Condensation
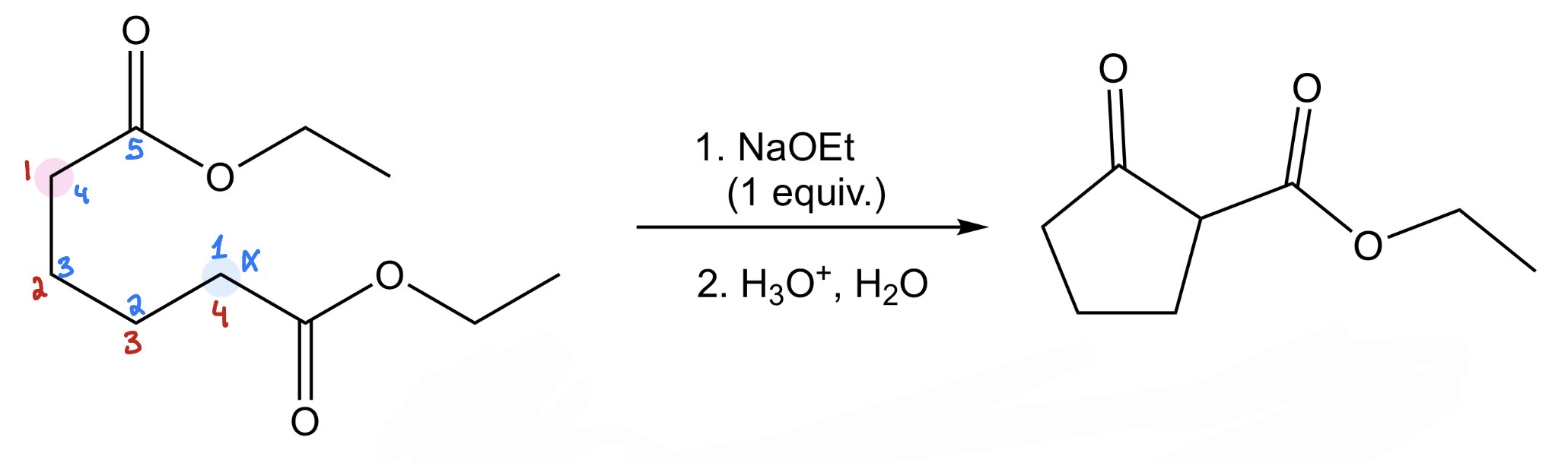
diester —> cyclic β-keto ester
Dieckmann Condensation
ester enolates/dimethyl carbonate w/ α hydrogens + w/ out—> β-keto esters/β-diesters
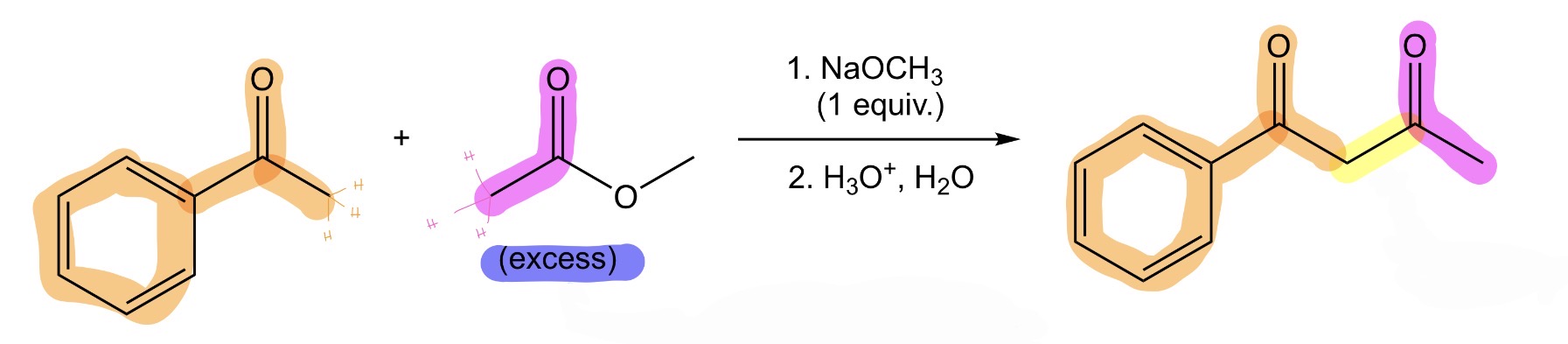
Crossed Claisen Condensation
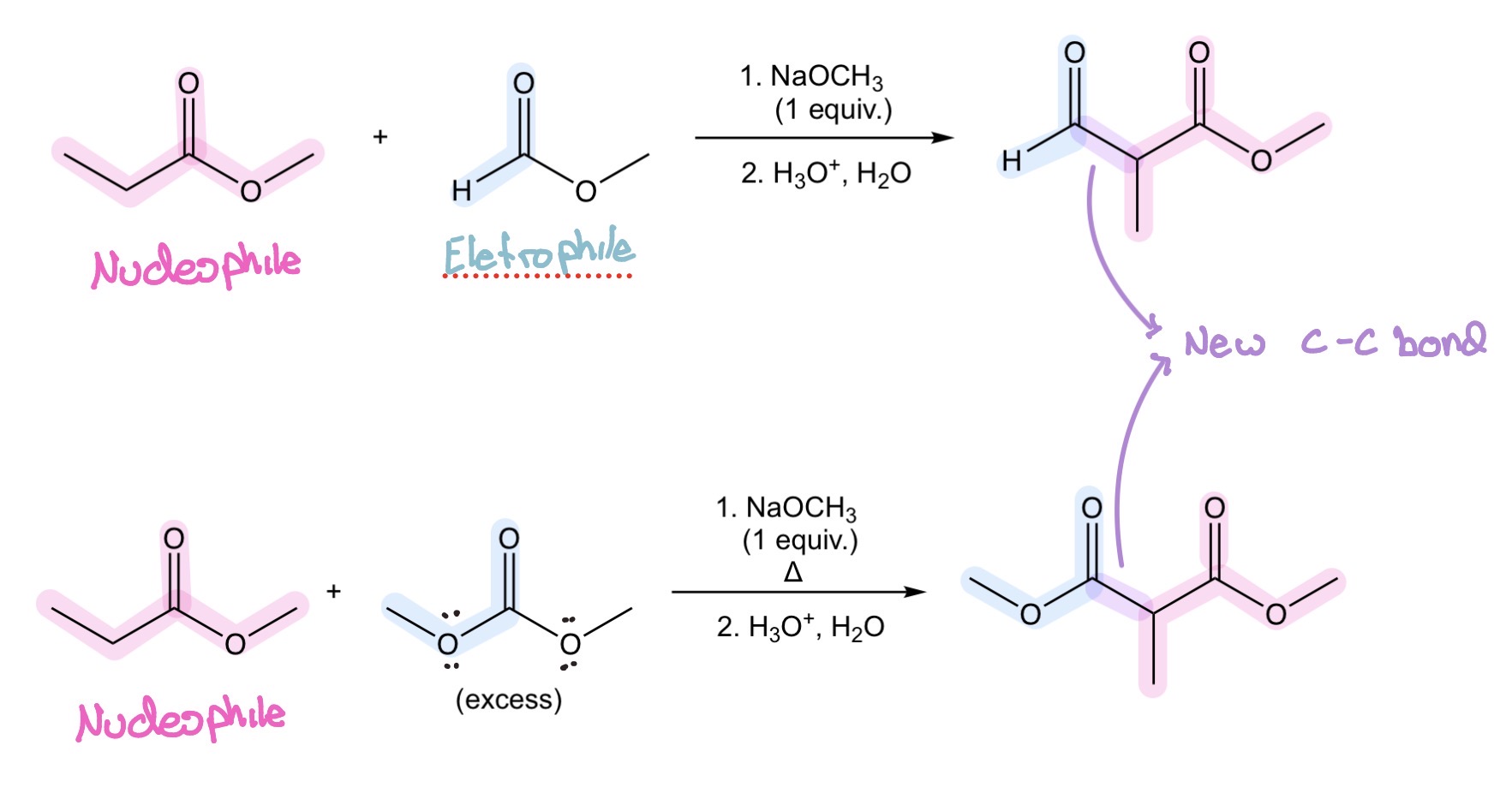
ketone + ester —> β-diketone