Quant exam 4
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
what are potentiometric methods based on?
potentiometric methods are based on measuring the potential of electrochemical cells without drawing appreciable current
what is the analyte in potentiometry?
the analyte is an electroactive species that participates in redox by donating or accepting an electron at an electrode within the galvanic cell
what is an indicator/working electrode?
electrons are transferred to or from the analyte through an electrode (i.e. a Pt wire) which is referred to as an indicator or working electrode
what is a reference electrode
the second half-cell is fixed (i.e., no unknown/analyte) providing constant potential, and thus called the reference electrode
what reference electrodes did we talk about in lecture
silver/silver chloride
saturated calomel (SCE)
standard hydrogen electrode (SHE)
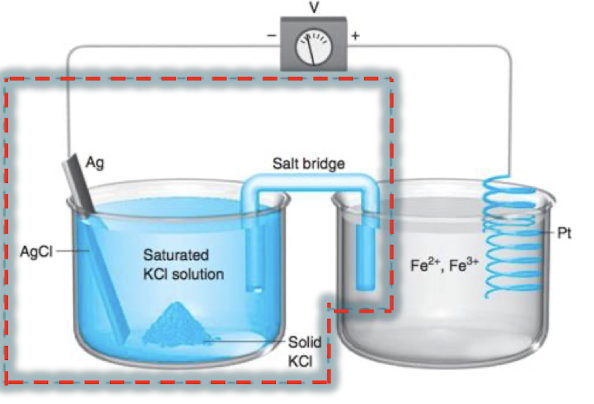
what type of cell is this?
galvanic cell
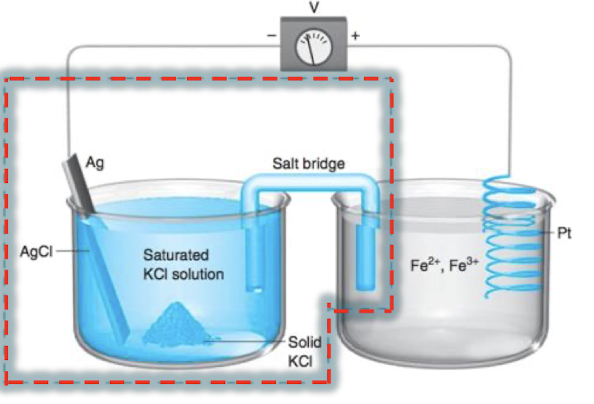
which is the anode?
the Ag/AgCl half cell
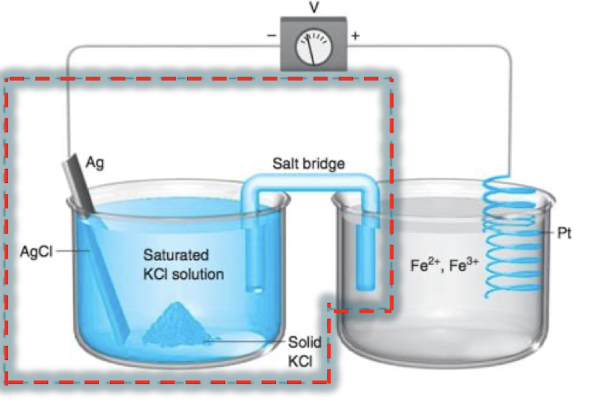
which is the working electrode?
the cell measuring electroactive species, the Pt/Fe2+, Fe3+ half cell
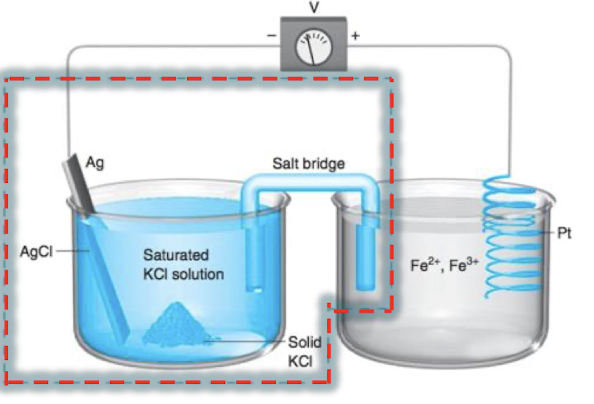
what is true at the reference electrode?
[Cl-] should be constant because it is in a saturated KCl solution. this means that the potential from this half cell is constant
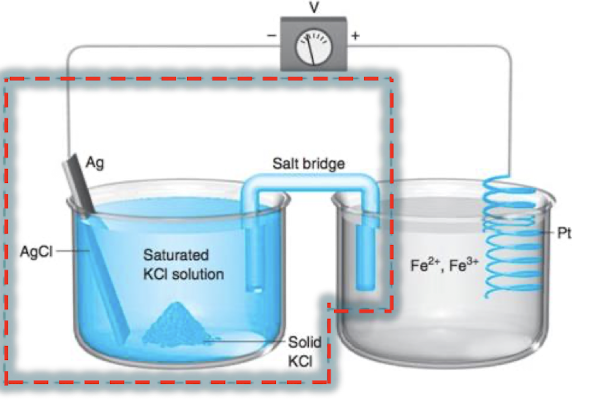
write the half reactions
right: Fe3+ + e- → Fe2-
left: AgCl + e- → Ag + Cl-
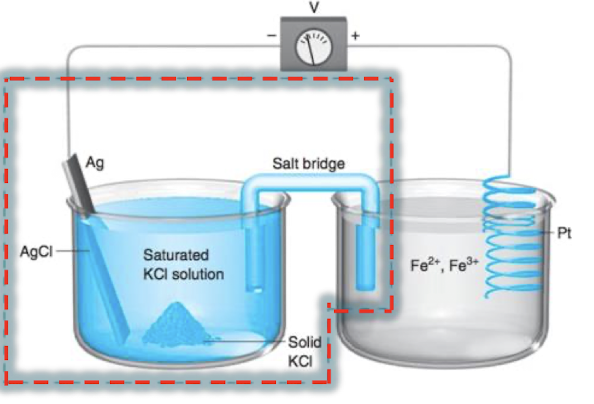
write the reactions in cell notation
Ag (s) | AgCl (s) | Cl- (aq) || Fe2+, Fe3+ (aq) | Pb (s)
what are the benefits of using double-junction reference electrodes?
they minimize interaction between reference KCl solution and sample solution
longer lifetime
best for samples with heavy metals, proteins etc. that can diminish performance over time
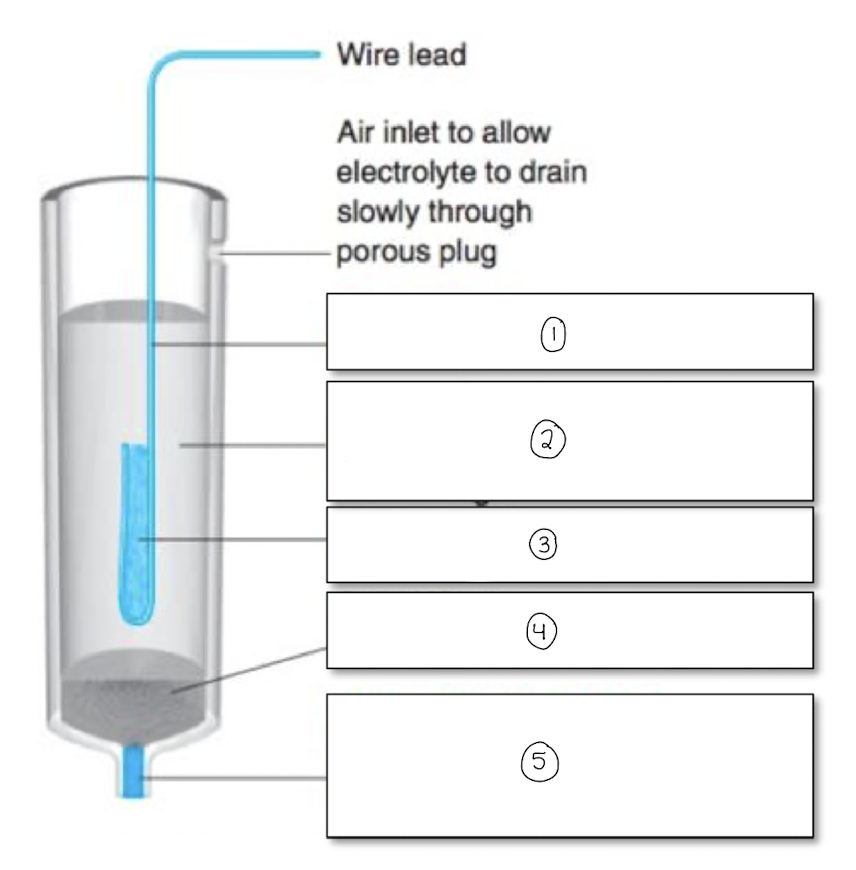
label the components of the silver/silver chloride reference electrode
Ag wire
Sat KCl and AgCl
AgCl paste
solid KCl (some AgCl)
porous plug/frit-salt bridge to external solution
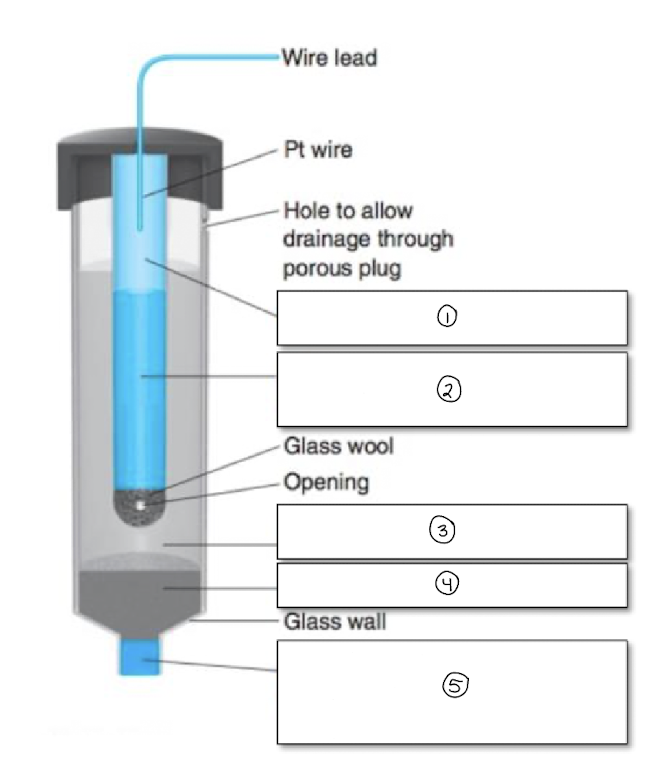
write the equation for the saturated calomel electrode
Hg2Cl2 + 2e- → 2Hg + 2Cl-
label the SCE
Hg (l)
Hg, Hg2Cl2, + KCl
sat. KCl
solid KCl
porous plug/frit-salt bridge to external solution
compare Ag | AgCl and SCE reference electrodes to SHE
these are the three common reference electrodes. SHE cannot be made into compact pen-sized devices, but it still serves as the zero point for comparing reference electrodes
when converting from SHE to Ag/AgCl or SCE, do you add or subtract?
subtract
what are the two types of indicator electrodes?
metal and ion selective
what are metal electrodes used for?
conventionally used for redox analyte detection
what are ion-selective electrodes used for?
conventionally used for ion-specific interaction (i.e., ion specifically absorbs/binds to the surface of the electrode membrane)
what are the common metal electrodes? what do you want in general for a metal electrode?
Pt is most common- very inert, easy to clean
Au is inert too
conductive carbon materials are also common
in general, you want a pristine electrode with a large surface area
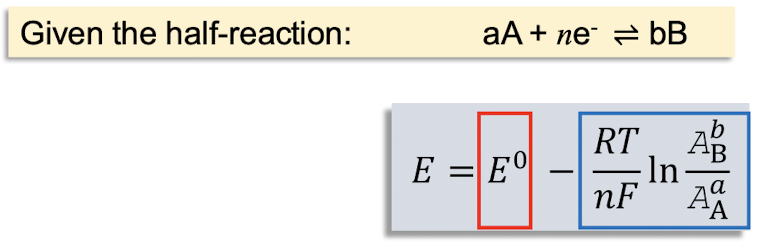
the nernst equation describes the net driving force for a redox. what do the red and blue components represent?
red: represents the driving force under standard conditions (i.e. activities = 1)
blue: shows the dependence on reagent concentrations
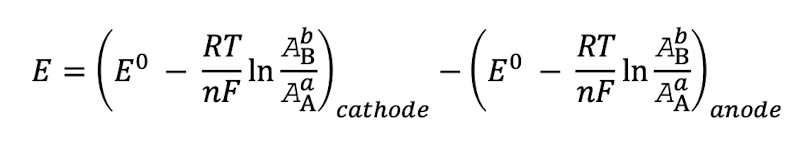
how do you find E for the entire cell?
what is a potentiometric titration
Ag+ + Cl- → AgCl
analogous to an acid-base titration curve
Ag+ is like H+ and Cl- acts like a base being titrated
effectively, the Ecell expression can be rearranged as a halide electrode
Ecell = 0.558V + 0.05916V log (Ksp/[Cl-])
in general, metal electrodes can be indicators for their given aqueous ions
though, metal-metal ion equilibrium can be difficult to establish for proper analysis
what is junction potential
a junction potential is a voltage difference occurring at the interface between dissimilar electrolyte solutions (i.e., at salt bridge termini)
what are the effects of junction potential
limits the accuracy of direct potentiometric measurements, as these contributions are difficult to measure precisely
what is a junction potential difference a result of? provide an example
a result of ion mobility difference
ex. Cl moves faster than Na, creating a defined charge interface
why is saturated KCl preferred for salt bridges?
K + Cl ions have similar ion mobility, this reduces junction potential
what is the response (mV) of a pH electrode per pH unit?
59.16 mV per 1 pH unit
what are the benefits of studying ISEs
many ions measured in blood chemistry are/can be analyzed by ISEs
chem 7 test constitute up to 70% of hospital lab tests. 4 of the 7 species are quantified by ISEs
what are glass membrane ISEs used for?
H+ and other monovalent cations
how do ISEs work?
diffusion of analyte ions from the membrane creates a slight electrical potential difference at the interface between the membrane and analyte solution
this potential difference changes variably in relation to the concentration of analyte ion in solution and can be calibrated for quantitation following a nernstian model
ISEs only measure the activity of free ions!! so complexed ions cannot be detected by ISE
can complexed ions be calculated by ISEs?
No!
what is an ionophore
the component providing selectivity in an ISE
how does a H+ ISE work?
Na+ within the glass network exchanges for H+
what is 𝛽
electromotive efficiency
how are 𝛽 and “constant” determined?
determined via calibration with at least 2 solutions of known pH. 𝛽 usually very close to 1
what is acc
describe the errors in pH measurement
the porous plug of the electrode leads to a junction potential, scales with the difference in ionic activity of standard vs unknown
junction potential drift- compensate by recalibrating every few hours
at high pH, [Na+] is high, the electrode responds to Na+ measuring a lower pH than true value. this is called alkaline error
at low pH, measured pH is often higher than true values because of complete protonation of the glass. this is called acid error
solutions must be stirred and allowed to equilibrate, generally for 30s to few minutes
electrode should always remain hydrated
keep clean, etc.
what is alkaline error
at high pH, [Na+] is high, the electrode responds to Na+ measuring a lower pH than true value.
what is acid error
at low pH, measured pH is often higher than true values because of complete protonation of the glass.
why is a selectivity coefficient necessary
ISEs are not completely exclusive, they are merely selective
ISEs have strong affinities for some ions, but interfering ions (of the same valence and comparable size) are common
what is selectivity coefficient
K potA,X = response to X/response to A
what is chromatography? what are the two types of chromatography discussed in class?
the separation of a mixture by passing through a medium in which components of differing properties travel at different rates
what is the mobile phase
the solvent moving through the column (can be liquid or gas)
what is the stationary phase?
often a viscous liquid, gel, or solid particle packed into the column
what gives rise to separation in chromatography
equilibration of solutes between mobile and stationary phases give rise to separation
what is the eluent
fluid entering the column
what is the eluate
fluid emerging from the column
in chromatography, column can be _______ or _______________
packed or open tubular
what kind of column is used for HPLC
packed column
what kind of column is used for GC
open tubular (most of the time
what is adsorption chromatography
exploits surface interactions to separate chemical mixtures
the more a solute interacts with the surface, the slower it adsorbs and desorbs from surface, the longer it is retained
ex. silica (polar molecules bind to silica gel and elute slower through column)
what is partitioning chromatography for a gas chromatography column
open tubular column, solute dissolved in liquid stationary phase
retention time is related to rate of partitioning in and out of stationary phase— GC capillary column
surface functionalized with C8
what is partitioning chromatography on a liquid chromatography column
retention time is related to rate of partitioning in and out of stationary phase - true for reversed-phase HPLC (NP column, polar mobile phase)
surface functionalized with C18
describe ion exchange chromatography
anion-exchange resin, anions are attracted to the positive resin
retention and retention tiem is related to the electrostatic interactions
describe size exclusion chromatography
retention time is related to the distance traveled. since large particles cannot penetrate the maze of pores in stationary phase, they elute first
describe affinity chromatography
one kind of molecule in a complex mixture becomes attached to a molecule that is covalently bound to stationary phase
all other molecules simply wash through
what is a chromatogram
plot of signal vs time
what is a chromatograph
instrument used to perform chromatography
requires an injector, a column and detector
what is the retention factor and what does it tell you
k = tr - tm / tm
time solute spends in the stationary phase/time solute spends in mobile phase
tells you equilibrium information
what is partition coefficient
K = cs / cm
what is band broadening-diffusion
as time increases or column lengthens, species separate but also bands naturally broaden
good chromatographic peaks are gaussian and thus, diffusion is limited
what is gas chromatography
in GC, gas analyte is transported through the column by a gaseous mobile phase, called the carrier gas
describe sample injection for GC
typical liquid injection volumes = ~1 uL
typical gas injection volumes = 10uL to 5 mL
leaving some headspace in micro-syringe prevents sample from immediately evaporating
carrier gas sweeps vaporized sample into the column
liners get changed every 20 to 100 injections
how can you control separation for GC? how do you improve resolution
you can buy columns with different properties
ex. high throughput: want a more robust column
narrower column, longer column, different stationary phase
describe retention time in GC
retention time in GC can be viewed as a thermodynamic process that depends on the enthalpy of vaporization and enthalpy of mixing with stationary phase
heat of vaporization scales linearly with boiling point
therefore, retention increases with boiling point for a given class of molecules
describe elution order in GC
a compound’s vapor pressure inside of a GC is a function of two opposing forces
the first opposing force is the vaporization of the compound. higher temperatures will increase a compound’s vapor pressure
the second opposing force is the strength of the intermolecular interactions between the compound and the stationary phase. stronger IMFs with the phase will decrease a compound’s vapor pressure
the easiest way to control a compound’s vapor pressure in a GC is through the GC oven temperature control