Unit 3
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
Synapsin
“Molecular Cowboy”
Sequesters/tethers vesicles in the reserve pool
Mechanism: binds to the vesicle and actin cytoskeleton
RIM
“Rab3 Interacting Molecule”
Scaffolding
Mechanism: binds to P/Q and N-Type voltage gated calcium channels and Rab3
RIM-BP
“Rab3 Interacting Molecule Binding Protein”
Recruits calcium channels for the active zone
Mechanism: binds to RIM and calcium channels
MUNC-13
The savior of syntaxin.
Exists in either an inactive-homodimer conformation or active conformation when bound to RIM.
The MUN domain helps kick MUNC-18 off of syntaxin, freeing the Habc domain and allowing syntaxin to become its active conformation.
Synaptotagmin
Calcium sensor
Couples with calcium to then interact with complexin and allow the trans-SNARE domain become the cis-SNARE domain.
C2 domains allow calcium binding (5 total sites)
Components of SNARE complex
Synaptobrevin (vesicular-SNARE)
Syntaxin (target-SNARE)
SNAP-25 (target-SNARE)
How is SNAP-25 associated with the plasma membrane?
Via lipid side chain interactions with the plasma membrane.
NSF
SNARE Complex disassembler; utilizes the energy of ATP hydrolysis plus adaptor proteins to take apart the complex.
Kiss and Run Model of Fusion
Phenomenon where some vesicles will not fully fuse with the membrane and instead form a transient pore which closes immediately following neurotransmitter release.
Clathrin
Associated with receptor mediated endocytosis (recycling process)
Honeycomb lattice structure
Clathrin Formation
Coat assembly and cargo selection
Bud formation
Vesicle formation
Uncoating
Triskelion
Unique structure of the honeycomb lattice
Contains one heavy chain and one light chain
Flexible
Concave
Connected to the membrane via adaptins
Endosomes
Sorting center
Allocates proteins for reloading, retrograde transport, etc.
Ba or Sr Release
Both barium and strontium CAN cause neurotransmitter release, however it is desynchronized and therefore does not produce a response.
Model Synapses
Calyx of Held (in the brainstem)
Giant squid synapse
Neuromuscular junction
Neuromuscular Junction (NMJ)
The junction between the nervous ad skeletomuscular systems.
The most commonly used synapse in experimentation.
Highly excitable
Motor End Plate
Where a nerve makes contact with muscle.
Junctional Fold
Invaginations beneath the presynaptic release sites. Increases the surface area of the postsynaptic cell which allows for a greater density of ion channels and receptors.
Basal Lamina
A structure in the synaptic cleft which holds enzymes that degrade acetylcholine (acetylcholinesterase) plus other molecules.
T-Tubule System
Network of invaginations in the muscle to help distribute action potentials everywhere.
Excitation-Contraction Coupling
Depolarization activates L-Type Ca2+ channels.
The L-Type Ca2+ channels are physically coupled to RyR (ryodine receptors, calcium channels on the smooth endoplasmic reticulum membrane).
The RyR opens
Calcium ions flow in and interact with the sarcomere.
Tubocurarine
Toxin used to keep the neuromuscular junction response below threshold which allows experimenters to measure the end plate potential.
End Plate Potential (EPP)
Dependent on time constant and how far away from the NMJ
Dependent on extracellular calcium concentration
k[CaX]^4
The theory that the binding of calcium to some unknown thing (X) to complete the complex.
Depends on multiple complexes
Calcium Conductance
Peaks during the afterhyperpolarization phase
Rises slowly
Family Tree of Calcium Channels
Voltage gated calcium channels (VGCCs) split into high voltage activated and low voltage activated channels.
Low voltage channels are T-Type channels.
High voltage activated channels split into L-Type or P/Q, N, or R-Type channels
ω Agatoxin
Binds to P/Q-Type calcium channels (Cav2.1)
ω Conotoxin
Binds to N-Type calcium channels
CD2
Toxin that blocks ALL calcium channels.
Most common calcium channel type?
P/Q-Type
Subunits of VGCCs
α1
α2
δ
γ
β
Transmembrane Subunits of VGCCs
α1
β
δ
γ
Intracellular Subunits of VGCCs
β
Extracellular Subunits of VGCCs
α2
Quantum Hypothesis
Bernard Katz; theory that postulated neurotransmitters were released in discrete packets called quanta.
This theory was confirmed by electron microscopy
Prostigamine
A toxin that blocks acetylcholinesterase, thereby increasing the available acetylcholine in the synaptic cleft.
Is used experimentally to look at the decay of minis.
How are mEPPs distributed and what does that mean?
Exponentially, that means they are random events. They require no stimulation and occur spontaneously.
What does changing the Ca/Mg Ratio Affect?
When lowers, it allows minis to be measured.
EPPs are _______ multiples of mEPPs
Integer
Residual Calcium Hypothesis
Katz proposed that calcium from an initial pulse may remain in the presynaptic terminal for some time even after the stimulus has diminished and the calcium of a second pulse will sum with the leftover calcium.
Quantal Analysis Assumptions
Enough receptors on the postsynaptic cell
Lots of release sites
Neurotransmitters are released in quanta
Independent probability of release
n
Quantal release sites
Neuromuscular junctions have lots
Central synapses have few
Pr
Release probability
Independent factor
Low when cell is at rest
High when calcium concentration increases
Evoked Response
Occurs when the calcium concentration increases.
Spontaneous Release
Occurs when the release probability is low, random event.
m
Quantal content
Quantal Content Equation
m=n*Pr
q
Quantal size
Defined as the postsynaptic response to the release of quanta
Dependent on the number of receptors and the number of neurotransmitters released (these are two separate factors but they are very difficult to differentiate)
Postsynaptic Potential Equation
PSP=q*n*Pr or m*q
Short Term Plasticity
Two stimuli delivered in rapid succession.
Different Types of Vesicle Pools and % Breakdown:
Reserve, 80-90%
Recycle, 10-15%
Readily releasable, 1%
Presynaptic Depression
Occurs when the readily releasable pool is depleted and there is not sufficient time to replenish it.
Glutamate Reuptake and Recycle Steps:
Glutamate is taken up from the synaptic cleft by astrocytes.
In astrocytes, glutamate is transformed into glutamine by glutamine synthetase.
Glutamine is transported BACK into the presynaptic cell.
In the presynaptic cell, glutamine is transformed back into glutamate by glutaminase.
Glutamate Receptor Family Tree
NMDA, AMPA, and Kinate.
AMPA Receptors Family Tree
GluR1, GluR2, GluR3, GluR4
Differ in their biophysical properties.
NMDA Receptors Family Tree
NR1, NR2, NR3
NR2 splits into NR2A, NR2B, NR2C, NR2D
Defining Features (topology) of AMPA Receptors:
Tetrameric
Can homotetramerize by incorporating different GluR subunits
Extracellular N-Terminus
Intracellular C-Terminus
Amino binding domain (NBD)
Ligand binding domain (LBD)
Transmembrane domain (TMD)
‘Line’ Structure of AMPA Receptor
N-Terminus ~ NBD ~ S1 ~ M1-3 ~ S2 ~ M4 ~ CBD ~ C-Terminus
S1 and S2 make up the ligand binding domain (LBD)
M1-3 and M4 make up the transmembrane domain (TMD)
AMPA I-V Plot
Reversal potential = 0 mV
Monovalent cation selectivity (K+ and Na+)
Slope = 8 pS
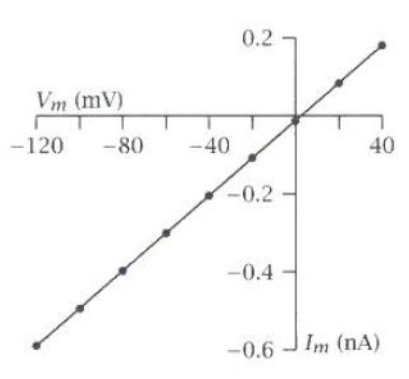
How does an AMPA receptor open?
In its closed conformation, the M3 helix occludes the pore.
When the ligand binding domain clamps down on glutamate, its linker rotates
This motion tugs on the M3 helix, causing it to open
Important structures of NMDA receptor?
Tetrameric
Two NR1 subunits and two NR2 subunits
Glycine is a co-agonist (required).
Mg can bind and block NMDA receptors and is only displaced by depolarization.
NMDA I-V Plot(s)
Reversal potential = 0 mV
Monovalent cation selectivity (K+, Na+, and Ca2+)
Slope w/ NO extracellular Mg = 20-50 pS
Slope w/ extracellular Mg = exponential
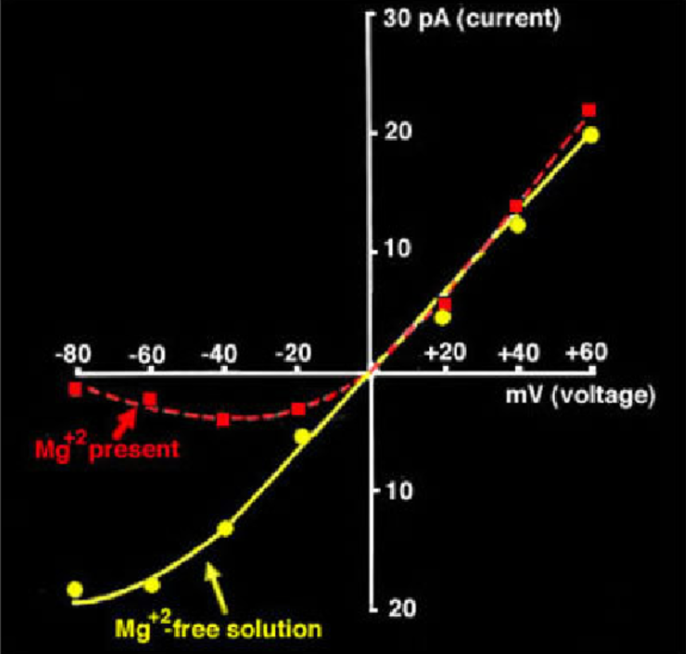
Defining difference between an EPSP and IPSP
The relationship between the reversal potential and threshold of the postsynaptic current.
Reversal potential < Threshold = IPSP
Reversal potential > Threshold = EPSP
EPSC of a Glutamatergic Cell
Defined by both the postsynaptic current of the AMPA and NMDA receptors.
AMPA is the fast component
NMDA is the slow component (voltage dependent)
Three Layers of Proteins in the Post Synaptic Density (PSD)
Neurotransmitter receptors
Primary scaffolds
Secondary scaffolds
Primary Scaffolding Protein for PSD
PSD-95
Defined by the binding of PSD-95 to the C-Terminus of NR2
GABA Synthesis
Glutamic acid is transformed into γ-amino-butyric-acid (GABA) by glutamic acid decarboxylase (GAD).
Synthesized in the presynaptic terminal
Where is GAD synthesized?
In the soma.
Two Types of GAD
67 and 65
67 is the main one
65 boosts GABA synthesis when needed
vGAT
Vesicular GABA transporter
Utilizes the v-ATPase to pump GABA into a vesicle (remember v-ATPase uses the energy from ATP hydrolysis to pump hydrogen ions into the vesicle)
GABA Recycling
GABA in the synaptic cleft is taken BACK to the presynaptic terminal by either astrocytes or GABA transporters (GAT).
It’s then converted into glutamate by GABA transaminase which produces glutamine.
What current mediates the IPSC?
Chloride
GABA Topology:
Pentameric: two α1 subunits, β2 subunits, and δ or γ subunits
Contains both a ligand binding domain (cys loop) and transmembrane domain (M1-4)
Extracellular N and C Termini
Importance of M2 in GABA
Has residues for chloride selectivity; makes up the pore interface.
Where does GABA bind?
Between the N-Terminal ligand binding domain and a subunit.
How interneurons are differentiated?
Calcium protein binding domains (CB, CR, and PV)
Shape (Basket, chandelier, …)
Synapse region
How is the PSD of GABAergic cells and glutamatergic cells different?
Glutamatergic cells postsynaptic densities are asymmetrical while GABAergic cells postsynaptic densities are symmetrical and therefore have pleomorphic synaptic vesicles.
How do benzodiazepines work?
They increase the frequency of GABA binding.
GABA I-V Plot
Reversal potential = 0 mV
Slope = 30 pS
Chloride selective