NEW Organic Chem II Exam 1
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Alkynes triple bonds consist of
2 pi bonds and 1 sigma bond
Nucleophile
“Nucleous Loving” typically have a negative charge or non-bonding e-pair available
Electrophile
“Electron Loving” typically have a positive charge or an electrong withdrawing group that puts a partial positive to an atom or have a polarizable pi bond
Mechanism arrows go from
Nucleophiles to electrophiles
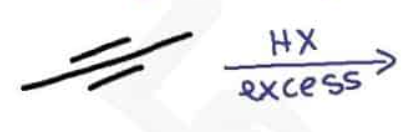
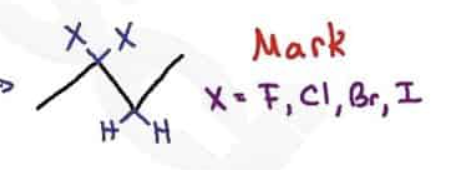

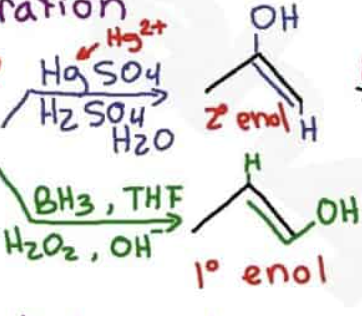
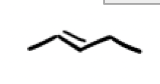
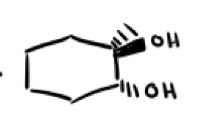
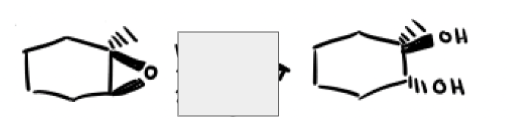
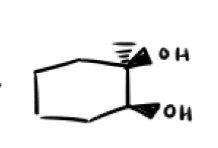
What are the reagents and intermediate
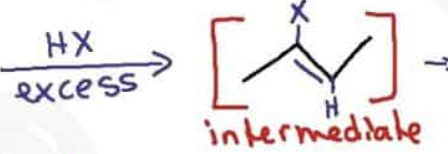
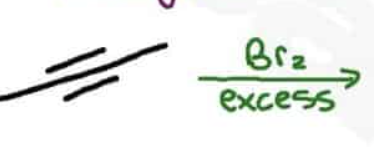
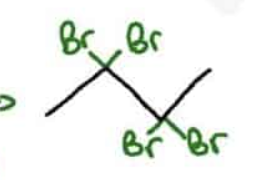

What are the reagents and intermediate
the interconversion between enol to its keto form is called
Tautomerization
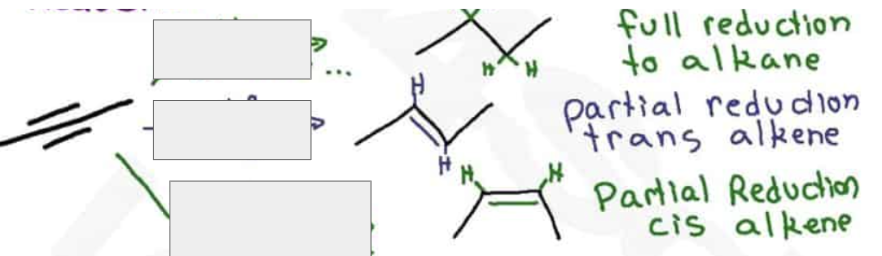
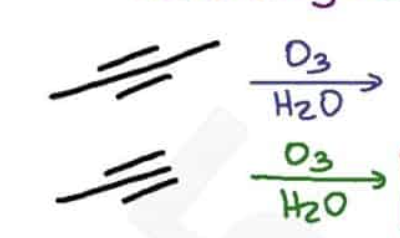
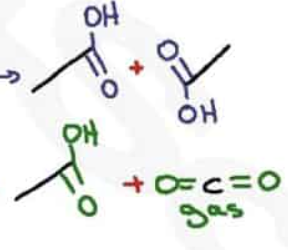
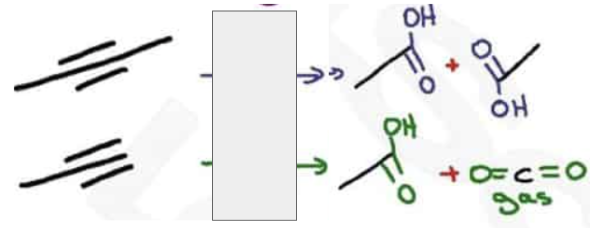
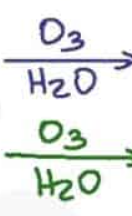
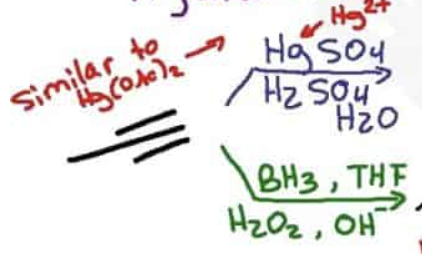
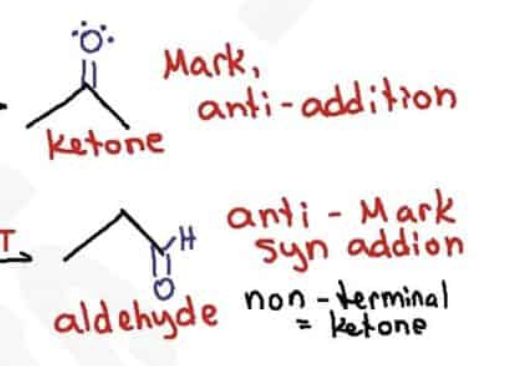
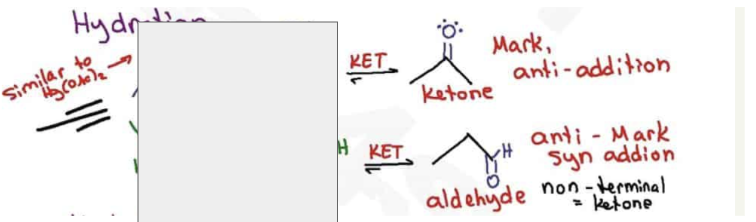
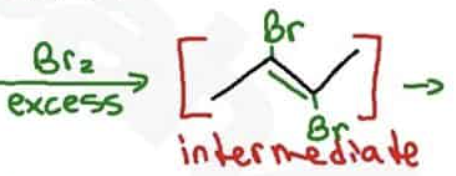
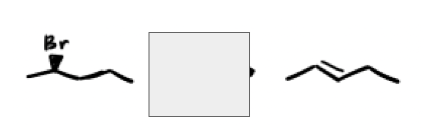
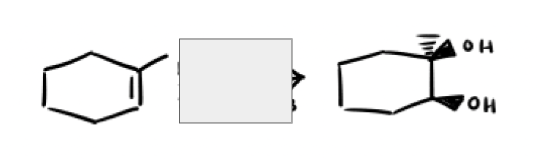
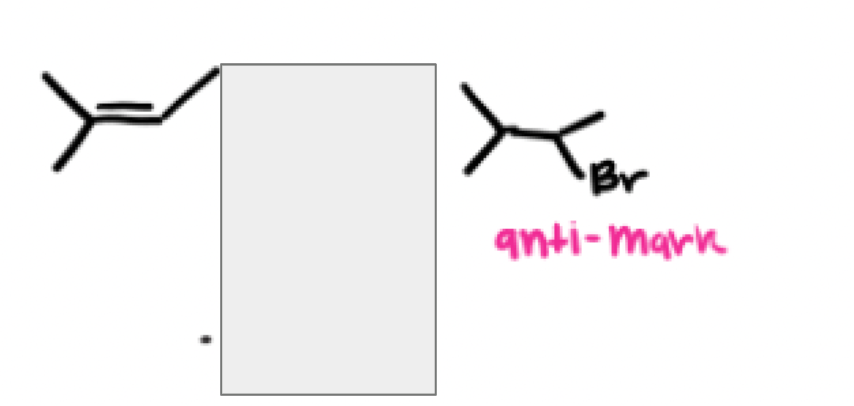
What are the reagents and intermediate
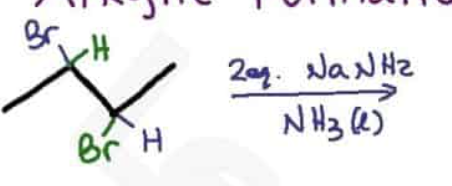


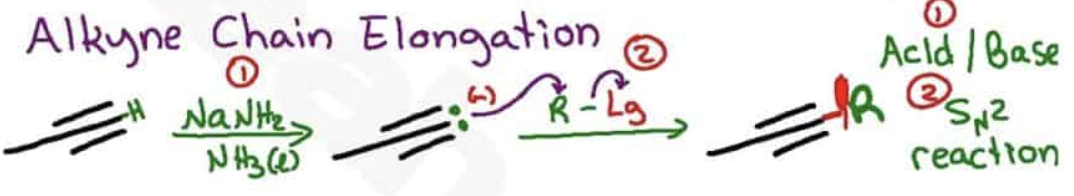
Using alkynes as nucleophiles process

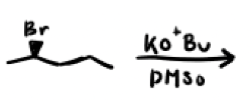

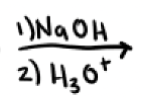
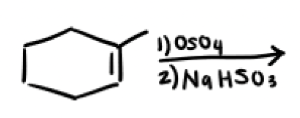
Rank halogens based on bond strength with a carbon
F > Cl > Br > I
Why is it that as bond length increases, bond strength decreases?
There is weaker overlap. The attraction between the nuclei and shared electrons decreases.
List Radical Stability
allylic ~ benzylic > tertiary > secondary > primary
In a radical substitution reaction what are the 3 steps, what happens in those three steps
INITIATION: light or heat promote homolytic cleavage of initiator molecule
PROPAGATION: radical removes H from an alkane
TERMINATION: 2 radicals meet + combine
Radical Substitution reactions using Br2 are _____
regioselective for a tertiary radical
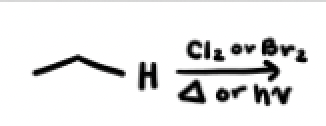
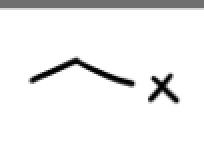
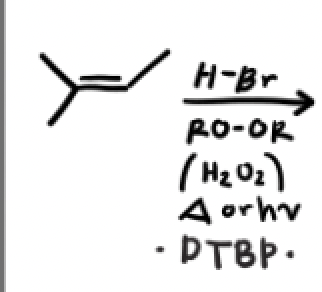
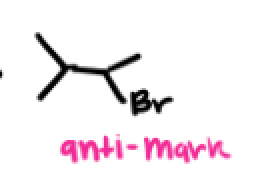
What’s another radical initiator, with what does it work with and why?
DTBP, it works only with HBr because iodine has a hard time breaking the pi bond and result in a slow endothermic reaction, additionally iodine prefers to bind to each other. We won’t even form a tertiary radical with chlorine because its endothermic (unfavorable) and is stuck in the initiation phase.
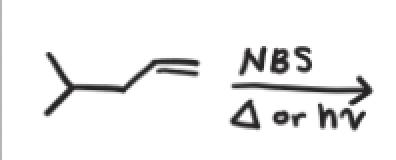
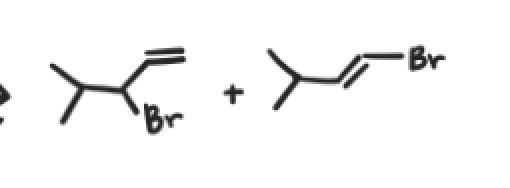

Cl2 or Br2 + heat or light
H-Br + Peroxide (ROOR, H2O2) or DTBP + heat or light
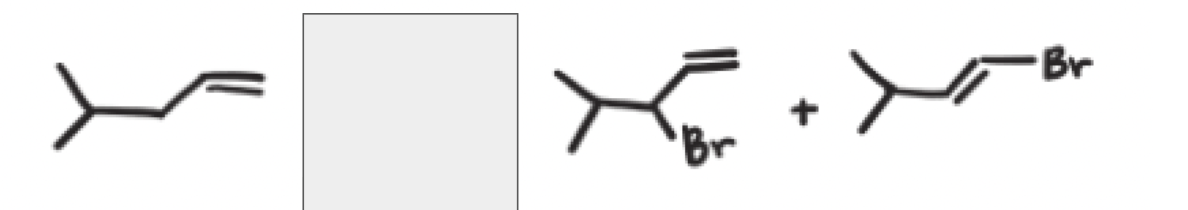
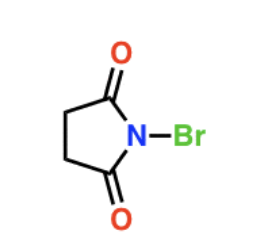
NBS + Heat or light
A reaction using NBS, the major product is?
the product with the most substituted alkene because that is the most stable product
Draw NBS
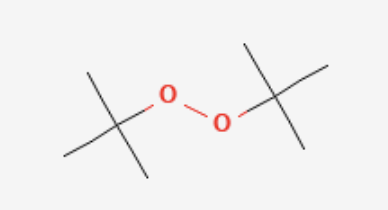
Draw DTBP
di-tertbutylperoxide
Polymer
Large molecules composed of repeating smaller units called monomers
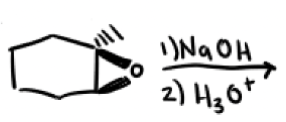
How do acetylides open epoxides?
Attack least substituted carbon
What are the rules for naming alkynes
1. Identify the parent chain
2. Identify and name substituents
3. Number so that the first substituent is given the lowest locant.
4. Arrange the substituents in alphabetical order
What is the first additional rule when naming alkynes
1. replace the suffix ane with yne
When naming a parent chain what determines which is the parent chain?
The parent chain that includes the triple bond
When numbering the parent chain of an alkyne how should the triple bond be numbered
it should be numbered so that the triple bond is on the lowest locant
How should you include the triple bond in the name
in the parent chain = hep-2-yne
or before the parent chain = 2-heptyne
What if there is an alkene and alkyne in chain?
alkene comes first in the name followed by the alkyne, takes priority
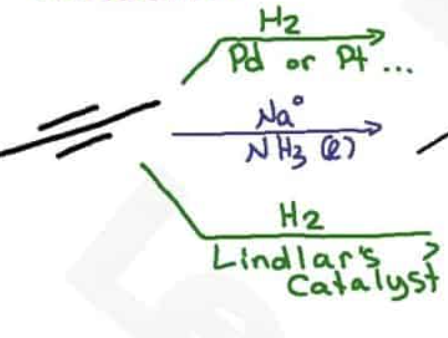
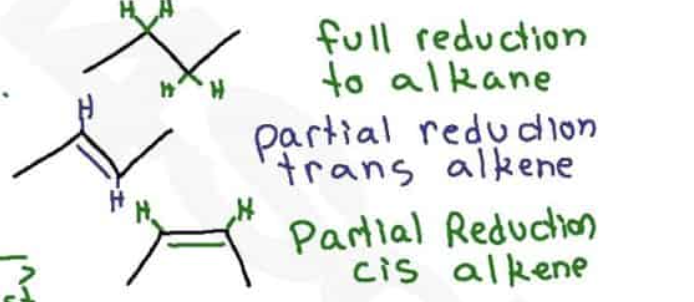
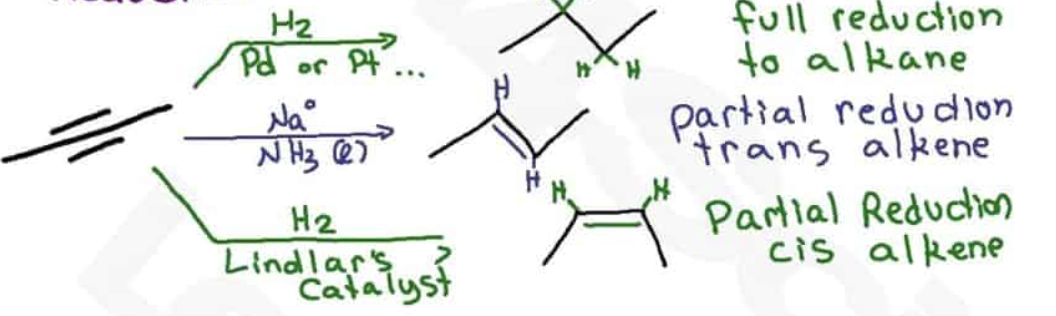
Lindlar’s catalyst is chemoselective, what does that mean?
Reagents react with one functional group, if there is an alkyne and alkene, it will go with the alkyne
In a metal reduction, what pushes the molecule into the trans position?
electron repulsion makes trans configuration more stable
steric hindrance between alkyl groups
Methyl and 1° halides undergo _____ reactions only
3° halides undergo _____ reactions only
SN2
SN1
Give an example of a polar protic solvent. What reaction do they favor?
H20, MeOH, EtOH, NH3, tBuOH, AcOH. They favor SN1 reactions
*Hydrogen bonding solvents tend to decrease the reactivity of nucleophiles.
*Quick N' Dirty Tip: Solvents with OH or NH groups are polar protic solvents
How do polar aprotic solvents favor SN2 reactions?
Because they exhibit dipole-dipole interactions but not hydrogen bonding, and as a result do not solvate anions very well. This means they can't weaken the nucleophile, and strong nucleophiles favor SN2 reactions.
In an SN2 reaction, what is the "big barrier" (the one key factor that can prevent the reaction from occurring)
In an SN2 reaction, steric hinderance can affect how the reaction occurs. Since the SN2 proceeds through a backside attack, the reaction will only proceed if the empty orbital is accessible. The more groups that are present around the vicinity of the leaving group, the slower the reaction will be. That's why the rate of reaction proceeds from 1° (fastest) > 2° > 3° (slowest)
In an SN1 reaction, the big barrier is?
Carbocation stability. Since the first step of the SN1 reaction is loss of a leaving group to give a carbocation, the rate of the reaction will be proportional to the stability of the carbocation. Carbocation stability increases with increasing substitution of the carbon (3° > 2° > 1°) as well as with resonance.
Remember that SN1 and SN2 reactions only occur for
Alkyl Halides. If the leaving group is directly attached to an alkene or alkyne, SN1 or SN2 will not occur!
What solvent is SN2 favored by? Give examples.
The SN2 reaction is favored by polar aprotic solvents - these solvents (acetone, DMSO, DMF, MeCN, HMPA, THF, acetonitrile, dichloromethane) are polar enough to dissolve the substrate and nucleophile but do not participate in hydrogen bonding with the nucleophile.
What solvent is SN1 favored by?
The SN1 reaction tends to proceed in polar protic solvents such as water, alcohols, and carboxylic acids. These also tend to be the nucleophiles for these reactions as well.
Stereochemistry of SN1 and SN2
Since the SN2 proceeds through a backside attack, if a stereocenter is present the SN2 reaction will give inversion of stereochemistry.
By contrast, if the SN1 leads to the formation of a stereocenter, there will be a mixture of retention and inversion since the nucleophile can attack from either face of the flat carbocation.
For SN2 reactions, in the transition state, the nucleophile and leaving group are
180° away from each other.
Racemix mixture, aka racemization, is
is the formation of 2 equal products, enantiomers, from a single starting material.
SN1 reactions
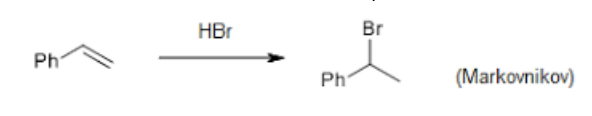
Markovnikov Rule
The halogen attaches to the carbon of the alkene with the fewest hydrogen atoms (or the most substituted)
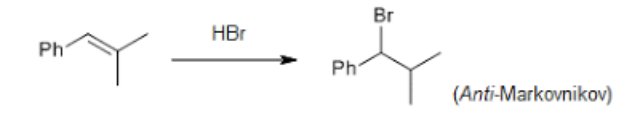
Anti-Markovnikov
Halogen attaches to the carbon of the alkene with the most hydrogen atoms (or the least substituted/)
In hydrohalogenation reactions (HBr, HCl) what is the rate reaction trend?
Methyl < Primary < Secondary < Tertiary
Rate increases as carbocation stability increases
What is an SN1 reaction?
Two-step reaction, the loss of the leaving group is first and the RDS, then nucleophilic attack on the carbocation
What is an SN2 reaction?
One-step reaction, backside nucleophilic attack
Concentration effects on SN1 and SN2 reactions:
SN1: increasing the concentration of the substrate (electrophile) increases the rate of reaction. Increasing concentration of the nucleophile has no effect
SN2: increasing concentration of either the nucleophile or electrophile (or both) will increase rate of reaction
Solvent effect for SN1 and SN2 reactions”
SN1: polar protic solvents stabilize leaving group as it leaves, which lowers the TS energy, and after it has left, preventing it from reattaching to the carbocation
SN2: polar aprotic solvents are best because aprotic solvents would also stabilize the negative charge of the nucleophile, which would slow the reaction
Leaving group effect on SN1 and SN2:
More stable leaving group increases reaction speed for both SN1 and SN2. Weak bases tend to be best leaving group.
F <<< Cl < Br < I
Rate law for SN1
rate = k[substrate]
Rate law for SN2
rate = k[substrate][nucleophile]
Rate Law E1
rate = k[substrate]
Rate law for E2
rate = k[substrate][base]
E2 reactions are a one-step mechanism, meaning
all bonds are broken and formed in a single step.
What is the relation of the base and the rate in E2 reactions?
Since the base appears in the rate equation for E2 reactions, the rate of an E2 reaction increases as the strength of the base increases, and vice versa.
What type of bases are used in E2 reactions?
E2 reactions are generally run with strong, negatively charged bases like -OH and -OR. Also DBN and DBU.
What type of solvent favors E2 reactions?
Polar aprotic solvents increase the rate of E2 reactions. DMSO, DMF, MeCN, HMPA, Acetone, THF, Dichloromethane, DMAc.
E1 reactions exhibit ____ kinetics, and is a _____ mechanism.
First-order kinetics, Two-step mechanism.
Strong bases like -OH and -OR favor ____ reactions, whereas weaker bases like H20 and ROH favor ____ reactions.
E2, E1.
SN1 and E1 reactions have exactly the same first step- formation of a carbocation. They differ in what happens to the carbocation. Explain
In an SN1 reaction, a nucleophile attacks the carbocation, forming a substitution product.
In an E1 reaction, a base removes a proton, forming a new pi bond.
Because the rate of both the E1 and E2 reactions increases as the number of R groups on the carbon with the leaving group increases, you cannot use the identity of the alkyl halide to decide which elimination mechanism occurs.
How then, does one decide whether a reaction is E1 or E2?
The strength of the base is the most important factor in determining the mechanism for elimination. Strong bases favor the E2 mechanism, while weak bases favor the E1 mechanism.
What type of solvent favors E1 reactions?
Polar protic solvents. H2O, MeOH, EtOH, NH3, tBuOH, AcOH
Carbocation rearrangements occur exclusively in ____ reactions
E1 reactions
They proceed via a distinct, free carbocation intermediate
Why don’t E2 reactions undego rearrangement?
E2 reactions do not undergo rearrangement as they are concerted, one-step mechanisms with no intermediate.