4. Intro to X-ray Spectroscopies
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
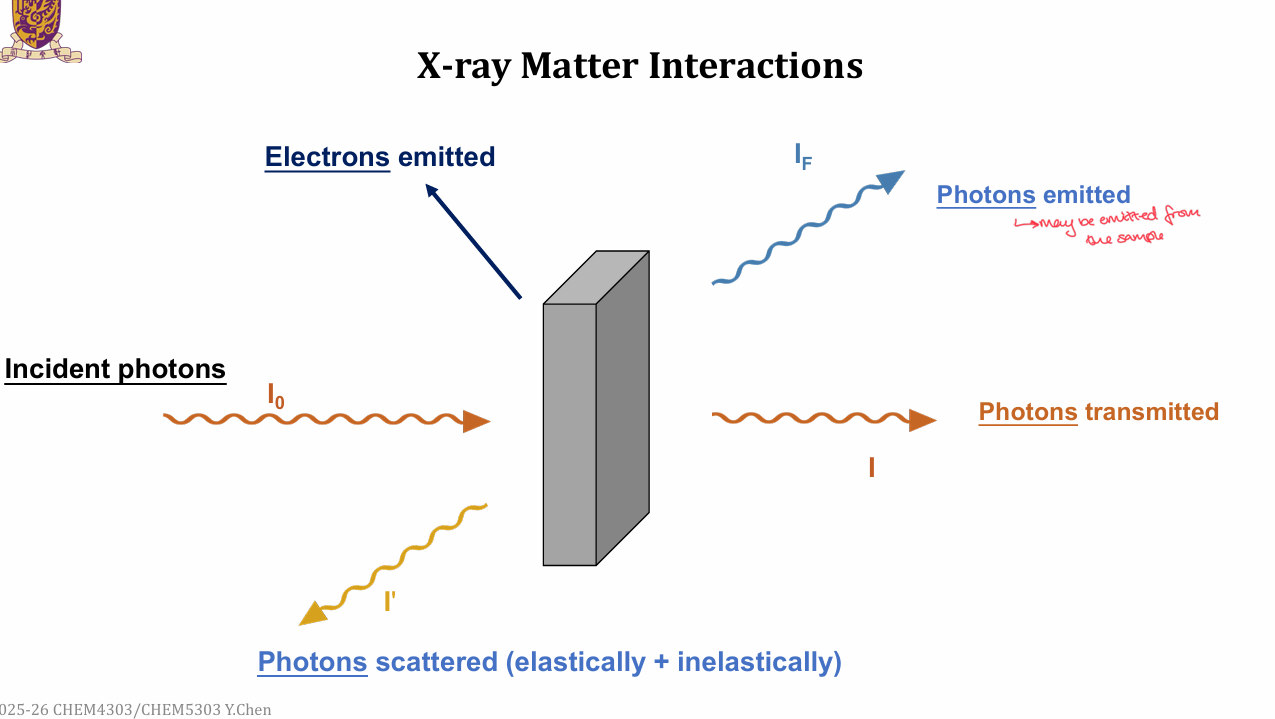
What are the basic X-ray matter interactions?
Incident photons, I0
Photons transmitted, I
Photons scattered, I’
Photons emitted, IF
What are the two fundamental photon group interactions?
elastic and inelastic scattering - photon scattered - the photons that are still there after the incident photons are transmitted
Photoelectric absorption - the photons that have disappeared
What are the elastic scattering techniques and uses?
There is no exchange of energy
can be diffraction of crystals: XRD or GIXRD
or scattering: in amorphous and liquids - WAXS and SAXS
Generally for the microscopic geometric structure
What are spectroscopy techniques and uses?
spectroscopy = photoelectric absorption or Inelastic scattering
Exchange of energy will take place
Absorption: XAS< EXAFS, XANES
Emission: XES HERFD
Photoelectron: PES, XPEEM, ARPES
Inelastic scattering: IXS, RIXS, X-Raman
Gives the electronic structure, the local structure and the dynamics, interatomic and subatomic
What x-ray interaction is measure for photoelectron spectroscopy, x-ray emission spec and x-ray absorption spec respectively?
photoelectron spec - electrons emitted from sample
x-ray emission spec - the photons emitted
x-ray absoprtion spec - photons transmitted through sample
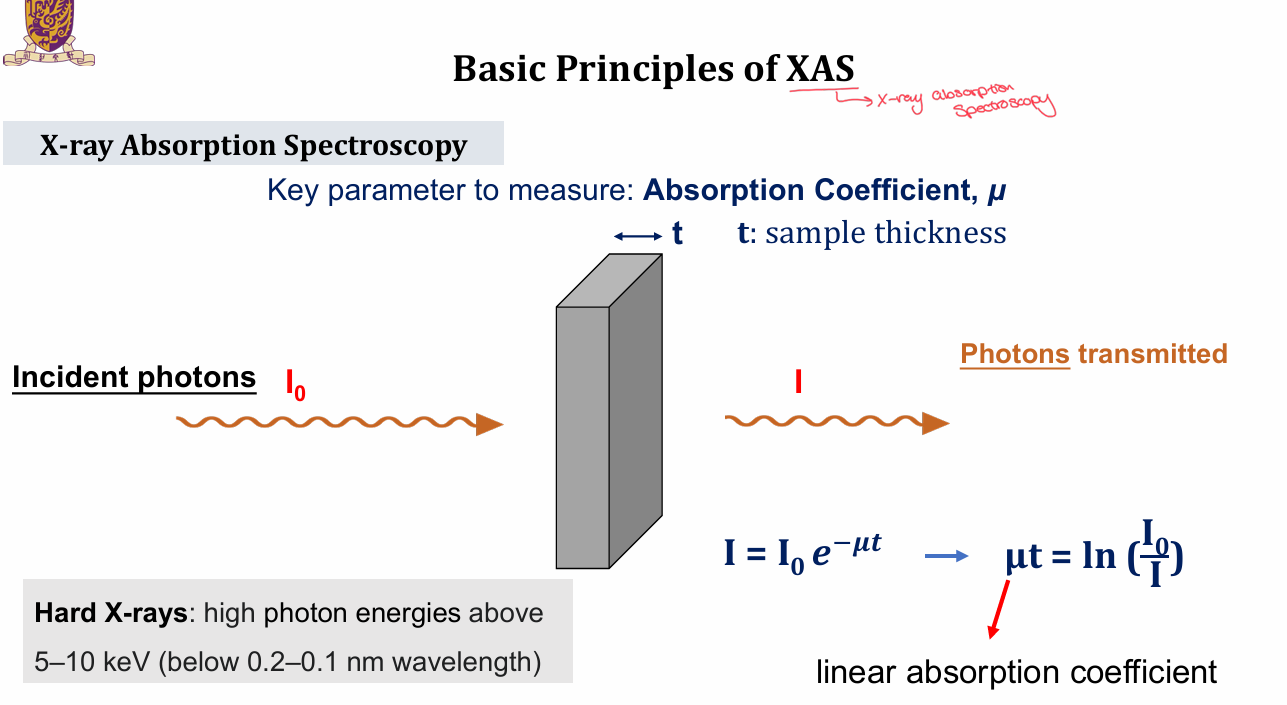
What are the basic principles of x-ray absorption spec’s (XAS) absorption coefficient?
key parameter measured is the absorptions coefficient, μ
done with hard x-rays/high photon energies of 5-10keV, below 0.2 - 0.1nm wavelength
I = I0e-μt
μt = ln (I0/I)
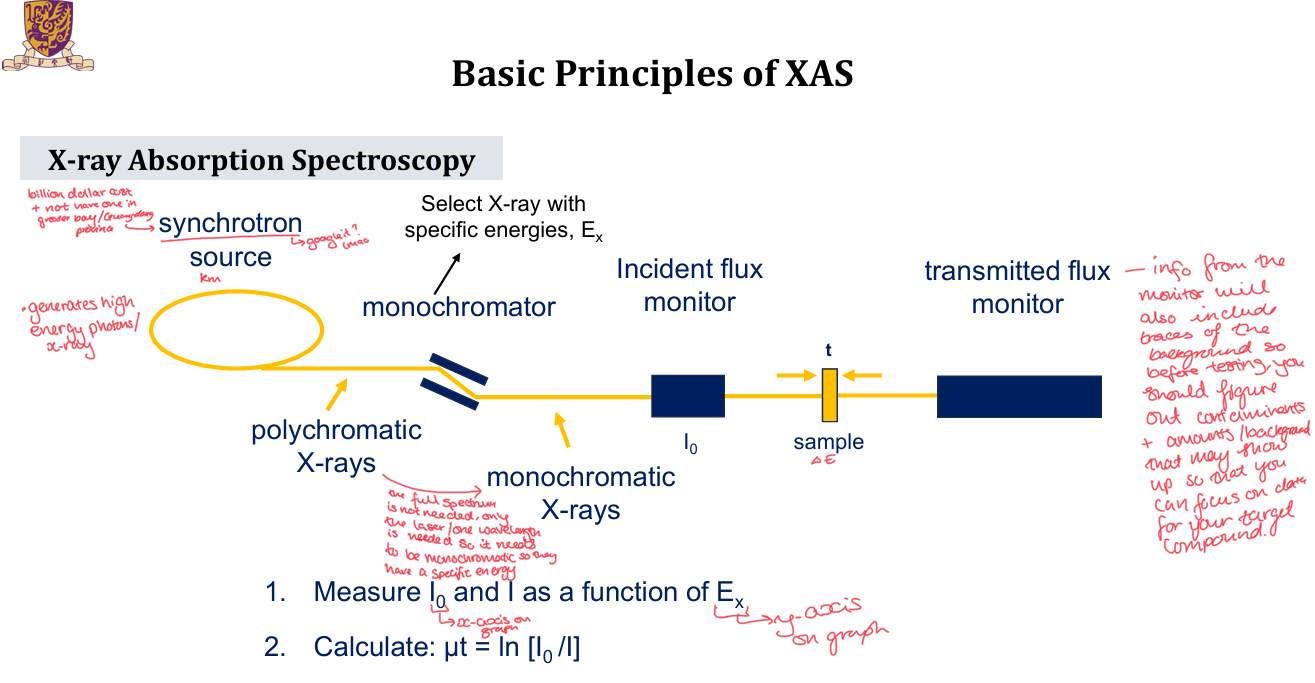
What is the setup of XAS?
synchotron source generates high energy photons/x-ray
polychromatic x-rays travel through a monochromator where x-rays of specifc energies are selected
monochromatic x-rays travel to incident flux monitor and then hit the sample
Transmitted flux monitor then sits after the sample
I0 and I are measured as a function of Ex - Ex will be the graph y-axis and I0 will be the x-axis
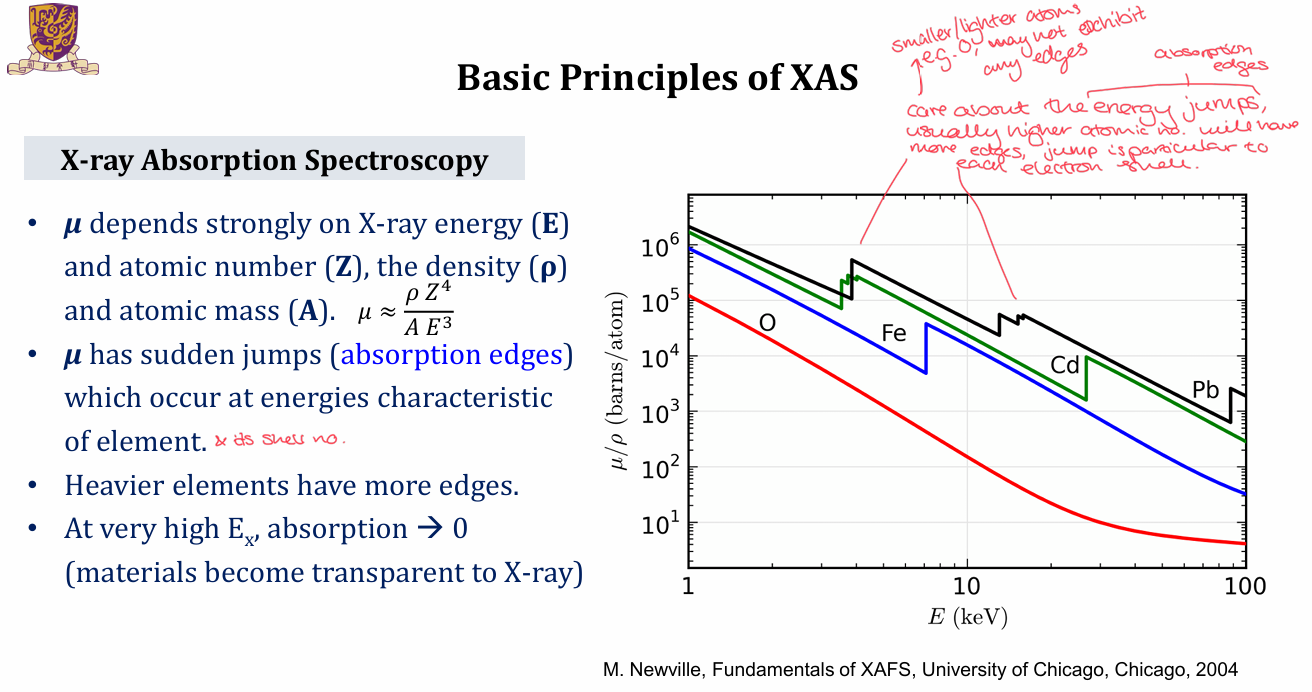
What does the absorbance graph for XAS look like and the equation to determine μt?
μ will depend on x-ray energy, E, atomic no, Z, density, ρ and atomic mass, A
μ will have sudden jumps - absorption edges - that occur at energies characteristic of the element and its shell numbers
heavier elements have more edges
At very high Ex, the absorption tends to 0 as the material will become transparent to x-ray
How is μ generated in XAS?
μ comes from the photoelectric absorptions
x-rays with energy 1-200 keV are all absorbed by matter through photoelectric effect
upon absorption of x-rays with certain energies, core-level electrons - K, L or M shells - from an atom are ejected
Ejected electrons = photoelectrons
The atom becomes excited and left with a core hole/absence of an electron
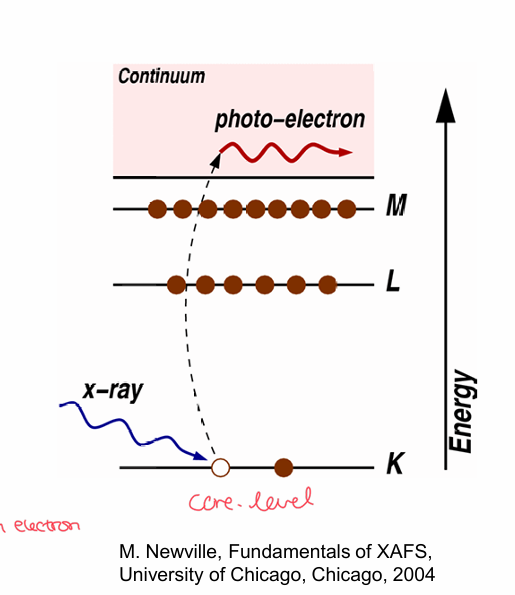
Which shell determines if the excited photoelectron is from K, L or M shells?
shell n = 1 are K-edges
2s shells ar L1 edges
2P shells are L2 and L3 edges
What happens to the excited core hole and what happens during the auger effect?
The excited core hole will return to ground state through having a higher level core electron fill the hole and emit its excess energy by fluorescent x-ray or auger electron
Auger effect is very uncommon and is a non-radiative way of coming to the ground state
energy of fluorescence or auger electron is the energy difference between the core levels

What data is measured/collected during XAS?
measure energy dependence of x-ray absorption coefficient, μ(E) at and above absorpition edge of element
It can be measured through transmission or fluorescence:
Transmission: gives and EXACT measurement of μ, absorption measured directly by measuring what is transmitted through the sample, limited by sample quality, thickness, conc etc
fluorescence: estimate of μ, radiative signals from re-filling of deep core hole is detected, μ(E) ~ If/I0
What is XAFS?
X-ray Absorption Fine Strucutre: oscillatory variation of x-ray absorption as a function of photon energy beyond absorption edge, AKA XAS
has 2 measuring regimes
XANES: x-ray absorption near-edge spec - for low energy photons
EXAFS: Extended x-ray absorption fine-structure - for high energy photoelectrons, 50 - 100 eV from edge
What do XANES and EXAFS measure or obtain data form?
XANES: covalency, local site symmetry, coordination chem, coord number, orbital hybridisation, orbital occupancy
EXAFS: local structure (bond distance, no of or type of neighbours), e.g. types of ligands, distance of neighbouring ligands and bond angle info
What is XES vs XAS?
XES is x-ray emission spec
XES is highly sensitive to local magnetic moments, spin transitions
XAS is x-ray absorption spec
XAS is sensitive to element (characteristic binding energies), chemical (shape and position of edge), spin (absorp coefficient is different for circular polarised x-rays in magnetic materials)