chem 2323 reactions
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
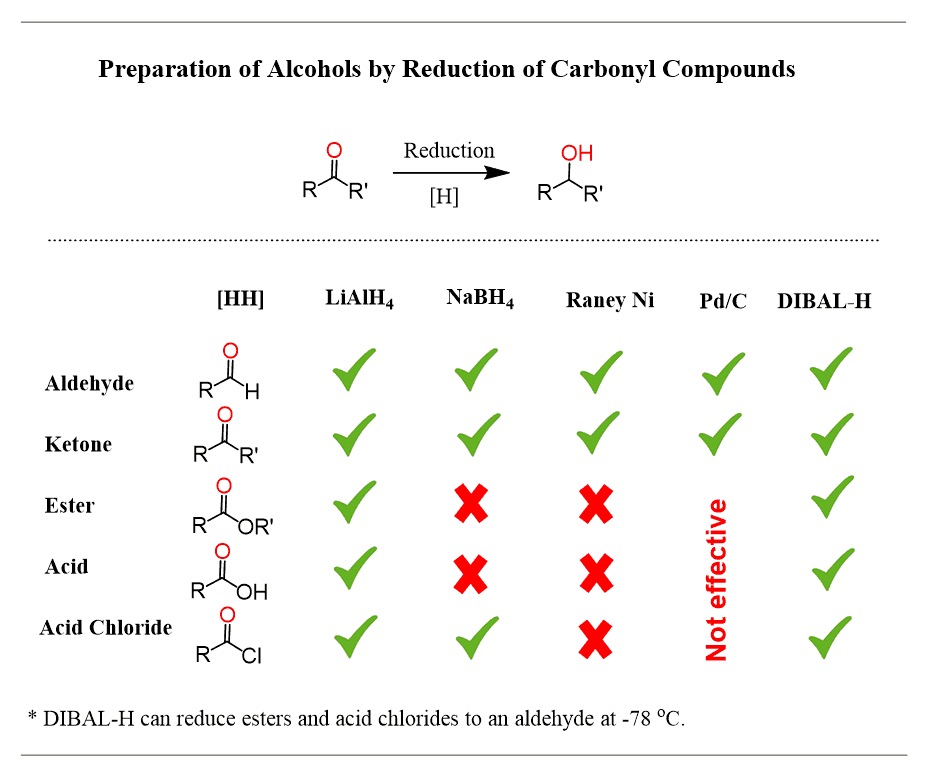
what does 1. LiAlH4 and 2. H2O do?
LiAlH4 is used in place of NaBH4 to convert functional groups like carboxylic acids, esters, ketones, aldehydes, and amides into primary or secondary alcohols and amines.
it must be used with H2O, as it needs the H+ proton
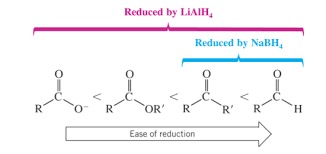
when MUST LiAlH4 be used?
when there are multiple oxygens present in the structure (still forms primary alcohol).
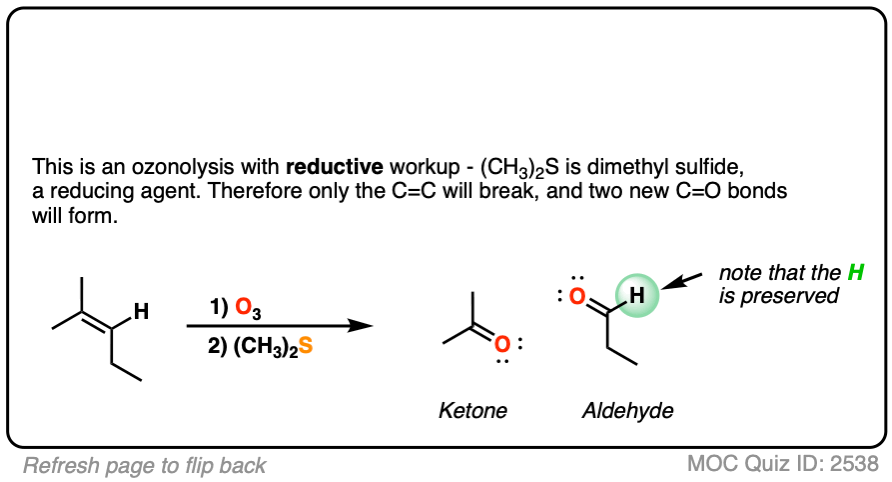
what does o3 do to alkene and alkyne structures?
it cleaves the double or triple bond, adding =O to each carbon involved in the bond. produces aldehydes or ketones
what does cl2/ccl4 do to an alkene?
anti-markovnikov addition of cl. Cl is added to each carbon in the double bond, but in trans directions (one wedged, one dashed)
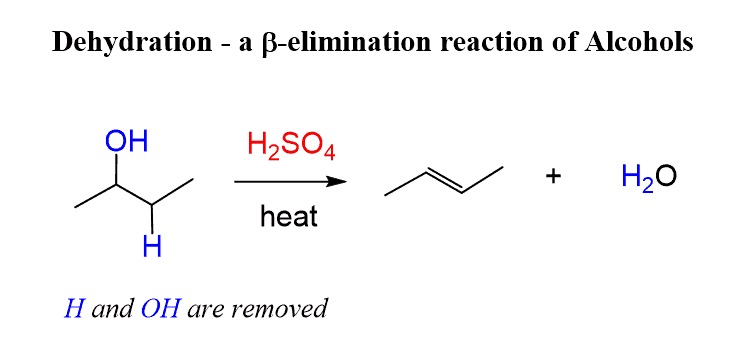
what can you use for the formation of a double bond/elimination of OH via zaitsev methods?
h2so4. it does zaitsev elimination to form the more substituted alkene
what does KMnO4 cold, OR OsO4 with peroxide, OR MCPBA/H3O+ do to ALKENES?
trans-addition of OH to carbons involved in the DOUBLE BOND
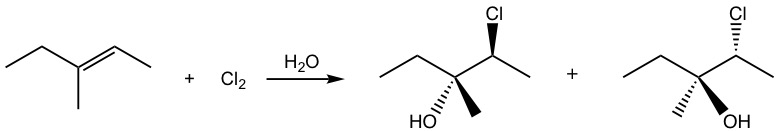
what does Cl2/H2O do to a double bond in an alkene?
it adds OH to the more substituted carbon in the double bond and Cl to the less substituted one
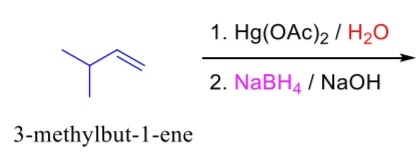
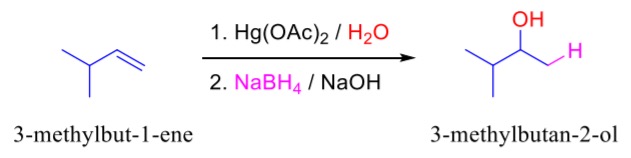
complete the following reaction.
Hg(OAc)2/H2O is responsible for the markovnikov addition of a =O (=O to the more substituted carbon in the double bond)
NaBH4 is responsible for converting =O to -OH.
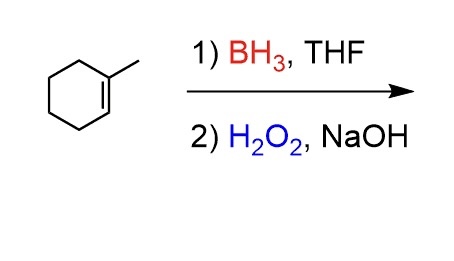
complete the following reaction.
BH3 * THF and H2O2 are responsible for hydroboration-oxidation, a two-step reaction that converts an alkene into an alcohol.
Anti-markovnikov. Adds -OH group to the less substituted carbon and an H to the more substituted carbon in a cis manner
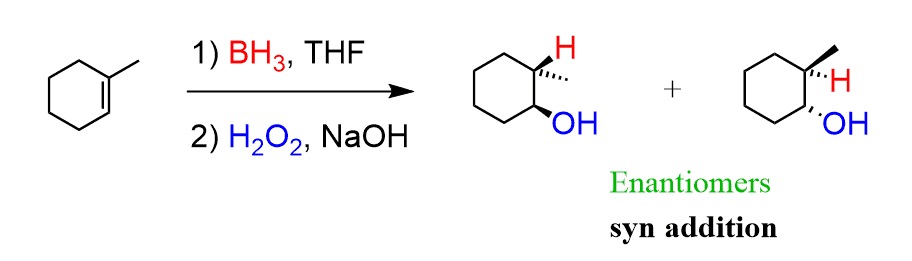
complete the given reaction
mcPBA breaks the double bond and forms a single bond. both the carbons are then bonded to the O.
H3O+ is then executed in anti-markovnikov fashion and adds trans OH to each carbon (wedged OH on one, dashed OH on the other)
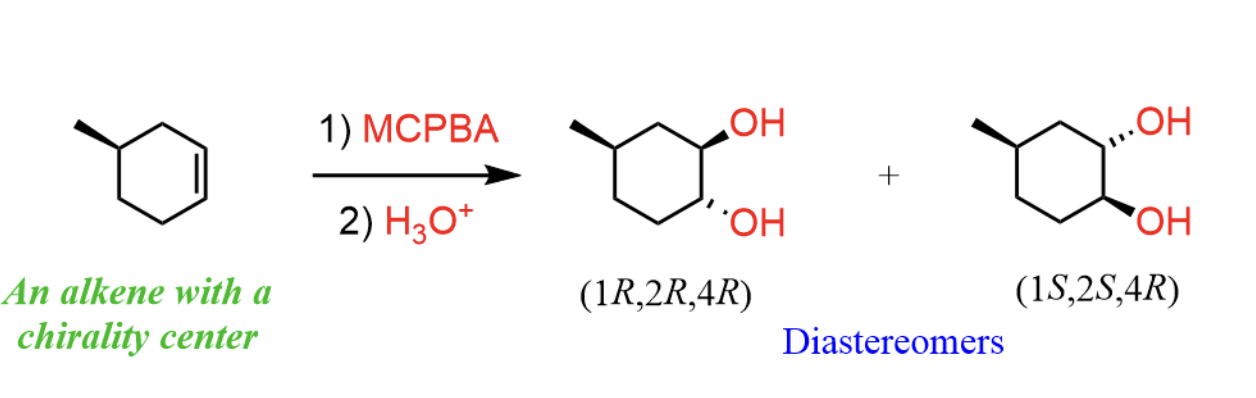
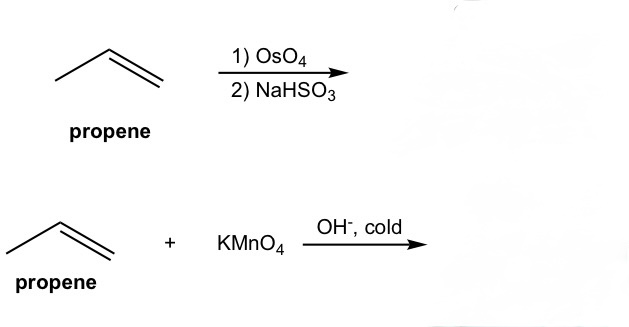
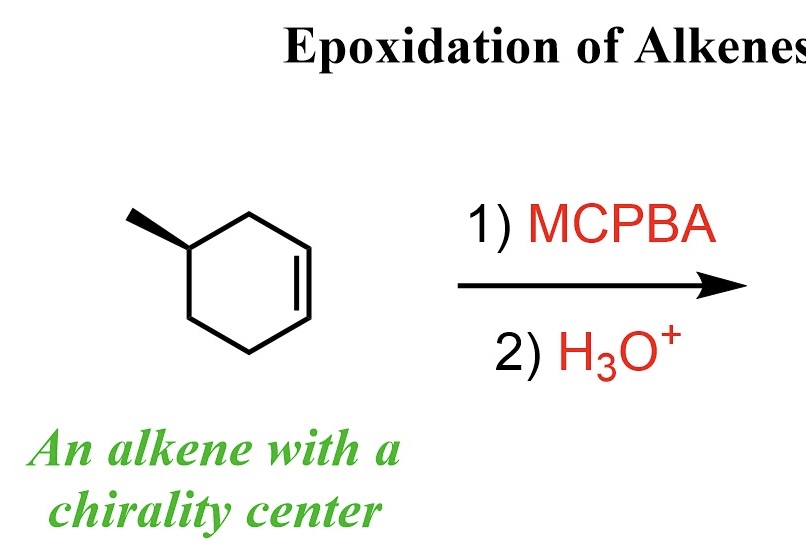
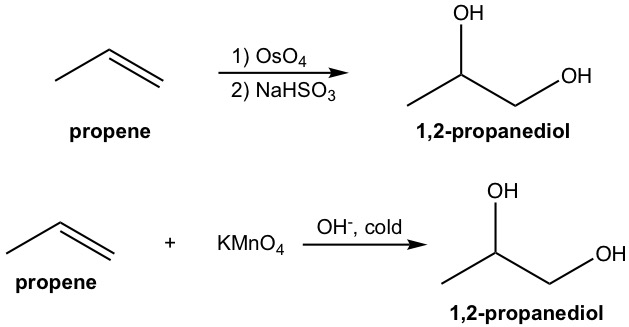
complete the reaction. note that it may be helpful to think of the first one as OsO4 and peroxide (the one learned in class)
OsO4 and peroxide and KMnO4 cold breaks the double bond in alkene and adds an -OH to each corresponding carbon in a cis manner
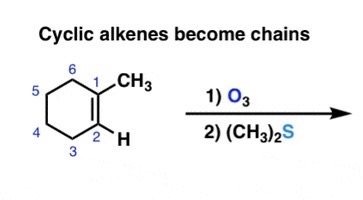
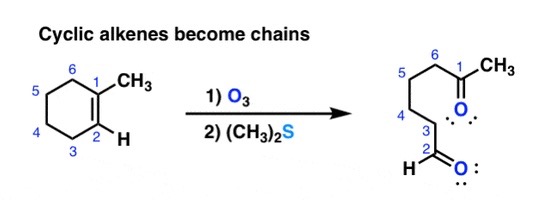
complete the following reaction.
o3 and (ch3)2S does ozonolysis. it completely cleaves the bond between two carbons and bonds each carbon to an =O.
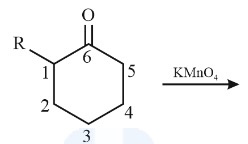
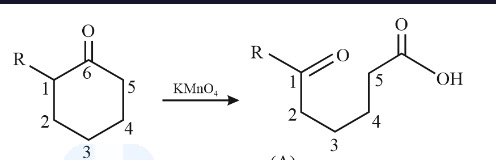
complete the following reaction
kmno4 cleaves the double bond completely, severing the bond between the carbons. it adds =O to the MOST substituted carbon and OOH to the least substituted one
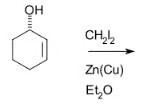
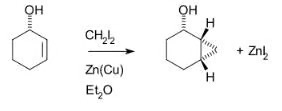
complete the following reaction
CH2I2 with ZnCu or CH2N2 reduces the double bond to a single bond, and then makes a bond form each of the carbons that were originally involved in the double bond to a CH2.
It creates a cyclopropyl like structure with the 3 carbons
what does CHX3/OH- do? assume X represents Cl or Br
it breaks the double bond and reduces it to a single bond between he two carbons. for the carbons previously engaged in the double bond, a halogen is added to each, as dashes or a dash to wedged halogen
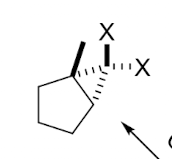
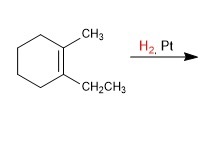
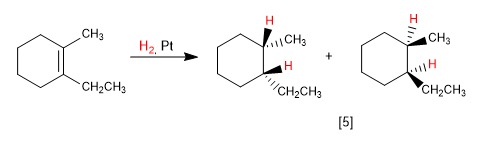
complete the following reaction.
H2/Pt or Pd performs catalytic hydrogenation. It reduces DOUBLE AND TRIPLE bonds to single bonds (alkane bonds) by adding hydrogens across the bonds.
it does cis/syn addition, adding the hydrogens to the same side of the molecule