2003: Fatty acid degradation/beta-oxidation and synthesis, palmitate synthesis, Triglyceride & cholesterol synthesis
1/97
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
98 Terms
What are the 4 primary physiological functions of fatty acids within the body?
Fuel Molecules: Fatty acids serve as a significant energy source.
They are mobilized from storage in fat tissue.
They are oxidized to meet the energy needs of a cell or organism.
During rest or moderate exercise, such as walking, fatty acids are our primary source of energy
Building Blocks: Fatty acids are building blocks of phospholipids and glycolipids.
Protein modification: Many proteins are modified by the covalent attachment of fatty acids. The attached fatty acids target proteins to membrane locations.
Hormones and Intracellular Messengers: Fatty acid derivatives serve as hormones and intracellular messengers
Where is the major site of triacylglycerol synthesis, storage, and mobilization into fuel molecules? What are the 2 notable deposits?
In white adipose tissue. Within the cytoplasm of fuel-rich adipocytes, commonly known as fat cells.
Located throughout the body, with subcutaneous (below the skin) and visceral (around the internal organs) deposits being most prominent.
How are triaclyglycerol’s stored and regulated within the adipose?
Triacylglycerols accumulate to form large structures called lipid droplets within adipocytes.
These droplets can occupy a significant portion of the cell's volume.
These droplets are surrounded by a monolayer of phospholipids and numerous proteins required for triacylglycerol metabolism.
These droplets are dynamic organelles essential for the regulation of lipid metabolism.
What are the 3-stage processing pathway that the peripheral tissues in the body access to utilizes the energy stored in lipids within adipose tissue?
Mobilization: In this process, triacylglycerols are degraded to fatty acids and glycerol.
Activation and transport: The fatty acids must be activated and transported into mitochondria for degradation.
Breakdown into acetyl CoA: The fatty acids are broken down in a step-by-step fashion into acetyl CoA, which is then processed in the citric acid cycle.
Acetyl CoA vs Acyl CoA
Acyl-CoA is a broad category of Coenzyme A derivatives featuring a fatty acid chain of any length (R-CO-SCoA), primarily involved in lipid metabolism and fatty acid oxidation.
Acetyl-CoA is a specific, small member of this family with a two-carbon chain (CH3-CO-SCoA), acting as the central hub connecting carbohydrate, fat, and protein metabolism to the citric acid cycle.
Stage 1 of fatty acid degradation - Mobilization: Lipolysis and how its triggered
Lipolysis: Before triacylglycerols can be used as fuel, they must be hydrolyzed (broken down with water) to yield isolated fatty acids and glycerol.
Epinephrine and glucagon, key hormones in energy regulation, trigger lipolysis. They also likely regulate how triacylglycerol stores are utilized in various tissues.
Stage 1 of fatty acid degradation - Mobilization: How are the products of lipolysis (fatty acids vs glycerol) released into the bloodstream?
Fatty acids are not water-soluble. To travel through the aqueous blood plasma, they bind to a blood protein called albumin, which delivers them to tissues requiring fuel and proceeds to fatty acid oxidation in the mitochondria.
The glycerol molecule, being water-soluble, is absorbed by the liver.
In the liver, glycerol can be converted into intermediates for glycolysis (glucose breakdown) or gluconeogenesis (glucose synthesis), depending on the body's energy needs.
Stage 2 of fatty acid degradation - Activation (1 of 4): How does fatty acids activated to acyl CoA? (location, enzyme, requirements, etc)
Fatty acids first separate from albumin in the bloodstream and enter target cell
Fatty-acid-transport proteins facilitate this process.
Inside the cell, they associate with fatty-acid-binding proteins for intracellular shuttling.
For degradation, fatty acids must be activated by reacting with coenzyme A (CoA) to form acyl CoA.
This activation takes place on the outer mitochondrial membrane.
It is catalyzed by the enzyme acyl CoA synthetase.
This reaction requires energy supplied by ATP.
Stage 2 of fatty acid degradation - Activation (2 of 4): Describe the 2 steps regarding how enzyme acyl CoA synthetase activates fatty acids into acyl CoA?
The fatty acid reacts with ATP to produce an acyl adenylate intermediate (and a PPi).
In the next step, the sulfhydryl group of coenzyme A attacks the acyl adenylate, resulting in the formation of acyl CoA and AMP.
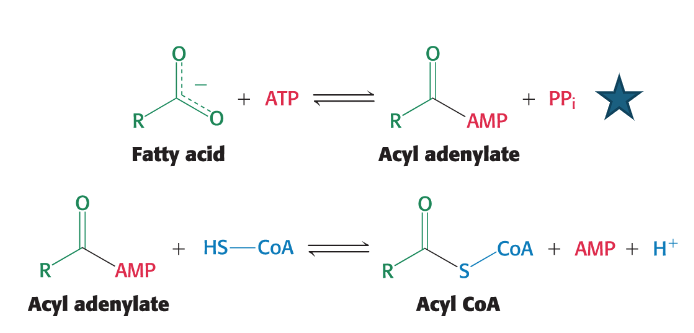
Stage 2 of fatty acid degradation - Activation (3 of 4): Describe the reversibility of the previous partial reactions and how they are driven forward
These two partial reactions are individually reversible under standard conditions.
However, the overall reaction is driven forward by the rapid and irreversible hydrolysis of pyrophosphate (PPi) by the enzyme pyrophosphatase into two inorganic phosphate molecules (2Pi). This hydrolysis makes the activation step energetically favorable.
Stage 2 of fatty acid degradation - Activation (4 of 4): What is the complete reaction for fatty acid activation?
RCOO⁻ + CoA + ATP → RCO-CoA + AMP + 2Pᵢ
Stage 2 of fatty acid degradation - Transport (1 of 2): How can the activated fatty acids (acyl CoA) cross the outer mitochondrial membrane and begin to cross the inner mitochondrial membrane?
Activated fatty acids can readily cross the outer mitochondrial membrane. However, activated long-chain fatty acids must be conjugated/attached to carnitine in order to cross the inner mitochondrial membrane.
The acyl group is transferred from the sulfur atom of CoA to the hydroxyl group of carnitine to form acyl carnitine.
This reaction is catalyzed by carnitine acyltransferase I (CAT I).
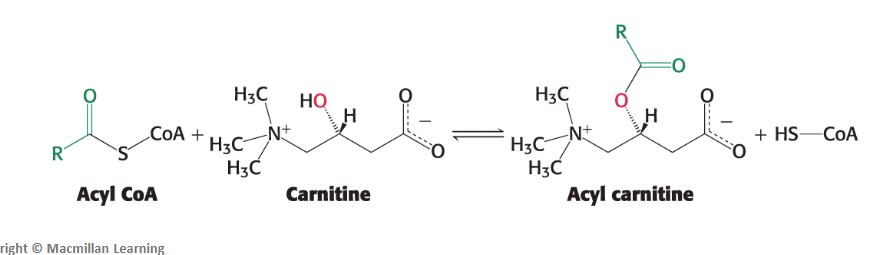
Stage 2 of fatty acid degradation - Transport (2 of 2): How is the long-chain fatty acids finally transported across the mitochondrial membrane after being converted to acyl carnitine? How can the cycle continue?
The acyl carnitine molecule is then shuttled across the inner mitochondrial membrane into the mitochondrial matrix by a transport protein called a translocase.
Once inside the matrix, the acyl group is transferred back to a molecule of CoA (which is abundant in the matrix) by the enzyme carnitine acyltransferase II (CAT II). This regenerates acyl CoA inside the mitochondrial matrix, ready for degradation.
Finally, the translocase returns carnitine to the cytoplasmic side of the inner membrane in exchange for another incoming acyl carnitine, allowing the cycle to continue.
CLINICAL INSIGHT: How do Pathological Conditions Result If Fatty
Acids Cannot Enter Mitochondria? What symptoms does this lead to?
Deficiencies in carnitine, carnitine transferase enzymes (CAT I or CAT II), or the translocase protein can lead to severe health issues.
Symptoms of carnitine deficiency vary, ranging from mild muscle cramping to severe weakness and, in extreme cases, even death.
An inability to synthesize carnitine might be linked to the development of autism in males.
Tissues heavily reliant on fatty acids for energy, such as muscle, kidney, and heart, are primarily affected.
Muscle weakness during prolonged exercise is a classic symptom of carnitine acyltransferase deficiency because muscles depend on fatty acids for sustained energy.
These conditions highlight that proper metabolite flow between cellular compartments is critical for health.
CLINICAL INSIGHT: How is carnitine normally obtained in our bodies?
Carnitine can be synthesized by humans and is also obtained from the diet, being particularly rich in red meat.
Despite its natural presence, carnitine is popular as a dietary supplement, marketed for increasing endurance, enhancing brain function, and promoting weight loss.
However, its effectiveness as a supplement in individuals without a diagnosed carnitine deficiency is not yet definitively established.
Stage 3 of fatty acid degradation - Breakdown (1 of 5): what are the main purposes of fatty acid degradation. What is the oxidation known as?
To oxidize the fatty acid into acetyl CoA (in which a saturated acyl CoA is degraded by a recurring sequence of four reactions)
To capture the high-energy electrons released during oxidation in the form of FADH2 and NADH, which will then power oxidative phosphorylation for ATP synthesis.
Because oxidation occurs at the beta (β)-carbon atom (the third carbon from the carboxyl end), this pathway is known as β-oxidation. The alpha (α) carbon is carbon 2, and the beta (β) carbon is carbon 3.
Stage 3 of fatty acid degradation - Breakdown (2 of 5): What is the first round of oxidation in fatty acid degradation?
The first reaction in each round of β-oxidation is the oxidation of acyl CoA by an acyl CoA dehydrogenase.
This reaction produces an enoyl CoA molecule with a trans double bond between carbon 2 and carbon 3 (referred to as Δ2 - position of 1st carbon in a double bond).
This enzyme requires flavin adenine dinucleotide (FAD) as an electron acceptor.
Acyl CoA+E-FAD→trans-Δ2-Enoyl CoA+E-FADH2
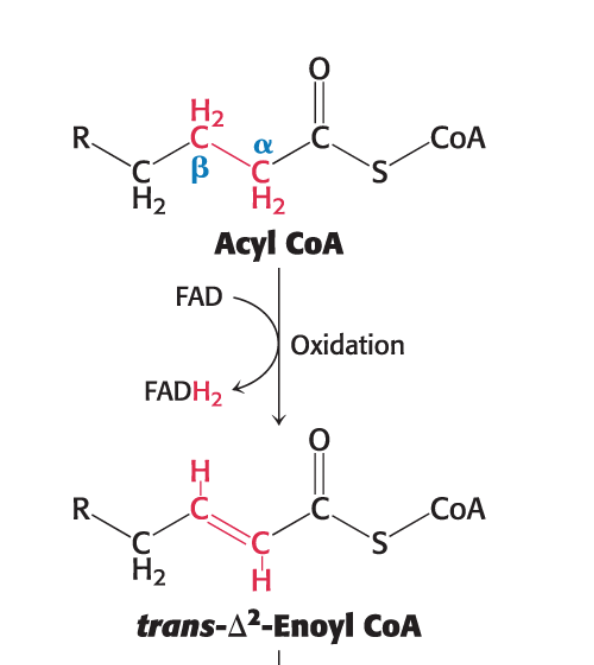
Stage 3 of fatty acid degradation - Breakdown (3 of 5): What is the second round of oxidation in fatty acid degradation? What’s special about this reaction?
The second reaction is the hydration of the newly formed double bond between carbon 2 and carbon 3.
This reaction is catalyzed by enoyl CoA hydratase.
This hydration is stereospecific; only the L isomer of 3-hydroxyacyl CoA is formed.
trans-Δ2-Enoyl CoA+H2O→L-3-hydroxyacyl CoA
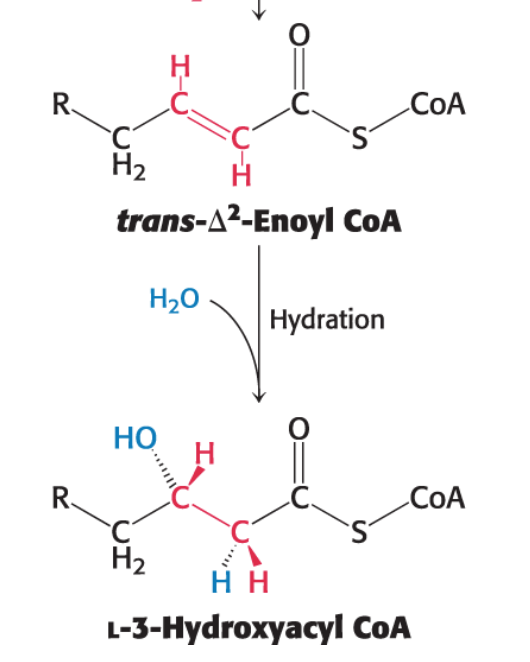
Stage 3 of fatty acid degradation - Breakdown (4 of 5): What is the third round of oxidation in fatty acid degradation?
A second oxidation reaction
Requires nicotinamide adenine dinucleotide (NAD+) as an electron acceptor.
Converts the hydroxyl group at carbon 3 into a keto group, generating NADH and 3-ketoacyl CoA.
Catalyzed by L-3-hydroxyacyl CoA dehydrogenase
L-3-Hydroxyacyl CoA+NAD+→3-Ketoacyl CoA+NADH+H+
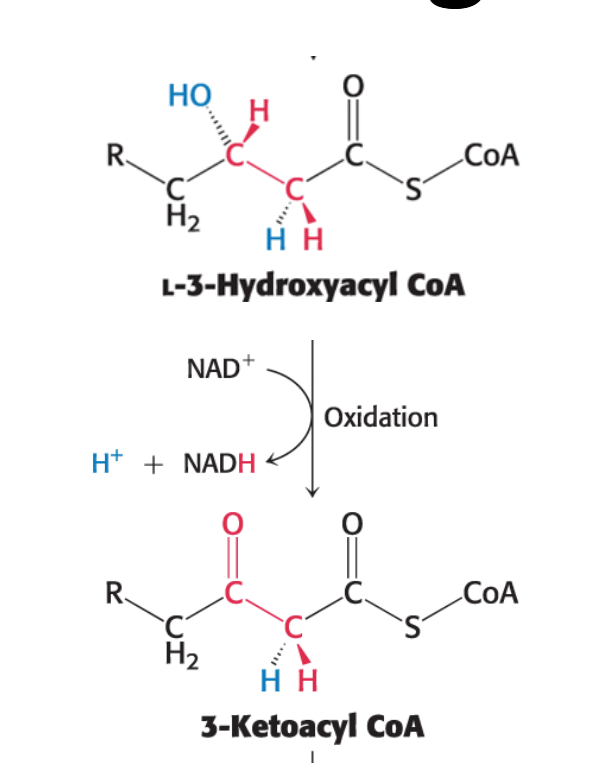
Stage 3 of fatty acid degradation - Breakdown (5 of 5): What is the forth round of oxidation in fatty acid degradation?
Thiolysis: The final step is the cleavage of 3- ketoacyl CoA by the thiol group of a second molecule of coenzyme A.
This yields acetyl CoA and an acyl CoA shortened by two carbon atoms.
This thiolytic cleavage is catalyzed by β-ketothiolase
3-Ketoacyl CoA+CoA→Acetyl CoA+Acyl CoA (n-2 carbon atoms)
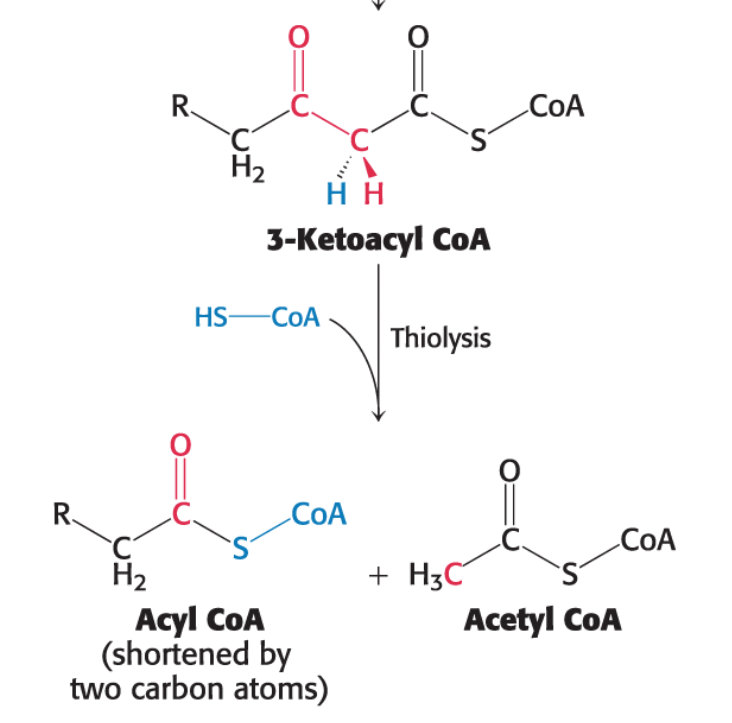
How many acetyl CoA will result from palmitate, a 16 carbon saturated fatty acid?
Acetyl CoA is a 2 carbon molecule.
For a 16-carbon saturated fatty acid like palmitate, it will undergo 7 rounds of β-oxidation, yielding 8 molecules of acetyl CoA .
How many ATP results from the complete oxidation of palmitate?
One round of β-oxidation: Produces 1 FADH2, 1 NADH, and 1 acetyl CoA (in addition to the shortened acyl CoA).
For palmitoyl CoA (C16), there are 7 rounds of β-oxidation, which yields:
7 FADH2
7 NADH
8 acetyl CoA
ATP generated:
Each FADH2 generates approximately 1.5 molecules of ATP through oxidative phosphorylation. So, 7×1.5=10.57×1.5=10.5 ATP.
Each NADH generates approximately 2.5 molecules of ATP through oxidative phosphorylation. So, 7×2.5=17.57×2.5=17.5 ATP.
Each acetyl CoA entering the citric acid cycle (TCA cycle) yields 10 molecules of ATP. So, 8×10=80 ATP.
Total ATP initially calculated: 10.5+17.5+80=108 ATP
However, 2 ATP are required for activation: so 108 - 2 = 106 ATP
What are ketone bodies? What’s the point of their synthesis?
An Alternative Fuel Source from Fats for some tissues
Under certain physiological conditions, such as prolonged fasting or uncontrolled diabetes, ketone bodies are synthesized from a buildup of acetyl CoA synthesized during fatty acid degradation in liver mitochondria, and secreted into the blood for use as fuel by some tissues such as heart muscles
Ketone bodies are carried from the liver mitochondria into the blood by transport proteins and are conveyed to other tissues such as heart and kidney
They allow acetyl-CoA (from fatty acids) to travel from the liver to other tissues.
In those tissues, they are converted back into acetyl-CoA to make ATP.
What are the 3 primary ketone bodies and how are they synthesized?
The three main ketone bodies are:
Acetoacetate
D-3-β-hydroxybutyrate
Acetone
The synthesis process involves:
The condensation of two acetyl CoA molecules to form acetoacetyl CoA, which then further reacts with 3-Hydroxy-3-methyl-glutaryl CoA to form acetoacetate.
D-3-β-hydroxybutyrate is formed when acetoacetate is reduced.
Acetone is produced by the spontaneous decarboxylation (removal of a carboxyl group) of acetoacetate.
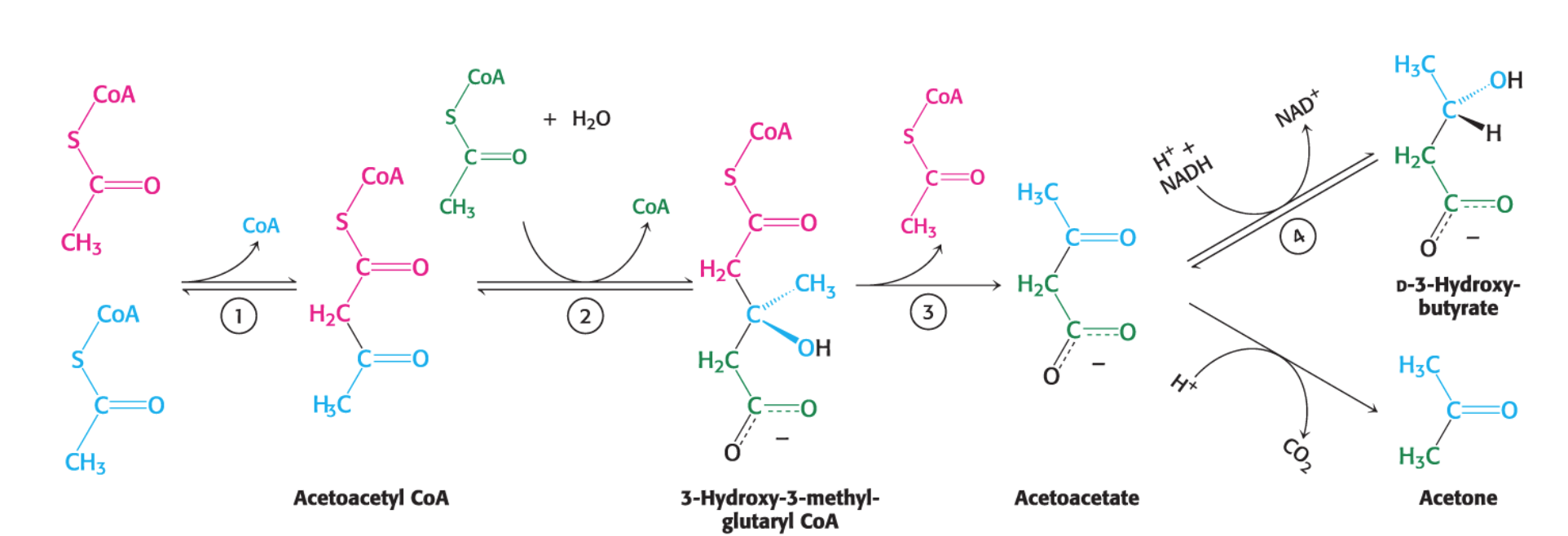
After being transferred to other tissues, how is D-3-beta-hydroxybutyrate converted back to acetyl CoA to be used to make ATP?
In tissues using ketone bodies, D-3-beta-hydroxybutyrate is oxidized to acetoacetate, which is ultimately metabolized to two molecules of acetyl CoA.
How are the ketone bodies, Acetoacetate and D-3-hydroxybutyrate, a major fuel source
Acetoacetate and D-3-hydroxybutyrate are known to be normal fuels of respiration.
They are not just indicators of impaired metabolism.
Heart muscle and the renal cortex use acetoacetate in preference to glucose.
They are utilized by the heart, muscle, kidney, and brain
The brain adapts to the utilization of acetoacetate during starvation and diabetes.
In prolonged starvation, 75% of the fuel needs of the brain are met by ketone bodies.
What’s the need for fatty acid (palmitate) synthesis?
While adult human diets typically provide sufficient fats, many tissues retain the ability to synthesize fatty acids.
Fatty acid synthesis is particularly important during embryonic development and lactation in mammary glands.
Most commonly, fatty acids are synthesized in the liver when the body's energy needs are met, allowing excess fuel to be stored.
What are two differences between Fatty Acid Degradation and Synthesis?
Location:
Synthesis primarily occurs in the cytoplasm.
Degradation primarily occurs in the mitochondrial matrix.
Carrier Molecules:
Intermediates in fatty acid synthesis are covalently linked to the sulfhydryl groups of a protein called acyl carrier protein (ACP).
Intermediates in fatty acid breakdown are covalently attached to the sulfhydryl group of coenzyme A (CoA).
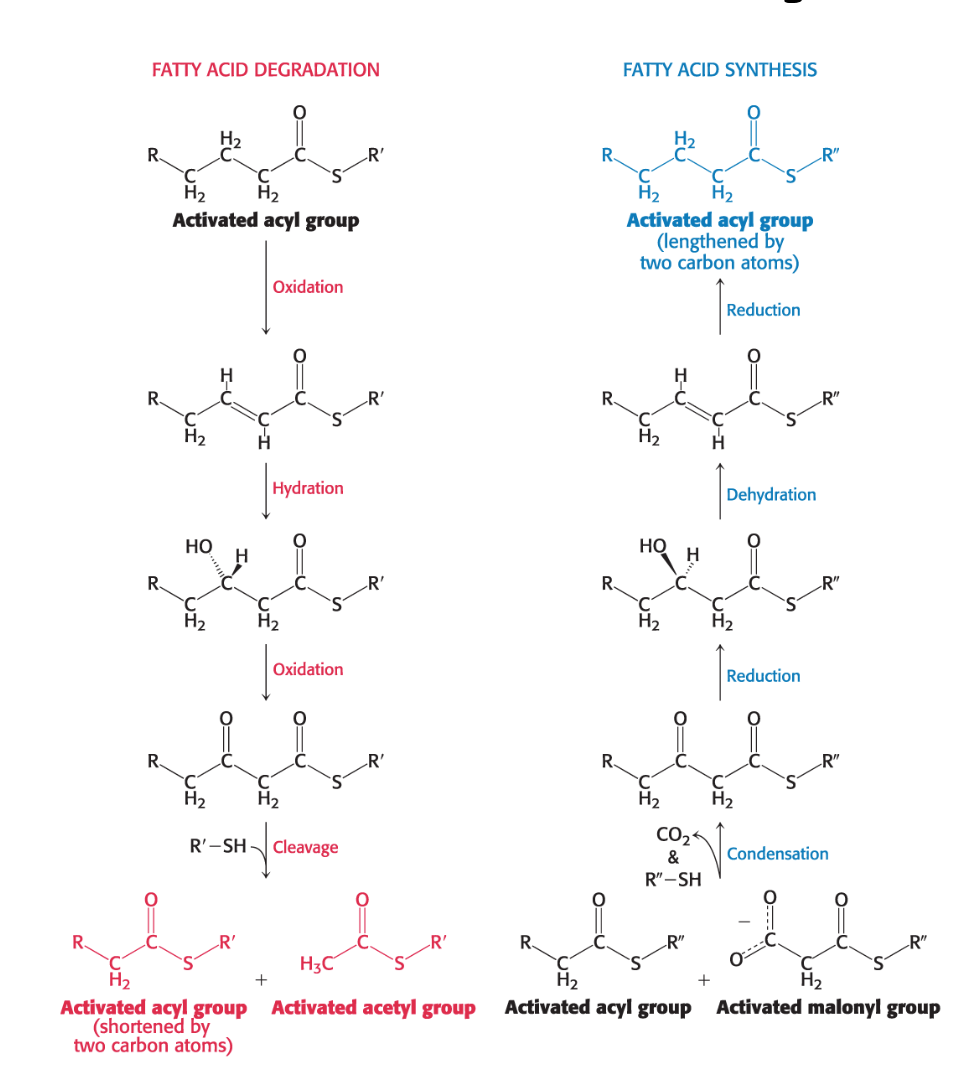
What are the 3 stages of fatty acid synthesis?
Transport: The first stage of fatty acid synthesis is transfer of acetyl CoA out of the mitochondria into the cytoplasm.
Citrate is transported into the cytoplasm and cleaved into oxaloacetate and acetyl CoA.
Activation: The second stage is the activation of acetyl CoA to form malonyl CoA.
Elongation: The third stage is elongation - repetitive addition and reduction of two carbon units to synthesize C16 fatty acid.
Synthesis occurs on an acyl carrier protein, a molecular scaffold.
Stage 1 of fatty acid synthesis - Transportation: Explain how acetyl CoA is transported when the mitochondrial matrix is impermeable to it
The primary challenge for fatty acid synthesis is that while the synthesis pathway operates in the cytoplasm, its precursor, acetyl CoA, is produced in the mitochondrial matrix (from pyruvate oxidation and fatty acid degradation).
This problem is solved by converting acetyl CoA into citrate within the mitochondrial matrix.
Citrate is formed by the condensation of acetyl CoA with oxaloacetate in the mitochondrial matrix, a reaction of the citric acid cycle.
Citrate can then be efficiently transported across the inner mitochondrial membrane into the cytoplasm. Once in the cytoplasm, ATP-citrate lyase cleaves citrate back into cytoplasmic acetyl CoA and oxaloacetate, using one molecule of ATP in the process.
Citrate+ATP+CoA→ATP-citrate lyaseAcetyl CoA+Oxaloacetate+ADP+Pi
What other molecule is produced in this citrate shuttle and how is it important for fatty acid synthesis?
NADPH. It is used in the reduction steps of fatty acid synthesis (during breakdown)
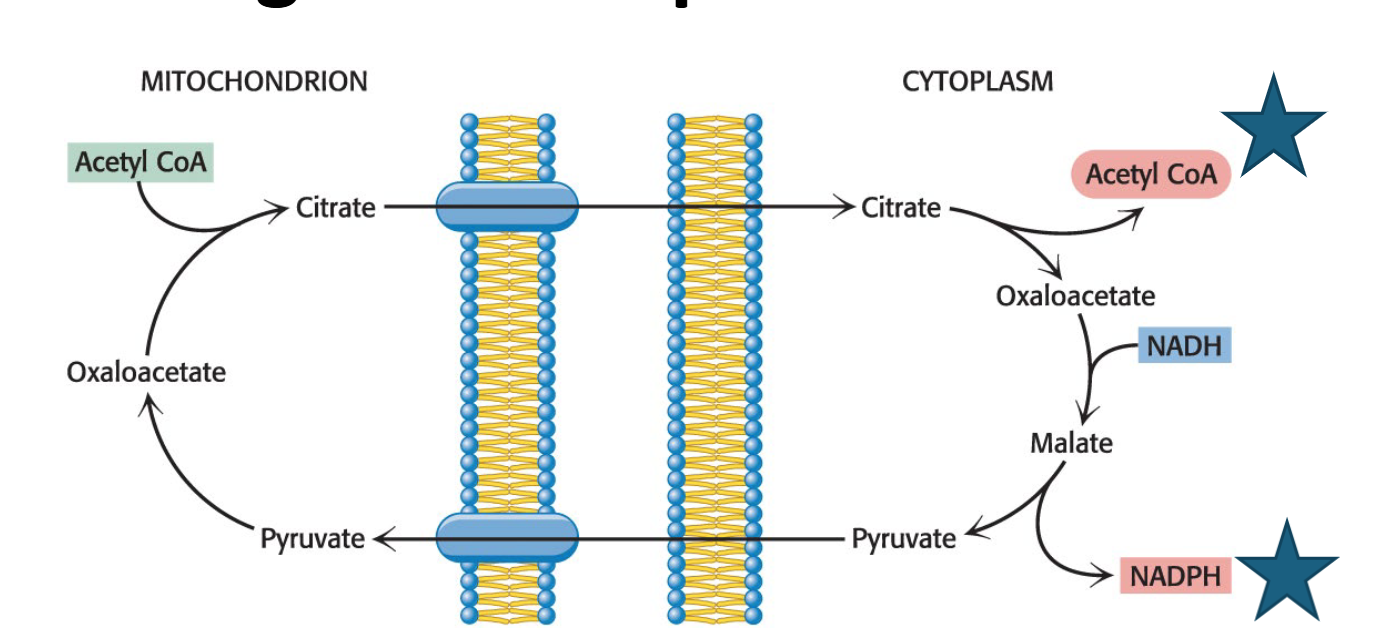
What are other sources of NADPH and ATP for Fatty Acid Synthesis? How does palmitate relate to this?
Other than the transport of citrate from mitochondria, and the subsequent conversion of oxaloacetate to malate and then pyruvate, generating NADPH (citrate shuttle), NADPH can be made from:
The pentose phosphate pathway and the combined action of the citric acid cycle
The necessary ATP for fatty acid synthesis is provided by glycolysis and oxidative phosphorylation.
The synthesis of palmitate requires 14 molecules of NADPH as well as the expenditure of ATP.
Stage 2 of fatty acid synthesis - Activation (1 of 3): Briefly explain the conversion of acetyl CoA to malonyl CoA
Fatty acid synthesis begins with the activation of acetyl CoA through carboxylation (adding CO2) to form malonyl CoA.
Malonyl CoA is considered the activated form of acetyl CoA.
Malonyl CoA is the actual carbon donor for all but two of the carbon atoms of palmitic acid.
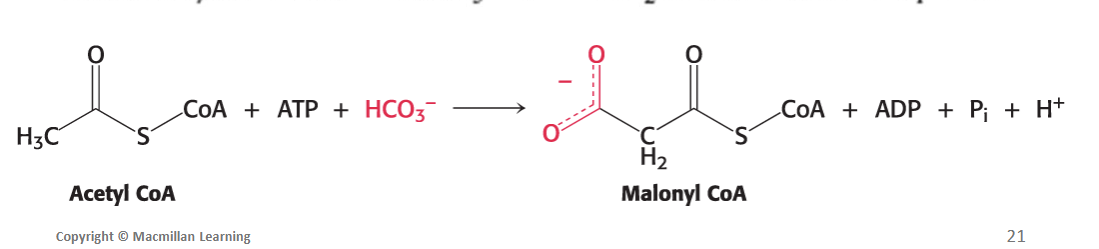
Stage 2 of fatty acid synthesis - Activation (2 of 3): Explain the specific 2 step process for the synthesis of malonyl CoA
Firstly, the biotin carboxylase (BC) activity of the enzyme (ACC1) catalyzes the formation of a carboxybiotin intermediate (activated CO2) at the expense of the hydrolysis of a molecule of ATP.
Secondly, the activated CO2 group is then transferred to acetyl CoA to form malonyl CoA by the carboxyltransferase (CT) activity of the enzyme (ACC1).
Stage 2 of fatty acid synthesis - Activation (3 of 3): Explain exactly how the carboxylation of acetyl CoA to malonyl CoA is catalyzed (which enzyme)
The carboxylation of acetyl CoA to malonyl CoA is catalyzed by acetyl CoA carboxylase 1 (ACC1).
ACC1 is a regulatory enzyme in fatty acid metabolism.
ACC1 is a biotin-dependent enzyme
ACC1 combines acetyl CoA with HCO3−, the form of CO2 in aqueous solutions, to form malonyl CoA.
ACC1 has BC and CT activity
Acetyl CoA+ATP+HCO3−→Malonyl CoA+ADP+Pi+H+
What are some Biotin Nutrition Facts?
Biotin is an essential vitamin, often produced by bacteria in the large intestine for human use. It is also found in various foods, including liver, eggs, cereals, and nuts. Biotin deficiency is rare, but symptoms can include lethargy, muscle pain, nausea, and dermatitis.
Describe the elongation process for fatty acid synthesis: primary enzyme
The enzyme system responsible for synthesizing saturated long-chain fatty acids from acetyl CoA, malonyl CoA, and NADPH is called fatty acid synthase (FAS).
FAS is actually a complex of distinct enzymes, each performing a different function in fatty acid synthesis.
It catalyzes a repeating four-step sequence that elongates the fatty acyl chain by two carbons at each step, using NADPH as the electron donor.
In vertebrates and fungi, FAS I is the primary system.
Fatty acid synthase consists of what two compartments?
Selecting and Condensing Compartment: This compartment binds the acetyl and malonyl substrates and catalyzes their condensation.
acetyl CoA is only used once at the beginning to make this 4-Carbon chain. ACP on FAS then keeps adding 2-C from malonyl to grow the chain
Modification Compartment: This compartment carries out the reduction and dehydration activities required for chain elongation.
What is the preparation step to the fatty acid elongation process? What is the initial carbon chain formed and what bond must be broken at the end of the fatty acid elongation process?
Chain Transfer/Charging (Preparation): Acetyl CoA and malonyl CoA must first be loaded onto FAS I. Then, catalyzed by β-Ketoacyl synthase:
Initially, the acetyl group from acetyl CoA is attached to the thiol group of ACP via thioester bond, releasing a CoA-SH. After which the acetyl group is transferred to the KS cysteine residue
The malonyl group attaches to ACP via thioester linkage and releases CoA-SH.
The ACP with malonyl group swings the malonyl closer to where the 2-C from acetyl are attached so that the two can condense together, and a CO2 gets removed. A 4-carbon chain is formed.
*In the next cycles, different malonyl CoA adds 2-C to the growing fatty acyl chain from previous cycles. Acetyl CoA will no longer be used
To end the chain and all 4 reactions (reductions and dehydration), the last 4-C molecule is called butyryl ACP and the thioester linkage at ACP must be broken at the end of the elongation process, particularly by thioesterase enzyme
What are the 4 steps to the fatty acid elongation process?
Step 1: Condensation (catalyzed by β-Ketoacyl synthase): A condensation reaction attaches the two carbons from the acetyl group (or a longer fatty acyl chain) to two carbons from the malonyl group. This releases CO2 and forms a β-ketoacyl-ACP.
Step 2: Reduction: The β-keto group is reduced to a hydroxyl group using NADPH as the electron donor.
Step 3: Dehydration: Water is removed, creating a double bond.
Step 4: Reduction: The double bond is reduced, saturating the chain, again using NADPH as the electron donor.
How is fatty acid synthesis of palmitate finally terminated
Fatty acid synthase continues to add two-carbon units, always from malonyl CoA, onto the growing fatty acyl group until it reaches a length of 16 carbons, forming a palmitoyl-ACP.
At this point, a thioesterase enzyme cleaves the palmitoyl group from ACP by hydrolysis, releasing palmitate.
The synthesis of palmitate requires how much acetyl CoA, NADPH, and ATP?
The synthesis of palmitate requires 7 bonds to be made, which requires:
Eight molecules of acetyl CoA, seven become the activated form of malonyl CoA
Seven molecules of ATP is required to activate acetyl CoA
7 Acetyl CoA+7 CO2+7 ATP→7 Malonyl CoA+7 ADP+7 Pi+7 H+
For each cycle, it requires 2 NADPH, so 14 molecules of NADPH in total
Overall: 8 Acetyl CoA+7 ATP+14 NADPH→Palmitate+14 NADP++8 CoA+6 H2O
Clinical Insight: Explain Fatty Acid Metabolism Is Altered in Tumor Cells
Tumors require large amounts of fatty acid synthesis to produce precursors for membrane synthesis.
β-Ketoacyl synthase inhibitors retard tumor growth.
Mice treated with β-ketoacyl synthase inhibitors also showed dramatic weight loss, suggesting that such drugs may be used to treat obesity.
Acetyl CoA carboxylase (rate limiting step to make malonyl-CoA) inhibitors may also be potential chemotherapy agents.
In lipid synthesis of triacylglycerols and phospholipids requires what? What is the common first step of this synthesis?
Lipid synthesis, including triacylglycerols and phospholipids, requires the coordinated action of gluconeogenesis and fatty acid metabolism.
The formation of phosphatidate (diacylglycerol 3-phosphate) is a common step in the synthesis of both phospholipids (for membranes) and triacylglycerols (for energy storage).
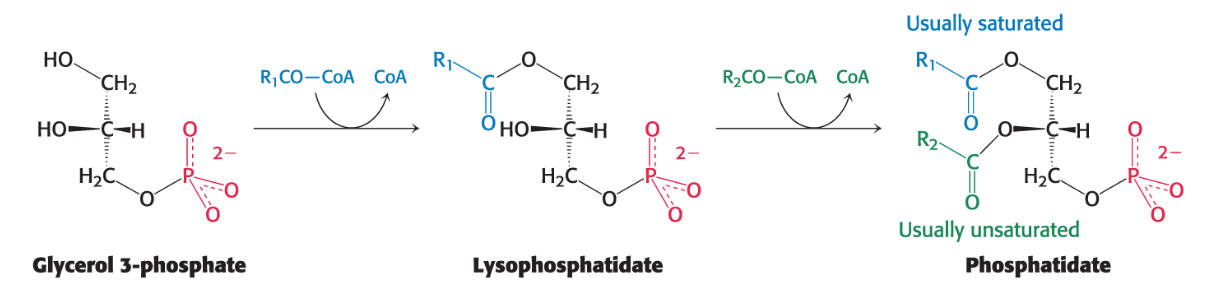
Describe the Biosynthesis of Phosphatidate
The pathway for phosphatidate biosynthesis begins with glycerol 3-phosphate.
Glycerol 3-phosphate is primarily formed by the reduction of dihydroxyacetone phosphate (DHAP), an intermediate synthesized by the gluconeogenic pathway.
*To a lesser extent, it can also be formed by the phosphorylation of glycerol.
In most cases, glycerol 3-phosphate is first acylated by a saturated acyl CoA lysophosphatidate (only 1 FA).
Lysophosphatidate is then acylated by an unsaturated acyl CoA to yield phosphatidate (2 FA).
Alternatively, phosphatidate can be formed by the addition of two fatty acids to glycerol 3-phosphate directly via acyltransferases.
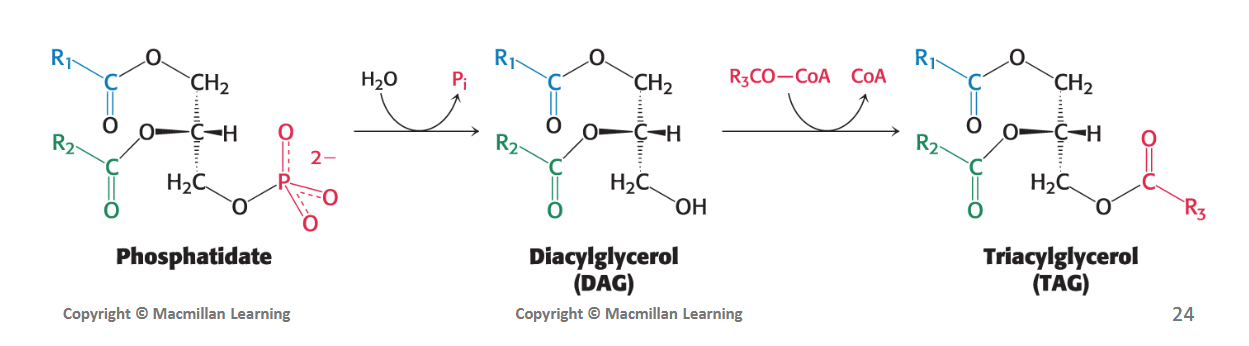
Describe Triacylglycerol Synthesis from Phosphatidate
The phospholipid and triacylglycerol biosynthesis pathways diverge at phosphatidate.
In the synthesis of triacylglycerols, phosphatidic acid phosphatase (PAP) enzyme hydrolyzes phosphatidate (removing a Pi) to produce a diacylglycerol (DAG).
This DAG intermediate is then acylated to a triacylglycerol (TAG) by the addition of a third fatty acid chain. This final step is catalyzed by diglyceride acyltransferase.
Where is the primary site for triacylglycerol, phospholipid, and cholesterol synthesis? Where are the triacylglycerols transported from there?
The liver is the primary site of triacylglycerol, phospholipid, and cholesterol synthesis.
From the liver, TAGs specifically are transported to muscles for fuel or to adipose tissue for storage (where it can also be synthesized, just to a lesser extent).
Approximately 85% of a person's energy is stored as triacylglycerols, predominantly in adipose tissue.
What’s the common activated precursor for triglycerides and phospholipids and how are triglycerides and phospholipids associated with the ER
Common precursor: Phosphatidate is the required precursor for TAG and phospholipid synthesis.
Triglycerides: TAG synthesis takes place on the ER because both PAP and diglyceride acyltransferase are often associated in a triacylglycerol synthetase complex bound to the endoplasmic reticulum (ER) membrane
Phospholipids: phospholipid synthesis also takes place in the endoplasmic reticulum
For phospholipid synthesis, phosphatidate must combine with what? What are the 4 types?
Phosphatidate is combined with an alcohol to generate the phospholipid.
– Choline = phosphatidylcholine
– Inositol = phosphatidylinositol
– Serine = phosphatidylserine
– Ethanolamine = phosphatidyletanolamine
What is cholesterol and what is its 3-stage process to its synthesis?
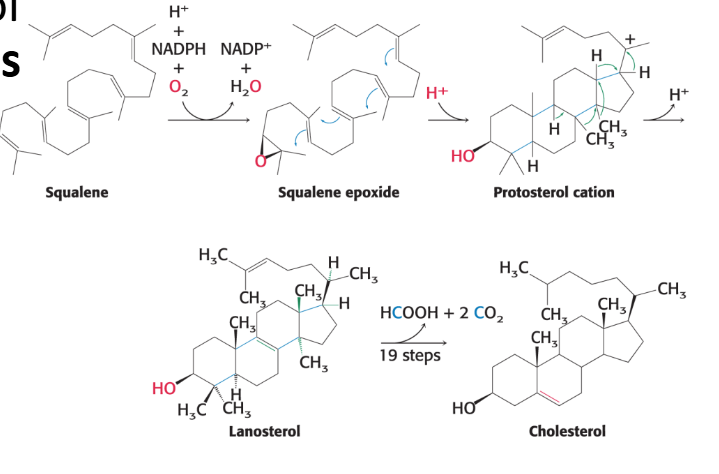
Cholesterol is a made of 27 carbon atoms derived from acetyl CoA through a three-stage synthetic process.
Stage 1: Isopentenyl pyrophosphate is synthesized from mevalonate.
Stage 2: Six molecules of isopentyl pyrophosphate condense to form squalene.
Stage 3: Squalene cyclizes and is converted into cholesterol.
• Stage 1 occurs in the cytoplasm and stages 2 and 3 in the endoplasmic reticulum
What is the importance of cholesterol and how is it regulated?
Importance of Cholesterol:
Membrane Fluidity: It maintains the proper fluidity of animal cell membranes.
Precursor: It is the precursor for important steroid hormones (e.g., progesterone, testosterone, estradiol, cortisol) and bile acids.
Regulation: The rate of cholesterol synthesis is highly responsive to the cellular level of cholesterol itself, ensuring homeostatic control.
Describe the stage 1 (1 of 3) of cholesterol synthesis: how is HMG-CoA formed and location
The first stage takes place in the cytoplasm.
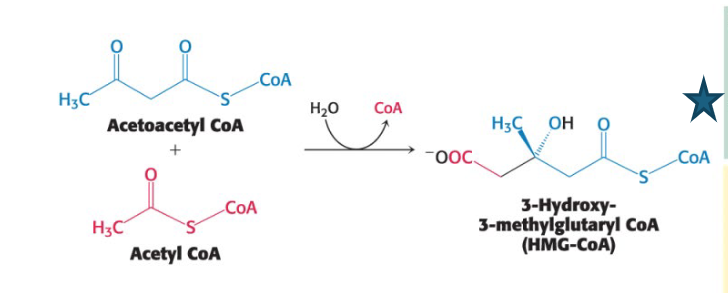
This set of reactions starts with the formation of 3- hydroxy-3-methylglutaryl CoA (HMG-CoA) from acetyl CoA and acetoacetyl CoA.
Describe the stage 1 (2 of 3) of cholesterol synthesis: how is mevalonate formed and what’s special about this step?
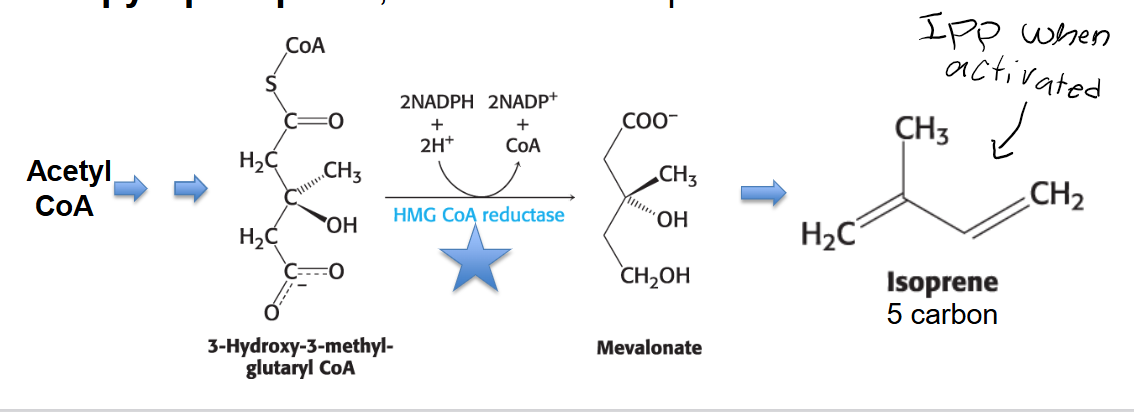
Mevalonate Synthesis (Rate-Limiting Step): HMG CoA is then reduced using NADPH to mevalonate. This is the committed and rate-limiting step in cholesterol formation.
The enzyme catalyzing this irreversible step is 3-hydroxy-3-methylglutaryl CoA reductase (HMG-CoA reductase).
HMG-CoA reductase is the key control site for cholesterol biosynthesis, and it requires NADPH.
Describe the stage 1 (3 of 3) of cholesterol synthesis: how is Isopentenyl Pyrophosphate formed?
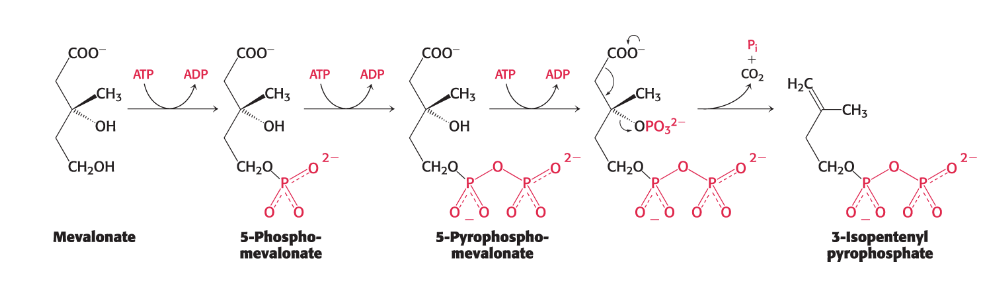
Mevalonate is then converted into 3-isopentenyl pyrophosphate in three consecutive reactions, each requiring ATP.
Stage 1 ends with the production of isopentenyl pyrophosphate, an activated five-carbon isoprene unit.
Describe the stage 2 of cholesterol synthesis: Describe how is squalene formed: location
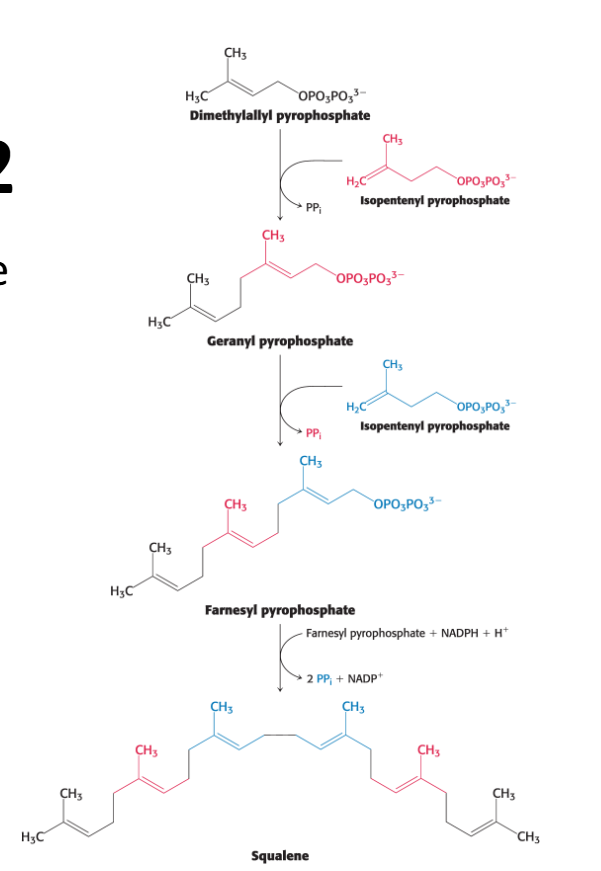
This stage occurs in the endoplasmic reticulum.
Squalene is synthesized from isopentenyl pyrophosphate.
Isopentenyl pyrophosphate isomerizes to dimethylallyl pyrophosphate.
The two isomer C5 units (one of each) condense to begin the formation of squalene (30 carbons)
Describe the stage 3 of cholesterol synthesis: Describe the Cyclization to Cholesterol: location and requirements
This stage occurs in the endoplasmic reticulum.
Squalene Activation: Squalene is first activated by conversion into squalene epoxide in a reaction that uses O2 and NADPH.
Cyclization: Squalene epoxide then cyclizes (forms rings) to lanosterol. Lanosterol has 30 carbon atoms.
Conversion to Cholesterol: Lanosterol (C30) is subsequently converted into cholesterol (C27) through a multistep process, during which three carbon atoms are removed.
Cholesterol is the Most Highly Decorated Small Molecule in Biology
DID YOU KNOW?
“Cholesterol is the most highly decorated small molecule in biology. Thirteen Nobel Prizes have been awarded to scientists who devoted major parts of their careers to cholesterol. Ever since it was isolated from gallstones in 1784, cholesterol has exerted an almost hypnotic fascination for scientists from the most diverse areas of science and medicine....Cholesterol is a Janus-faced molecule. The very property that makes it useful in cell membranes, namely its absolute insolubility in water, also makes it lethal.”
What are statins in terms of the how HMG-CoA reductase enzyme can be controlled?
Statins, or HMG-CoA reductase inhibitors, structurally resemble mevalonate (the rate limiting step in cholesterol synthesis) and are a class of medications used to lower cholesterol by blocking the liver enzyme responsible for producing it.
What are the fates of cholesterol after synthesis?
In vertebrates, most cholesterol is synthesized in the liver, then exported.
Can be exported as bile acids, biliary cholesterol, cholesteryl esters, or as lipoproteins.
Differ between the physical properties (polarity) of cholesterol, TAGs, and phospholipids
Cholesterol and Phospholipids: These are amphipathic molecules, meaning they have both hydrophobic (water-fearing) and hydrophilic (water-loving) regions.
Phospholipids are reasonably soluble in water due to their polar head groups.
Cholesterol, while amphipathic, has a significant hydrophobic portion that prefers not to associate with water.
Triacylglycerols: These are generally hydrophobic molecules, with virtually no solubility in water.
Why do triacylglycerols and cholesterol require lipoproteins for transport?
Due to TAGs hydrophobic nature, and cholesterol mostly hydrophobic nature require specialized transport mechanisms in the body's aqueous fluids (blood plasma). This is where lipoproteins come into play.
What are lipoproteins and their surface versus interior structure?
Lipoproteins are spherical particles that transport hydrophobic lipids (like triacylglycerols, cholesterol, and cholesteryl esters) through the aqueous environment of the plasma.
Interior structure: Contains hydrophobic lipids, primarily triacylglycerols, cholesterol, and cholesteryl esters.
Surface Shell: Composed of more-polar lipids (a phospholipid monolayer) and proteins called apolipoproteins.
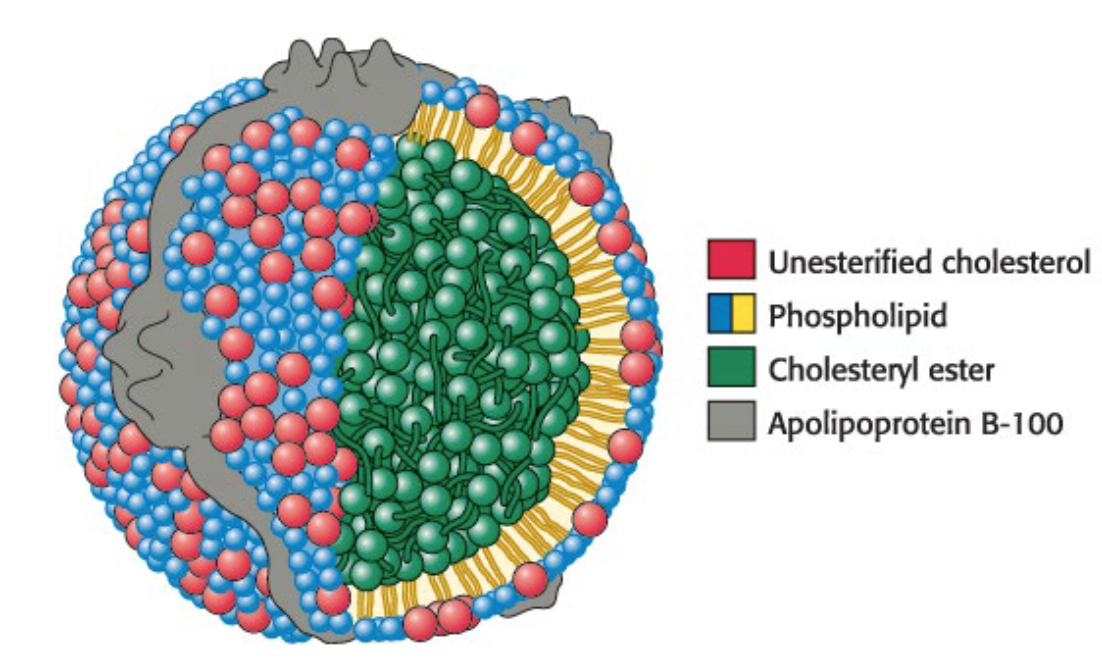
What are the 2 crucial roles of apolipoproteins?
They solubilize hydrophobic lipids.
They contain cell-targeting signals
What are the different lipoproteins and how are they classified? Can this classification be modified?
Lipoproteins are classified according to increasing density.
This can be modified: Lipoproteins can shift between classes as they release or pick up cargo, thereby changing their density.
chylomicrons (X60,000)
chylomicron remnants
very low-density lipoproteins (VLDLs) (X180,000)
intermediate-density lipoproteins (IDLs)
low-density lipoproteins (LDLs) - bad cholesterol (X180,000)
high-density lipoproteins (HDLs) - good cholesterol (X180,000)
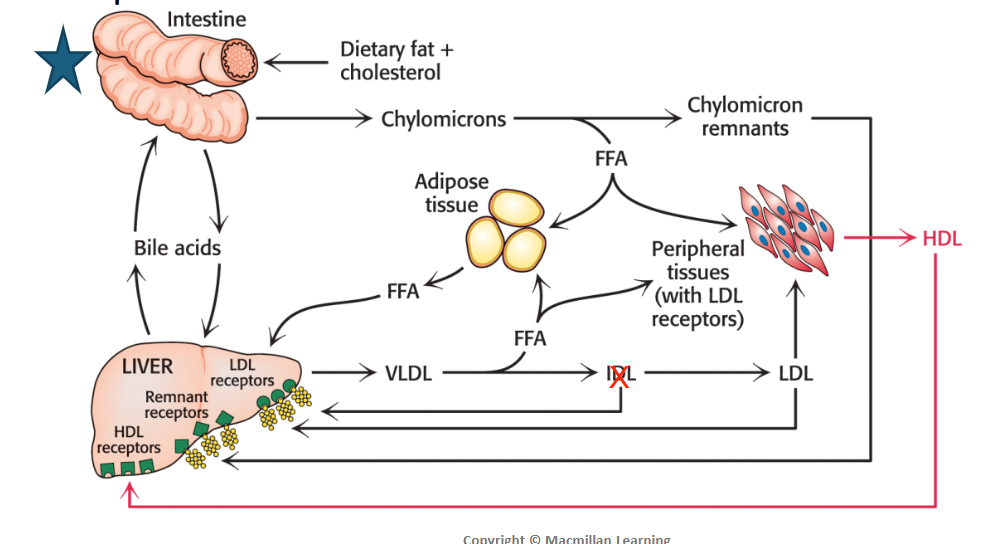
What is the specific role of Chylomicrons and associated proteins?
Role: Packaged in the intestine. Deliver triglycerides from the intestines that are derived from food intake
ApoB-48 is associated
The triacylglycerols associate with specific proteins and a small amount of phospholipid and cholesterol to form lipoprotein transport particles called chylomicrons (CM).
ApoC-II activates lipoprotein lipase to release free fatty acids from TAGs for fuel in adipose tissue, heart, and skeletal muscle.
Chylomicron remnants go to the liver for absorption
They are released into the lymph system and then into the blood.
How are both TAGs and cholesterol exported in the blood when they are produced in excess in the liver?
Triglycerides and cholesterol in excess of the liver’s own needs are exported into the blood in the form of very low-density lipoprotein (VLDL).
What is the specific role of VLDL, associated proteins, and what happens to the TAGs versus cholesterol that they contain after being exported to blood?
Role: Packaged in the liver from diet derived and de-novo synthesized lipids. Deliver triglycerides derived from the liver to non-hepatic (not liver) tissues
ApoB-100 is associated
After export to blood:
Triglycerides: ApoC-II activates Lipoprotein lipase, which hydrolyzes the triacylglycerols in VLDLs, releasing fatty acids that are taken up by cells in non-hepatic tissues like adipocytes for storage.
Cholesterol: The cholesterol-rich remnants, called intermediate-density lipoproteins (IDLs), are rapidly converted into low-density lipoprotein (LDL) by the removal of more TAGs
What is the specific role of LDL and well as their structure, function, and any associated proteins
Function - Major Cholesterol Carrier: deliver cholesterol and cholesteryl esters derived from the liver to non-hepatic tissues and plays a role in regulating de novo cholesterol (not from diet, body makes it itself) synthesis at these sites.
Structure: It has a core of cholesterol molecules linked by ester bonds to fatty acids (cholesterol ester), surrounded by phospholipids, unesterified cholesterol, and a single copy of ApoB-100 recognized by LDL receptors.
What is the specific role of HDL and what protein is associated?
Role: Removes excess cholesterol from non-hepatic tissues
They pick up cholesterol from non/extra-hepatic tissues and bring it back to the liver in a process termed reverse cholesterol transport.
Apolipoprotein-A1 is associated
Cholesterol metabolism must be regulated in order to what?
Cholesterol metabolism must be precisely regulated to prevent atherosclerosis, the thickening of arterial walls with a subsequent loss of elasticity.
How do LDL lipoproteins play a central role in cholesterol metabolism in terms of receptor-mediated endocytosis
The primary source of cholesterol for peripheral tissues is the LDL.
High concentrations of LDL in the blood play a role in setting the conditions for a heart attack.
LDL is normally removed from the blood in a process called receptor-mediated endocytosis, which serves as a paradigm for the cellular uptake of many molecules.
Receptor-mediated endocytosis of LDL is accomplished in three steps.
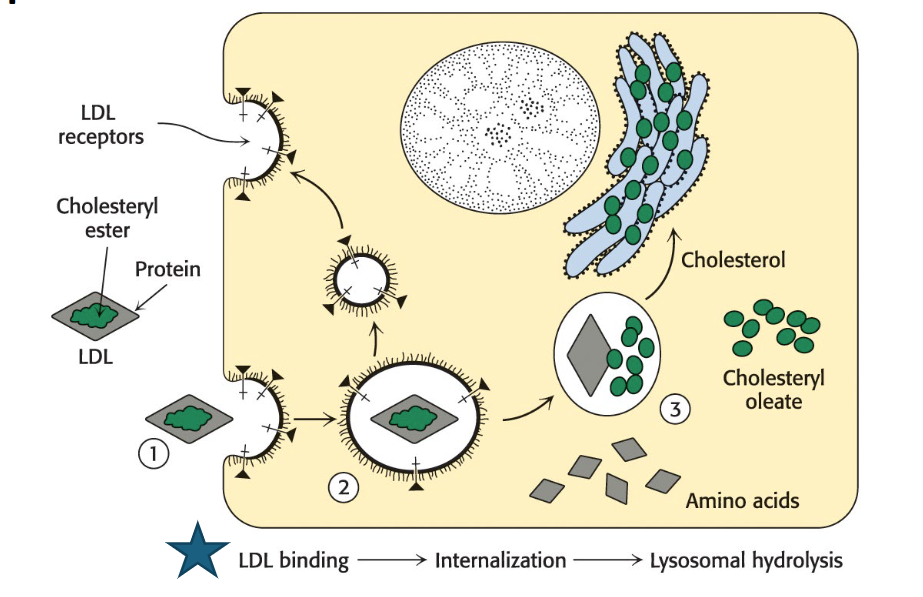
Describe the 3 steps of the receptor-mediated endocytosis of LDL
LDL binding: ApoB-100 on the LDL particle binds to a specific LDL-receptor protein on the plasma membrane of non-liver cells.
Internalization: The plasma membrane invaginates (folds inward) in the vicinity of the receptor-LDL complex, forming an endocytic vesicle (endosome) that encloses the complex. This is called endocytosis.
Hydrolysis in lysosomes: The vesicles fuse with lysosomes, which contain degradative enzymes.
The protein (ApoB-100) component of LDL is hydrolyzed to free amino acids.
The cholesteryl esters in LDL are hydrolyzed by lysosomal acid lipase to free cholesterol and fatty acids.
The LDL receptor usually returns to the plasma membrane to pick up more LDL.
The round-trip time for a receptor is about 10 minutes; in its lifetime of about a day, it brings many LDL particles into the cell.
What is the fate of the released unesterified cholesterol from the receptor-mediated endocytosis of LDL?
The released unesterified cholesterol can be used for membrane biosynthesis or re-esterified for storage inside the cell.
Stored cholesterol must be re-esterified because high concentrations of free cholesterol can disrupt cell membrane integrity.
Describe the feedback regulation of the LDL receptor
The synthesis of the LDL receptor itself is subject to feedback regulation.
When cholesterol is abundant inside the cell, new LDL receptors are not synthesized, which blocks the uptake of additional cholesterol from plasma LDL.
Clinical Insight: Describe what is Niemann–Pick Disease and how it relates to the transport of Cholesterol from the Lysosome
Niemann–Pick diseases are a group of lipid storage disorders of varying severity. One fatal variety is caused by the accumulation of cholesterol in lysosomes that results in multiple organ failure
Clinical Insight: Describe what is Familial Hypercholesterolemia (FH), how it’s caused, and how it affects homozygotes and heterozygotes differently
FH is a genetic disorder characterized by high concentrations of cholesterol and LDL in the plasma (3-4 times the desired amount). In familial hypercholesterolemia, cholesterol is deposited in various tissues because of the high concentration of LDL cholesterol in the plasma (possibly leading to atherosclerosis)
Molecular Defect: The primary defect is an absence or deficiency of functional LDL receptors.
Homozygotes: Have almost no functional LDL receptors and often die of coronary artery disease in childhood.
Heterozygotes: Have about half the normal number of receptors, with a milder but variable clinical course (affecting about 1 in 500 people).
Clinical insight: What exactly occurs when LDL accumulates under the skin and endothelial cells lining the blood vessels?
Pathogenesis: This impaired LDL entry into liver and other cells leads to increased plasma LDL. This cholesterol then accumulates:
Excess LDL can become oxidized LDL (oxLDL), which stimulates an inflammatory response by the immune system, initiating plaque formation.
Immune system cells called macrophages take up oxLDL, becoming foam cells.
These foam cells get trapped in blood vessel walls, contributing to atherosclerotic plaques that narrow arteries and lead to heart attacks.
Clinical insight: Describe 3 ways HDL seems to protect against atherosclerosis
HDL and its role in returning cholesterol to the liver are important in mitigating these life-threatening circumstances.
Anti-atherogenic Properties: HDL has several properties that protect against atherosclerosis, including inhibiting LDL oxidation.
Reverse Cholesterol Transport: HDL's best-characterized role is removing cholesterol from cells, particularly macrophages, and returning it to the liver for excretion (as bile or in feces). This is crucial for mitigating plaque formation.
Foam Cell Prevention: When reverse cholesterol transport fails, macrophages become foam cells and facilitate the formation of plaques. The more HDL, the more readily this transport takes place and the less likely the macrophages are to develop into foam cells.
What are dietary strategies for reducing CVD risk?
Lowering dietary cholesterol generally has only a minor impact on blood cholesterol for most people. So instead we try these:
Strategies to reduce cardiovascular disease (CVD) risk include:
Increasing HDL activity for cholesterol scavenging (e.g., n-3 fatty acids).
Decreasing cholesterol synthesis (e.g., polyunsaturated fatty acids).
Consuming nutrients like fiber, which increase bile acid and cholesterol excretion.
How should we make efforts to maintain a healthy lipids and lipoprotein metabolism?
Make efforts to lower total triglycerides, total cholesterol and LDL-cholesterol levels
Make efforts to increase HDL-cholesterol levels
Cholesterol is a precursor to what?
Bile salts, steroid hormones, and vitamin D.
How is bile synthesized, stored, and secreted, and what's their function in the body?
Synthesis: Bile salts are steroid-based molecules synthesized from cholesterol in the liver. They are composed of cholesterol, phospholipids, and the breakdown products of heme.
Storage and Secretion: Stored in the gallbladder and secreted into the small intestine.
Function: Act as detergents that emulsify dietary lipids, making them more accessible for digestion by lipases and for subsequent absorption.
Explain what excess cholesterol in bile can lead to. What might have to be done in this case?
Excess cholesterol in bile can precipitate to form gallbladder stones. These stones can block bile secretion and cause inflammation of the gallbladder, a condition called cholelithiasis, leading to pain and nausea.
If need be, the gallbladder is removed, and bile flows from the liver through the bile duct directly into the intestine.
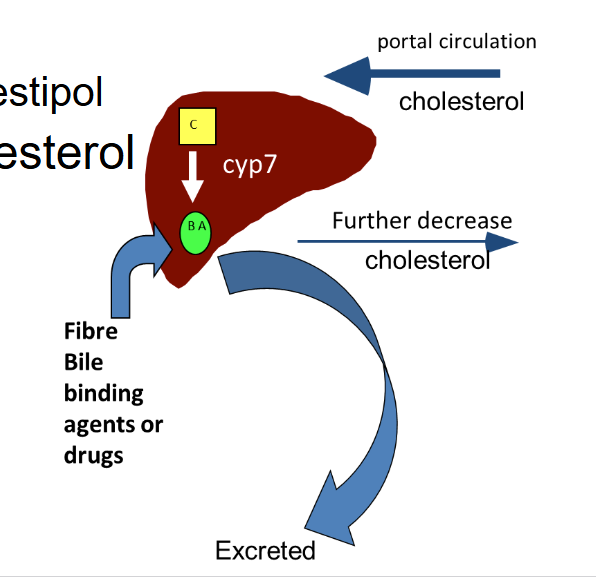
How can Bile Salt Binding Drugs and fiber reduce lower LDL cholesterol?
Medications like cholestyramine and colestipol are bile acid/salt binding resins. They lower cholesterol by removing it from the liver in the form of bile acids.
Fiber or medication uses Cyp7 enzyme to bind to bile acids and excrete them from the liver.
By increasing their excretion, they also promote reverse cholesterol transport to return cholesterol back to liver to synthesize more bile acids, further decreasing cholesterol.
How are steroid hormones derived and what is their primary function in the body?
Derivation: Steroid hormones are derived from cholesterol and share a common four-fused-ring structure called the steroid nucleus.
Function: Powerful signal molecules that regulate numerous organismal functions by binding to and activating receptor proteins that act as transcription factors (proteins that regulate gene expression).
What are the five major classes of steroid hormones?
1. Progestagens
2. Glucocorticoids
3. Mineralocorticoids
4. Androgens
5. Estrogens
Five major classes of steroid hormones: What is the function of Progesterone (a Progestagens)?
prepares the lining of the uterus for the implantation of an ovum and is essential for the maintenance of pregnancy.
Five major classes of steroid hormones: What is the function of Glucocorticoids (e.g., Cortisol)?
Promote gluconeogenesis and glycogen formation.
Enhance the degradation of fat and protein.
Inhibit the inflammatory response.
Crucial for responding to stress; absence can be fatal.
Five major classes of steroid hormones: What is the function of Mineralocorticoids (e.g., Aldosterone)?
Act on the kidney to regulate salt balance, blood volume, and blood pressure.
Five major classes of steroid hormones: What is the function of Estrogens (e.g., Estradiol)?
Required for female secondary sex characteristics and participate in the ovarian cycle along with progesterone.
Five major classes of steroid hormones: What is the function of Androgens (e.g., Testosterone)?
Responsible for male secondary sex characteristics, maintenance of testes, and muscle mass development.
Clinical insight: what’s the relation between Androgens steroid hormones and Athletic Performance? What are the side effects in males versus females?
Some athletes take androgens because the anabolic effects of androgens increase lean muscle mass.
Side effects in males: Decreased testosterone secretion, testicular atrophy, and sometimes breast enlargement (gynecomastia) due to conversion of excess androgen to estrogen.
Side effects in females: Decreased ovulation and estrogen secretion, breast regression, and facial hair growth.
How is vitamin D synthesized into its active form?
Cholesterol is the precursor of vitamin D.
• The ultraviolet light of sunlight breaks a bond in 7- dehydrocholesterol to form previtamin D3.
• This initiates the synthesis of calcitriol (1,25- dihydroxycholecalciferol), the active hormone.
Clinical Insight: Vitamin D and Bone Development: What are diseases related to the deficiency of vitamin D?
Rickets (in childhood): Inadequate calcification of cartilage and bone, characterized by weakened bones and bowed legs.
7-dehydrocholesterol in the skin is not cleaved to previtamin D3 because of little exposure to sunlight for many months and/or diets provided little vitamin D supplementation.
Osteomalacia (in adulthood): Softening and weakening of bones.
What’s the recommended intake of vitamin D daily and from what?
10 μg daily, often obtained from fortified foods (e.g., milk).
What are broader roles of vitamin D, other than bone development?
Research suggests vitamin D has roles beyond bone metabolism, potentially enhancing muscle performance, preventing cardiovascular disease, reducing cancer incidence, and protecting against autoimmune diseases like diabetes.
What is the prevalence (commonness) of the deficiency of vitamin D?
Vitamin D deficiency appears to be more common than thought.
People living in northern climates may not be exposed to enough sunlight during certain times of the year to synthesize adequate amounts of vitamin D.
Dark skin pigmentation blocks UV radiation, people with dark complexions living in such areas may be especially susceptible to vitamin D deficiency.
In the United States, some studies suggest that 75% of people with dark-complected skin have insufficient blood levels of vitamin D.
Explain how the Isopentenyl pyrophosphate (C5), the activated five-carbon isoprene unit generated during cholesterol synthesis can be used as Building Blocks for Diverse Biomolecules. Give examples
Terpenes: Many plant fragrances come from volatile C10 (two isoprene units) and C15 (three isoprene units) compounds called terpenes:
Myrcene: 2 isoprene units (C10H16) from bay leaves.
Limonene: 2 isoprene units (C10H15) from lemon oil.
Zingiberene: 3 isoprene units (C15H24) from ginger oil.
Menthol from peppermint oil,
Citronellal
Natural Rubber: A linear polymer of cis-isoprene units.