brunetti parenterals 2
1/75
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
why the parenteral route?
- rapid onset of drug action in emergency situations
- patients who are nil per os; nothing by mouth for medical reasons
- unconscious
- for drugs, like proteins, that cannot be absorbed effectively from the GI
practical considerations for parenteral administration
- intradermal almost never used for treatment
- IM and SQ used fairly often, but they must be absorbed into systemic circulation
- more volume in IM than SQ
- more delayed drug delivery with IM; allows for oil formulations to sit and slowly release over time
cleansing the injection site
- 70% isopropyl alcohol
- povidone iodine [Betadine]
- chlorhexidine-alcohol
choosing the appropriate needle gauge and length
- length is based on route of administration
- gauge of needle is thickness; lower is thicker and higher is thinner
- tend to use higher gauge
general characteristics of IV preparations
- provides rapid onset action
- bioavailability 100% in IV because no absorption phase
IV bolus vs infusion
- bolus with 21 g needle
- bolus undiluted directly in systemic circulation
- infusion for patient safety with diluted drug slowly administered over time to minimize toxicity
- bolus achieves effective Cp immediately, followed by IV infusion to sustain Cp level
- some drugs can never be given bolus, but some drugs must be given low doses as bolus
nurses often dilute IV push meds
- nurses do this to diminish pain and irritation at injection site and to avoid drug reactions
- sometimes they do this because undiluted solution too viscous or more concentrated solution is too difficult to measure the appropriate dose in the syringe
- because usually nurses perform this dilution outside of a laminar flow workstation, possible that sterility of injectable solution may be compromised
- very quick process, so do not have to worry about class sterility
1 liter=
1 kg
IV preps in large volumes
- LVPs up to 1000 mL are administered via infusion via an indwelling catheter (needle)
- infusion flow rates
- there are risks associated with IV administration and indwelling catheters
infusion flow rates
- can be up to 150 mL/hr or greater, unless KVO/keep vein open the slower
- rate also depends on the specific drug. once determined, the rate is accurately controlled with infusion pumps
common structure of catheter
- one spike in you, splits off into y or w shape
- multiple entry points, so you can infuse multiple drugs at once
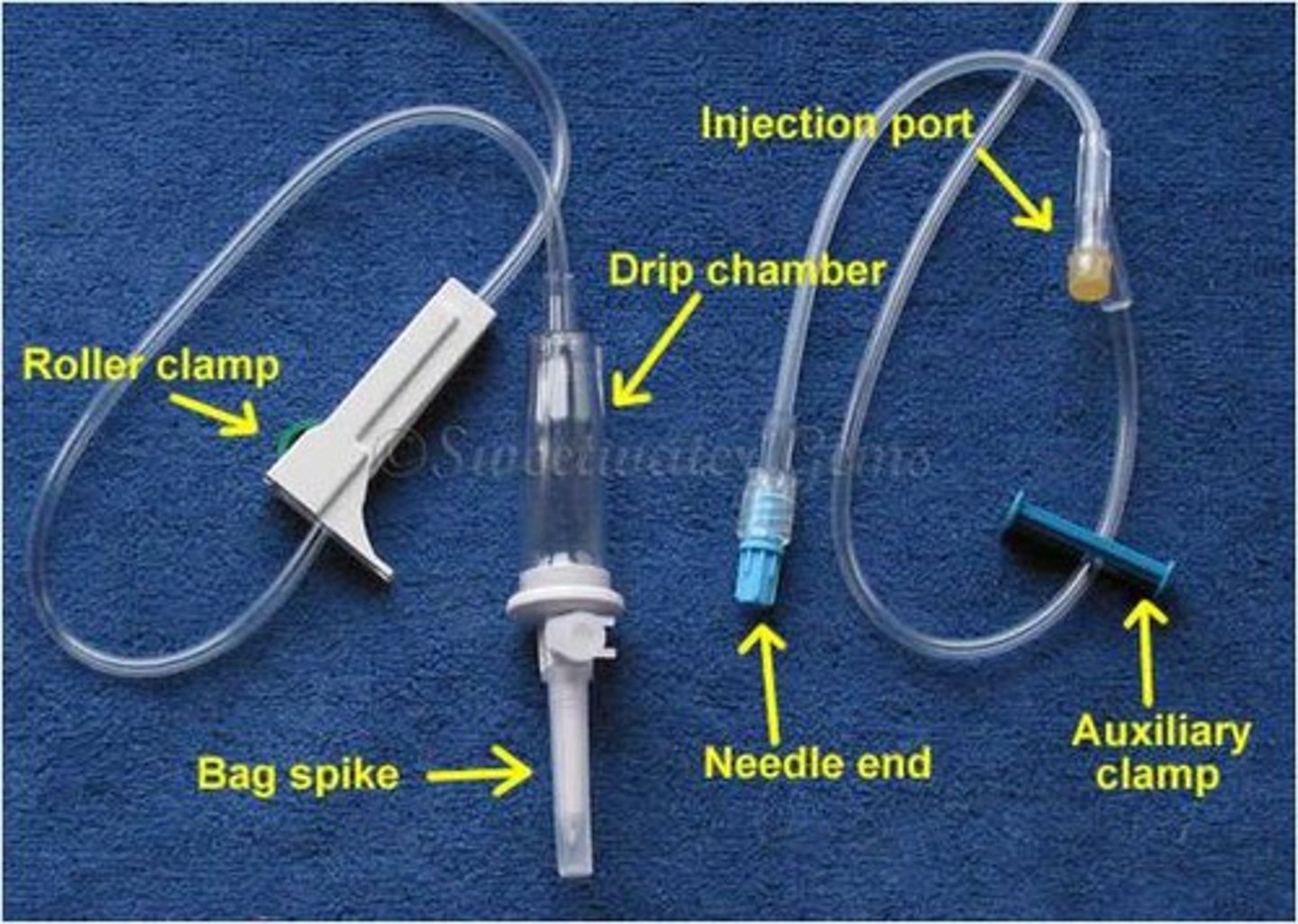
risks associated with IV administration and indwelling catheters
- infection
- thrombophlebitis
- extravasation
- central line associated blood stream infections (CLABSIs)
extravasation
- could have devastating consequences depending on the physiochemical characteristics of the drug
- drug that is caustic leaks out into dermal area and causes necrosis
CLABSIs and venous thromboembolisms
- there are 2 adverse complications that are associated with IV infusions, indwelling catheters and central venous catheters
- there are problems that become life-threatening
- when there products occur, it detracts from the hospital's quality index ratings
infiltration
- leakage to surrounding tissue if non-vesicant solution
Vesicants
- something that causes tissue necrosis
- ex: many therapeutic agents
- may need surgical intervention
how to handle extravasated drug
- antidote preferred over saline
peripheral IV
- inserted into peripheral veins, but do not terminate in central veins
- may feel burning
- regular peripheral IVs can cause fewer events compared to central, but regular peripheral IVs are short term
Central Venous catheters (CVC)
- for patients who need longer-term venous access
- also to minimize pain and risk of harm for some patients
- are catheters that are inserted directly into central veins
- usually inserted by physicians into a vein in the neck and are often reserved the ICU
- the lines eventually reach the superior vena cava
CVC increase the risk of
- CLABSI
- venous thromboembolisms
PICC line (peripherally inserted central catheter)
- a more commonly used variation of CVC line
- can be inserted by physicians or specially trained nurse
- are inserted into peripheral veins but terminate in central veins live CVCs
- not limited to the ICU
- may require heparinization or KVO slow infusion with isotonic LVPs to prevent thrombus formation
advantage of PICC lines
- can be kept in longer that peripheral IVs
disadvantages of PICC line
- poses greater risk of infection and embolism than ordinary peripherally venous catheters
intra-arterial
- less common form of the intravascular route
- for targeting a drug to a specific region or tissue
- high risk of injury
onset for IM
- more slowly absorbed and slow onset
- effects of the drug will be prolonged
IM needle
- lower gauge, around 20, so that you can draw up the formulation
drugs you can only use with IM
- Benzathine Penicillin G
- an aqueous suspension, so it can never be administered IV
- the preferred treatment for syphilis
where can you inject IM
- deltoid muscle for smaller volume up to 2 mL
- gluteus maximus for larger volume up to 5 mL
- MUST avoid hitting nerves or blood vessels
SQ needles
- 21g
- shorter than IV bolus or IM
SQ injection volume
- limited to 2 mL, but usually lower
other names of SQ injection
- hypodermic injections
SQ injection sites
- upper arm or the anterior thigh
- need to rotate the site of injection for frequently administered SC injections, as with insulin, to reduce risk of lipoatrophy
- "pinch and inch"
what is SQ not suitable for
- irritating drugs
intradermal points
- 23g needles
- shorter needle length
- often used for diagnostic tests and some immunizations
- not as common as IV, IM, or SQ
- limited to less than 2 mL
- not intended for producing systemic effects
safe injection practices
- use new needle and syringe each time you reuse MDV
- make sure to wipe the rubber top before each use every time with alcohol swab
Safe syringe and assistance for substance abuse issues
- hypodermic syringes and needles may be sold from the pharmacy department behind the counter in NJ pharmacies under the supervision of a licensed pharmacist
- the purchaser needs to show proof of 18 years of age, and then purchase is limited to a maximum of 10 units at a time
- the pharmacy must provide the purchaser with a state-approved info sheet on proper disposal, including locations, as per the NJ department of health and human services
industrial manufacture or large-scale compounding of parenteral dosage forms
- as enforced by FDA for all US manufacturing facilities
industrial general compounding considerations
- adhere to all applicable cGMPs/current good manufacturing practices
- subject to FDA inspections which could result in recalls or closures if violations are found
Hospital/infusion pharmacy general considerations of parenteral compounding
- requirements are enforced by the state board of pharmacy
- monitored by the joint commission, the hospital accrediting agency
similarities between industry and healthcare facilities
- environmental controls: ISO class 5 environmental control in Direct compounding areas
- aseptic technique/processing: there must be no obstruction between HEPA filter's "first air" and any "critical sites"
- have only essential objects on the working surface of the Hood/direct compounding area to avoid the creation of dead spaces that obstruct laminar air flow
- training programs required by all involved personnel, with periodic recertification
- overall process validation to be conducted at the end of each batch preparation. more routine in industrial manufacturing or in 503b outsourcing compounding pharmacies
- policy and procedures manual required in industry, hospitals, and 503b facilities
- industrial processing involves adherence to similar requirements as in hospital CSP work. USP chapter 797 and 800 requirements apply
lyophilized powder
- cake does not only have the drug; it also has excipients
- a powder
- has longer stability and shelf life, but once you mix it, it needs to be refrigerated and used quick
- reconstitute with sterile water for injection SWFI
- once powder in solution, it must be further diluted before injected into patient
- most go into solution rapidly, except for daptomycin
common excipients in lyophilized powder
- mannitol and lactose as fillers
- phosphate, citrate, and acetate salts as buffers to adjust pH for solubility and stability
Lyophilization
- an industrial process; not an extemporaneous process in hospitals or compounding pharmacies
- requires low pressure vacuum technology
- does no require or usually involve high temperatures, so many drugs, including biologics, that are thermolabile can be formulated into lyophilates
- a relatively expensive industrial manufacturing process
water for injection
- not sterile, but must be pyrogen free
- do not typically use this in hospital setting
- its method of preparation and ultimate quality depend on the original water source
- employed in industrial operations to manufacture products that are then sterilized by a terminal method of sterilization like autoclaving
treatments that water for injection undergoes if impure
- classic distillation to remove pyrogens
- filtration
- reverse osmosis
- de-ionization
technical requirements for water for injection
- limits on total solids upon evaporation
- must be stored in well-closed containers at controlled low temperature to limit bacterial growth that will lead to pyrogen formation
- must be used within 24 hours to limit possibility of pyrogen formation
sterile water for injection
- cannot be used for IV infusion of large volumes
- hemolysis of patients RBCs lead to death if given plain sterile water infusion because hypotonic
- discard unused portion, because it is single use
- must be pyrogen free
bacteriostatic water for injection
- has preservative Benzyl Alcohol
- 30 mL volume limitation to prevent the patient from receiving big dose of preservative
- name and concentration of bacteriostatic agent must be stated on the label
- useful vehicle in MDVs
- not more than 5 mL can be injected into patient
never use bacteriostatic water for injection in infant
- they can't metabolize benzyl alcohol
- causes gasping syndrome, which is a CS toxicity
sodium chloride injections
- if patient has high sodium don't use this
- when there is specific chemical or physical incompatibility between Na+ and API as indicated on package insert of product
- the diluting vehicle of choice for large volume parenteral infusion for drug administration and also for hydration, electrolyte replenishment therapy and for "keep vein open" purposes
sodium chloride injection concentrations
- normal saline or isotonic saline is 0.9% NaCl solution
- 3 is hypertonic, and used in hyponatremic patients
- 23.4 used to make other concentrations of sodium chloride injection, but never directly injected
dextrose for injection
- refers to 5% dextrose solution, of D5W, which is approximately isotonic
- becomes the diluting vehicle of choice for large volume infusions when NaCl injection not available or contraindicated
don't use dextrose for injection when...
1. The patient needs to avoid a glucose spike - as in patients with diabetes.
2. The Active Pharmaceutical Ingredient (API) is not compatible with dextrose
(usually indicated on the package insert)
3. The patient is hyponatremic
ringer's injection/lactated ringer's injection
- more physiologic because it has electrolytes that aren't just sodium and dextrose
- has NaCl, KCl, CaCl2
- bounce back and forth between the preferred fluid historically, but we prefer Lactated Ringer's right now
slide 30 image
- dextrose and normal saline are approximately isotonic
non-aqueous vehicles
- could be toxic at large volumes
- must be diluted further so that body can clear it before it becomes toxin
- sterile glycerin may also be administered IV for osmotic diuresis to remove fluid form the brain as in treating Reyes syndrome
- vegetable oils, olive oil, sesame oil, substitutions if it doesn't work for the person unless TPN; only works for IM
total parenteral nutrition
- microemulsion so that body is able to handle lipids and this special formulation can not be exchanged with other oils
preservatives
- benzyl alcohol 1% is most used
- benzalkonium Cl 0.01%
- thimerosal 0.01%, which was suspected to have caused autism in children who got vaccines but hasn't been proved yet
- methyl and propyl parabens 0.01% to 0.3%
terminal methods of sterilization
- want to achieve probability of less that 1/10^6
- involves the application of a lethal process to sealed containers
dry heat sterilization
- a terminal sterilization method not used for drugs
- 150-170 degrees centigrade for 2 hours or more
- sterilization and depyrogenization of surgical instruments and glassware
- sterilization of non-aqueous materials like glycerin and liquid petrolatum/mineral oil
pressurized steam sterilization
- terminal
- autoclave
- steam acts as sterilant
- higher pressure and higher temperature means less time to sterilize
- the preferred method of sterilization for aqueous CSPs
- closed systems, like solutions in flexible plastic bags, that are sterilized by this method must contain water in order to generate the steam that acts as the sterilizing agent
- method of choice for sterilizing most parenteral drug products unless the API is easily destroyed by heat
radiation sterilization
- terminal
- employs ionizing radiation, usually limited to industrialized applications to sterilize gauze, bandages, possible some drugs
- UV radiation, a wavelength of 253.7 um is not actually classified as a method of sterilization and is a special case
UV radiation
- can be used to reduce bacterial counts in a room where sterile products are prepared and stored
- used more often in industrial settings
- use is limited because personnel need to wear protective eye covering to avoid harm to the eyes
ethylene oxide sterilization
- terminal
- strictly industrial
- sterilizes disposable syringes, needles, and membrane filtration devices
- ethylene oxide gas penetrates sealed pouches to sterilize the contents
- 2 weeks to de-sorb the ethylene oxide gas residue which is toxic if in contact with human skin
filtration sterilization
- not a terminal method
- used very often in clinical setting
- use filters to filter out what you think is sitting in the fluid
- USP 797: passage of solution through a sterilizing grade membrane to produce a sterile effluent
membrane filters
- 0.22 and 0.45 um are most common sizes
- screen or sieve filters
- the ones used today are much smaller compared to older depth-style sterilizing filters
- commercially available in wrapped disposable pouches
USP 797 definition of sterilizing membranes
- Membranes that are documented to retain 100% of a culture of a strain of Brevundimonas (Pseudomonas) diminuta [diameter 0.3μm] per cm2 of membrane surface under a pressure of not less than 30 psi. Such filter membranes are usually 0.22 μm (or 0.2 μm)
nominal pore size, depending on the manufacturer
0.45 microns
- for more viscous fluids
sterilizing membrane filters
- remove but don't kill bacteria
- produce a sterile effluent(filtrate) with very low particulate matter counts
- commercially available in wrapped disposable pouches
types of membrane filters
- polymers and cellulose
polymers for membrane
- cellulosic esters, polyvinylidene fluoride, polytetrafluoroethylene
cellulose ester nitrate or acetate for membrane filter
- most commonly used for routine sterilizations
- could be problematic for sterilizing solutions containing proteins that might bind to the membrane.
- The other types of membranes mentioned above are appropriate for dealing with solutions of extreme pH values or for non-aqueous solutions.
positive vs negative pressure on filters
- filtration sterilization using a disposable syringe with a mounted filter for positive pressure AFTER you have initially draw up the solution, so that when you push the liquid out the dirty stuff gets left in the syringe
- never reuse filter because the crud is stuck
- positive pressure u are left with dirty in the filter
- negative is when u draw up clean drug in the filter
when is vacuum pressure/negative used
- for filtering larger volumes and has industrial applications
in-line filters
- frequently attached to administration sets to ensure that solutions being transfused into the veins of high-risk patients do not contain any bacteria
- filter built within the tubing
6 inch rule
- when you are in the hood, you have a 6 inch frame to work into so that the HEPA filter can work and can constantly clear particulate matter
- do not work at the edge of the hood