Biological Molecules Lecture Notes
1/39
Earn XP
Description and Tags
Comprehensive vocabulary flashcards covering biochemistry, carbohydrates, lipids, proteins, and the properties of water based on the textbook transcript.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Biochemistry
The study of the chemical reactions of biological molecules.
Metabolism
The sum total of all the biochemical reactions in the body.
Organic molecules
Molecules that contain carbon, often forming long chains or ring structures.
Macromolecule
A giant molecule, such as a polysaccharide, protein, or nucleic acid.
Polymer
A giant molecule made from many similar repeating subunits joined together in a chain.
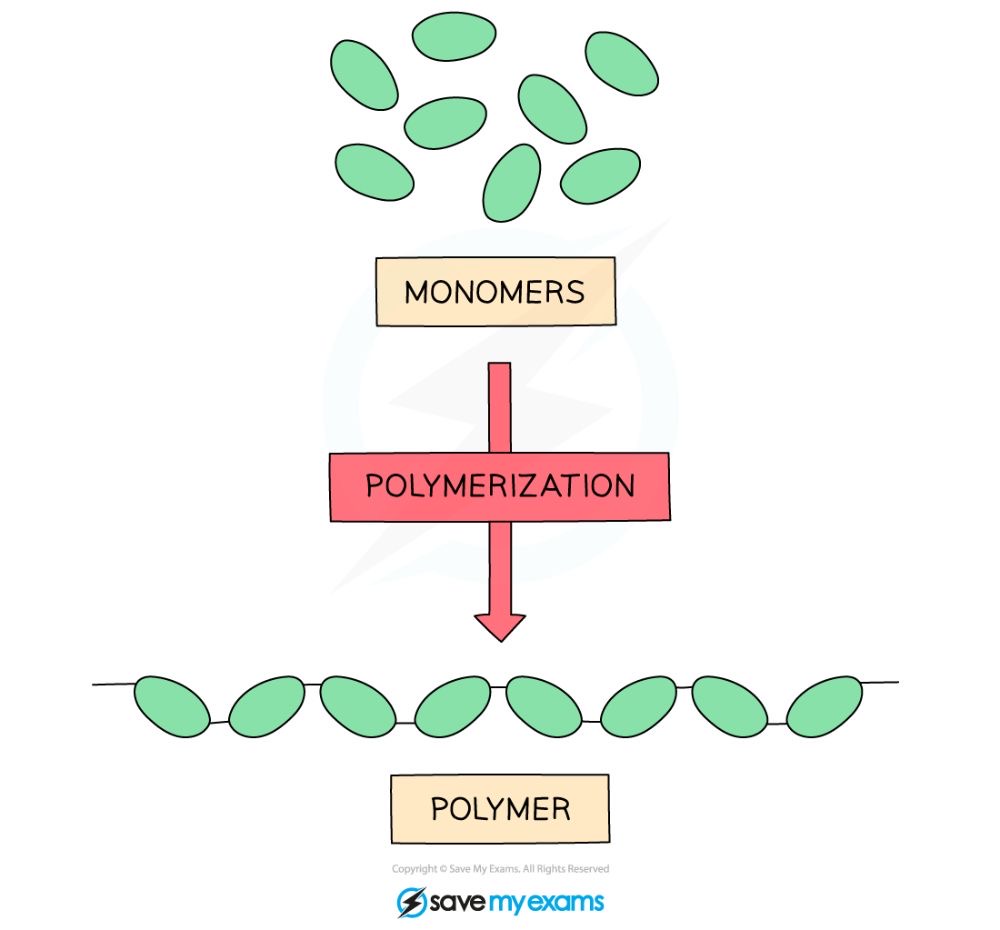
Monomer
A simple molecule which is used as a basic building block for the synthesis of a polymer.
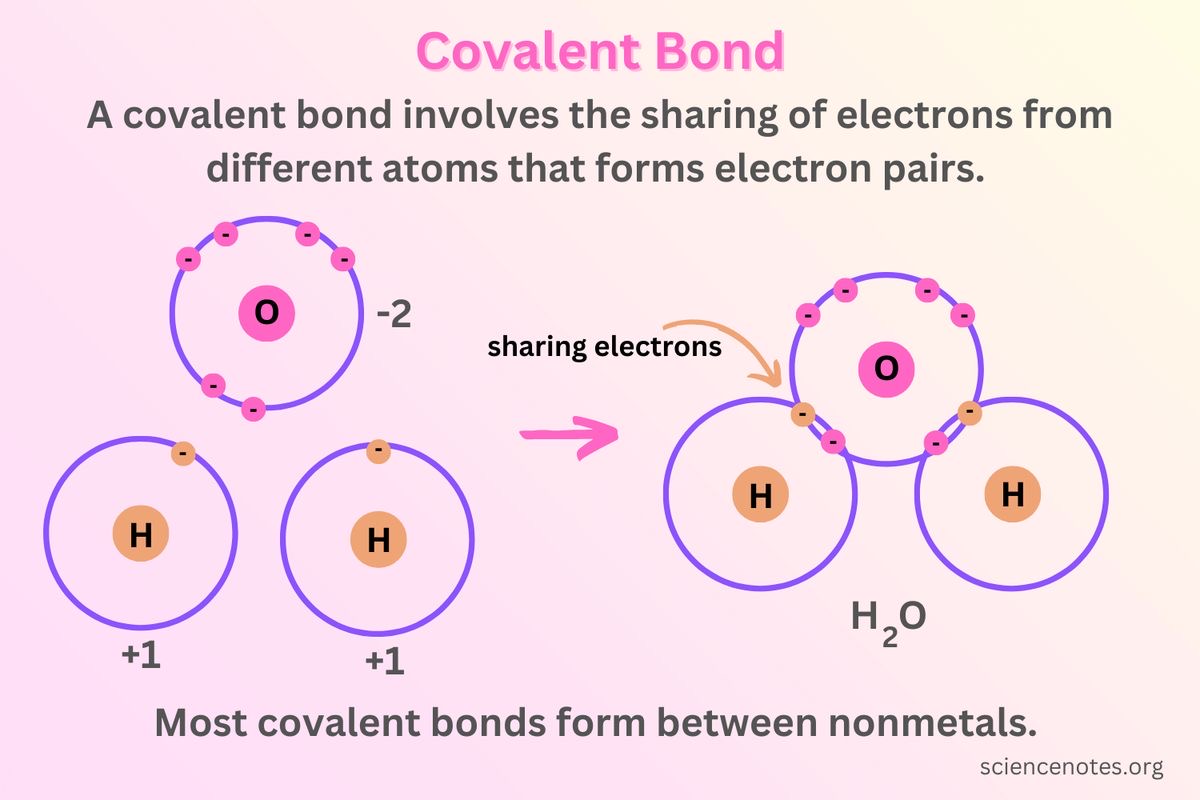
Covalent bond
A bond in which atoms are joined by sharing electrons; examples include glycosidic, ester, and peptide bonds.
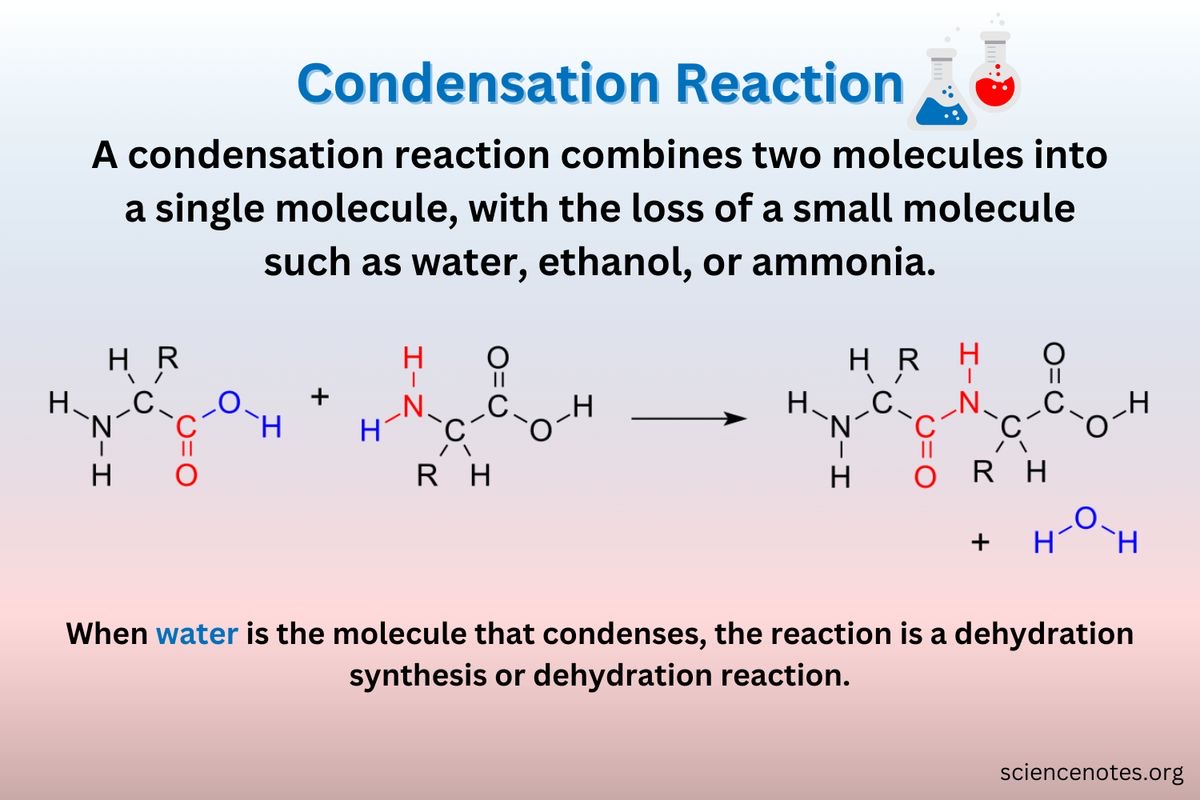
Condensation reaction
A chemical reaction involves the joining together of two molecules by the removal of a water molecule.
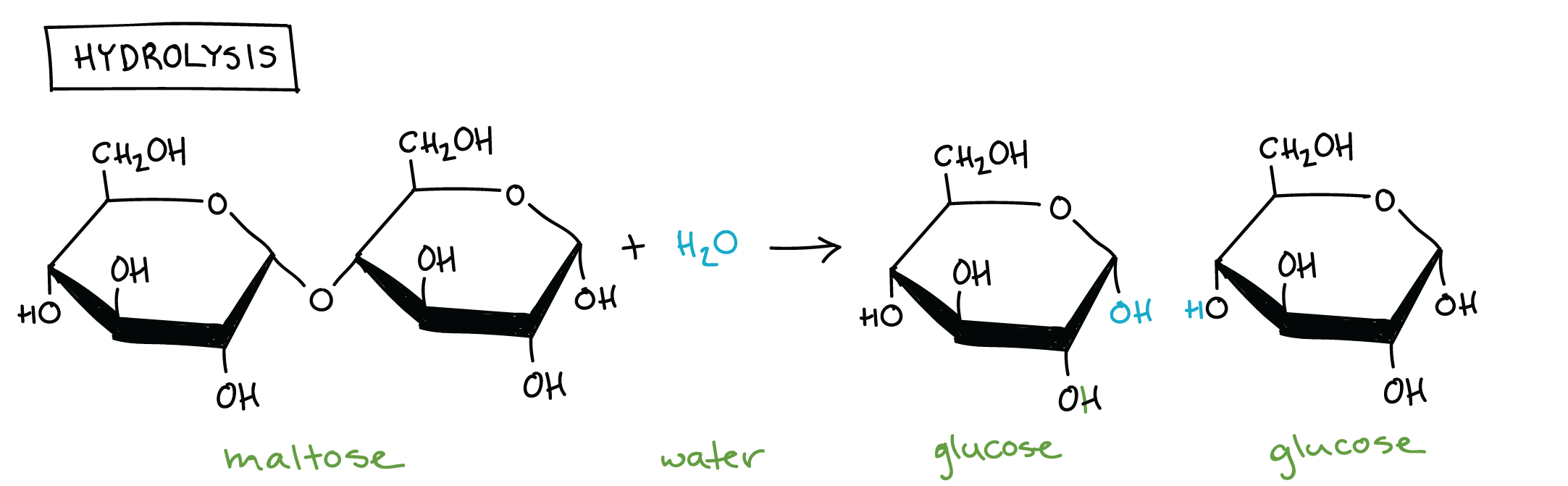
Hydrolysis
A chemical reaction in which a chemical bond is broken by the addition of a water molecule.
Monosaccharide
A molecule consisting of a single sugar unit with the general formula (CH2O)n.
Hexose
A monosaccharide containing 6 carbon atoms, such as glucose, fructose, and galactose, with the molecular formula C6H12O6.
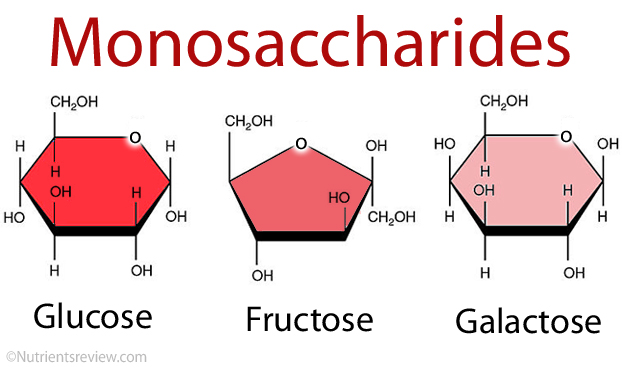
Isomers
Two forms of the same chemical, such as α-glucose and β-glucose, which provide variety for biological structures.
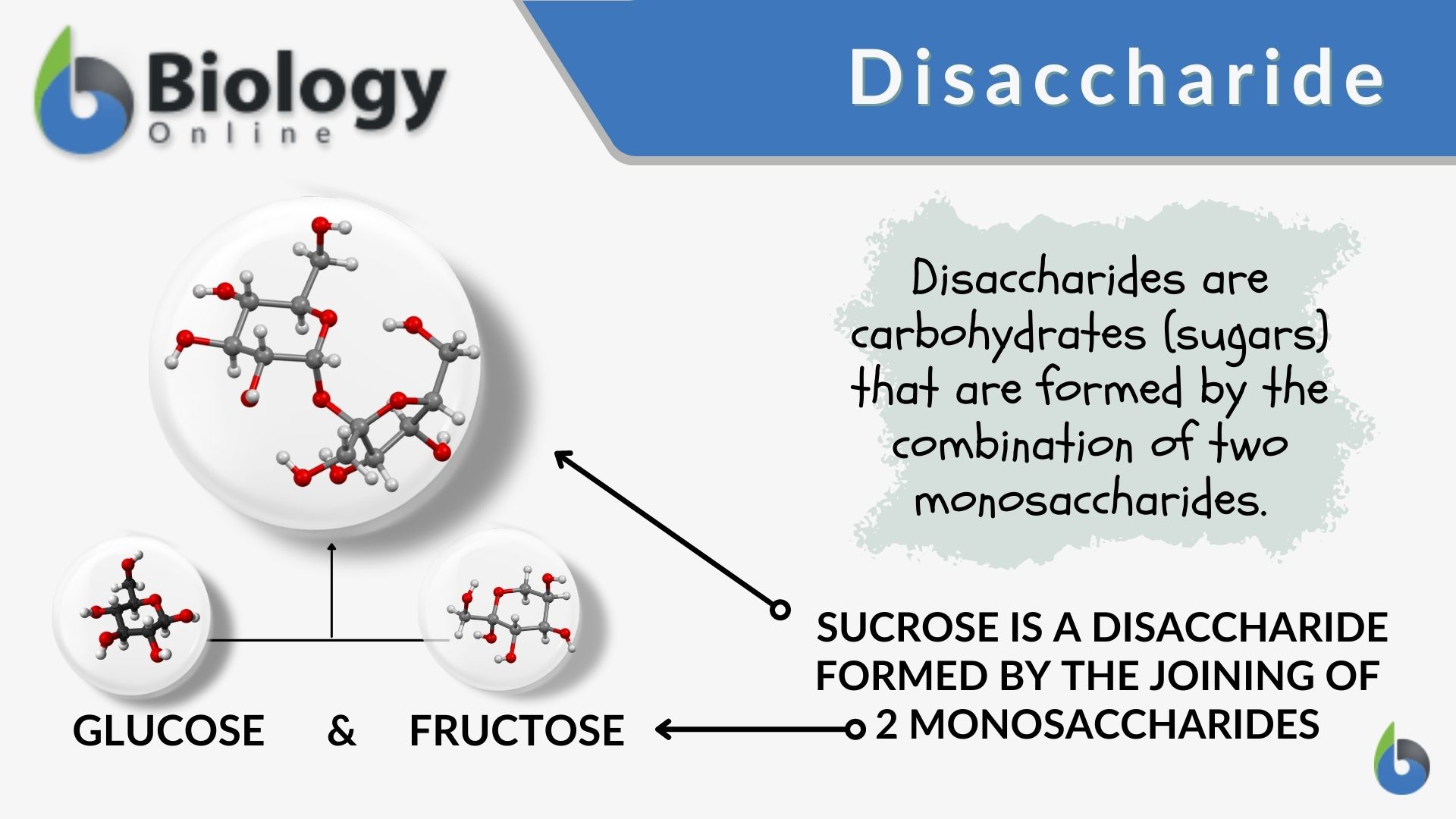
Disaccharide
A sugar molecule consisting of two monosaccharides joined together by a glycosidic bond.
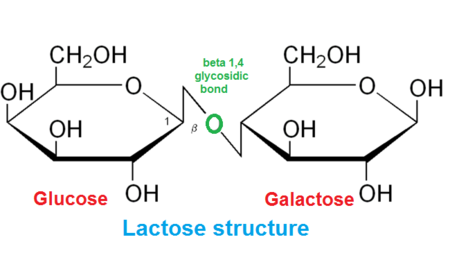
Glycosidic bond
A covalent C−O−C link between two sugar molecules formed by a condensation reaction.
Benedict's test
A test for reducing sugars where a substance is heated with Benedict's reagent, changing from blue to a yellow, red, or brown precipitate.

Polysaccharide
A polymer made by joining many monosaccharide molecules by condensation, such as starch, glycogen, and cellulose.

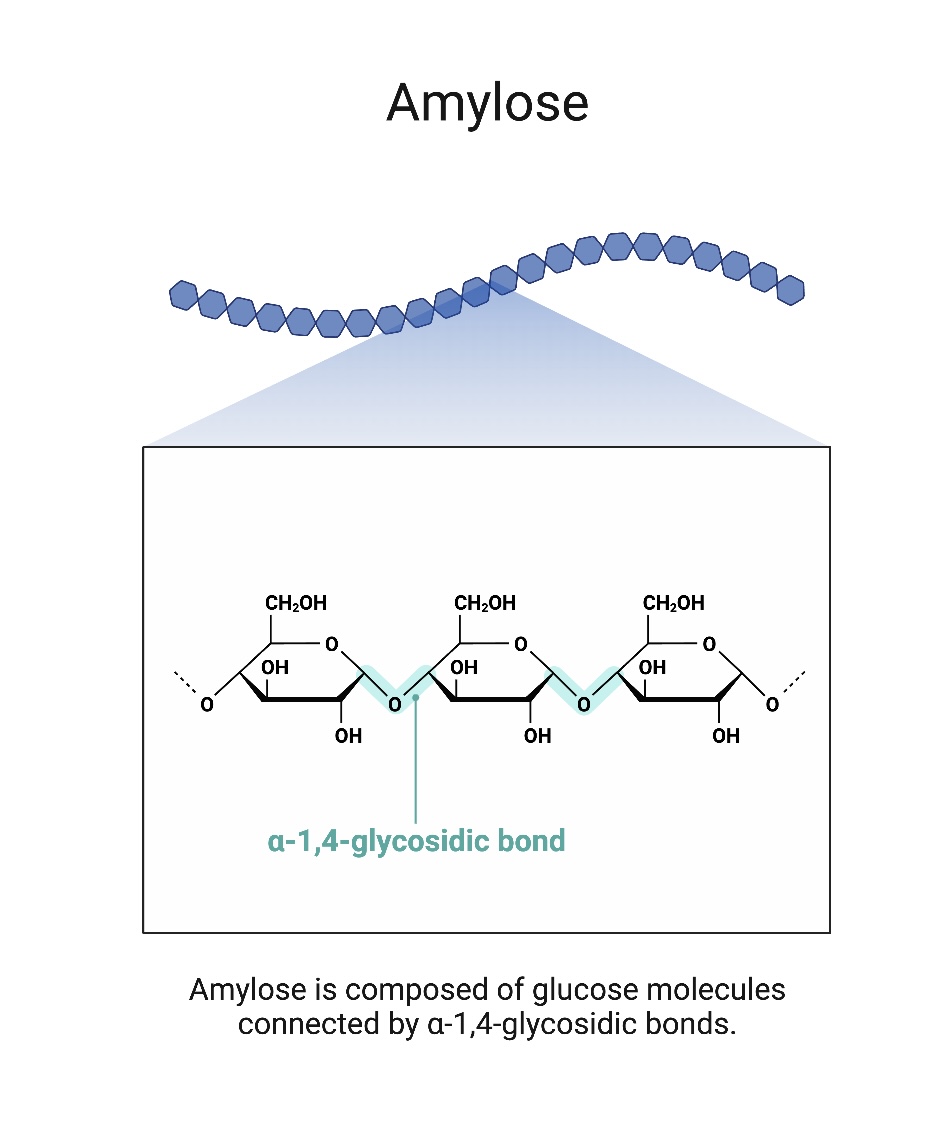
Amylose
A component of starch made of unbranching chains of several thousand 1,4 linked α-glucose molecules that coil into helical structures.
Amylopectin
A component of starch made of shorter 1,4 linked α-glucose chains with side branches formed by 1,6 linkages.
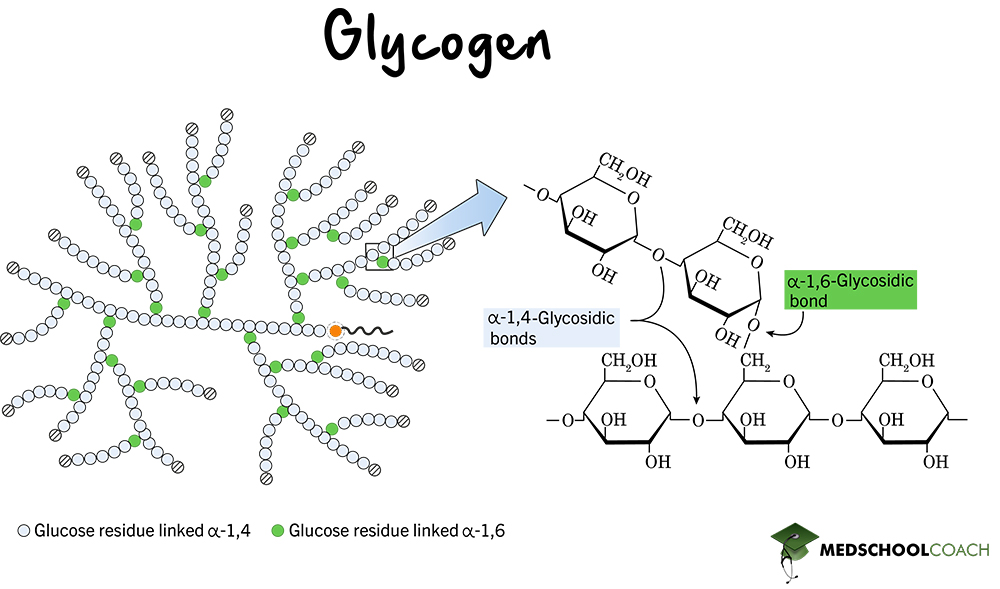
Glycogen
A storage polysaccharide in animals made of α-glucose chains with many 1,6 linkages, resulting in a highly branched structure.
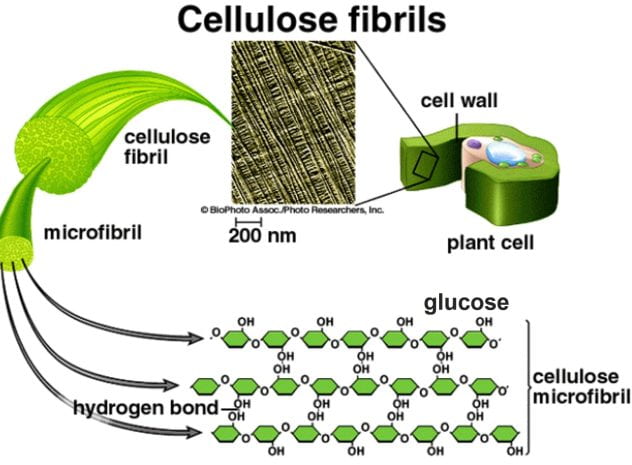
Cellulose
A polysaccharide made from β-glucose subunits that acts as a strengthening material in plant cell walls.
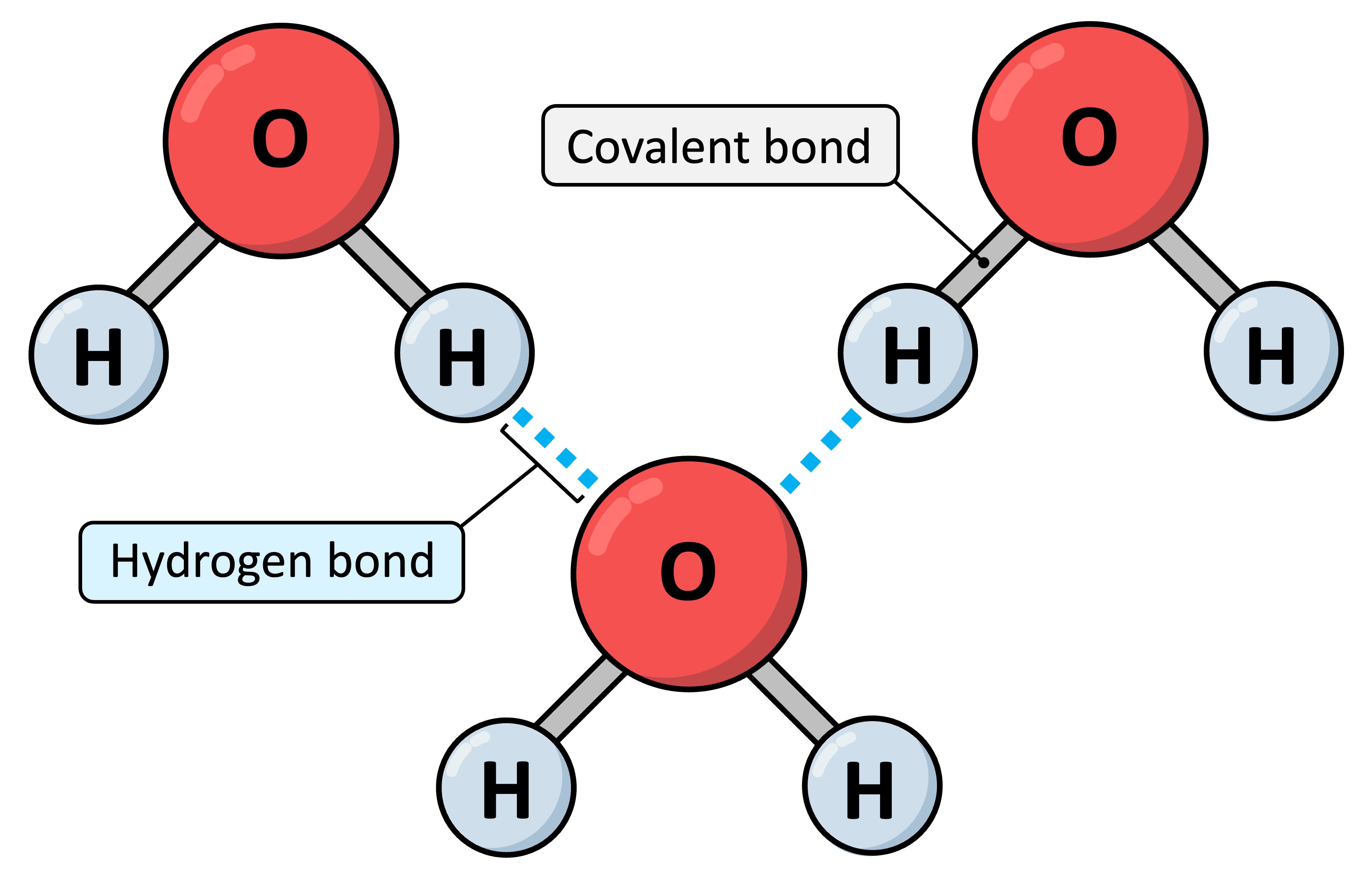
Hydrogen bond
A weak bond formed by the attraction between a group with a small positive charge (δ+) on a hydrogen atom and a group with a small negative charge (δ−).
Microfibrils
Bundles of 60 to 70 cellulose molecules tightly cross-linked by hydrogen bonding.
Dipole
An unequal distribution of charge in a molecule, leading to positively and negatively charged regions.
Hydrophilic
Water-loving polar molecules that are attracted to water and tend to be soluble.
Hydrophobic
Water-hating non-polar molecules that are not attracted to water and are insoluble.
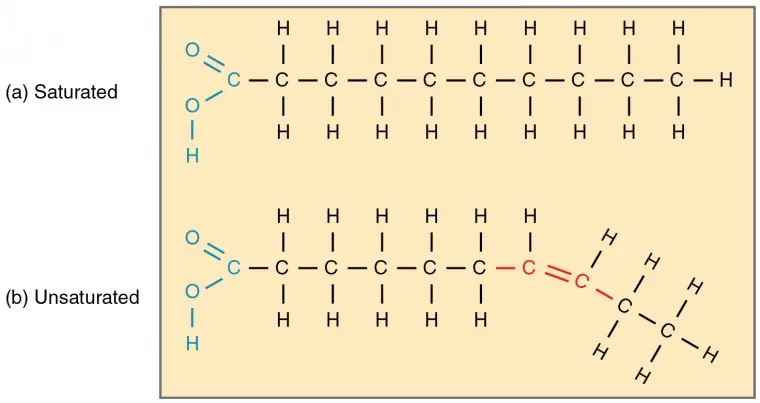
Fatty acids
Molecules containing a carboxyl group (−COOH) 'head' and a long hydrocarbon tail.
Ester bond
A chemical bond, represented as −COO−, formed when the carboxyl group of an acid reacts with the hydroxyl group of an alcohol.
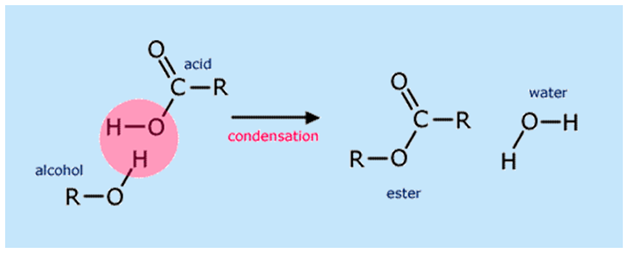
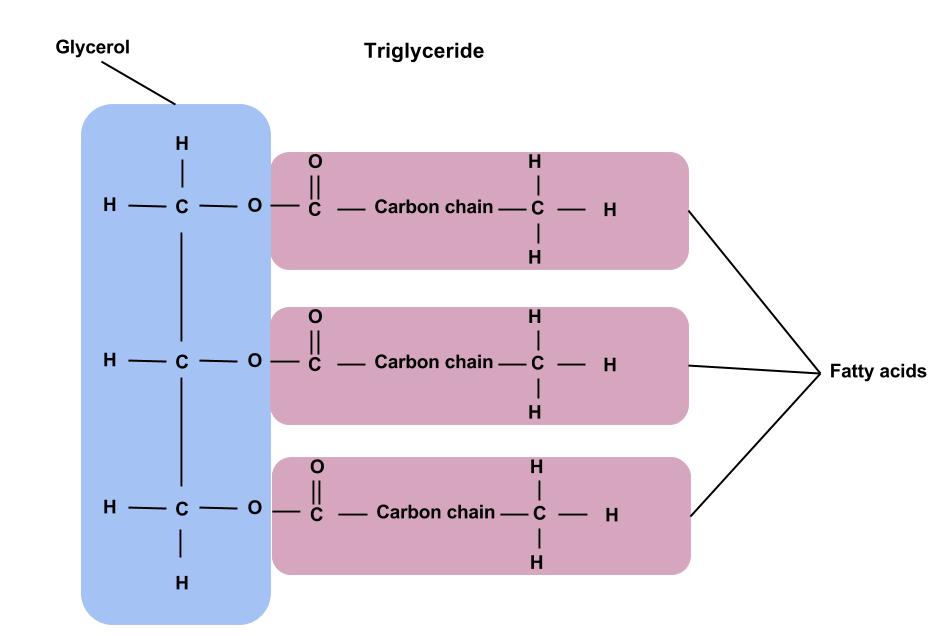
Triglyceride
A lipid formed when three fatty acid molecules combine with one glycerol molecule through condensation reactions.
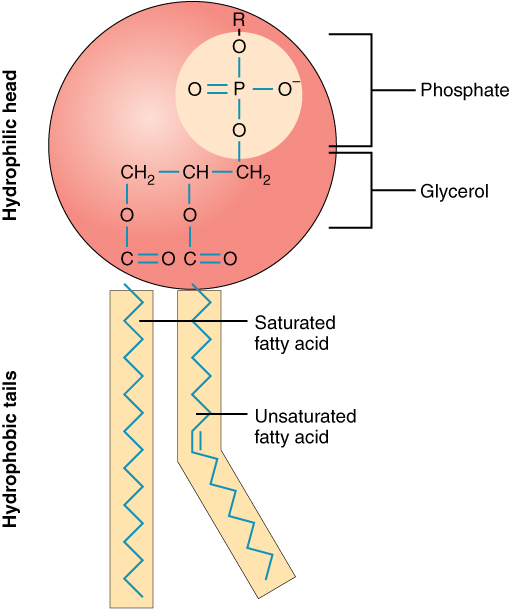
Phospholipid
A lipid where one fatty acid is replaced by a polar phosphate group, creating a molecule with a hydrophilic head and two hydrophobic tails.
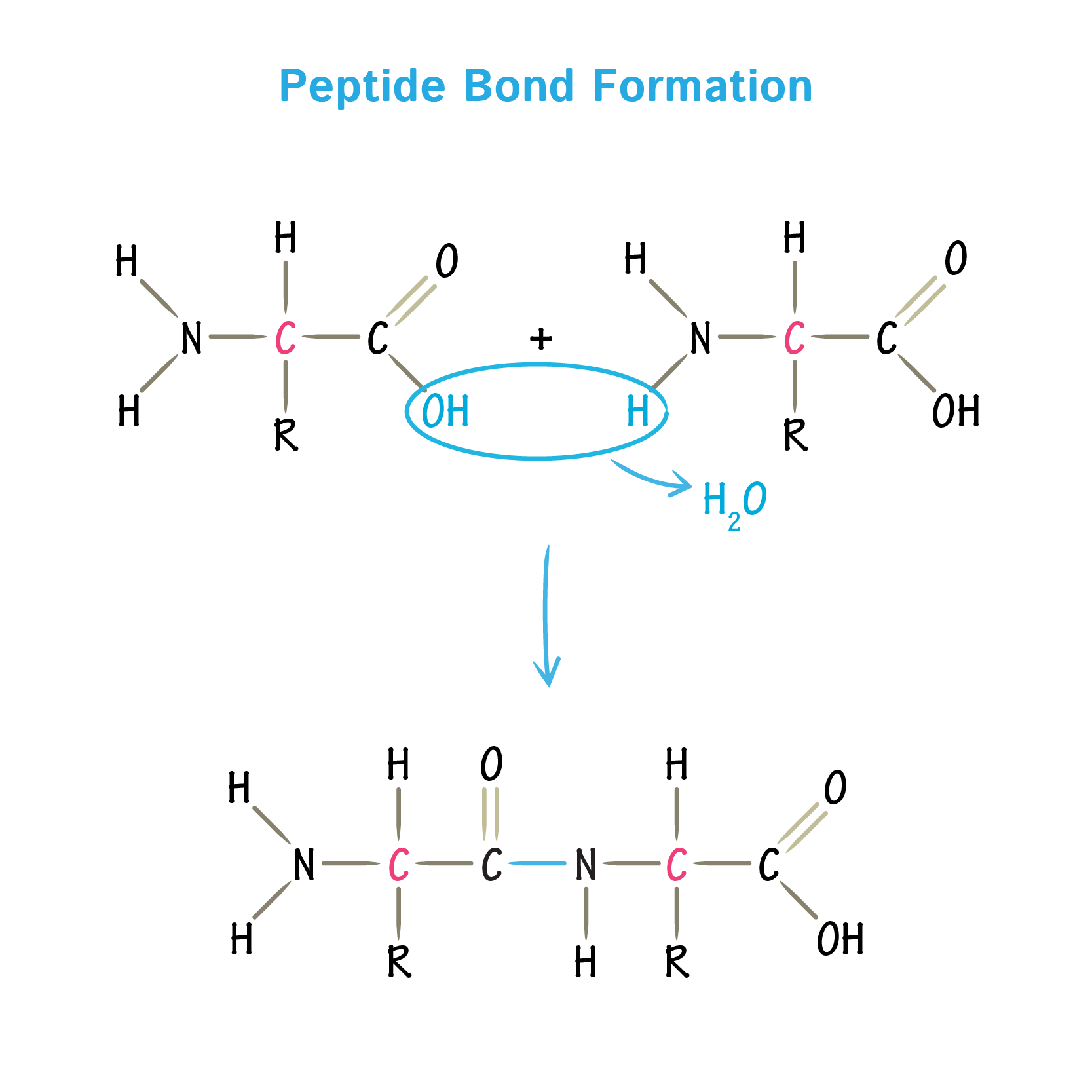
Peptide bond
A covalent C−N link joining neighbouring amino acids together in proteins, formed by a condensation reaction.
Primary structure
The specific sequence of amino acids in a polypeptide or protein.
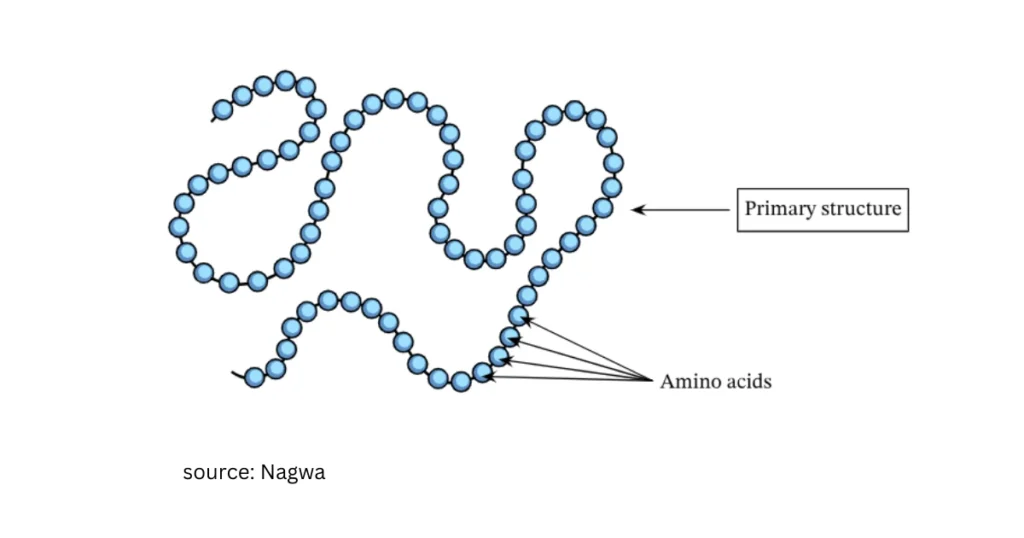
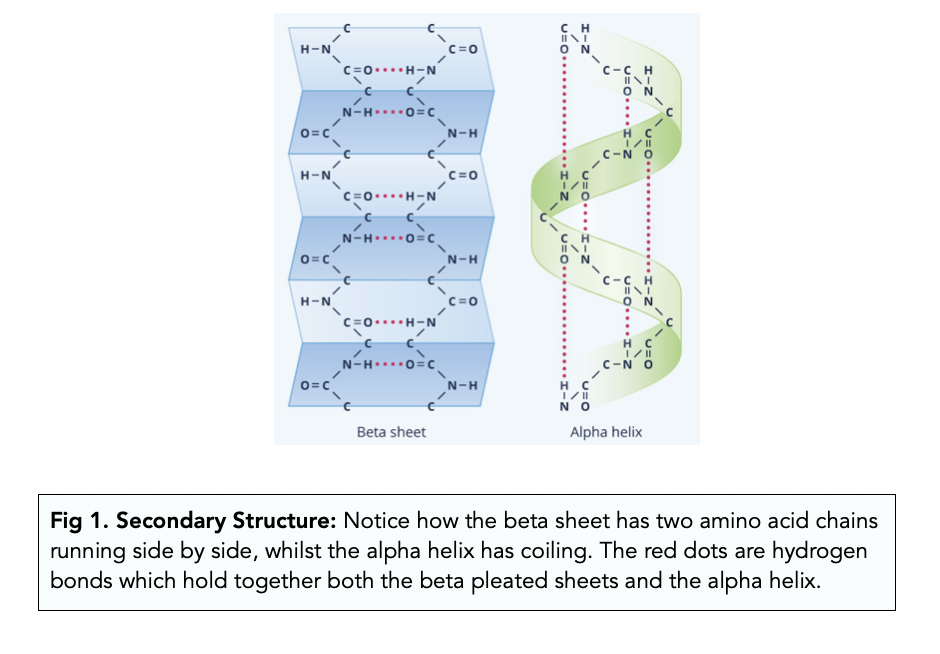
Secondary structure
The regular coiling or folding of an amino acid chain, such as an α-helix or β-pleated sheet, held by hydrogen bonds.
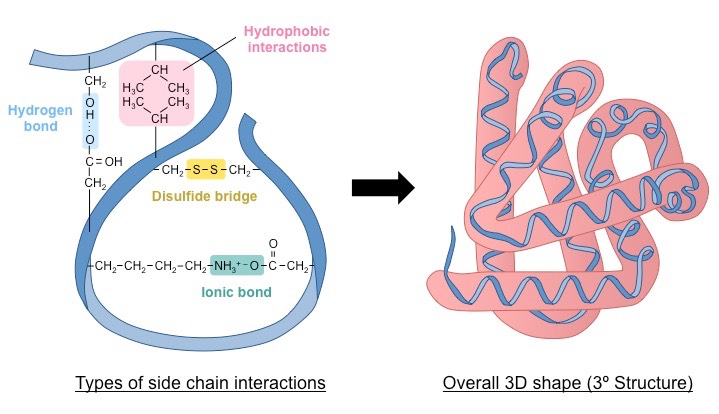
Tertiary structure
The compact three-dimensional coiling of a polypeptide chain held by hydrogen bonds, disulfide bonds, ionic bonds, and hydrophobic interactions.
Quaternary structure
The overall structure formed by the arrangement of two or more polypeptide chains in a protein molecule.
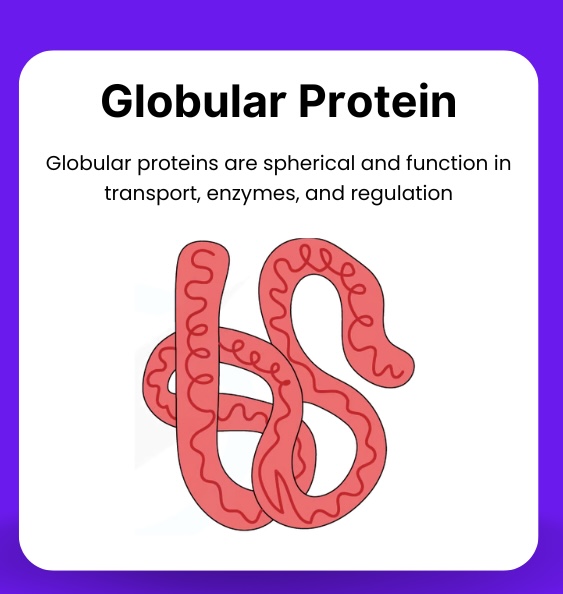
Globular protein
A protein folded into a relatively spherical shape, usually water-soluble and metabolically active, like enzymes and haemoglobin.
Fibrous protein
A protein with a long, thin structure that is generally insoluble and performs structural roles, like keratin and collagen.

Prosthetic group
A non-protein component that is a permanent part of a protein molecule, such as the iron-containing haem group in haemoglobin.
Biuret test
A test for the presence of protein where the reagent turns from pale blue to purple in the presence of peptide bonds.

Specific heat capacity
The amount of heat energy required to raise the temperature of 1kg of a substance by 1∘C.
Latent heat of vaporisation
A measure of the heat energy needed to change a liquid into a gas.