13. Heating products
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Why do we heat our food?
To kill bacteria
To make it safer to store
To detoxify lectins
To change its texture and taste
To make it easier to digest
What does pyrolysis/heating of fat create?
PAHS
Furans
Acrolein
MCPD
Glycidyl esters
What does pyrolysis/heating of proteins create?
HAAs
N-nitrosos compounds
What does pyrolysis/heating of carbohydrates create?
Acrylamide
Furans
Polycyclic aromatic hydrocarbons (PAHs)
Genotoxic carcinogens formed during incomplete combustion of organic matter
Mainly found in smoke, also cigarette smoke (therefore smokers have higher chance of cancer)
Bay region theory: PAHs with bay regions are carcinogenic. (see picture)
It becomes dangerous when metabolized in the liver, leading to liver tumors.
Metabolization = more reactive, they are electrophilic and react with EVERYTHING
PAH and bbq
PAHs formed in food during pyrolysis of fat, e.g. when fat comes in contact with open fire
Fat falls apart in open carbon chains which recondense in the smoke to generate PAHs
PAH sources on BBQ:
Pyrolysis of fat in food product
Combustion of charcoal/wood
Smoke of fat drips on charcoal/wood
MOEs for average consumers and high consumers PAH
MOEs for average consumers: 17500 for PAH4
MOEs for high-level consumers: 9900 for PAH4
Generally not a concern, but approaching concern for high consumers, especially of smoked or grilled foods → regulatory limits
Furans
Formed through several thermal degradation pathways, involving polyunsaturated lipids, ascorbic acid, carbohydrates, and Maillard intermediates
Found in many food products
Toxicity of Furans
Reactive metabolite: cis-2-butene-1,4-dial forms DNA adducts
High incidence hepatocellular tumours in rodents
Possible epigenetic mechanism of carcinogenesis through phase II metabolism
Maillard Reaction
Reaction between reducing sugars and amino acids during heating > 120 C in low-moisture conditions (typical of frying, baking and roasting)
Browning, characteristic flavors in baked and roasted products
Formation of acrylamide, heterocyclic amines (HCAs, incl. IQ, meIQ, PhIP), AGEs
In what foods is acrylamide mostly found?
Potato chips
French fries
Biscuits, cakes
Coffee powder
Crisp bread
Breakfast cereals
At what temperature does acrylamide form?
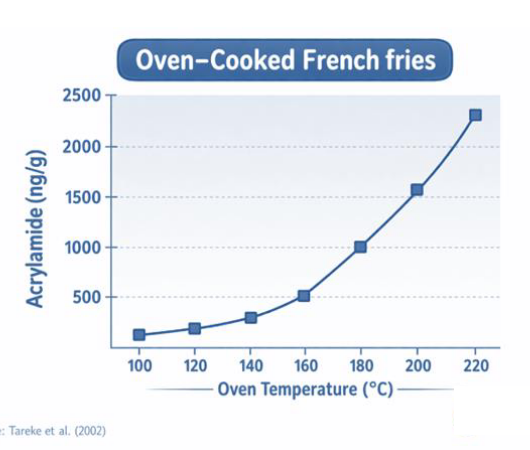
Acrylamide in potato products?
Asparagine nitrogen reservoir for acrylamide in potatoes
Levels of reducing sugars is limiting factor
Fresh potatoes have low levels of reducing sugars
Reducing sugar levels increase during storage, if stored below 8 C or if potato becomes green
Acrylamide toxicity
It is an IARC group 2A carcinogen
This means that it is probably carcinogenic to humans, based on incidences
Vegetable oils
Free fatty acids formed when food containing fats, especially vegetable oils, are subjected to high-temperature processing methods such as refining, deodorization, and baking
Main components formed upon heating:
MCPD esters = heat + glyceries + chloride sources
Glycidyl esters = heat + glycerides, @ harsher deoderization conditions
3-MCPD: non-genotoxic carcinogen (vegetable oils)
Indirect mechanism (whole mechanism not yet clear)
We are definitely exposed more than the tolerable daily intake
Information about 2-MCPD is not yet available

Glycidyl esters: genotoxic carcinogen
Particularly formed in refined oils
