Chemistry final
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
Calculate the mass of a cube of platinum of edge length 4.00 mm (density = 21.4 g/cm3).
1.37 g
This calculation should include how many significant digits?
1.80 + (2.1 × 0.5896) =
2
Dioxin is a highly toxic compound that has been shown to increase the risk of cancer in humans. The U.S Environmental Protection Agency limits the levels of dioxin in drinking water to a maximum of 30 ng/L. If a 1500 mL sample of water contains 1.2×10–8 g of dioxin, is the dioxin concentration in the sample above, below or at the maximum level?
below the maximum level
One gram of gasoline produces 45.0 kJ of energy when burned. Gasoline has a density of 0.77g/mL. Calculate the amount of energy produced by burning 3600 mL of gasoline.
1.2×10^6 kJ
Rolls of aluminum foil are 30.4 cm wide and 0.0016 cm thick. What is the maximum length of
aluminum foil that can be made from 680 g of aluminum? (The density of aluminum is 2.70
g/cm3.)
5200 cm
Which of the following values has exactly 4 significant figures?
1.030
A laboratory instructor gives samples of a particular metal to each of four students, I, II, III, and IV, and they each weigh their sample three times. Their masses for the three trials are:
Student I: 8.40 g, 8.40 g, 8.40 g
Student II: 8.38 g, 8.42 g, 8.40 g
Student III: 8.56 g, 8.66 g, 8.91 g
Student IV: 8.03 g, 8.03 g, 8.04 g
Given that the true mass of each of the metal samples is 8.40 g, which student’s values demonstrated good accuracy but poor precision?
Student II
One isotope of a metallic element has a mass number of 64 and 34 neutrons in the nucleus. The cation derived from the isotope has 28 electrons. What is the element and what is the charge of the cation?
Zn, 2+
What is the formula of chromium(II) nitrite?
Cr(NO2)2
Which of the following is a correct formula for an ionic compound containing elements from the alkaline earth metal and the halogen groups?
BaCl2
Which is the correct formula for the compound formed between the 6th period alkali metal and the hypobromite ion?
CsBrO
Select the compound that is incorrectly named.
K3PO4 potassium phosphide
If a sample of CaI2 contains 2.45×1020 calcium ions, how many iodide ions does it contain?
4.90×10^20 ions
Humans need to consume 150. μg of I– per day. Usually this happens by ingesting KI, which is 76.45% iodine by mass. How much KI should a person consume in a day to meet the nutritional recommendation of 150. μg I–/day?
196 μg
Two stable isotopes of Ag exist; Ag-107 has a mass of 106.9051 amu and a percent abundance of 51.84%. Determine the atomic mass of the other isotope.
108.909 amu
Calculate the mass of Cr in 550. g of Cr2O3.
376 g
10.0 moles of C10H8 contain which of the following?
4.82×10^25 H atoms
Which statement is incorrect?
A 10.0 g sample of H2O contains the same number of molecules as a 10.0 g sample of H2S.
A compound has the empirical formula CH2O. Which of the following cannot be the molecular formula of this compound?
C4H8O6
In the order given, what are the coefficients in the balanced reaction?
___FeS + ___O2 → ___Fe2O3 + ___SO2
4, 7, 2, 4
Calculate the number of carbon atoms in a 0.750 carat diamond. Diamond is composed of carbon atoms. (1 carat = 200 mg)
7.52×10^21 atoms
Determine the empirical formula for propiolic acid based on its elemental mass percent composition.
C: 51.44%, H: 2.88%, O: 45.68%.
C3H2O2
What mass of AgCl (molar mass = 143.32 g/mol) can be formed in the complete reaction of 24.6 g of Ag2S (molar mass = 247.80 g/mol) with excess HCl?
Ag2S(s) + 2HCl(aq) → 2AgCl(s) + H2S(g)
28.5 g
In a 50.0 g sample of K2SO4, the number of moles of potassium is
0.574 mol
A 14.98 g sample of an element contains 3.71×10^23 atoms. Identify the element.
Mg
What mass of Li3N (molar mass = 34.83 g/mol) can be obtained when 5.88 mol of Li is reacted with 0.900 mol of N2?
6Li(s) + N2(g) → 2Li3N(s)
62.7 g
Lindane has the empirical formula CHCl and a molar mass of 290.8 g/mol. How many total atoms does a molecule of lindane contain?
18
Hydrazine, N2H4, is synthesized according to the following equation:
2NaOH + Cl2 + 2NH3 → N2H4 + 2NaCl + 2H2O
You combine 100. g each of NaOH (molar mass = 40.0 g/mol), Cl2 (molar mass = 71.0 g/mol), and NH3 (molar mass = 17.0 g/mol). When the reaction is complete, which reactant(s) remain(s)?
Cl2 and NH3
The final step in the synthesis of ammonia, NH3(g), occurs according to the reaction:
N2(g) + 3H2(g) → 2NH3(g)
What is the percent yield if 11.2 g of H2(g) (2.02 g/mol) reacts with excess N2(g) (17.04 g/mol) to form 48.2 g of NH3(g)?
76.5%
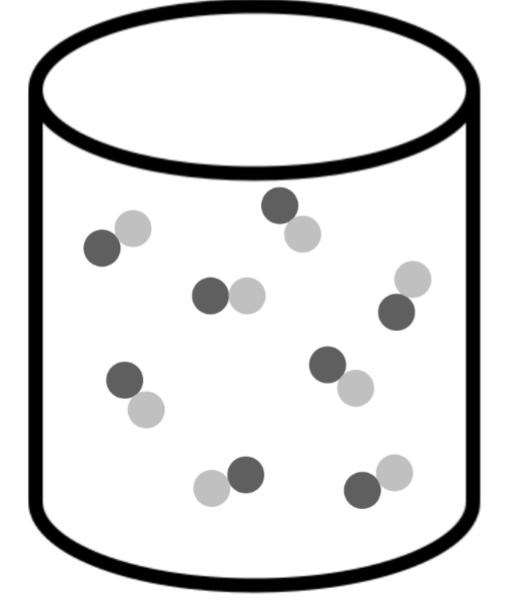
What classification best describes the contents of the container in the drawing?
pure compound
A sample of oxalic acid, H2C2O4 (90.04 g/mol), weighs 1.692 grams. It is placed in a 100. mL volumetric flask and filled with water to the mark on the neck. What is the molarity of the resulting solution?
0.188 M
Calculate the volume of a 1.420 M Ba(OH)2 solution needed to titrate 37.60 mL of a 1.500 M HNO3 solution.
19.86 mL
Find the volume of a 0.202 M HI solution needed to react with 21.0 mL of a 0.398 M KMnO4 solution according to the following equation:
12HI(aq) + 2KMnO4(aq) + 2H2SO4 (aq) → 6I2(s) + Mn2SO4(aq) + K2SO4 (aq) + 8H2O(l)
248 mL
Aqueous solutions of KOH and MgCl2 are mixed. Which of the following lists best represents the resulting mixture?
K+(aq), Cl–(aq), Mg(OH)2(s)
Which of the following processes will result in a precipitation reaction
Mixing a AgNO3 solution with a K2SO4 solution.
An aqueous solution contains 1.5 moles of HCl. How many total moles of ions are in solution?
3.0 mol
What volume of 2.00 M HCl is needed to prepare 0.280 L of a 0.900 M HCl solution?
126 mL
An aqueous barium chloride solution was added to a 15.0 mL sample of a sodium sulfate solution according to the reaction below. If 1.83 grams of solid were recovered, what was the molarity of the original sodium sulfate solution?
Na2SO4 (aq) + BaCl2 (aq) → 2NaCl + BaSO4
0.523 mol/L
What are the spectator ions in the reaction between nitrous acid and sodium hydroxide?
Na+
What happens when you mix an aqueous solution of NaNO3 with an aqueous solution of BaCl2?
There is no reaction. All possible products are soluble.
If 4.39 g AlCl3 (133.33 g/mol) is dissolved in enough water to make exactly 100.0 mL of solution, what is the molar concentration of chloride ion?
0.988 M
Which of the following is both an acid and a weak electrolyte?
H2SO3
Which of these substances when dissolved in water is a strong electrolyte?
NaNO3
Which of the following compounds is not soluble in water
FeCO3
Describe the following reactions as being a precipitation reaction, an acid-base reaction, or neither.
I: Fe(NO3)3(aq) + 3KOH(aq) → Fe(OH)3(s) + 3KNO3(aq)
II: 2Na(s) + Cl2(g) → 2NaCl(s)
III: HNO3(aq) + LiOH(aq) → LiNO3(aq) + H2O(l)
I: precipitation, II: neither, III: acid-base
A 100 g sample of an aqueous solution contains 4.00 g of NH3. The density of this solution is 0.978 g/mL. What is the molarity of NH3 in the solution?
(molar mass of NH3 = 17.03 g/mol)
2.30 mol/L
A balloon is filled with 1.92 g of He and occupies a volume of 12.5 L. What is the balloon’s volume after 0.85 g of He has leaked out though a small hole (assuming constant pressure and temperature)?
6.97 L
How does the density of phosgene gas, COCl2, compare with the density of hydrogen sulfide gas, H2S, in a 1.0 L vessel at the same pressure and temperature?
The density of COCl2 is approximately three times bigger than the density of H2S
Aluminum reacts with chlorine gas to form aluminum chloride.
2Al(s) + 3Cl2(g) → 2 AlCl3(s)
What minimum volume of chlorine gas at 298 K and 0.296 atm is required to completely react with 7.85 g of aluminum?
36.1 L
A mixture of gaseous S2F2, N2F4 and SF4 is placed in an effusion apparatus. Rank the gases in order of increasing effusion rate, slowest to fastest.
rate of SF4 < rate of N2F4 < rate of S2F2
The mole fractions of a gaseous mixture is given below. What is the partial pressure of hydrogen? The mixture is at STP.
Gas: Mole Fraction
N2: 0.786
O2: 0.209
H2O: 0.00046
CO2: 0.00004
H2: ?
3.42 torr
A 1200 mL sample of helium gas is at a pressure of 350 mm Hg and a temperature of 300 K. What volume will this gas sample occupy if the pressure is increased to 700 mm Hg and the temperature is increased to 400 K?
800 mL
Phosphorous (P4) and Oxygen react together to form P4O10 according to the following
reaction: P4(g) + 5O2(g) P4O10(s)
How many moles of P4 react with 40.0 L of O2 at STP?
0.357 mol
What would happen to the average kinetic energy of the molecules of a gas if the temperature of the sample increased from 20 ºC to 40 ºC?
It would increase.
A sample of 35.1 g of methane gas, CH4, has a volume of 5.20 L at a pressure of 2.70 atm. Calculate the temperature.
–195 ºC
Dissolving 4.19 g of an impure sample of calcium carbonate in hydrochloric acid produced 0.974 L of carbon dioxide measured at 293 K and 0.986 atm. Calculate the percent by mass of pure calcium carbonate (molar mass = 100.1 g/mol) in the impure sample.
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) + CO2(g)
95.4 %
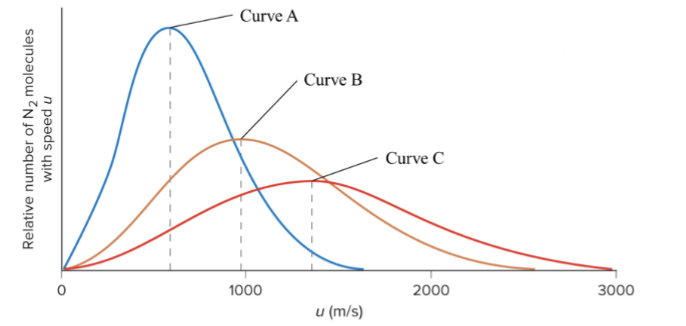
The following graph shows the distribution of molecular speeds for N2 gas at three different temperatures. Which statement is true?
Curve C represents N2 at the highest temperature
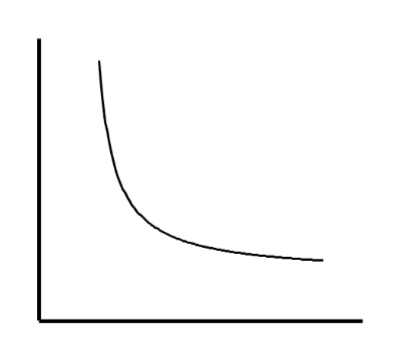
In a sample of an ideal gas, this graph could represent a plot of
P vs. V at a given constant T
At the same temperature and pressure, CH4 effuses
two times as fast as SO2
Given a mixture of gases: 1.00 g He, 14.0 g N2 and 10.0 g NO. What is the total pressure at 27.0 °C if the gases are confined in a 2.00 L container?
13.3 atm