Peptides
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
General structure of amino acids
α‑carbon, amino group, carboxyl group, H, and an R‑group that “differs in each amino acid” .
Are amino acids amphoteric
yes
Amphoteric meaning
can accept or donate protons
How are amino acids amphoteric
ability to donate protons from the carboxyl group and accept protons at the amino group
What pH groups are both functional groups charged?
pH 6.5-7.5
What are zwitterions?
Zwitterions are molecules that contain both positive and negative charges, resulting in no net charge overall.
When are amino acids zwitterions in the pH
physiological pH,
Classes of side chains
non-polar
polar uncharged
charged
non-polar side chains
no charge or electronegative atoms
therefore do not form hydrogen bond
not soluble in water
are typically found in the interior of proteins to help stabilize their structure.
Polar or uncharged/neutral amino acids
have partial charges that can form hydrogen bonds
are soluble in water
Charged amino acids
charged side chains which form hydrogen and ionic bonds
very soluble in water
What is chiral in AA
alpha-carbon
What amino acid is not chiral
glycine is not chiral
What amino acids do proteins only contain
L-amino acids
What configuration to L AA have
L-amino acids have an S configuration except for cysteine, which has an R configuration.
Isoelectric point
the pH at which a molecule carries no net electrical charge,
How are peptides formed
amino acids join via the peptide bond, which is “resonance stabilised” and has partial double‑bond character
how many AA do peptides have
<50
How many AA do proteins have
Typically over 50
How amino acids are separated
Electrophoresis (separation by pI)
Amino acids migrate in an electric field depending on charge.
Chromatography separation by polarity
Ion‑exchange chromatography separates amino acids by charge/polarity.
How is isoelectric point calculated
x
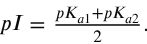
How are peptides bonds resonace stabilised
electrons are shared unequally
therefore, greater electron density on the oxygen
with orbital overlap imparting partial double bond character to the amide bond
Primary Proteins
linear sequences of amino acids linked by peptide bonds, forming the basic structure of proteins.
secondary protein
the folding and twisting of a single polypeptide chain into alpha helices and beta sheets, stabilized by hydrogen bonds between backbone atoms.
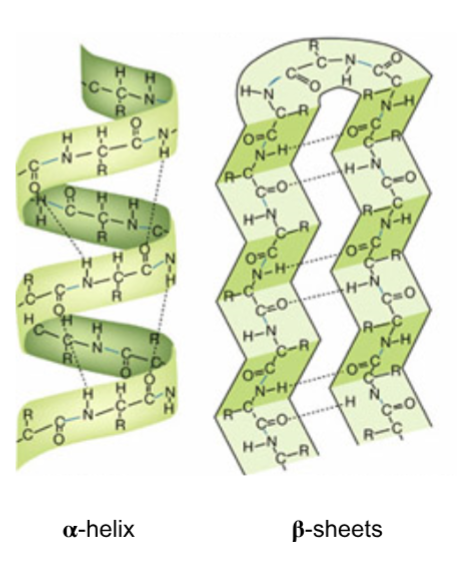
α‑helix
right‑handed coil; H‑bonds between C=O of residue i and NH of i+4
β‑sheets
parallel or antiparallel strands; R‑groups alternate above/below the sheet.
Tertiary structure
the overall 3D shape of a polypeptide, formed by interactions between including hydrogen bonds, ionic bonds, hydrophobic interactions and disulfide bridges.
Quaternary protein
When 2 or more polypeptide chains interact via the same forces as tertiary proteins
what are the 2 categories of proteins
Fibrous and globular proteins.
What is a common quaternary protein
Haemoglobin