15A: principles of transition metal chemistry
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Deduce the electronic configurations of atoms and ions of the d-block elements of period 4, given the atomic number and charge.
Note that copper and chromium have unexpected configurations due to the stability associated with half full and full d subshells.
Note that electrons are removed from the 4s subshell before the 3d subshell as when filled the 4s subshell has a slightly higher energy than the 3d subshell

Definition of a transition metal:
D-block elements that form one or more stable ions with incompletely filled d-sub shells.
Why do transition metals show variable oxidation number?
the 3D subshell and 4s subshell are quite close in energy
Electrons can be removed/lost from both sub shells
Empty 3d orbitals allow electrons to be gained into these (therefore it can be reduced)
Successive ionisation energies steadily increase
Definition of a ligand
small molecule or ion which has a lone pair of electrons which can be used to form a dative bond with a central metal ion
Atom/ion/molecule/species dative covalently bonded to a central metal ion
What type of bonding is involved in the formation of complex ions?
Dative covalent bonding (formed when both of the electrons in the bond come from the same atom)
What is a complex ion?
A central metal ion surrounded by ligands
How does the colour of aqueous ions, and other complex ions, form?
the difference in energy levels between the higher and lower d orbitals determines the wavelength of light absorbed
D orbitals/subshell are/is split in energy by ligands
Light energy / photons are needed for electron promotion, from lower energy d orbital to higher energy d orbital
The colour not absorbed is the colour seen
Why is there a lack of colour in some aqueous ions and other complex ions?
If the wavelength of light absorbed is outside the visible range, the salt is white / solution is colourless
Also some non transition metals have full d orbitals when split therefore electrons cannot be promoted from lower to higher energy d orbitals.
Where do changes in transition metal ion colours arise from?
Changes in:
oxidation number
Ligand
Coordination number
Definition of coordination number:
the number of dative covalent bonds to a central metal ion
Name some monodentate ligands
H2O
OH-
NH3
Monodente ligands = form one coordinate bond to a metal ion
Name some bidentate ligands
en (1,2-diaminoethane)
Ox (ethanedioate ions)
Bidentate ligands form two coordinate bonds to a metal ion
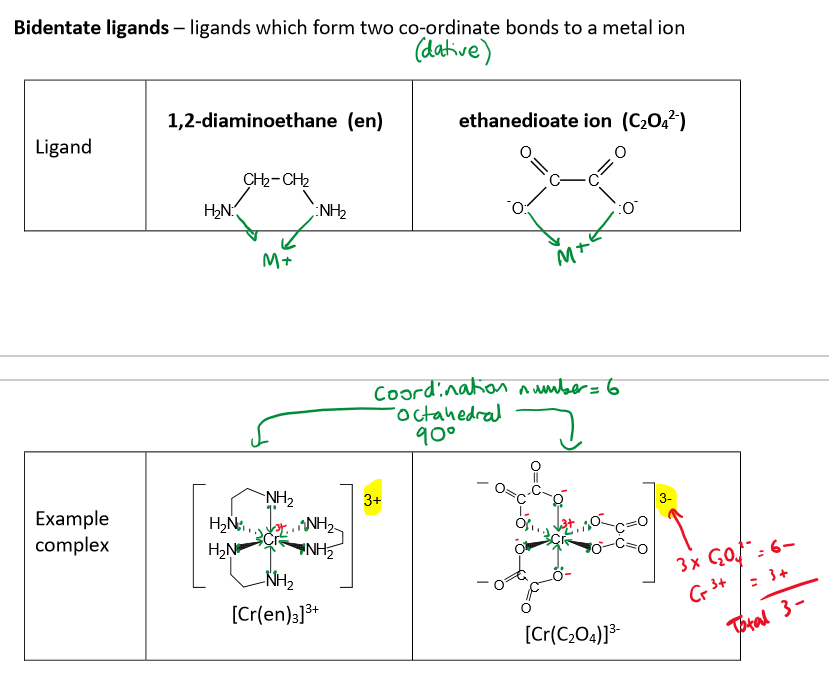
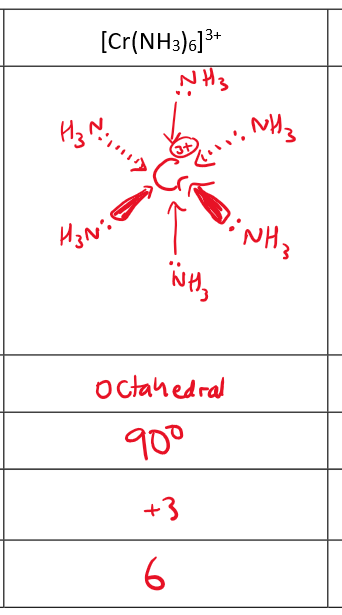
What is the shape of a complex formed with 6 monodentate ligands e.g. H2O, OH- or NH3
octahedral
6-fold coordination
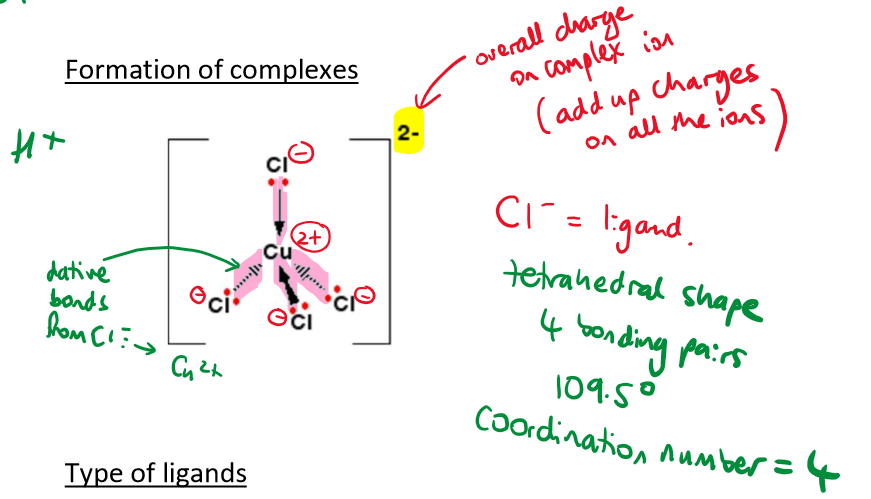
What shape complex will transition metals form with 4 Cl- ligands
Cl- is relatively large
Therefore forms tetrahedral complex
When do square planar complexes form?
4 ligands; 2 large and 2 small
E.g. cis-platin [Pt(Cl)2(NH3)2]
![<p>4 ligands; 2 large and 2 small</p><p>E.g. cis-platin [Pt(Cl)2(NH3)2]</p>](https://assets.knowt.com/user-attachments/8ed60852-de3c-4efb-901c-0251000a4697.png)
Why is cis-plat used in cancer treatment supplied as a single (stereo/geometric) isomer and not in a mixture with the trans form?
Trans form is not active drug and causes side effects
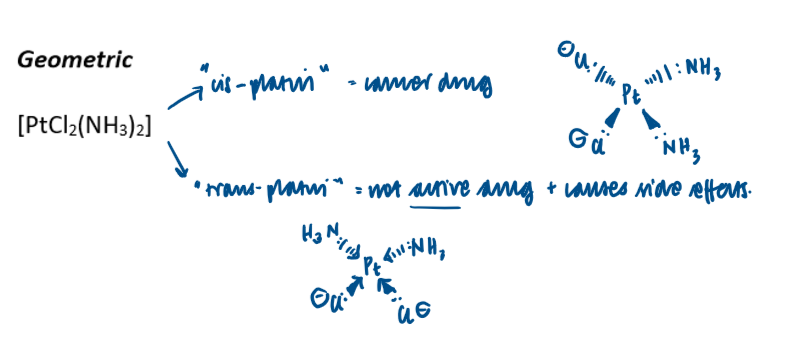
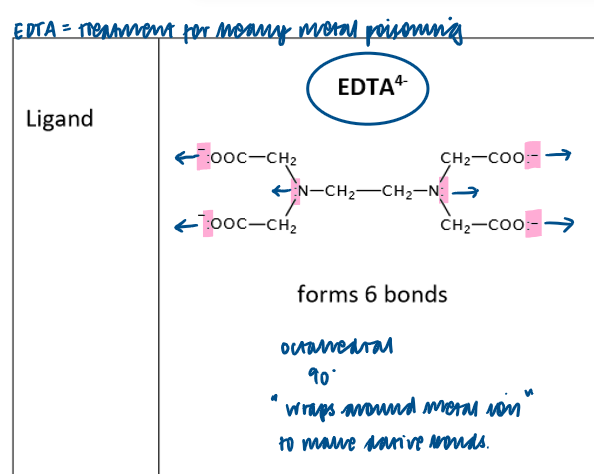
What is an example of a multidentate ligand?
EDTA4-
(Note forms octahedral shaped complex with 6 coordinate bonds- so coordination number 6)
What is haemoglobin an example of?
An iron (II) complex containing a multidentate ligand.
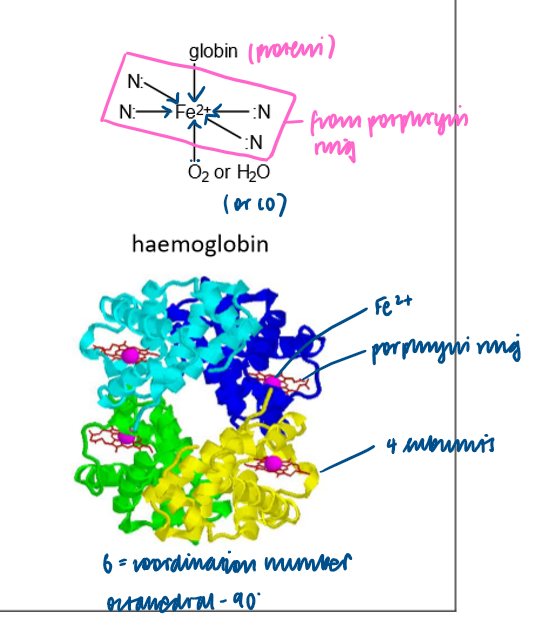
What happens when carbon monoxide is present in bloodstream?
A ligand exchange reaction
An oxygen molecule bound to haemoglobin is replaced by a carbon monoxide molecule
Carbon monoxide binds irreversibly so prevents oxygen from being transported by blood around body.
What are the key properties of transition elements:
can act as catalysts
can make coloured compounds
can have variable oxidation states
can form complexes (with ligands)