C6 : The Rate and Extent of Chemical Change
1/31
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
In simple words what does the rate of chemical reaction mean?
How fast the reactants are changed into products
What is the equations for the mean rate of reaction?
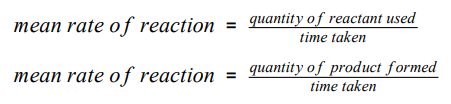
On a graph, when the line is steep what does this tell us about the rate of reaction?
The faster the rate of reaction is
On a graph, when the line is less steep what does this tell us about the rate of reaction + why?
The rate of reaction has slowed because the reactants are being used up
On a graph, when the line is stationary what does this tell us about the rate of reaction + why?
The reaction has stopped because the reactants are used up
What does the collision theory suggest?
Chemical reactions can occur only when reacting particles collide with each other with sufficient energy
What is activation energy?
The minimum amount of energy that particles must have to react
What are the 4 things rate of reaction depends on + how do these 4 methods increase the rate of reaction??
Temperature
Concentration of a solution or pressure
Surface area
Catalysts
They increase the rate of reaction because they increase the number of successful collisions between reacting particles
Explain how an increase in temperature increases the rate of reaction (3)
When the temperature is increased the particles have more energy and move faster
Because they are moving faster there will be more frequent collisions
Also the faster they move, the more energy they exert + that energy fuels the reaction
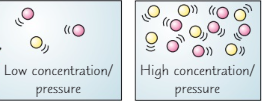
Explain how an increase in concentration or pressure increases the rate of reaction (2)
An increase is concentration means there are more particles in a given volume so more collisions
Similarly when pressure is increased the same number of particles are in a smaller volume so once again more collisions
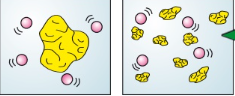
Explain how an increase in surface area increases the rate of reaction (2)
If one of the reactants is a solid, breaking it up will increase its surface area to volume ratio
This means that for the same volume of the solid the particles will have more area to work on so there will be more collisions
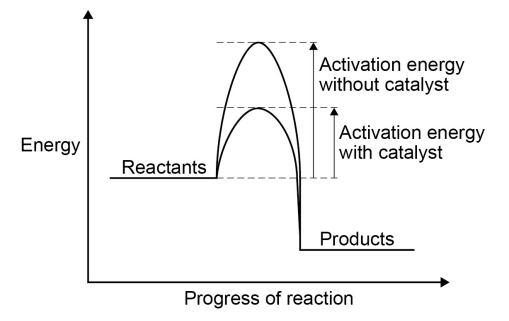
What is a catalyst + how does a catalyst increases the rate of reaction (3)
A catalyst is a substance that speeds up a reaction without being used up in the reaction itself (its not part of the overall reaction)
Different catalysts are needed for different reactions and they all work by decreasing the activation energy needed for the reaction to occur
They do this by providing an alternate reaction pathway with a lower activation energy
What is the fancy way of saying cloudy?
Turbidity
Explain the disappearing cross reaction experiment (9)
Use a measuring cylinder to put 10cm3 of sodium thiosulfate solution into a conical flask
Place the conical flask onto a printed black cross
Next add 10cm3of hydrochloric acid into the conical flask
Swirl the solution and start a stopwatch
Look down through the top of the flask
Stop the stopwatch when the solution has become cloudy and the cross is no longer visible
Carry this experiment out using lower concentrations of sodium thiosulfate
Repeat 3 times and calculate a mean and remove anomalies for each concentration of thiosulfate solution
Plot a graph
Explain how to measure the volume of a gas produced by a reaction (8)
Use a measuring cylinder to place 50cm3 of hydrochloric acid into a conical flask
Attach the conical flask to a bung and delivery tube
Then place the delivery tube into a container filled with water
Place an upturned measuring cylinder also filled with water over the delivery tube
Add a 3cm strip of magnesium to the hydrochloric acid and start a stopwatch
The reaction produces hydrogen gas which is trapped in the measuring cylinder
Every 10 seconds, measure the volume of the hydrogen gas in the measuring cylinder + continue until no more hydrogen is given off
Repeat the experiment with different concentrations of hydrochloric acid
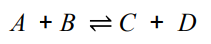
What is meant by a reversible reactions?
When the products of the reactants can react to produce the original reactants
(Forward reaction and backward reaction)
What is meant by equilibrium?
When the forward and reverse reactions occur at exactly the same rate in a closed system
As reactants react what happens to their concentrations?
It decreases
Explain how an equilibrium is achieved (4)
As the reactants react, their concentrations fall so the forwards reaction will slow down
But as more and more products are made their concentrations will rise and the backward reaction will speed up
After a while the rate of the forward reaction will be the same as the rate of the backward reaction
The system is now at equilibrium
When does equilibrium take place?
When a reversible reaction takes place in a closed system
Why is one reaction exothermic + the other endothermic in a reversible reaction?
Because the energy absorbed from the surroundings by the endothermic reaction is equal to the energy transferred to the surrounding by the exothermic reaction
What does it mean if the equilibrium lies to the right? (in terms of concentration)
The concentration of the products is greater than the concentration of the reactants
What does it mean if the equilibrium lies to the left? (in terms of concentration)
The concentration of the reactants is greater than the concentration of the products
What 3 things does the position of equilibrium depend on?
Temperature
Pressure (effects gases only)
Concentration
What is Le Chatelier’s Principle?
If the conditions of a reversible reaction are changed then the position of equilibrium will shift to try to counteract the change
How does a temperature increase effect the equilibrium + how does this effect the amount of products? (3)
The equilibrium shifts to the endothermic direction to try to decrease the heat so
the amount of products increases for an endothermic reaction
the amount of products decreases for an exothermic reaction
How does a temperature decrease effect the equilibrium + how does this effect the amount of products? (3)
The equilibrium shifts to the exothermic direction to produce more heat so
The amount of products decreases for an endothermic reaction
The amount of products increases for an exothermic reaction
What does a pressure increase cause the equilibrium to do?
An increase in pressure causes the equilibrium position to shift towards the direction of the smaller number of molecules
What does a pressure decrease cause the equilibrium to do?
A decrease in pressure causes the equilibrium position to shift towards the direction of greater number of molecules
If the concentration of one of the reactants or products is changed what happens to the system in terms of equilibrium?
The system is no longer at an equilibrium
How does a concentration increase of reactants effect the equilibrium + how is equilibrium reached again?
If the concentration of a reactant is increased, the equilibrium will shift to the left + reactants will react to form more products until equilibrium is reached again
How does a concentration decrease of reactants effect the equilibrium + how is equilibrium reached again?
If the concentration of a reactant is decreased, the equilibrium shifts to the right + more products will react until equilibrium is reached again