DSA Neuro (CS, Medulla, MB, BS interactions) Pons in atlas + need synaptic transmission
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Syringomyelia
Cavitation of the central regions of the spinal cord, as in a small syringomyelia, will frequently damage fibers crossing in the anterior white commissure ( Fig. 9.3 ). This bundle conveys fibers from the posterior horn across the midline to enter the ALS on the opposite side ( Fig. 9.6 ). Consequently, a lesion of this structure will damage fibers coursing in both directions, resulting in a bilateral loss of pain and thermal sensations that correlate with the damaged levels of the spinal cord. For example, if the lesion is in mid to low cervical levels, the pain and thermal sensory deficits will fall over the shoulders and arm in a “cape distribution.” A large syrinx that involves the anterior white commissure and extends into the anterior horn results in a bilateral sensory loss, as noted earlier, and weakness of the corresponding extremity. Because these lesions are usually in the cervical levels, extension of the syrinx into one anterior horn results in an ipsilateral weakness of the upper extremity; if both anterior horns are involved, the weakness is bilateral. In syringomyelia, the cavity that develops in central areas of the spinal cord does not have a lining of ependymal cells and therefore is not an enlargement of the central canal. This is sometimes called a noncommunicating syringomyelia to differentiate it from a cystic structure that may connect with the central canal ( communicating syringomyelia ). On the other hand, a cavitation of the central canal is called a hydromyelia (or hydrosyringomyelia ). Syringomyelia is commonly seen in patients with Chiari malformations in the posterior fossa but may also be a consequence of trauma to the spinal cord, tumors, and infections. The symptoms are highly variable but most frequently include loss of pain and temperature sensations, extremity weakness, and unsteady gait.
Brown-Séquard Syndrome
A functional hemisection of the spinal cord results in a clinical picture that reflects damage to the lateral corticospinal tract, the ALS, and the posterior columns (a Brown-Séquard syndrome ). A lesion on the right at C4 to C5 will result in muscle weakness or paralysis ( hemiparesis, hemiplegia ) on the right side (corticospinal damage); loss of pain and thermal sensations on the left side (ALS damage; these fibers cross in the anterior white commissure); and loss of proprioception, vibratory sense, and discriminative touch on the right (gracile and cuneate fasciculi injury; these fibers are not crossed in the spinal cord). These lesions are frequently called functional hemisections in recognition of the fact that the cord is not perfectly cut halfway across but may be injured or deformed by, for example, pieces of a damaged vertebra. The net result is a loss of function on half of the spinal cord.
High Cervical Cord Lesion
Injury to high cervical levels of the spinal cord is, in general, a catastrophic event. In addition to the potential for a total loss of sensation for the body below the lesion and of voluntary motor control below the lesion, there is another important complicating factor. The phrenic nucleus is located in the central regions of the anterior horn at levels C3 to C6. This cell group innervates the diaphragm and in high cervical lesions is disconnected from the centers of the medulla that control breathing. Consequently, in patients with high cervical lesions, preservation of the ability to breathe becomes a major factor in care.
Acute Central Cervical Spinal Cord Syndrome
The acute central cervical spinal cord syndrome, commonly called the central cord syndrome, is an incomplete spinal cord injury. This may result from hyperextension of the neck (sometimes in a patient with bone spurs on the vertebrae) that momentarily occludes blood supply to the cord via the anterior spinal artery. Consequently, the deficits reflect the territory served by the branches of this vessel. The results are bilateral weakness of the extremities (more so of the upper than of the lower), varying degrees and patterns of pain and thermal sensation loss, and bladder dysfunction. Many of these patients recover most or all of their function within 4 to 6 days. In general, function of the lower extremities returns first, bladder function next, and function of the upper extremities last. Pain and thermal sensations may return at any time, and posterior column sensations are not affected in these patients.
Variations on these main themes may occur. For example, a spinal cord hemisection at T8 would affect the body below that level but would spare the upper trunk and upper extremity. A lesion involving the posterior columns bilaterally would result in proprioceptive and discriminative touch losses below the level of the lesion but would spare pain and thermal sensations. In our study of systems neurobiology, we shall explore these and other examples of dysfunction resulting from spinal cord lesions.
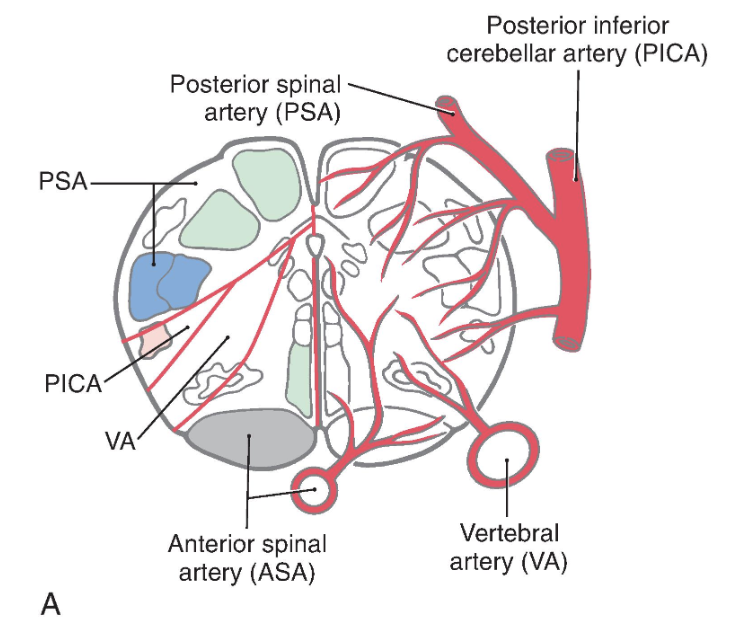
Identify each of the vascular territories in the illustrations.
Identify the motor and sensory nuclei found within the territories served by each vessel.
Identify the motor and sensory tracts, or fiber bundles, found within the territories served by
each vessel. Describe the location of the cells of origin and the site(s) of termination for these
tracts/fibers. Be sure to specify laterality.
Determine what motor and sensory deficits (if any) would result from occlusion of the vessels
supplying the medulla oblongata. See text for help
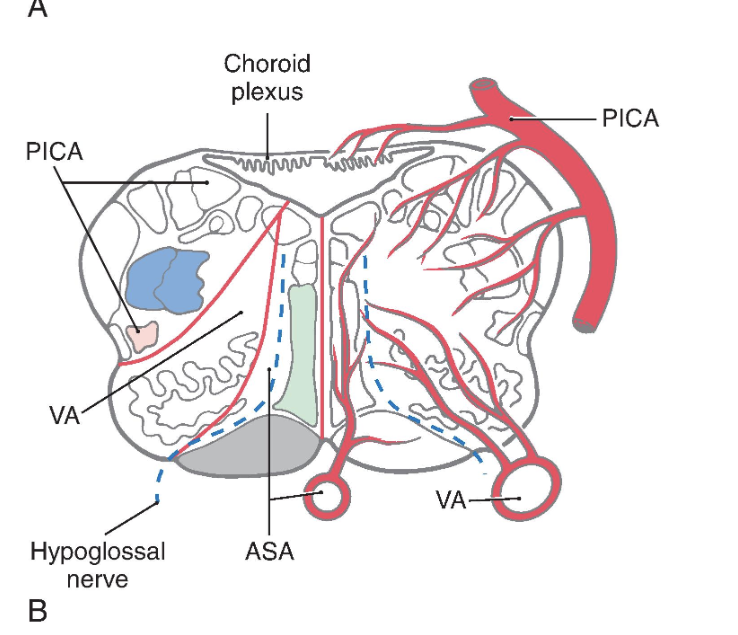
Identify each of the vascular territories in the illustrations.
Identify the motor and sensory nuclei found within the territories served by each vessel.
Identify the motor and sensory tracts, or fiber bundles, found within the territories served by
each vessel. Describe the location of the cells of origin and the site(s) of termination for these
tracts/fibers. Be sure to specify laterality.
Determine what motor and sensory deficits (if any) would result from occlusion of the vessels
supplying the medulla oblongata. See text for help
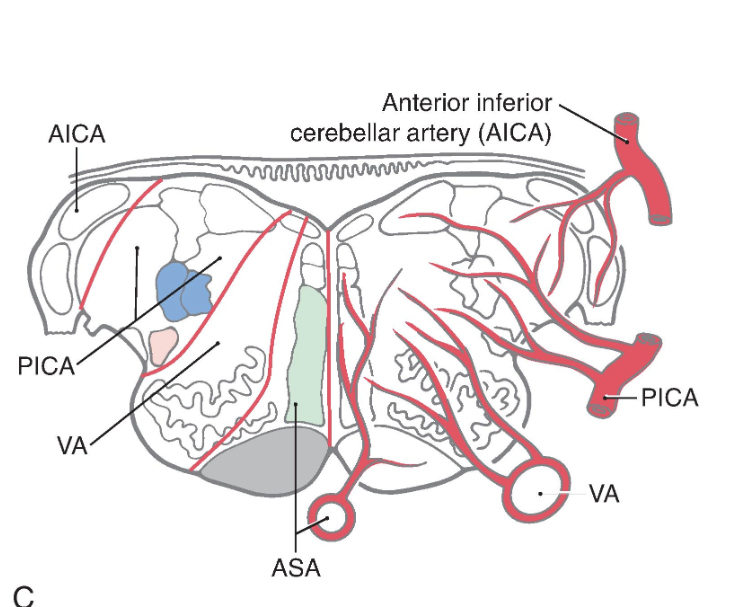
Identify each of the vascular territories in the illustrations.
Identify the motor and sensory nuclei found within the territories served by each vessel.
Identify the motor and sensory tracts, or fiber bundles, found within the territories served by
each vessel. Describe the location of the cells of origin and the site(s) of termination for these
tracts/fibers. Be sure to specify laterality.
Determine what motor and sensory deficits (if any) would result from occlusion of the vessels
supplying the medulla oblongata. See text for help
medial medullary syndrome ( Dejerine syndrome )
Medial structures of the medulla at all levels, including the pyramid, medial lemniscus, and hypoglossal nucleus and roots, are served by penetrating branches of the anterior spinal artery ( Fig. 11.16 ). The branches of the anterior spinal artery that penetrate into medial portions of the medulla tend to alternate to the right and left. Occlusion of these penetrating branches to one side of the medial medulla may result in a pattern of deficits characteristic of the medial medullary syndrome ( Dejerine syndrome ). The deficits and corresponding structures damaged in this syndrome include a contralateral hemiparesis (pyramidal and corticospinal damage), a contralateral loss of proprioception and vibratory sense (medial lemniscus), and a deviation of the tongue to the ipsilateral side when it is protruded (hypoglossal root or nucleus injury). On the other hand, occlusion of the anterior spinal artery may result in bilateral deficits reflecting damage to both pyramids, both medial lemnisci, and both of the hypoglossal nuclei or their exiting roots.
The posterior medulla caudal to the obex is served by branches of the posterior spinal artery ( Fig. 11.16A ). Major structures in this area include the posterior column (gracile and cuneate) nuclei and the spinal trigeminal tract and nucleus. Although vascular lesions of the posterior spinal artery are rare, they may produce an ipsilateral loss of proprioception and vibratory sense on the body (damage to posterior columns and nuclei) coupled with an ipsilateral loss of pain and temperature sensation from the face (spinal trigeminal tract).
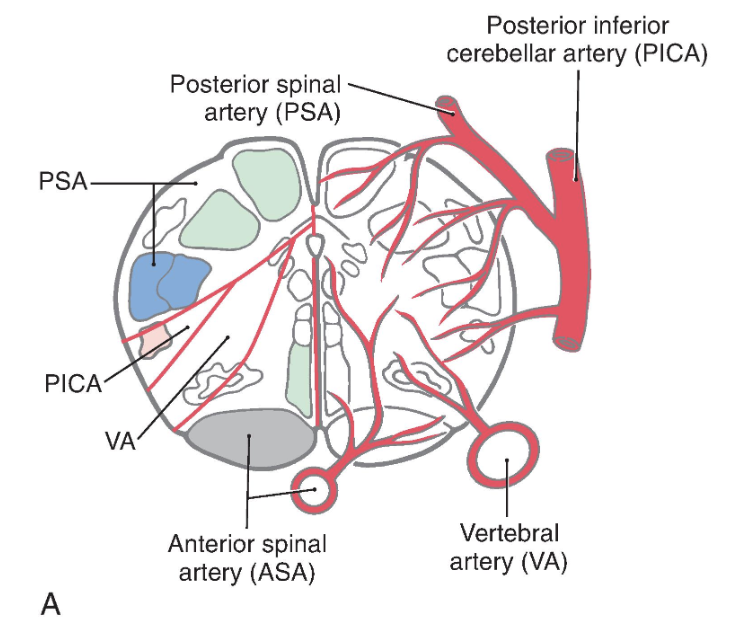
lateral medullary syndrome, PICA syndrome, or Wallenberg syndrome
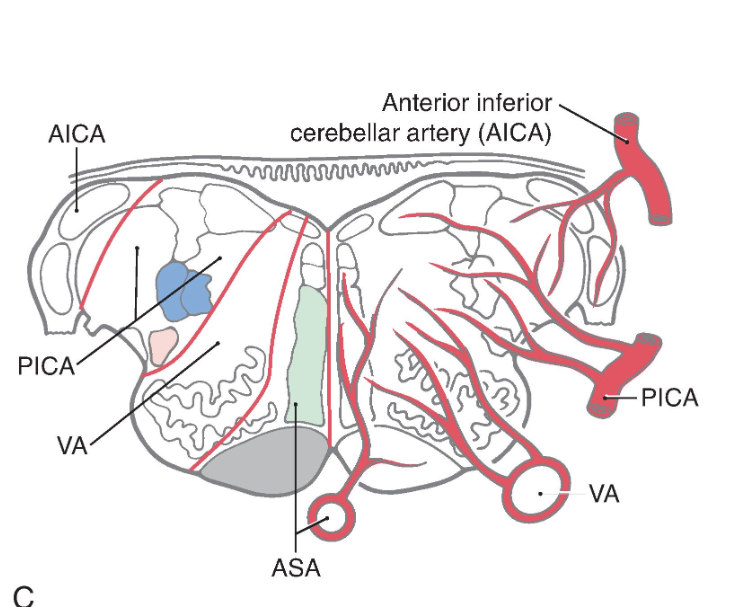
Rostral to the obex, the entire posterolateral medulla is served by branches of the PICA ( Figs. 11.16B , C and 11.17 ). Included in the territory served by this vessel are the anterolateral system, spinal trigeminal tract and nucleus, vestibular nuclei, solitary tract and nucleus, and nucleus ambiguus. Vascular insufficiency of the PICA territory (or blockage of one vertebral artery) gives rise to a characteristic set of sensory and motor deficits commonly called the lateral medullary syndrome, PICA syndrome, or Wallenberg syndrome ( Fig. 11.17 ). The deficits seen and the corresponding structures involved are (1) contralateral loss of pain and temperature sensation from the body (anterolateral system), (2) ipsilateral loss of pain and temperature sensation from the face (spinal trigeminal tract and nucleus), (3) some vertigo and nystagmus (vestibular nuclei), (4) loss of taste from the ipsilateral half of the tongue (solitary tract and nucleus), and (5) hoarseness and dysphagia (nucleus ambiguus or roots of cranial nerves IX and X) ( Fig. 11.17C ). Patients with the lateral medullary syndrome may also have Horner syndrome owing to injury to hypothalamospinal fibers descending through the lateral areas of the medulla. We shall further explore the details of these clinical syndromes in later chapters.
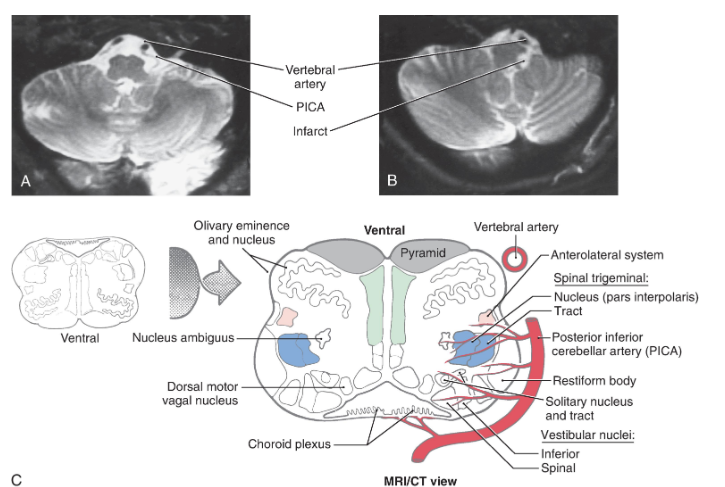
Lateral medullary (Wallenberg) syndrome
Lateral medullary (Wallenberg) syndrome. A normal magnetic resonance image (MRI, T2 weighted) (A) shows the vertebral and posterior inferior cerebellar arteries in relation to the medulla. The patient whose MRI is shown in (B) (MRI, T2 weighted) had an occlusion of the PICA, which resulted in an infarct of the territory of the medulla served by this vessel. The structures damaged in this lesion are shown in (C). Compare with Fig. 11.16 .
In addition to this broad expanse of the medulla, branches of the PICA also serve the choroid plexus of the fourth ventricle. At the pons-medulla junction, the cochlear nuclei and a small adjacent part of the restiform body are served by branches of the anterior inferior cerebellar artery ( Fig. 11.16C ).
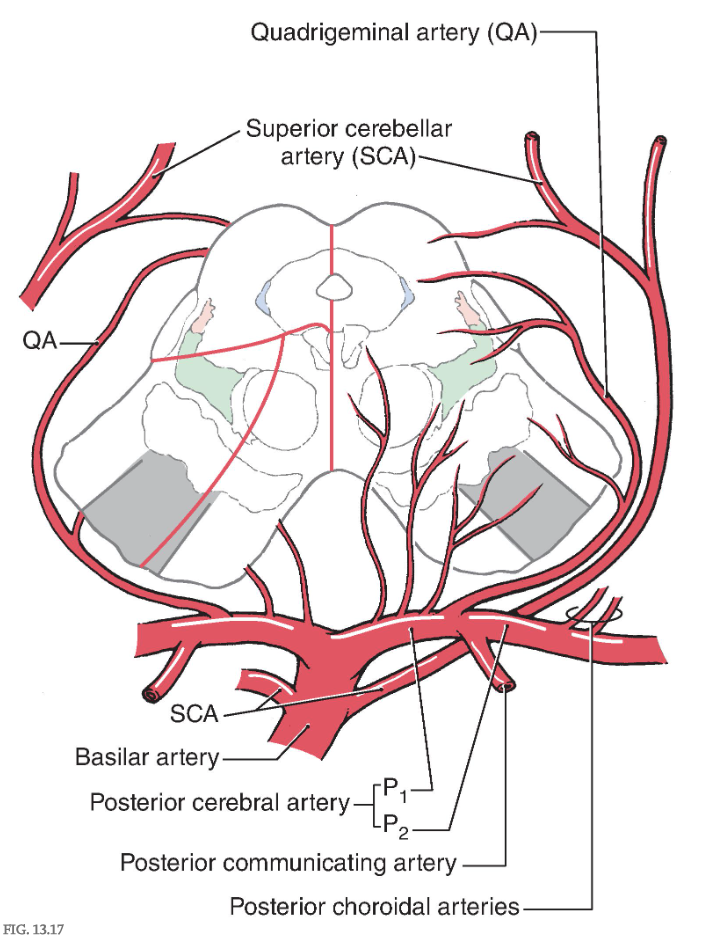
Internal Vasculature of the Midbrain
The blood supply to the midbrain originates from the basilar artery and its major branches (the quadrigeminal and superior cerebellar arteries ), from the anterior choroidal artery, which is a branch of the internal carotid, and from the medial posterior choroidal artery, which is usually a branch of P 2 ( Fig. 13.17 ). Medial regions of the midbrain receive numerous small branches from the P 1 segment of the posterior cerebral artery and from the posterior communicating artery. These paramedian branches constitute the posteromedial group of branches from the circle of Willis. Included in their territory are the oculomotor, trochlear, and Edinger-Westphal nuclei; the exiting oculomotor fibers; the red nucleus; and medial aspects of the substantia nigra and crus cerebri ( Fig. 13.17 ).
Blood supply of the midbrain. Arteries are shown mainly on the right and the territories served by each on the left. The anterior choroidal artery, which is a branch of the internal carotid and follows the general route of the optic tract ( Fig. 13.3B ), also sends branches to the lateral portions of the midbrain.
Ventrolateral regions of the midbrain are served by penetrating branches of the quadrigeminal artery ( Fig. 13.17 ), the anterior choroidal artery, and the medial posterior choroidal artery. The region served by these branches includes the lateral parts of the crus and substantia nigra and the medial lemniscus.
The posterior midbrain is served primarily by the quadrigeminal artery ( collicular artery ), which typically arises from P 1 ( Fig. 13.17 ). Much of the periaqueductal gray, the nuclei of the superior and inferior colliculi, the anterolateral system, and the brachium of the inferior colliculus are served by quadrigeminal branches. Additional blood supply to the area surrounding the exit of the trochlear nerve and the inferior colliculus arises from medial branches of the superior cerebellar artery.
Vascular Syndromes of the Midbrain
Weber syndrome
Occlusion of vessels serving the medial portions of the midbrain may result in the Weber syndrome ( Fig. 13.18A ). This produces an ipsilateral paralysis of all extraocular muscles except the lateral rectus and superior oblique, reflecting damage to the exiting root of the oculomotor nerve and a paralysis of the contralateral extremities, indicating damage to corticospinal fibers in the crus cerebri. The ipsilateral pupil is also dilated. This lesion also includes damage to corticonuclear fibers in the crus, resulting in a weakness of the facial muscles of the lower half of the face and a deviation of the tongue when it protruded, both on the side contralateral to the lesion (see also Chapter 25 ).
Vascular Syndromes of the Midbrain
Claude syndrome
If the vascular lesion is located in the more central area of the midbrain, the structures damaged include the fibers of the oculomotor nerve, the red nucleus, and the cerebellothalamic fibers. Collectively, the deficits seen after a lesion in this area constitute the Claude syndrome ( Fig. 13.18B ). Deficits include an ipsilateral paralysis of most eye movements; the eye is directed down and out with a dilated pupil (oculomotor nerve) and a contralateral ataxia, tremor, and incoordination that results from the damage to the red nucleus and cerebellothalamic fibers (see Chapters 24 and 25 ).
Vascular Syndromes of the Midbrain
Benedikt syndrome
A large lesion that includes the territories of both the Weber and Claude syndromes results in a constellation of deficits called the Benedikt syndrome ( Fig. 13.18C ). These include an ipsilateral paralysis of most eye movements, contralateral weakness of the extremities, and contralateral tremor and ataxia. In each of these lesions, it is possible that the VE preganglionic parasympathetic fibers arising in the Edinger-Westphal preganglionic nucleus may be damaged. Thus, the pupil on the side of the lesion will be dilated because of the action of the intact sympathetic input to the dilator muscle of the iris.
Decerebration
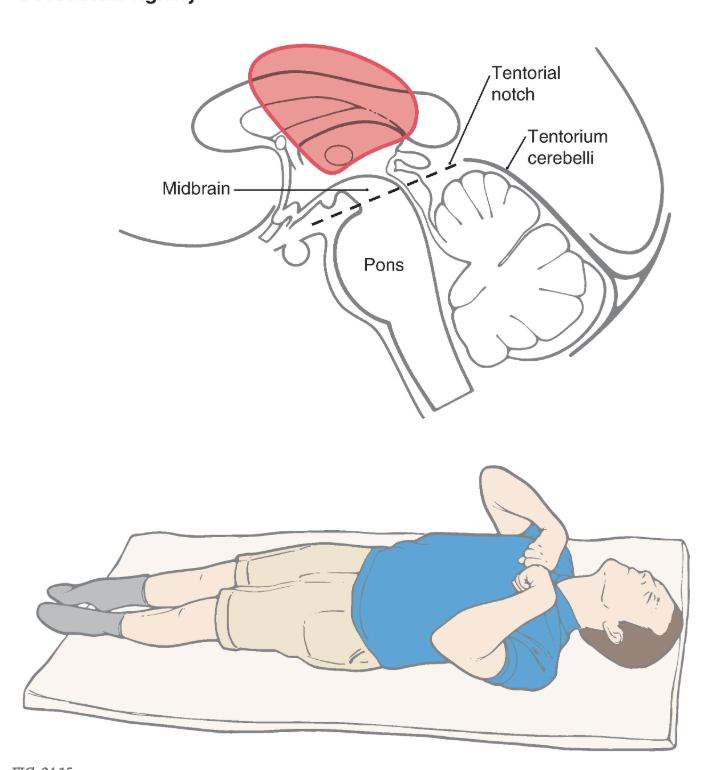
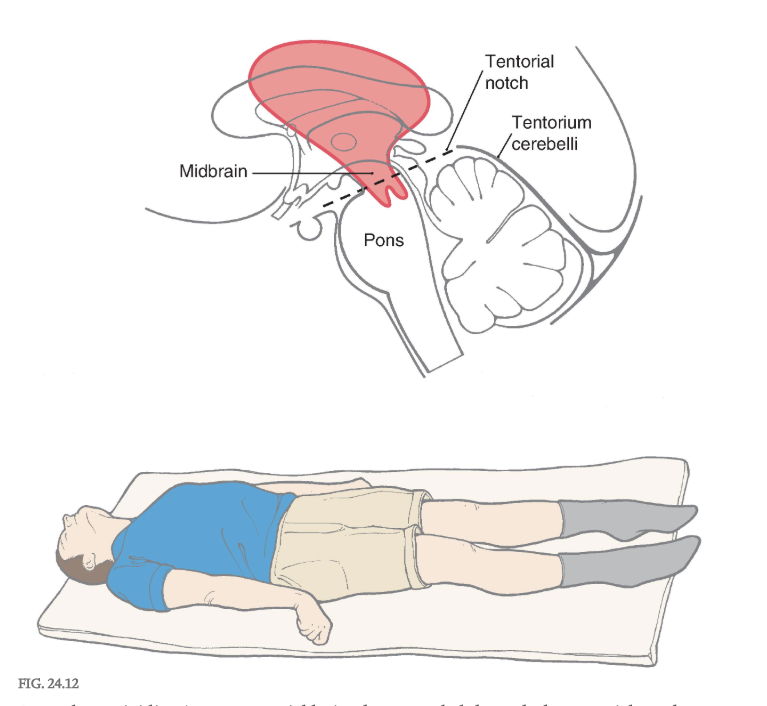
Removal of the influence of the cerebral cortex and other higher centers on brainstem-spinal systems was the premise in this experiment, with the idea that whatever functions remained were controlled predominantly by the brainstem-spinal systems. In the basic experiment, under deep anesthesia, the brainstem was completely transected bilaterally between the superior and the inferior colliculi ( Figs. 24.9A and 24.10A ). This procedure resulted in a constellation of deficits that closely resemble those seen in patients with supratentorial lesions that cause herniation of the midbrain downward through the tentorial notch, a process referred to clinically as central, or transtentorial herniation ( Fig. 24.11 ). The experimental lesion in animal models, like the comparable lesion in humans, results in unopposed hyperactivity in extensor musculature in all four extremities, a condition called decerebrate rigidity ( Fig. 24.12 ).
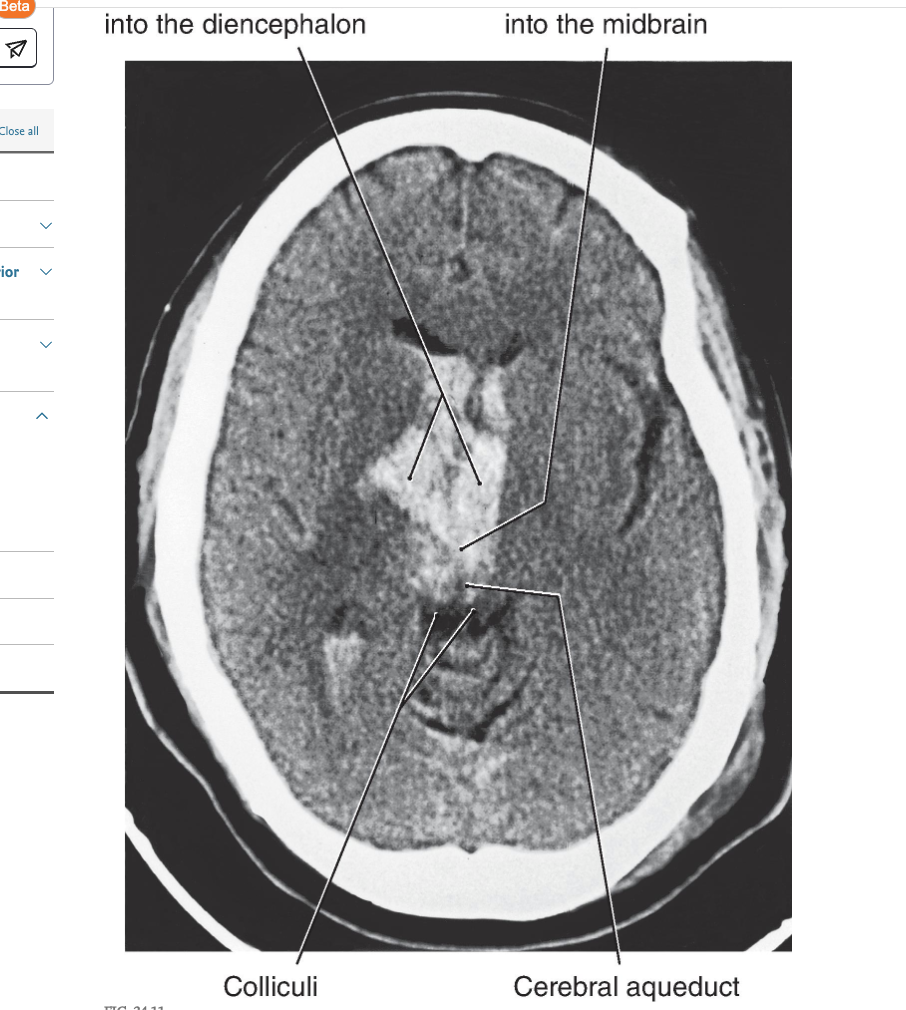
Decerebration
Rigigity
Decerebrate rigidity. A supratentorial lesion has extended through the tentorial notch. The patient’s lower extremities are extended, with the toes pointed inward; the upper extremity is extended, with the fingers flexed and the forearms pronated; and the neck and head are extended. The rigidity may be so extreme that the patient’s back is arched up off the bed. A patient may become decerebrate after a period of decorticate posturing (see Fig. 24.15 ).
In this situation, all descending cortical systems are interrupted, including the corticospinal tract as well as the corticorubral and corticoreticular projections. In addition, the red nucleus and rubrospinal tract are damaged. However, the vestibulospinal system, which lacks a cortical projection, remains intact. Similarly, the cells of origin for the excitatory and inhibitory components of the reticular formation lie caudal to the level of the lesion, thus both pontine and medullary reticulospinal projections remain intact ( Fig. 24.9 and 24.10 ), although they have lost their excitatory cortical input. Importantly, the ascending nociceptive input from the anterolateral system that is preferentially directed to the pontine elements of the reticular formation remains intact, and thus the excitatory component of the reticulospinal system remains functional. Conversely, the inhibitory reticulospinal component from the medulla is rendered nonfunctional because it is left with no significant excitatory drive. Consequently, a noxious stimulus delivered to a decerebrate patient may activate the excitatory reticulospinal system and exacerbate decerebrate rigidity that is already present or evoke decerebrate posturing when it is otherwise not apparent.
Central (or transtentorial ) herniation may be seen in patients with large tumors in the hemisphere or after a large hemorrhage in the hemisphere ( Figs. 24.11 and 24.12 ). In the diencephalic stage (before herniation through the tentorial notch), the patient may have a decreased level of consciousness, lethargy, small but poorly reactive pupils, and eye movement disorders. In addition, the withdrawal reflex to noxious stimuli is intact, reflexes are hyperactive, and there is a bilateral Babinski response. Once the herniation occurs, there is a rapid decline. The patient exhibits decerebrate posture, ( Fig. 24.12 ) is comatose, has dilated and fixed (do not react to light) pupils, and eye movement is absent. As the damage extends downward through the midbrain, respiration is compromised ( Cheyne-Stokes, tachypnea, followed by shallow rapid rates), and survival is highly unlikely.
Posterior (Dorsal) Root Section
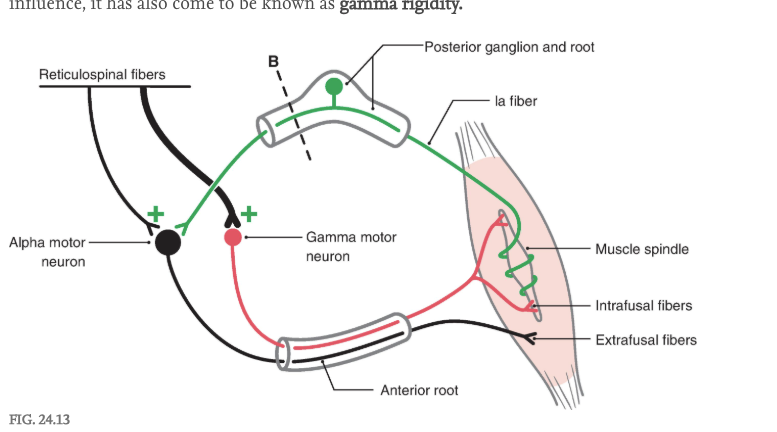
The extensor hypertonus observed in decerebrate individuals could be the result of excessive activation of either alpha or gamma motor neurons. To answer this question, the posterior root input from one extremity was interrupted in a decerebrate animal ( Figs. 24.9A, B , 24.10A, B , and 24.13 ). Immediately, the extensor hypertonus in that limb collapsed. This suggests that the descending reticulospinal or vestibulospinal influence on extensor motor neurons is focused primarily on gamma rather than on alpha extensor motor neurons ( Fig. 24.13 ). Lesioning of the posterior root would not affect the integrity of the descending reticulospinal or vestibulospinal projections onto alpha motor neurons. However, sectioning of the posterior root interrupts the gamma loop and thus eliminates the circuit that would be used by the gamma motor neuron to produce indirect activation of extensor alpha motor neurons (via activation of Ia sensory fibers) and subsequently the stimulation of extensor extrafusal muscle fibers ( Fig. 24.13 ). Thus, because decerebrate rigidity is associated with loss of the gamma motor neuron influence, it has also come to be known as gamma rigidity.
The gamma loop is formed by (1) gamma motor neurons that innervate intrafusal muscle fibers; (2) intrafusal muscle fibers that contract, stretching the sensory terminal encircling the central region of the spindle with consequent activation of the Ia fiber; (3) Ia fibers entering the posterior root and activating alpha motor neurons in the anterior horn; and (4) increased Ia activity, causing increased alpha motor neuron activity with consequent extrafusal muscle contraction. Sectioning of the posterior root ( broken line, B ) removes the Ia fiber, and gamma motor neurons no longer “indirectly” produce contraction of extrafusal muscle fibers; the gamma loop is interrupted.
Other studies aimed at evaluating the role of the vestibulospinal system in the extensor hypertonus involved removal of the anterior lobe of the cerebellar cortex ( Fig. 24.9A, B, C and 24.10A, B, C ). The net effect of the cerebellar cortex (inhibitory Purkinje cell influence on vestibular nuclei and inhibitory Purkinje cell influence on fastigial nucleus activation of vestibulospinal system) on the vestibulospinal system is inhibitory, thus removal of this influence is overall excitatory on vestibulospinal systems. Under these conditions, the extensor hypertonus was increased compared to decerebration alone, and further, this hypertonus remained when the posterior root was subsequently lesioned. This suggests that the hypertonus in that limb is not due to enhanced excitatory input to gamma motor neurons from the gamma loop, but instead there is enhanced direct input to extensor alpha motor neurons resulting from increased excitatory activity in the vestibulospinal system.
Decortication
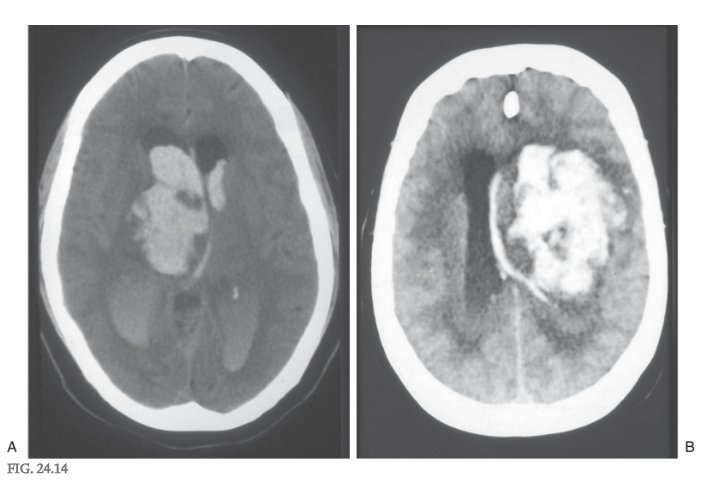
A second type of posturing frequently observed in human patients is referred to as decorticate posturing or decorticate rigidity ( Figs. 24.9D and 24.10D ). The clinical events that lead to this type of posturing are frequently large lesions of the cerebral hemisphere ( Fig. 24.14 ). These may be vascular or rapidly growing tumors that are located in one hemisphere, damage major tracts on that side, efface the midline, and compromise tracts on the opposite side by mass effect and compression. The net result is a removal of cortical influence on brainstem motor nuclei.
In the clinic, the patient presents with flexion of the upper extremities (intact rubrospinal fibers) at the elbow combined with extensor hypertonus in the lower extremities (intact reticulospinal fibers), an altered state of consciousness and respiration, oculomotor deficits, and a range of motor responses from weakness to motionless ( Fig. 24.15 ).
Decortication
rigidity
Decorticate rigidity. The lesion is in a supratentorial location. The lower extremities are extended, with the toes pointed slightly inward, and the upper extremities are flexed against the chest. The head is extended. A lesion in the supratentorial location may produce decorticate rigidity or posturing that may proceed to decerebrate rigidity or posturing as the lesion expands inferiorly (caudally) through the tentorial notch.
In experimental animals, this posture can be mimicked by transection of the brainstem (decortication) at a level just rostral to the superior colliculus ( Figs. 24.9D and 24.10D ). This lesion leaves the rubrospinal tract intact while eliminating the cortical input to the red nucleus. The rubrospinal system can still be activated because excitatory projections to the red nucleus from the cerebellar nuclei are unaffected by the lesion ( Fig. 24.10C ). The rubrospinal tract influences primarily flexor muscles, and most of this activity, in humans, is limited to the upper extremity. The upper extremities do not exhibit extensor hypertonus but instead show an increase in flexor tone, presumably due to the intact rubrospinal system. In some patients, the lower extremities might exhibit a minimal level of extensor hypertonus for the same reasons as in decerebration. This characteristic type of posturing is called decorticate rigidity.
These two conditions, decerebration and decortication, are not uncommon ( Figs. 24.12 and 24.15 ), and knowledge of these signs and the underlying brain pathologic process is important in the diagnosis and clinical management of these patients. In some cases, the patient may be comatose and initially exhibit decorticate posturing that subsequently converts to decerebrate posturing. This is an ominous sign, as it suggests that the lesion has continued to progress and now involves more caudal portions of the brainstem. The patient’s cardiovascular and respiratory control centers in the medulla may soon be compromised, necessitating prompt intervention.