BIOCHEM 4M03 BANK
1/721
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
722 Terms
normal blood sugar level (fasting)
70mg/dL (3.9 mmol/L) and 100m/dL (5.6 mmol/L), but usually maintained at around 80-90mg/dL (5 mM) (conversion factor is around 18 bw mM and mg/dL)
grams of glucose in an adult human compared to grams of sugar in a can of cola
4.5 grams of blood glucose vs around 40 grams of coke glucose
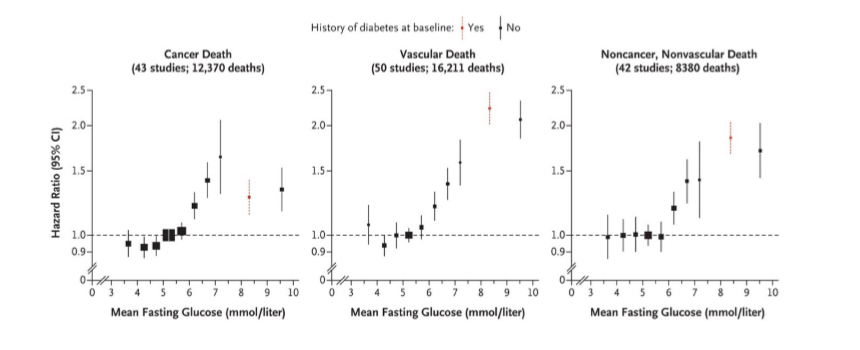
health with blood glucose
higher and longer elevations of blood glucose means a greater risk of chronic diseases (used as an indication of pre-diabetes or diabetes). it is a highly regulated key aspect of health
blood glucose comes from
internal stores: liver and kidneys (glycogen). food: complex carbs (polysaccharides), simple sugars (sucrose and glucose)
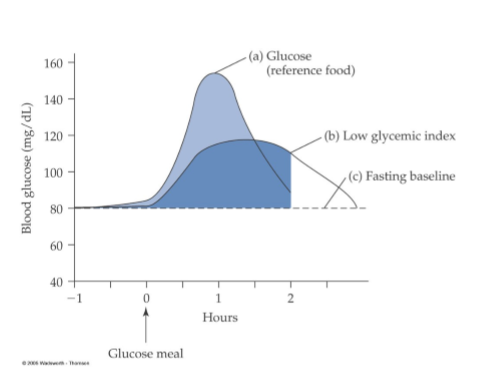
glycemic index
ranks carbohydrate-containing foods by how quickly they raise blood sugar levels, using a scale of 0 to 100, with glucose as 100. High-GI foods (55+) cause rapid spikes, while low-GI foods (55 or less) lead to slower, steadier increases, aiding blood sugar control and fullness
factors affecting the glycemic index of food
highly processed food, soluble fibres, fat and proteins, and acids
how highly processed foods affect glycemic index
more easily gelatinized (engages more water) and digested faster (higher GI)
how soluble fibres affect glycemic index
slow down interaction between carbohydrates and enzymes
how fat and protein affect glycemic index
slow stomach emptying and slow down carb digestion
how acids in food affect glycemic index
slow down rate of digestion and absorption of carbohydrate
carbohydrate classification
memorize (except for a few)
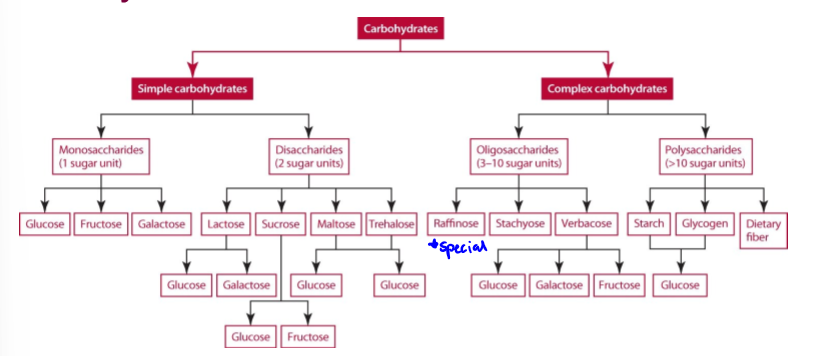
simple sugar examples
monosaccharides: glucose, galactose, and fructose. disaccharides: maltose, lactose, and sucrose
complex sugar examples
oligosaccharides and polysaccharides
oligosaccharides examples and foods
raffinose (trisaccharide of galactose, fructose, and glucose); generally in: beans, veggies, bran, whole grains.
polysaccharides examples and foods
cellulose (most common organic compound on earth), and glycogen (animal starch, more branched and compact)
places where carb digestion happens
mouth, duodenum, small intestine
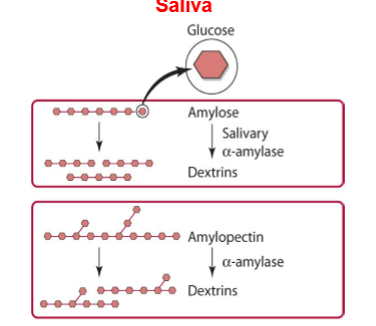
carb digestion in mouth
amylose -(salivary alpha amylase hydrolyzes alpha 1,4 glycosidic bonds in amylose)→ dextrins. amylopectin -(salivary alpha amylase hydrolyzes alpha 1,4 glycosidic bonds in amylopectin)→ dextrins
carb digestion in stomach
not much action. the acidity of the gastric juice destroys the enzymatic activity of the salivary amylase, so there is no further digestion of amylose and amylopectin (dextrins)
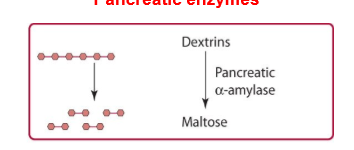
amylose digestion in duodenum
pancreas releases pancreatic alpha amylase which hydrolyzes alpha (1,4) glycosidic bonds. dextrins broken down into maltose into small intestine.
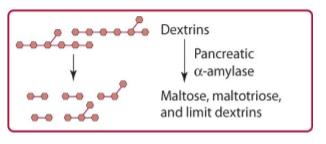
amylopectin digestion in duodenum
pancreas releases pancreatic alpha amylase which hydrolyzes alpha (1,4) glycosidic bonds. dextrins broken down into maltose, maltotriose, and limit dextrins. hydrolysis stops 4 residues away from the alpha (1,6) bond
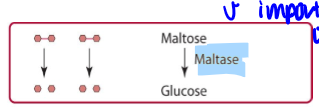
amylose digestion in small intestine
maltose hydrolyzed by maltase (brush border enzymes, forming free glucose)
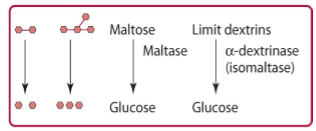
amylopectin digestion in small intestine
maltose, maltotriose, and isomaltose are further hydrolyzed in the brush border by the enzyme maltase or isomaltase (alpha dextrinase) to glucose. isomaltase is the sole carbohydrase capable of hydrolyzing alpha (1,6) glycosidic bonds
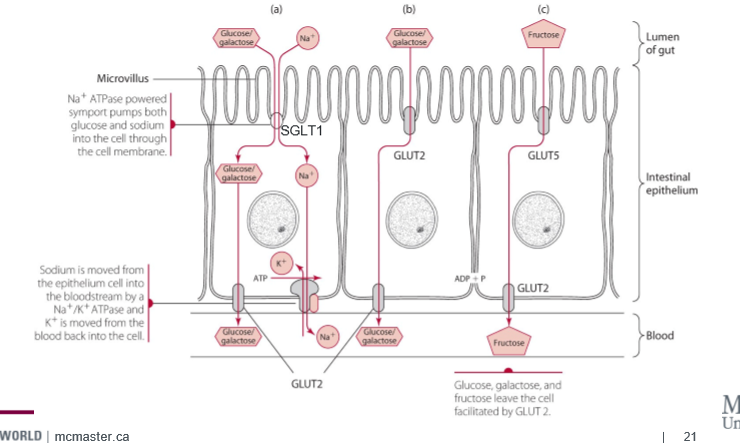
absorptions, transport, and distribution of carbs
once digested down to simple sugars (or eating simple sugar), glucose transporters GLUT and SLGT are required for glucose to cross membranes
GLUT
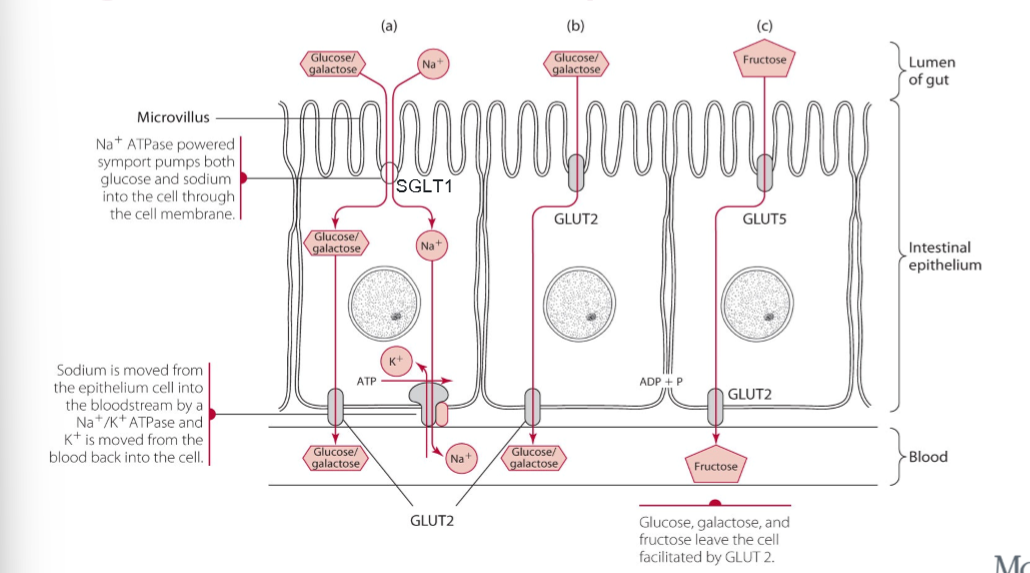
glucose transporter isoforms- GLUT2 and GLUT5. facilitated transport
SLGT
sodium dependent glucose cotransporters, active. active transport. SLGT1 is for glucose and galactose, Na+/K+ ATPase maintains sodium balance in cell
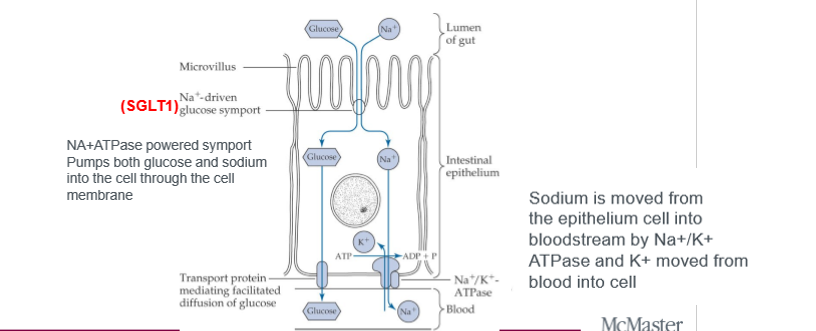
GLUT2
for glucose and galactose. especially after large carb meal. glucose conc in the gut lumen is greater than in blood, and this transporter goes to the apical membrane (lumen border)
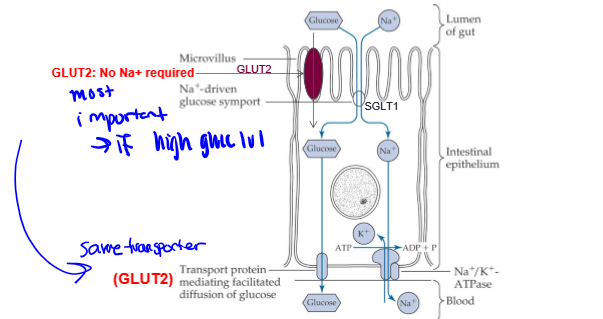
GLUT5
only one for fructose. independent of glucose concentration, independent of active transport
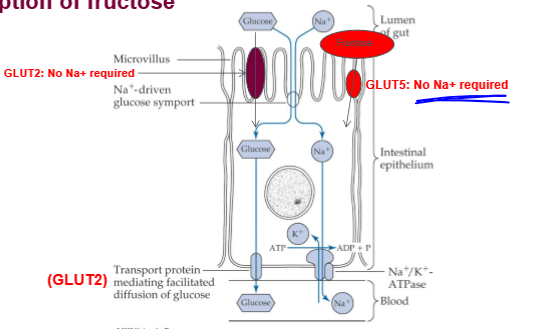
intestinal glucose and fructose transport
hormones altering blood glucose
major regulators of blood glucose, insulin receptor signaling stimulates more GLUT4 to reside at the cell surface, hence more glucose uptake into tissues
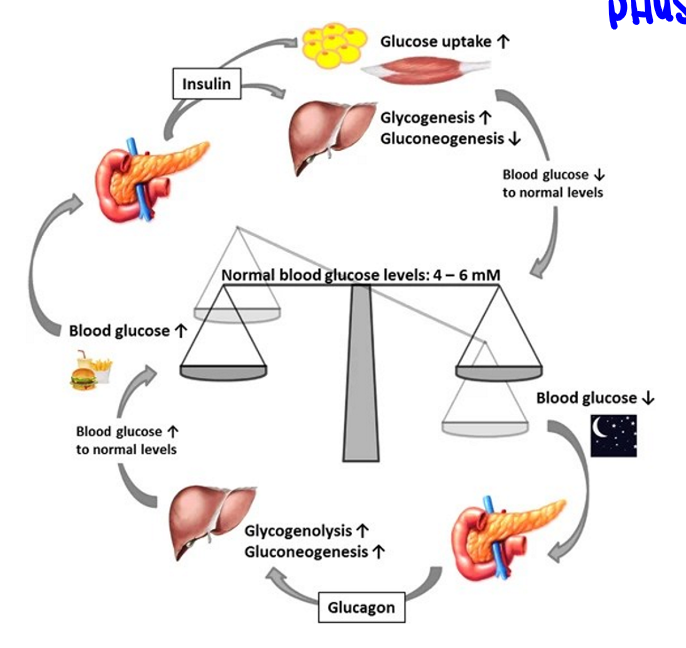
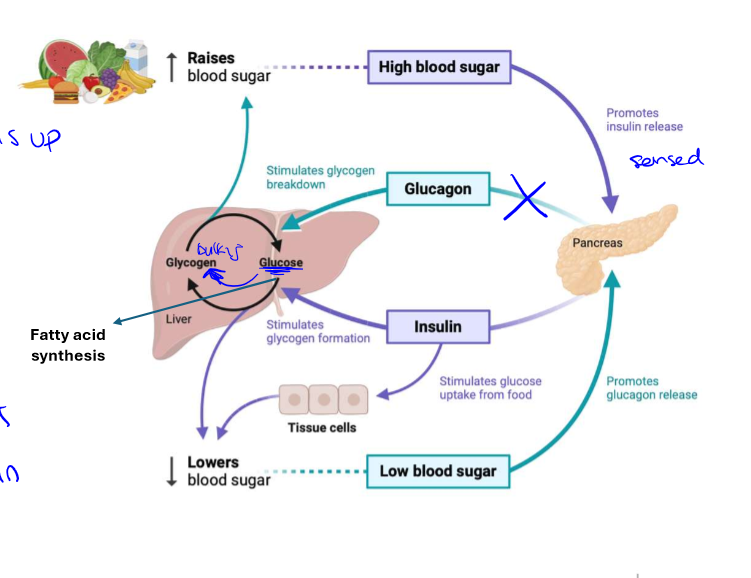
elevated blood glucose
sensed by pancreas which releases insulin, insulin promotes blood uptake into tissues, this lowers glucose production in the liver
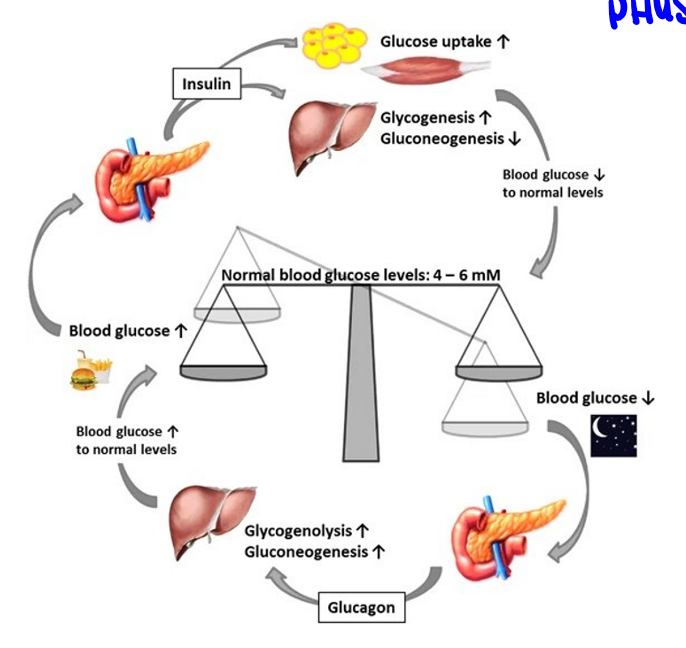
lowered blood glucose
sensed by pancreas which released glucagon, glucagon promotes release of glucose from carbohydrate reserves, increasing blood glucose (counterbalances insulin)
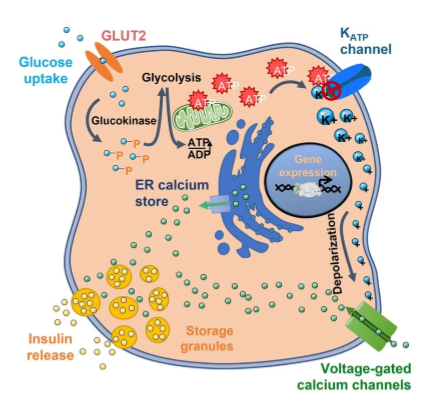
steps for insulin release
1) blood glucose above 5mM enters pancreatic beta cell via GLUT2. 2) increase in ATP via glucokinase reaction. 3) K+ ATP sensitive channel: K+ channel closes, K+ efflux suppressed leads to depolarization of beta cell. 4) Ca2+ channel opens leads to increased Ca2+. 5) insulin granules move to surface. 6) exocytosis of insulin and c-peptide. leads to higher insulin in blood can act on tissues
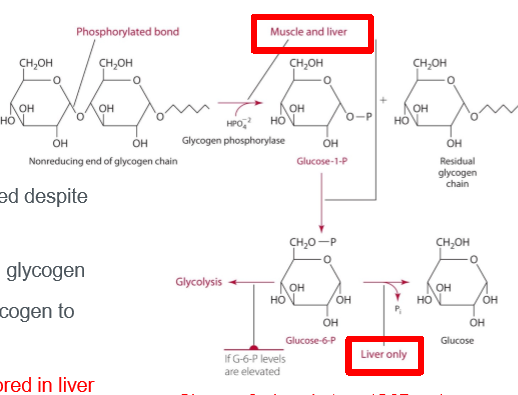
sources of glucose aside from food
blood glucose must be maintained despite periodic meal/sugar ingestion, muscle and liver major stores of glycogen, liver and kidney can convert glycogen to glucose (enzyme G6Pase), and releasable/usable glucose stored in liver
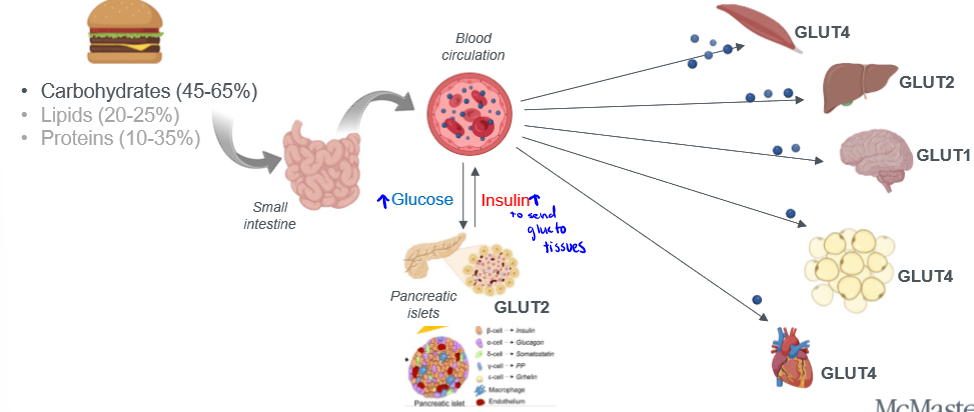
the macronutrients and their percentages in the body
carbs: 45%-65%; lipids: 20%-25%; proteins: 10%-35%. fats have increased over time
dietary roles of lipids
high energy source: 1g fat - 9 kcal (very high); satiety: energy density, slower digestion, hormones, etc; nutrient absorption: vitamin A, D, E, K (diluted in the fat)
physiological roles of lipids
energy storage: white adipose tissue; cell structure: phospholipids; insulation/protection: thermal insulation, cushioning of vital organs (visceral fat); hormone production: cholesterol, steroid hormones (sex hormones); nerve function: myelin sheath; temperature regulation: brown adipose tissue
types of dietary lipids
triglycerides, phospholipids, and sterols
triglycerides simple
1 glycerol and 3 fatty acids. up to 24 carbon atoms, differed by saturation and chain length. structure is
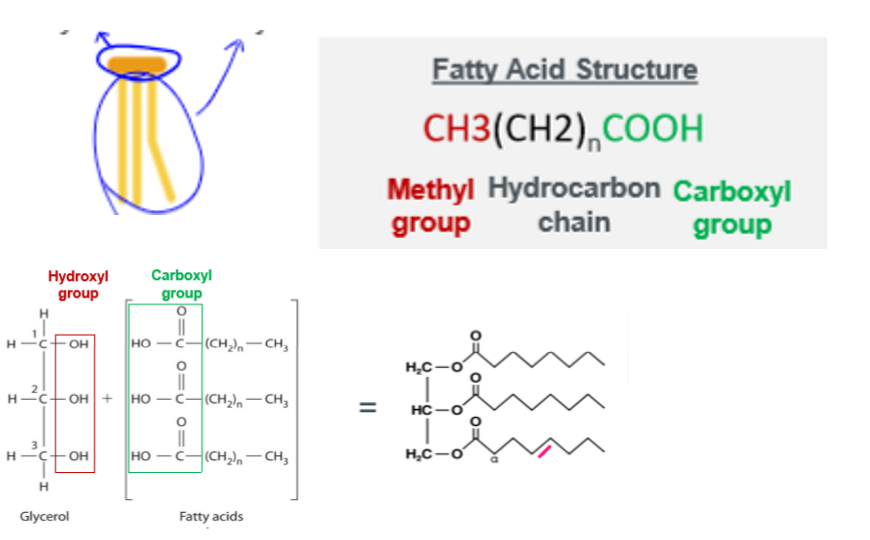
phospholipids simple
1 glycerol, 2 fatty acids, and 1 phosphate. head is hydrophilic, tails are hydrophobic

sterols simple
multiple carbon rings such as cholesterol
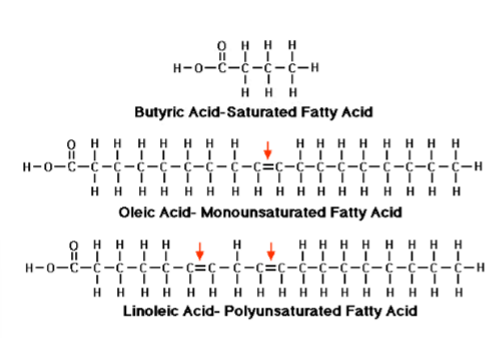
fatty acid saturation
saturation means no double bonds; unsaturated is presence of a double bond (mono for 1, poly for more than 1). saturated means hydrogen saturation
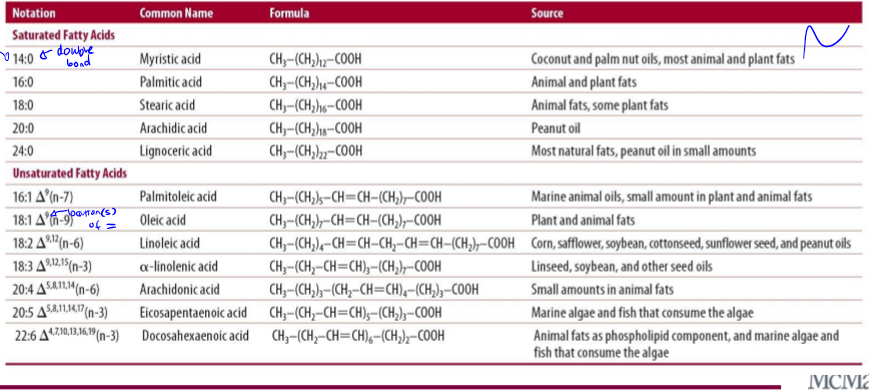
dietary lipid ratios
dw about common names and formula, know some examples
types of unsaturated fats
cis and trans
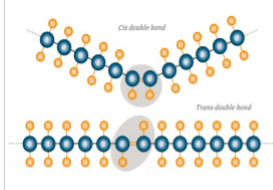
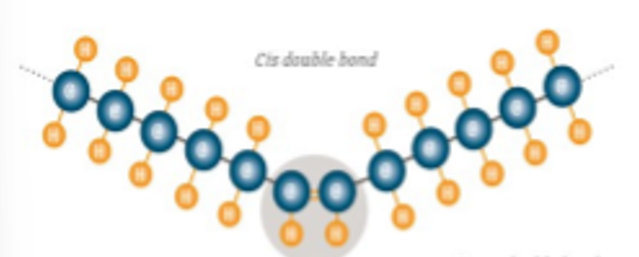
cis fat
naturally occurring, bent, liquid (bc cant stack). type of unsaturated fat
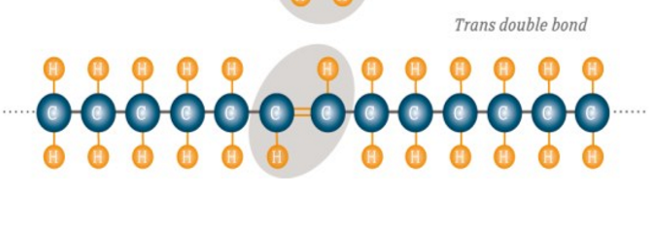
trans fat
mostly modified, food industry makes it through hydrogenation. eg: margarines, cookies, muffins, fried food, cinnamon roll (1 has 5g trans fats). purpose: longer shelf life, but increases cardiovascular risks. some naturally occurring ones are dairy and meats
essential fatty acids
omega 3 and 6 fatty acids (polyunsaturated fatty acids). some sources are: fatty fish, flax seeds, walnuts, leafy veggies, etc. a proper balance of 1:1 between omegas 3 and 6 is best
omega 3 fatty acids
means that first double bond is on the third carbon. it is an unsaturated cis fat so has many health benefits
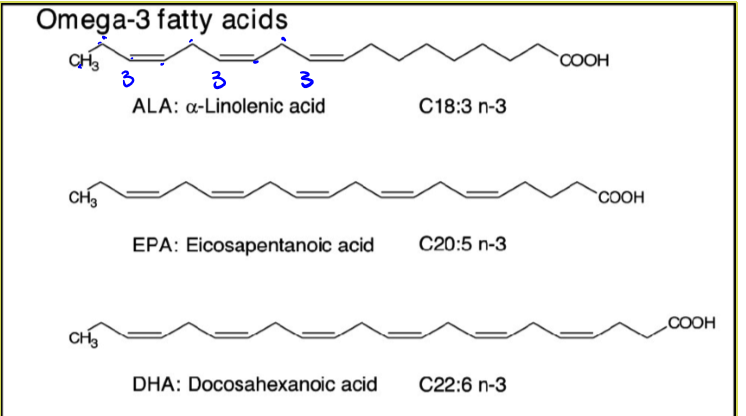
omega 6 fatty acids
means that first double bond is on the sixth carbon. it is an unsaturated cis fat so has many health benefits
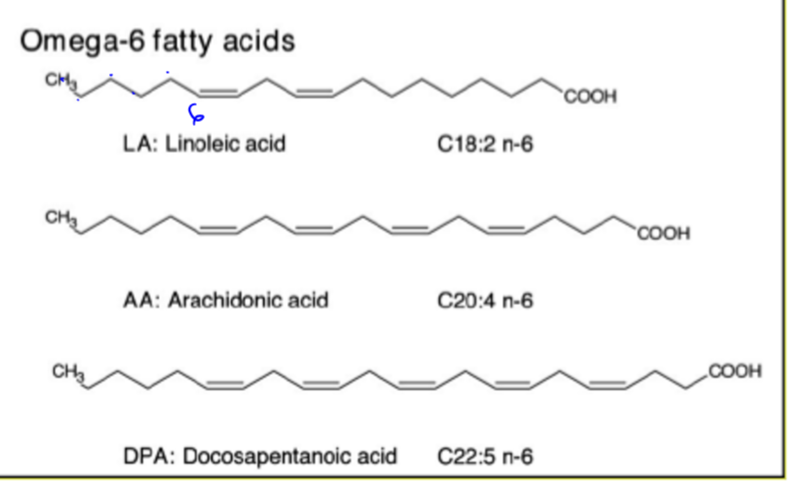
short fatty acid chain length
less than 6 carbon atoms, relatively water soluble compared to long chain FAs. sources: fermentation of fibres in the colon
medium fatty acids
between 6 and 12 carbon atoms, relatively soluble compared to long chain FAs. sources: coconut oil, palm kernel oil. goes to the blood circulation
long fatty acids
over 12 carbon atoms. primary dietary lipid in a typical diet. not very water soluble compared to short and medium chained FAs. sources: animal fat, olive/corn/sunflower/soybean oils. nuts/seeds, avocado. transported to lymphatic system
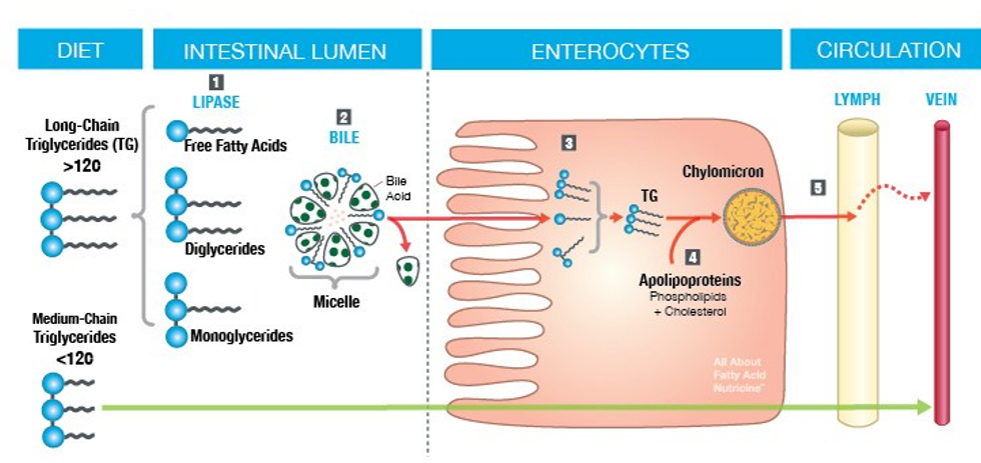
digestion of triglycerides
in the mouth, stomach, and small intestine
digestion of fats in the mouth
mechanic: chewing. chemical: lingual lipase
digestion of fats in the stomach
mechanical: mixing. chemical: lingual/gastric lipase
digestion of fats in the small intestine
mechanic: peristalsis. chemical: pancreatic lipase
fats before absorption
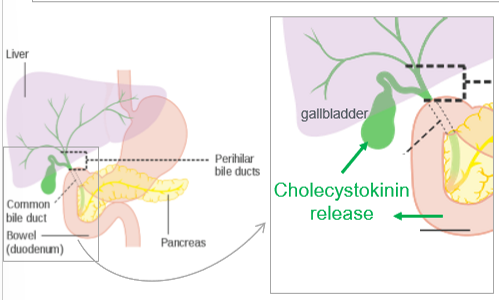
broken down, large fatty deposits are emulsified by bile which is released from the gallbladder. bile is then broken down into bile salt, bilirubin, cholesterol, phospholipids, water, etc
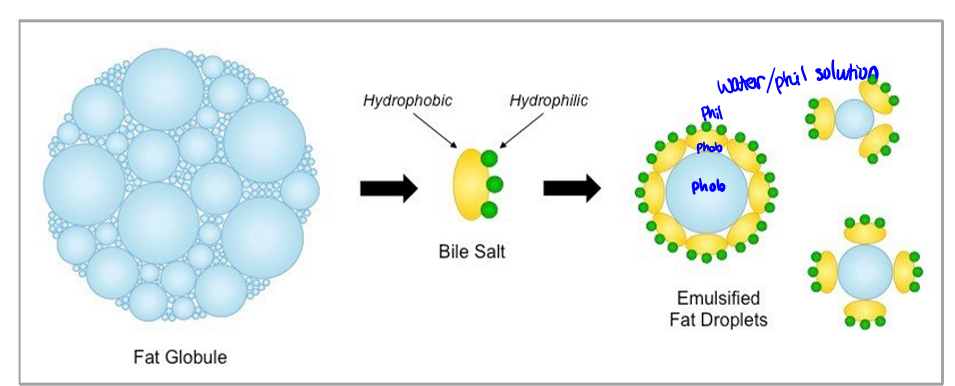
emulsification
of fats. bile salts are amphipathic (hydrophobic body, hydrophilic heads), increases SA for digestion, micelle formation for absorption
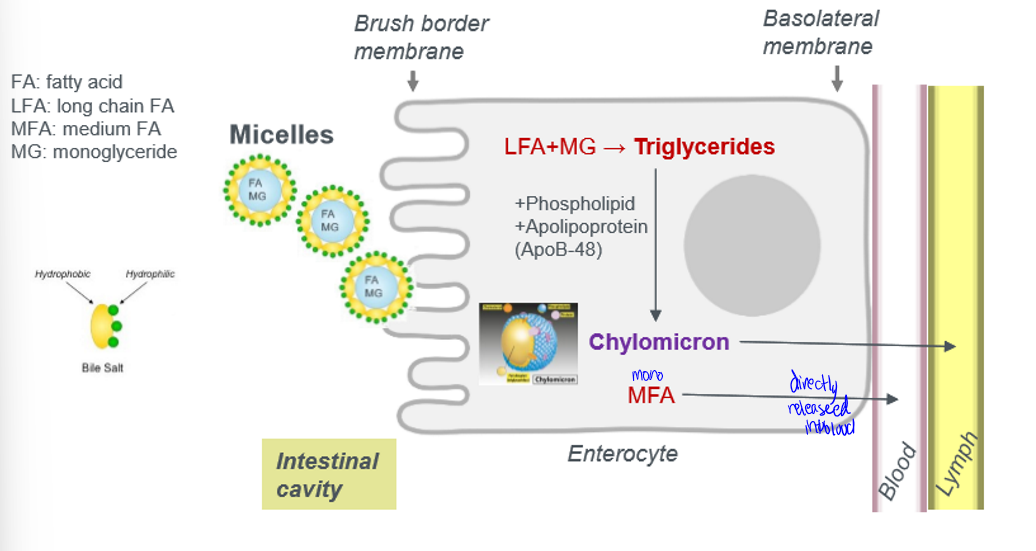
fat absorption
micelles formed by bile salts allow for stability and efficiency.
digestion and absorption pathway
chylomicron
a large, triglyceride-rich lipoprotein produced in the small intestine to transport dietary fats (lipids) and fat-soluble vitamins from food into the lymph and bloodstream
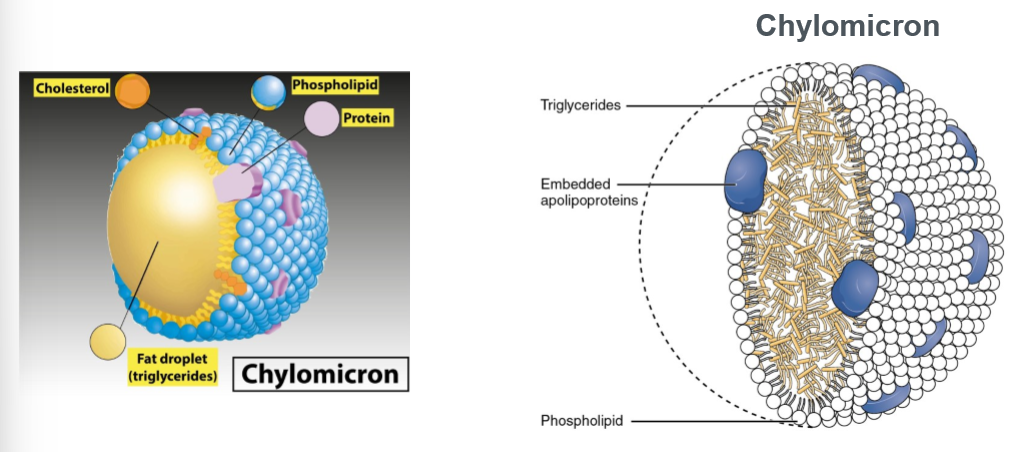
blood lipid levels
triglycerides must be less that 150 mg/dl, and cholesterol must be less than 200 mg/dl. higher blood lipid levels leads to a higher risk of disease
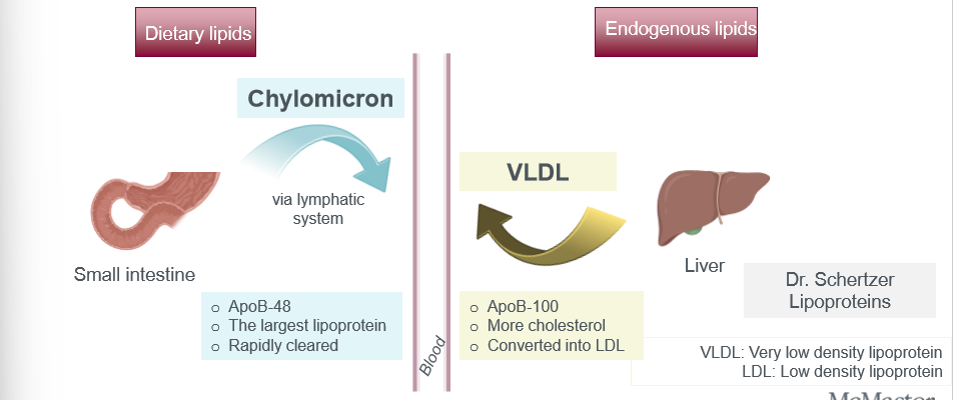
dietary vs endogenous lipids
triglycerides in the blood circulation is derived from dietary lipids and endogenous lipid synthesis
major glucose uptake organs and their primary glucose transports
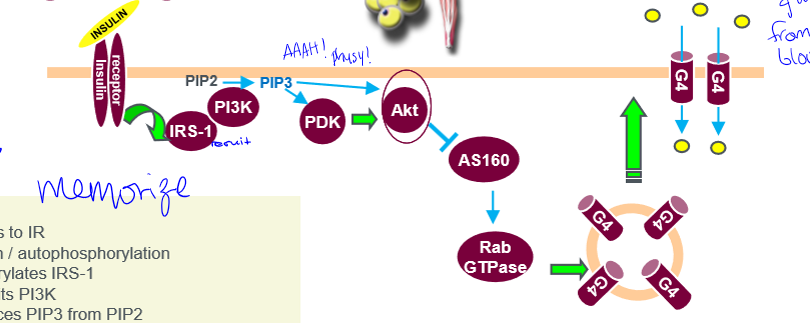
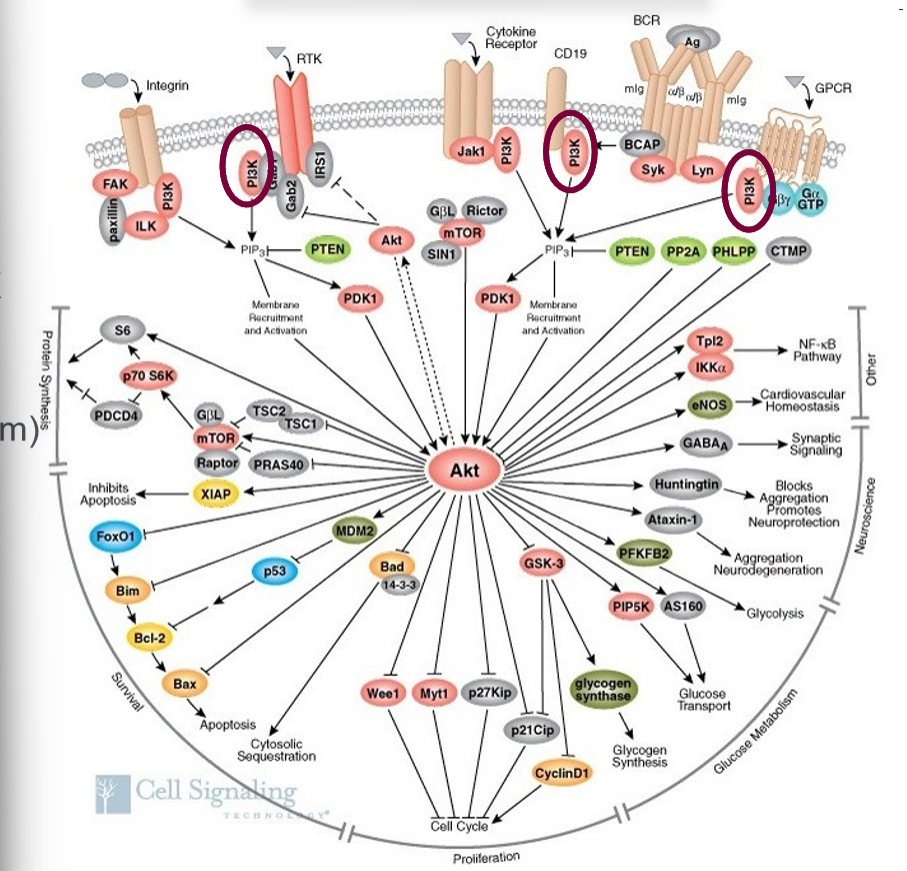
insulin signaling pathway
1) insulin binds to insulin receptor (IR); 2) IR activation/autophosphorylation; 3) IR phosphorylates insulin receptor substrate (IRS-1); 4) IRS-1 recruits PI3K; 5) PI3K produces PIP3 from PIP2; 6) PIP3 recruits PDK and Akt; 7) PDK activates Akt; 8) Akt inactivates AS160 leading to activate Rab proteins (a type of GTPase); 9) promote the translocation of GLUT4
Akt
protein kinase B. master regulator of cellular metabolism, a key player in Akt activation is an enzyme called PI3K, has 3 isoforms (Akt1, Akt3 growth, and Akt2 glucose metabolism), Akt2 KO mice develop diabetes
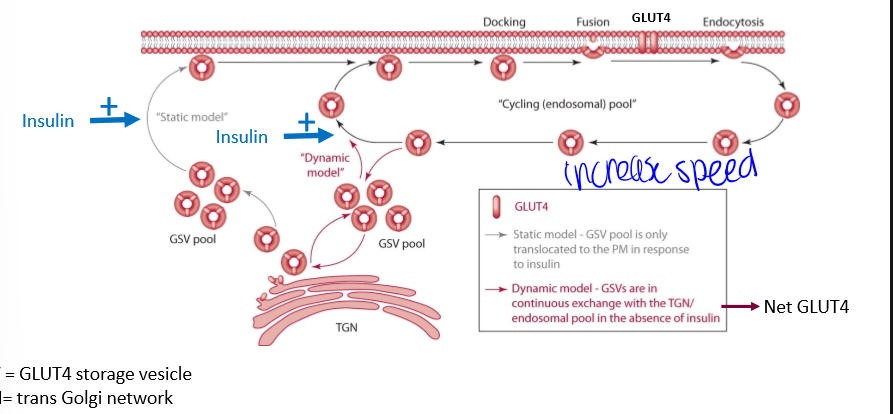
GLUT4 translocation
just know it dont memorize it
what does glucose do once inside the cell
1) glucose oxidation (glycolysis) for energy; 2) glycogen synthesis (glycogenesis) for storage; 3) fatty acid formation (de novo lipogenesis) for storage
glycolysis important proteins
HK, PFK, PK, and PDH
glycolysis path
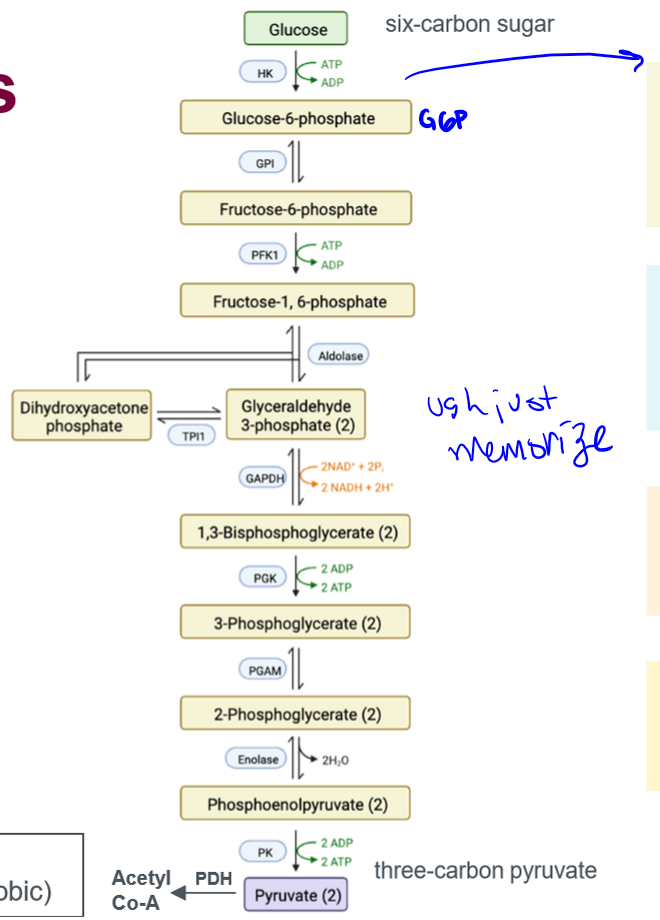
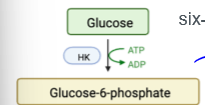
HK
glycolysis protein; hexokinase in muscle/fat, glucokinase in liver. glucokinase in liver increased by insulin, both are allosterically inhibited by G6P (the protein they produce)
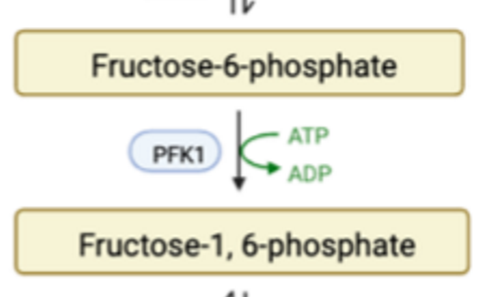
PFK
glycolysis protein; phosphofructokinase. first committed step in glycolysis; activated by insulin, AMP, and ADP; inhibited by ATP and citrate
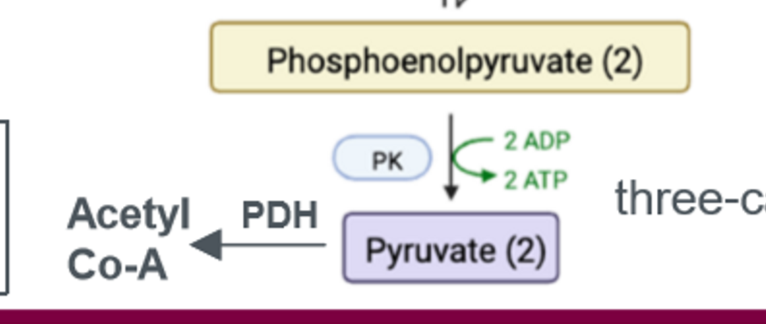
PK
glycolysis protein; pyruvate kinase. activated by AMP, F-1,6-BP; inhibited by ATP, acetyl-CoA, alanine
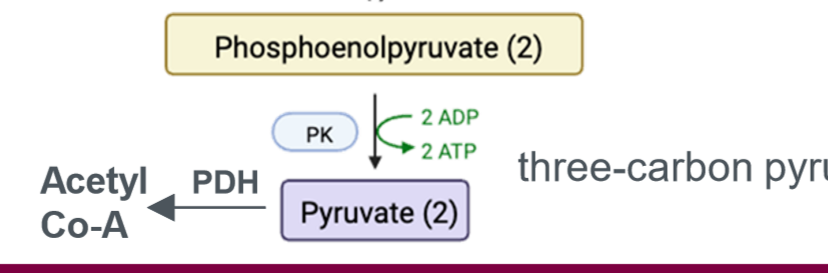
PDH
glycolysis protein; pyruvate dehydrogenase. activated by insulin, NAD, and ADP; inhibited by ATP, acetyl-coa, NADH
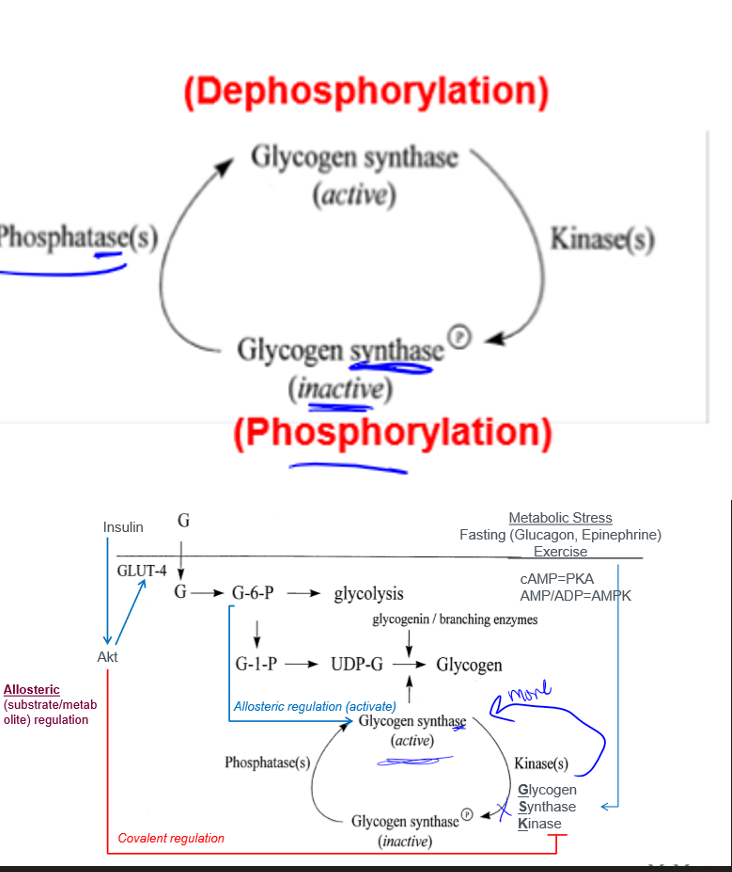
glycogenesis
conversion of glucose to glycogen. steps 1 and 4 are rate limiting.
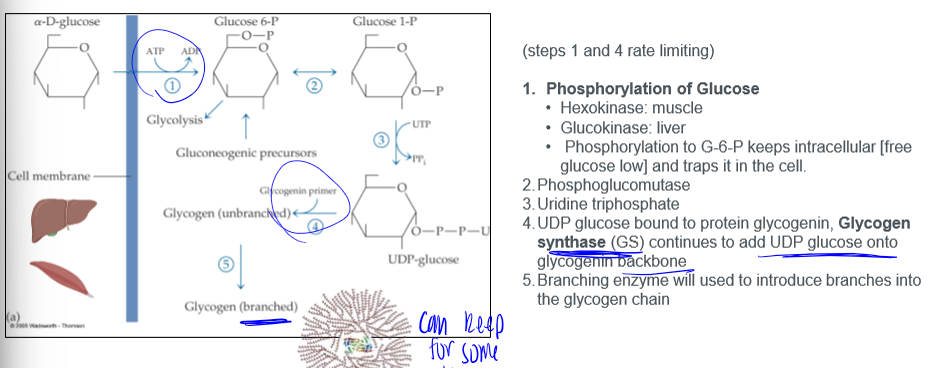
glycogen synthase
activity controlled by: covalent regulation involving protein phosphorylation, and allosteric (substrate/metabolite) regulation
product and action of increased muscle glucose uptake
increased glucose transporter
product and action of increased liver glucose uptake
increase glucokinase
product and action of increased liver and muscle glycogen synthesis
increased glycogen synthase
product and action of increased liver and muscle glycolysis, acetyl CoA production
increased phosphofructokinase-1 and increased pyruvate dehydrogenase complex
why do athletes use a sugar loading strategy 1-4 days prior to participating in prolonged endurance activities such as a marathon and why
to get glucose to store into glycogen that they can then use it as glucose when it is time. we dont use fats bc digesting fatty acids takes longer than sugars
glucose homeostasis post meal
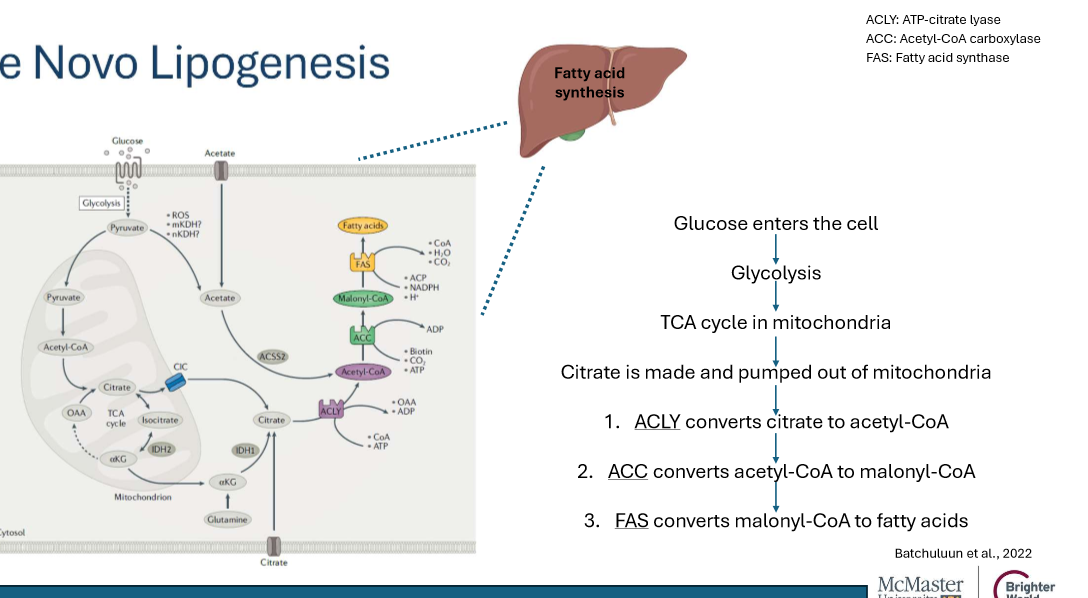
glycogen is bulky and requires water to store. to enhance storage of glucose, the body converts glucose (as liver fills up) to fatty acids that are more energy dense
de novo lipogenesis
MASH and MAFLD
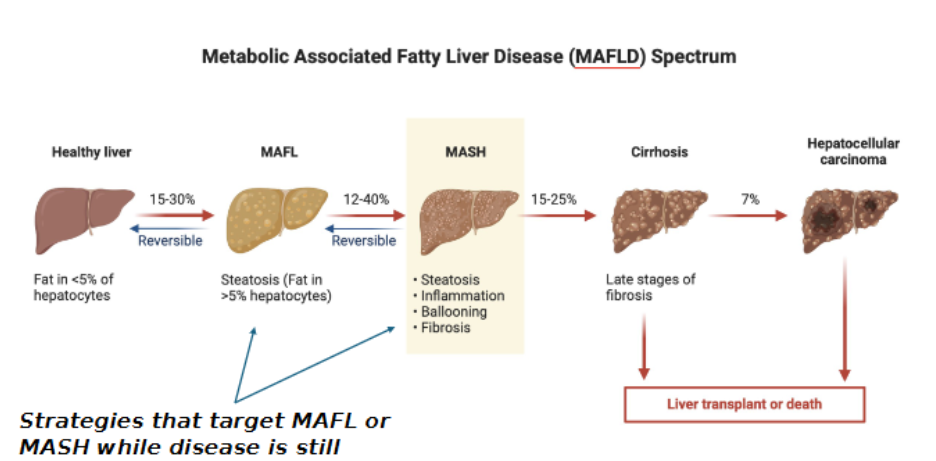
what biochemical/physiological process in the liver is linked to MAFLD progression
de novo lipogenesis
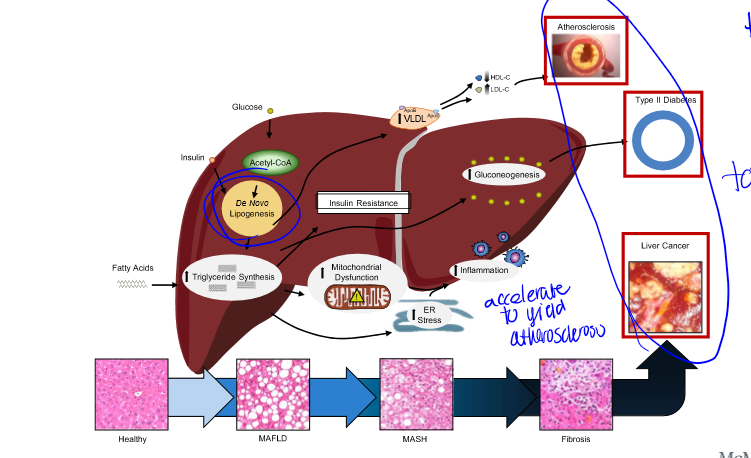
What kind of diseases can arise from liver fat synthesis
atherosclerosis, type II diabetes, liver cancer, etc. this is why reducing fat synthesis is beneficial
reason of MASH study
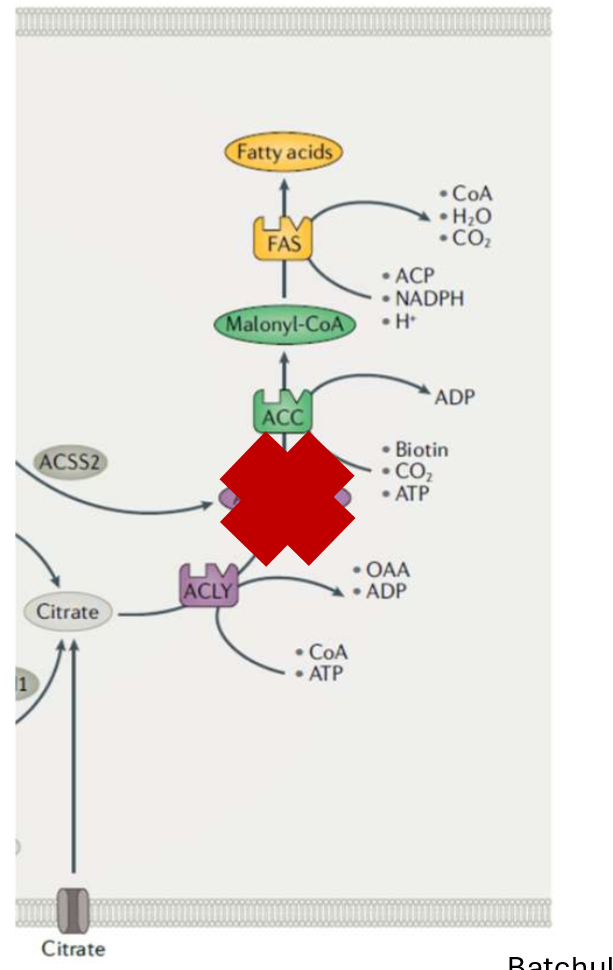
protein ACC contributes to MAFLD progression, pharm ACC inhibitors reduce DNL and liver steatosis but lead to increased circulating triglycerides (increasing CVD risk), so scientists must find another way to solve initial issue
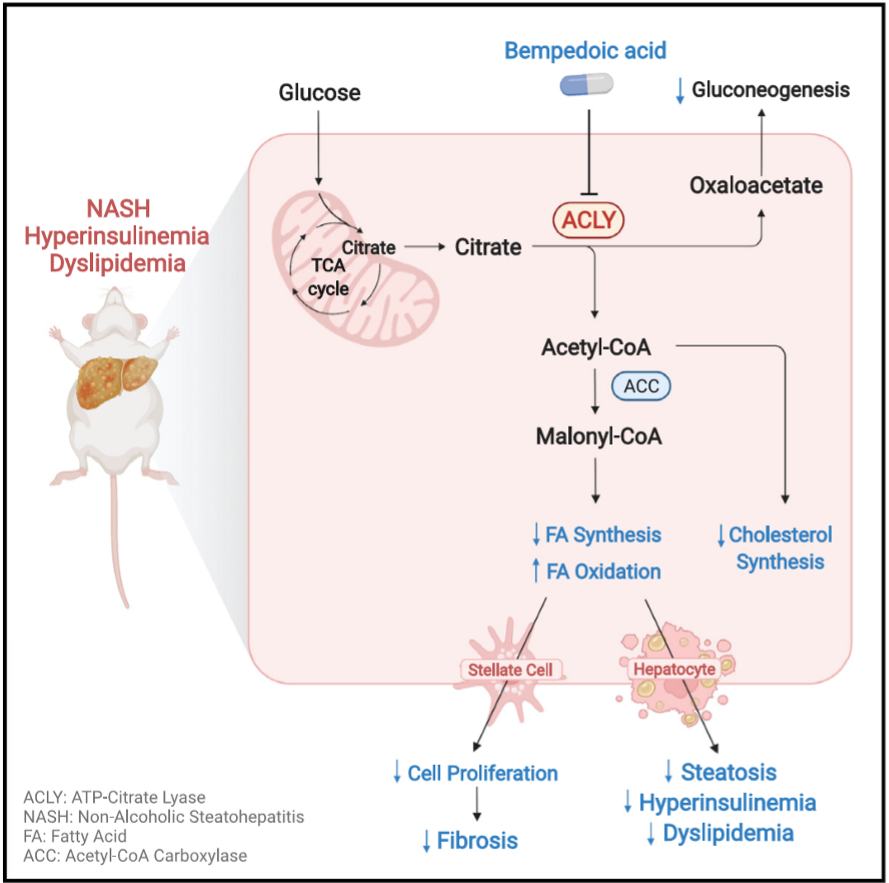
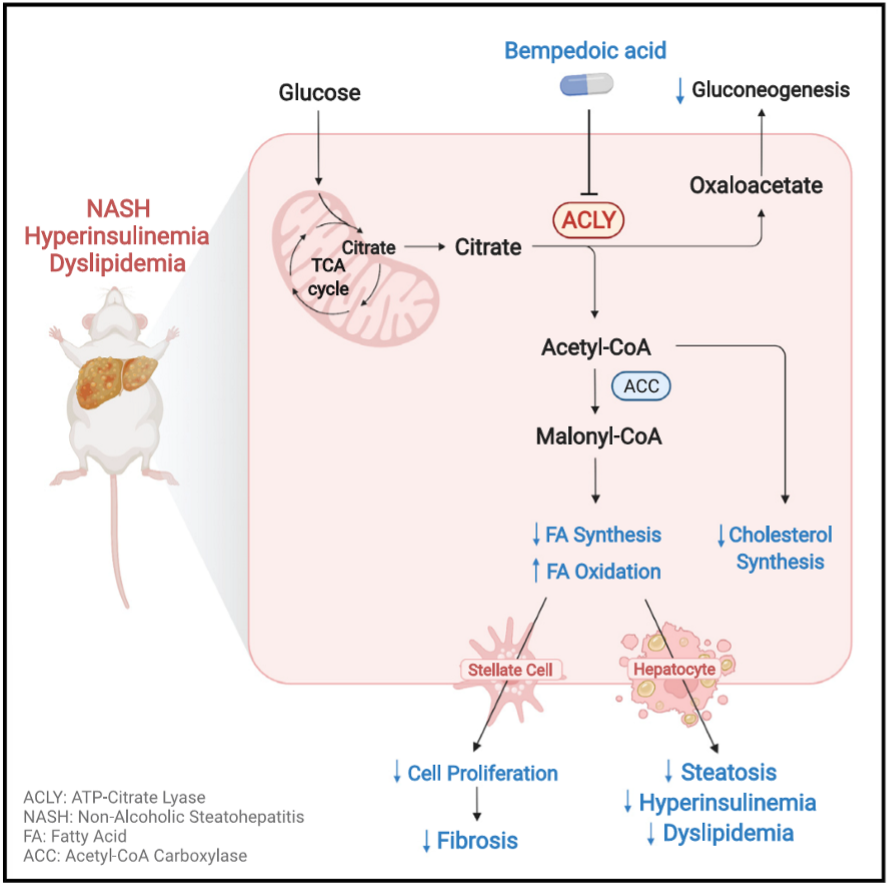
purpose of MASH study
ACLY (upstream of ACC) is a promising target but unknown if effective for MASH. scientists need to investigate whether inhibiting ACLY can mitigate MASH progression
how to model human MASH on mice
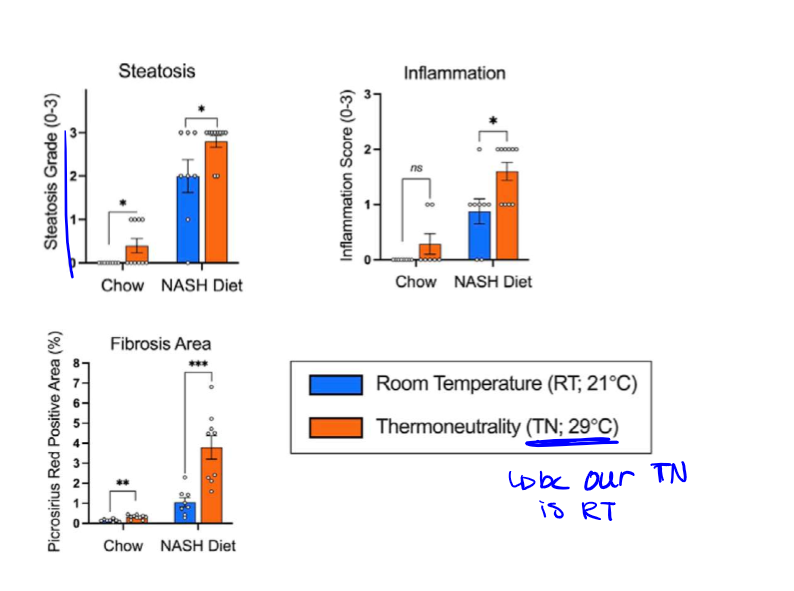
include a “NASH diet” consisting of: 1) high fat (40%), 2) high fructose (20%), 3) high cholesterol (0.02%); to increase lipid flux to the liver, DNL, inflammation, and fibrosis; and house at TN (29 degrees)
TN of a mouse
instead of housing the mice at our TN which is RT, these mice are held at 29 so they can maintain core temp without having to produce extra heat (burn calories), because we need that fat to stay
types of testing for ND vs Chow and RT vs TN
done with hematoxylin & eosin (stains nuclei and cytoplasmic components, fat remains unstained and appears clear/white) and picrosirius red (stains collagen red (surrounding) which is the major component of fibrosis). as well: steatosis, inflammation, and fibrosis area
results of H&E and PSR tests
to test mouse models, if ND & TN offer best resemblance to MASH in humans, and it did
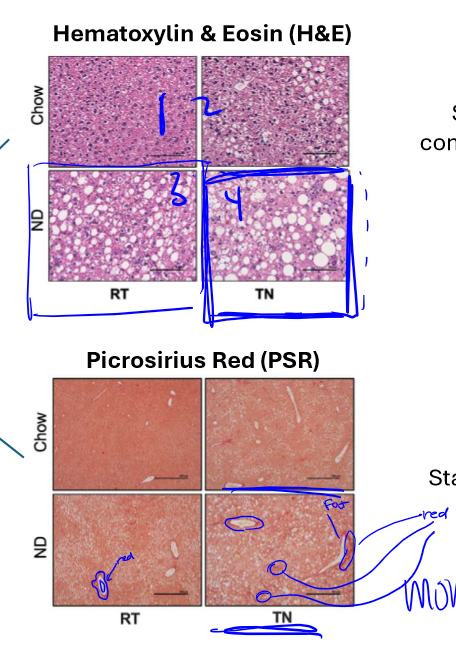
results of steatosis grade, inflammation score, and PSR % tests
again showed that NASH diet at TN offers the best resemblance to human MASH
inhibition of fatty acid synthesis through ACLY deletion
deletion of ACLY gene through knockout technique (ACLY hKO (Cre (cut enzyme)) this means that mice grow up with the enzyme and then the KO is induced when the ND starts
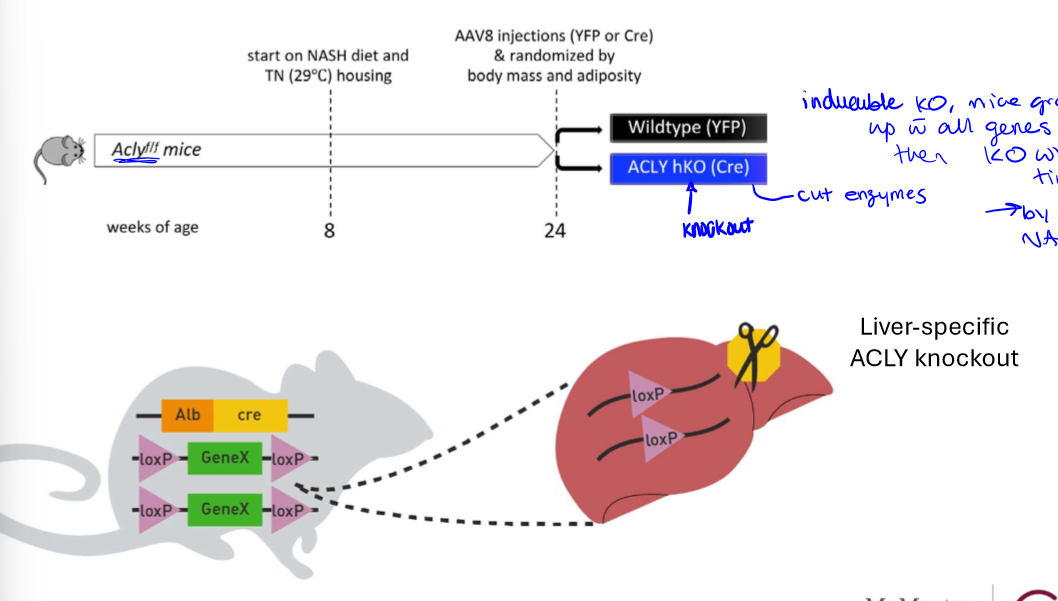
why was ACLY gene KO vs WT done
to test whether the inhibition of the gene would even work to reduce MASH symptoms
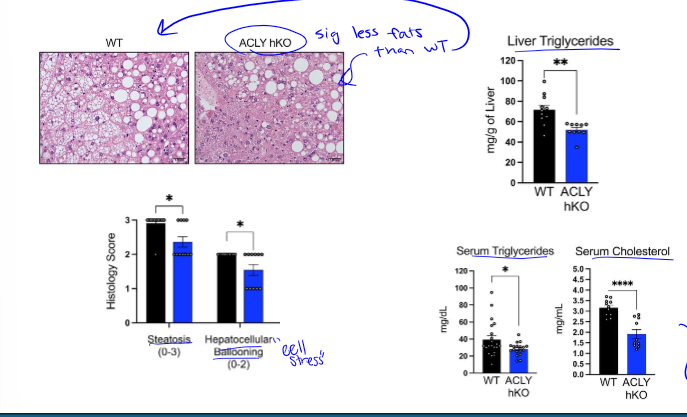
results of ACLY gene deletion
liver ACLY deletion reduces steatosis, reduces liver triglycerides, reduces hepatocellular ballooning, and also leads to less serum triglycerides and cholesterol (which was the issue w inhibition of ACC)
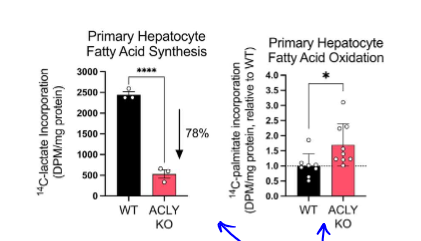
how is reduction in MASH with ACLY deletion happening
in KO, more fat is being oxidated than synthesized, which is what we want to see. this was found by giving radioactive substrates to primary liver cells
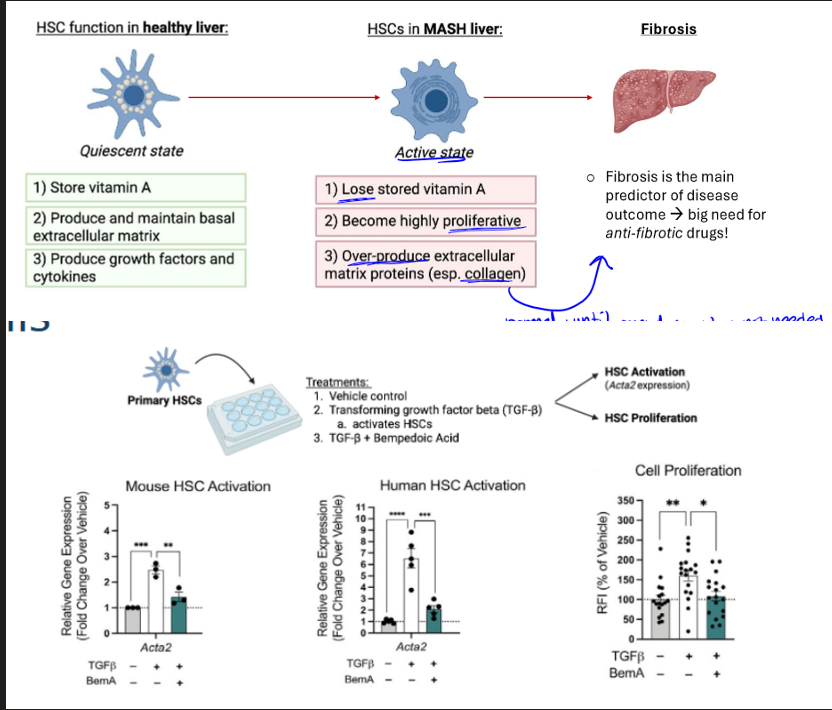
bempedoic acid and testing
pharmacologically inhibits ACLY in hepatocytes, and so this should recapitulate some of the effects we saw w the genetic KO model. mice in this test were given NASH diet, housed at TN, and then either given drug or vehicle control (like placebo)
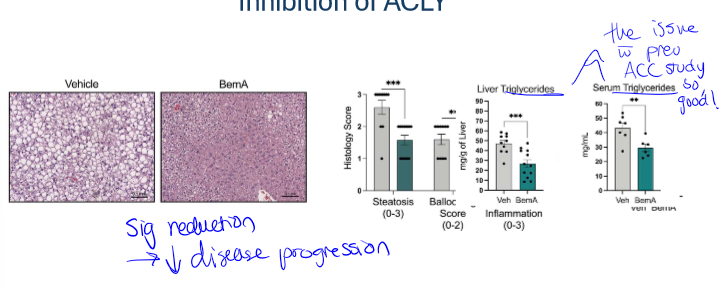
results of bempedoic acid test
significant reduction in disease progression for those with drug rather than vehicle control. it also showed reduced steatosis and ballooning, as well as less liver and serum triglycerides (which was the issue w ACC inhibition, so this is shown to work well)
steatosis and fibrosis with bempedoic acid
the drug lowers both through inhibition of ACLY (sig less red stains for collagen)
cell type involved in bempA study
the drug inhibits hepatic stellate cells (HSCs), which are non-parenchymal cells that make up 5-10% of liver cells.