Lecture 8/Ch 7: Monosaccharide and Disaccharides
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
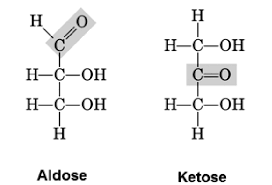
Aldose vs ketose
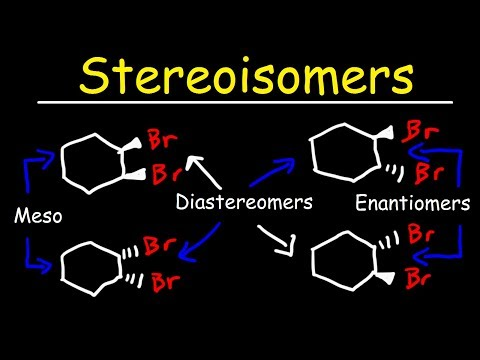
stereoisomers
position of one of the molecules in a chiral center is flipped
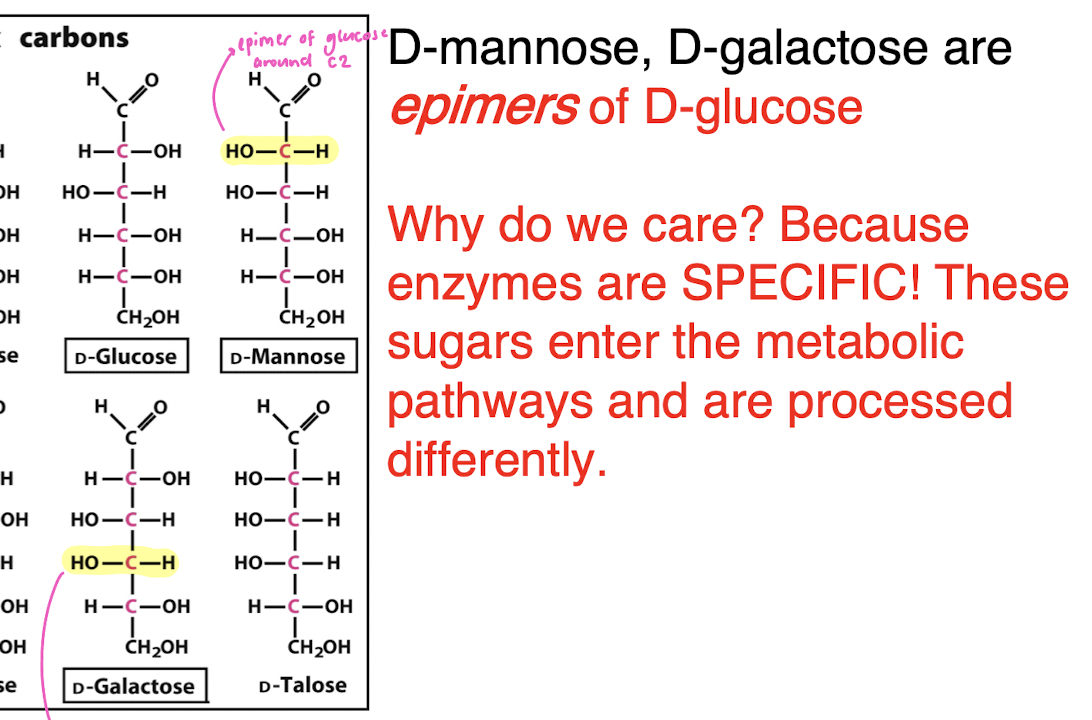
epimers
a subclass of stereoisomers that differ in the configuration of exactly one chiral center, while all other stereocenters remain identical
because enzymes are SPECIFIC and these sugars therefore enter metabolic pathways and are processes differently
Why do we care about epimers?
enantiomers
mirror images, switched at every chiral center
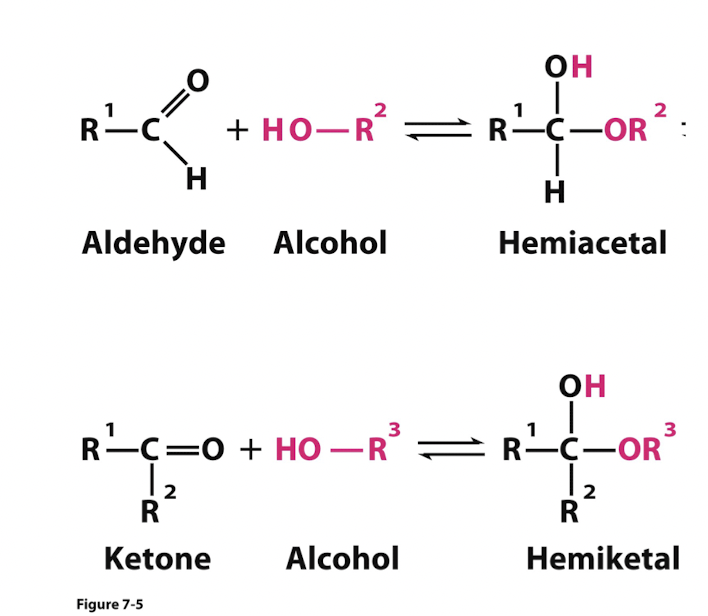
If intramolecular reactions occur, they make ring structures since sugars have both carbonyl and hydroxyl groups
Hemiacetal/Hemiketal reaction
anomeric carbon
the chiral center in a cyclic sugar created by the intramolecular reaction of a carbonyl group with a hydroxyl group to form a cyclic hemiacetal or hemiketal
anomers
a specific type of diastereomer (and epimer) found in cyclic sugar molecules, differing only in their configuration at the anomeric carbon (alpha vs beta)
alpha
at anomeric carbon when -OH is below the plane of the ring
beta
at anomeric carbon when -OH is above the plane of the ring
pyranose
6-membered ring
furanose
5-membered ring
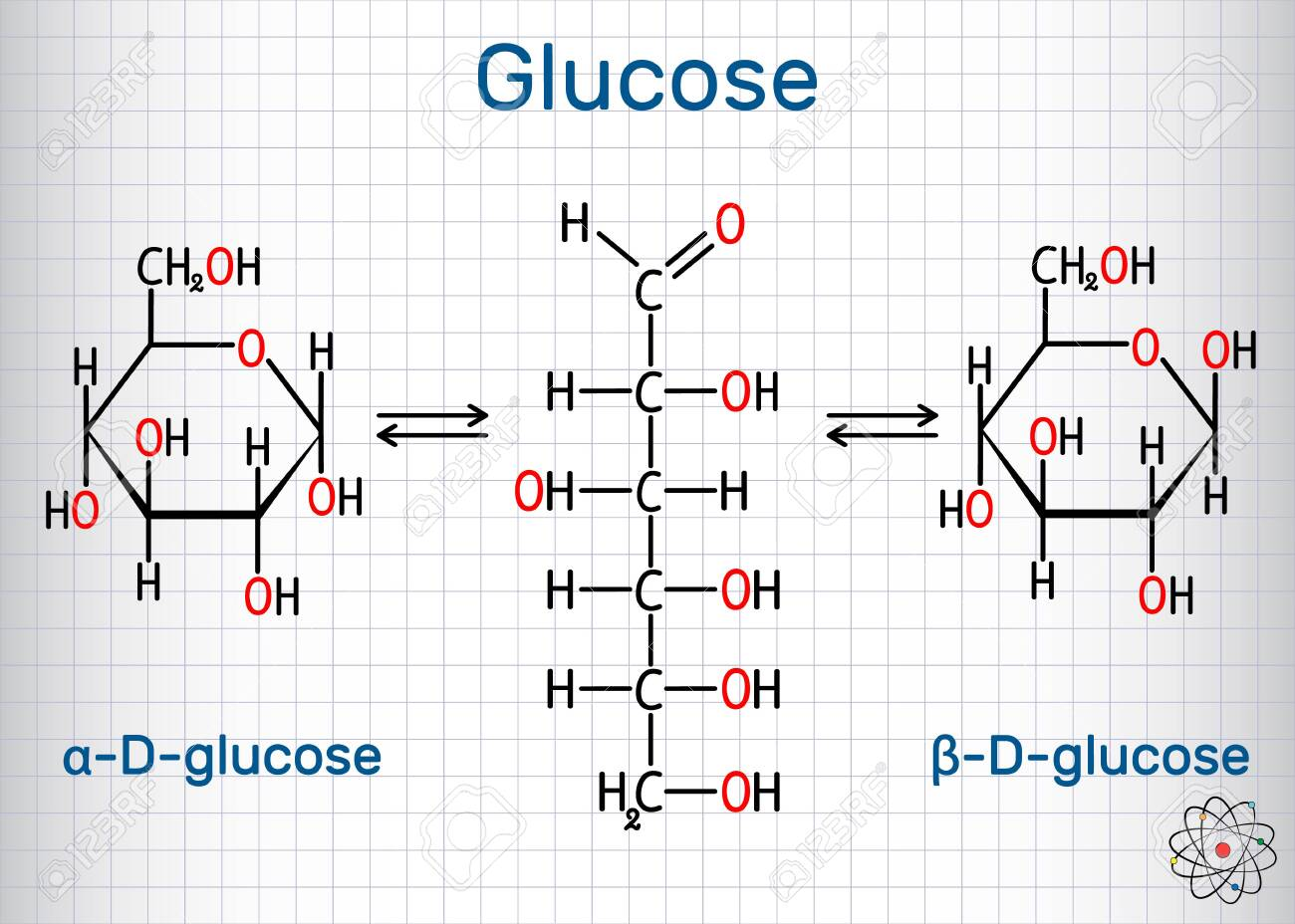
glucose
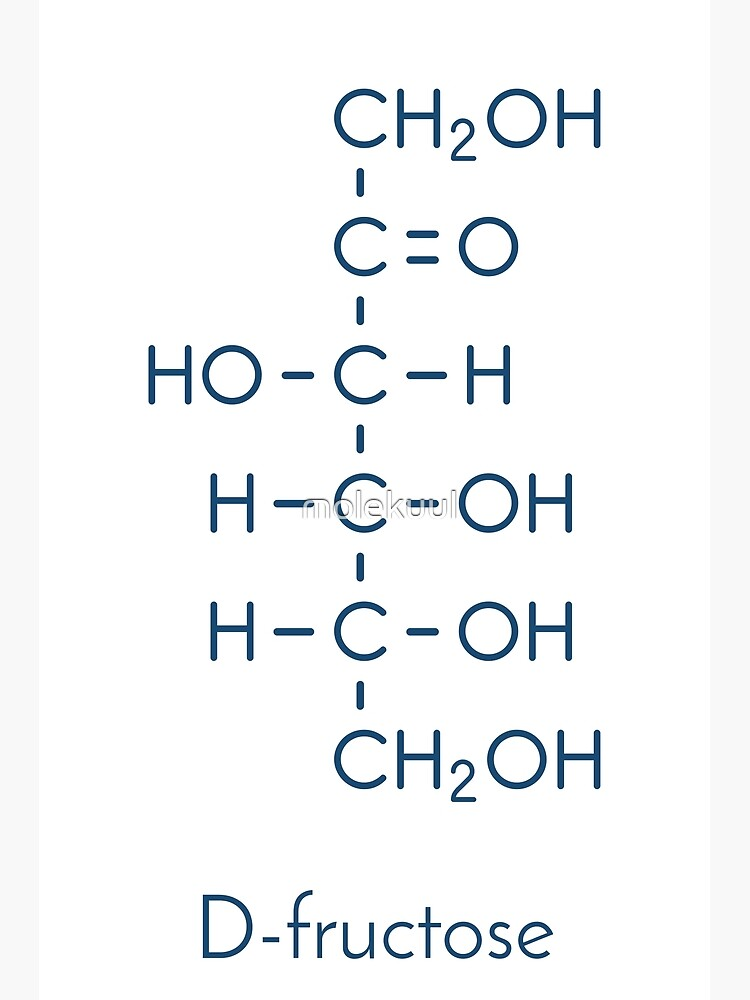
fructose
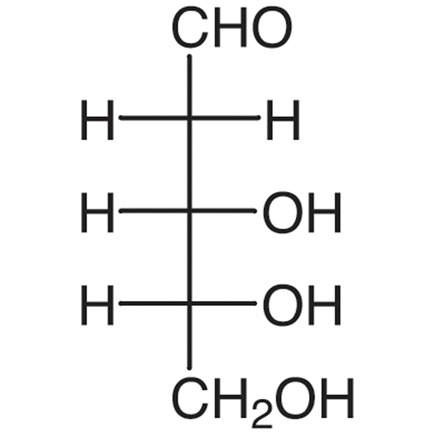
ribose
condensation reaction, hemiacetal/hemiketal + 2nd alcohol
how is a glycosidic bond made?
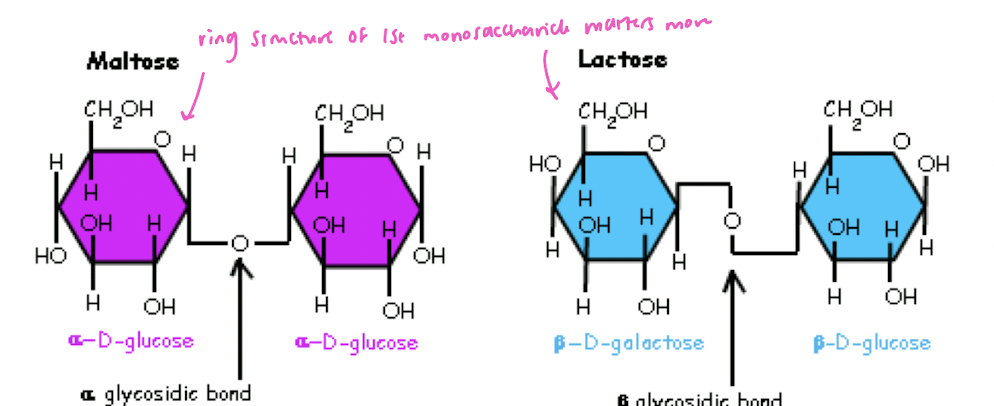
alpha vs beta glycosidic bond
reducing sugars
carbohydrates that possess a free aldehyde or ketone group, allowing them to act as reducing agents by donating electrons to other molecules, usually when reducing end C1 linearizes
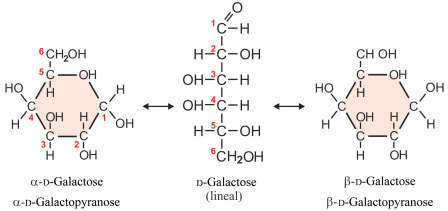
galactose
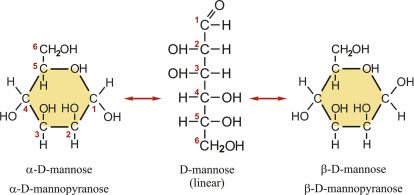
mannose